In Vitro and in Field Response of Different Fungicides against Aspergillus flavus and Fusarium Species Causing Ear Rot Disease of Maize
Abstract
:1. Introduction
2. Results
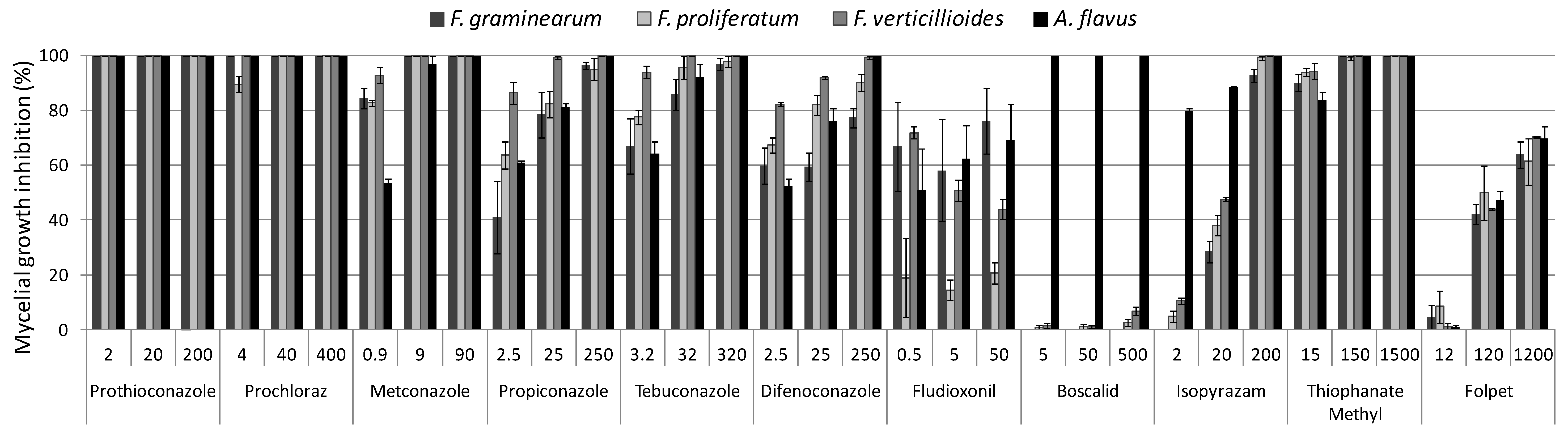
2.1. In Vitro Colony Growth Inhibition
2.1.1. DMIs
Metconazole
Propiconazole
Tebuconazole
Difenoconazole
2.1.2. SDHIs
Boscalid
Isopyrazam
2.1.3. PPs
Fludioxonil
2.1.4. MBCs
Thiophanate-methyl
2.1.5. Phthalimides
Folpet
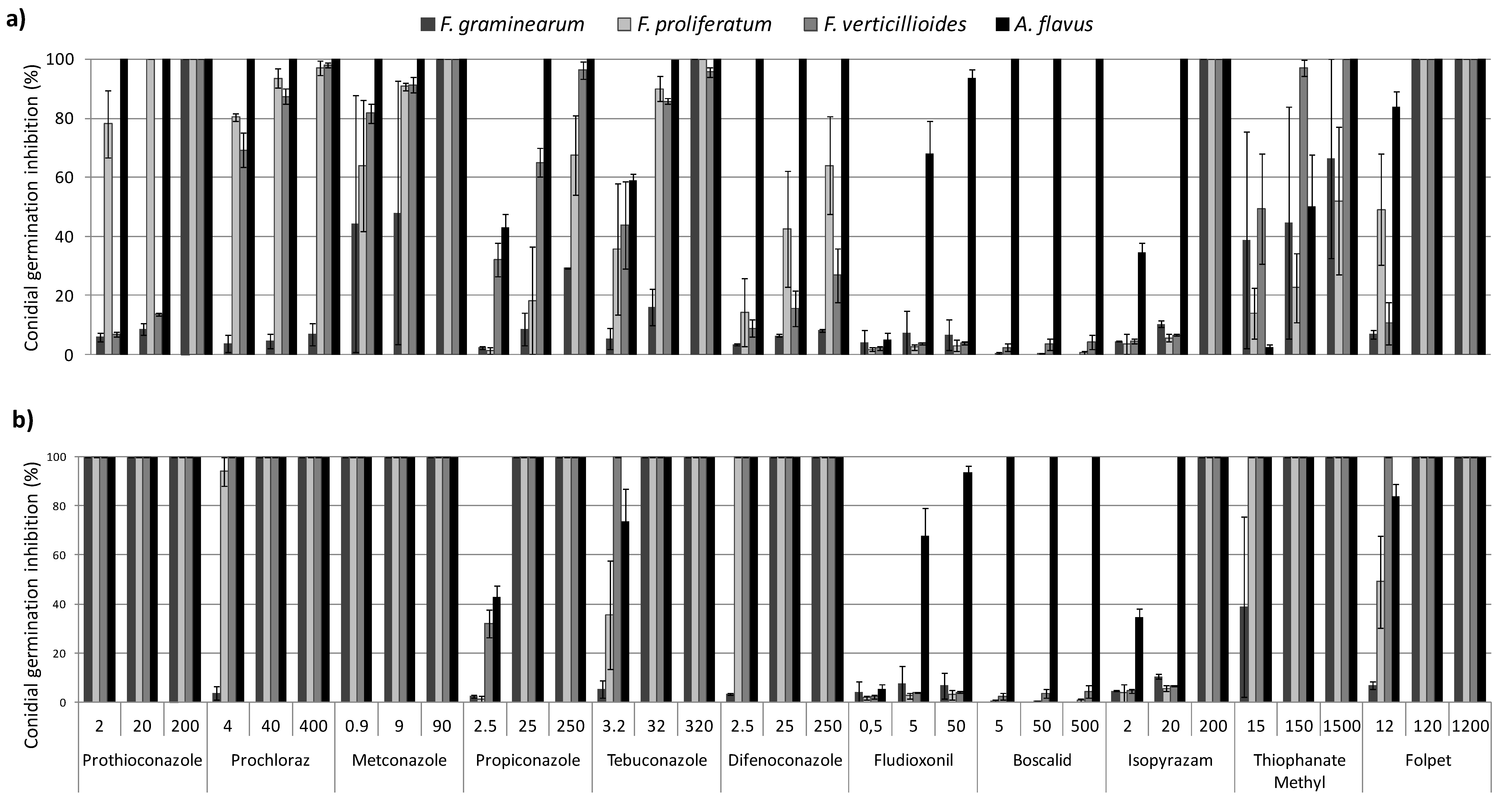
2.2. In Vitro Conidial Germination Inhibition
2.2.1. DMIs
2.2.2. SDHIs
2.2.3. PPs
2.2.4. MBCs
2.2.5. Phtalimides
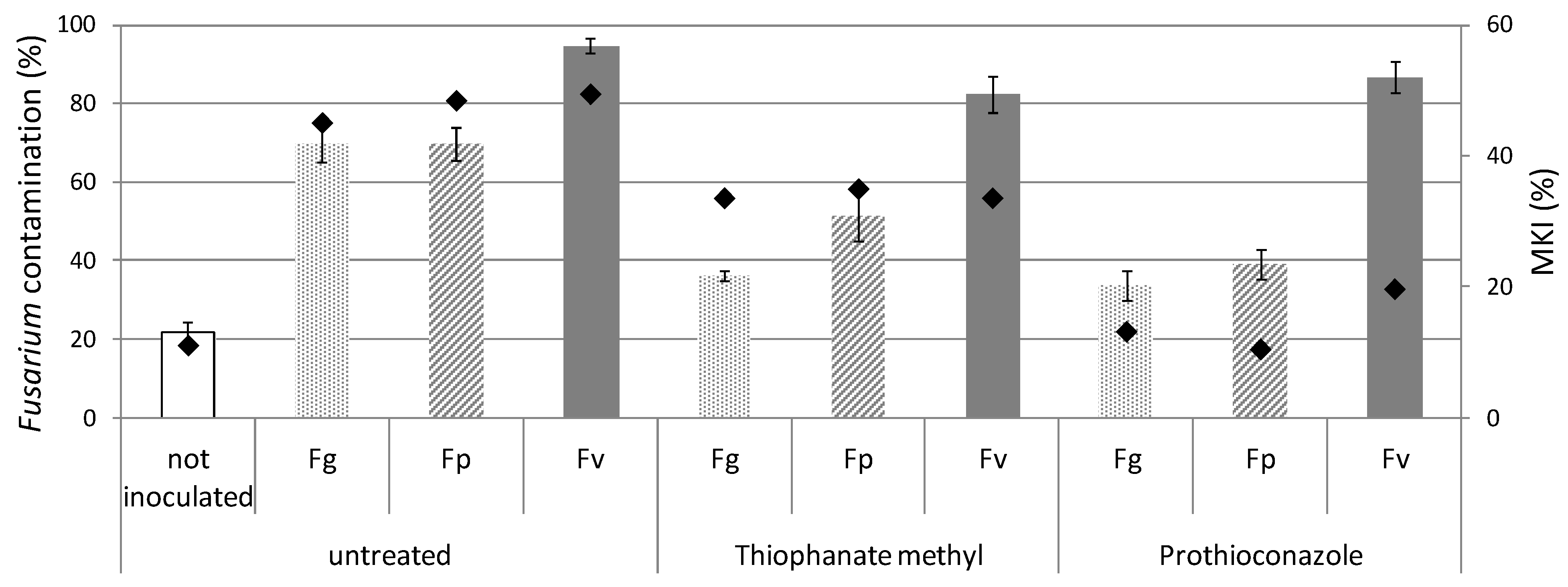
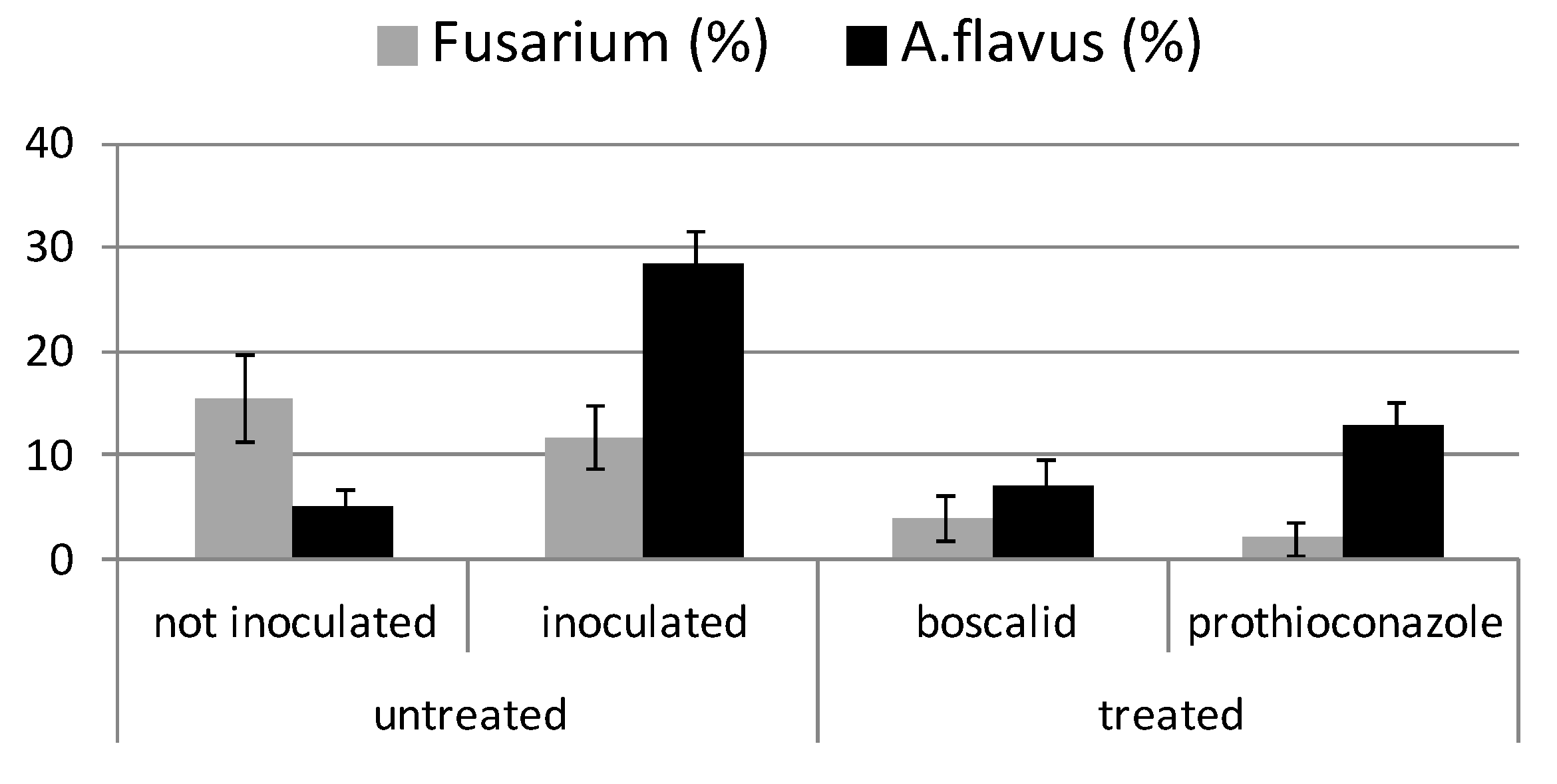
2.3. Fungal Symptoms Assessment on Maize Plants in Field Trials
2.4. Re-Isolation of Fungal Species from Maize Plants by Mycological Analyses
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Fungal Strains
5.2. Fungicides Tested In Vitro
5.3. Mycelial and Conidial Germination Assays
5.4. Field Experiments Settings
5.4.1. Fungicide Application and Fungal Inoculum
5.4.2. Evaluation of Fungal Symptoms on Maize Plants
5.4.3. Fungal Re-isolation and Growth Conditions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. Available online: www.fao.org/faostat/en (accessed on 11 November 2018).
- Latterell, F.M.; Rossi, A.E. Gray leaf spot of corn: A disease on the move. Plant Dis. 1983, 67, 842–847. [Google Scholar] [CrossRef]
- Leslie, J.F.; Pearson, C.A.S.; Nelson, P.E.; Toussoun, T.A. Fusarium spp. from corn, sorghum and soybean fields in the central and eastern United States. Phytopathology 1990, 80, 343–350. [Google Scholar] [CrossRef]
- Zhang, B.; Chen, W.; Yang, X. Occurrence of Pythium species in long-term maize and soybean monoculture and maize/soybean rotation. Mycol. Res. 1998, 102, 1450–1452. [Google Scholar] [CrossRef]
- White, D.G. Compendium of Corn Disease, 3rd ed.; American Phytopathological Society: St. Paul, MN, USA, 1999; ISBN 0890542341. [Google Scholar]
- Treikale, O.; Javoisha, B.; Pugacheva, E.; Vigule, Z.; Feodorova-Fedotova, L. Northern leaf blight Helminthosporium turcicum on maize in Latvia. Commun. Agric. Appl. Biol. Sci. 2014, 79, 481–485. [Google Scholar] [PubMed]
- Munkvold, G.P. Epidemiology of Fusarium disease and their mycotoxins in maize ears. Eur. J. Plant Pathol. 2003, 109, 705–713. [Google Scholar] [CrossRef]
- Brefort, T.; Doehlemann, G.; Mendoza-Mendoza, A.; Reissmann, S.; Djamei, A.; Kahmann, R. Ustilago maydis as a Pathogen. Annu. Rev. Phytopathol. 2009, 47, 423–445. [Google Scholar] [CrossRef]
- Vollmeister, E.; Schipper, K.; Baumann, S.; Haag, C.; Pohlmann, T.; Stock, J.; Feldbrügge, M. Fungal development of the plant pathogen Ustilago maydis. FEMS Microbiol. Rev. 2012, 36, 59–77. [Google Scholar] [CrossRef]
- Logrieco, A.F.; Mulè, G.; Moretti, A.; Bottalico, A. Toxigenic Fusarium species and mycotoxins associated with maize ear rot in Europe. In Mycotoxins in Plant Disease; Logrieco, A., Bailey, J.A., Corazza, L., Cooke, B.M., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002; pp. 597–609. ISBN 0-4020-0871-6. [Google Scholar]
- Logrieco, A.; Moretti, A.; Perrone, G.; Mulè, G. Biodiversity of complexes of mycotoxigenic fungal species associated with Fusarium ear rot of maize and Aspergillus rot of grape. Int. J. Food Microbiol. 2007, 119, 11–16. [Google Scholar] [CrossRef]
- Desjardins, A.E. Fusarium Mycotoxins. Chemistry, Genetics, and Biology; APS Press: St. Paul, MN, USA, 2006; ISBN 9780890543351. [Google Scholar]
- Escobar, J.; Lorán, S.; Giménez, I.; Ferruz, E.; Herrera, M.; Herrera, A.; Ariño, A. Occurrence and exposure assessment of Fusarium mycotoxins in maize germ, refined corn oil and margarine. Food Chem. Toxicol. 2013, 62, 514–520. [Google Scholar] [CrossRef]
- De Boevre, M.; Jacxsens, L.; Lachat, C.; Eeckhout, M.; Di Mavungu, J.D.; Audenaert, K.; Maene, P.; Haesaert, G.; Kolsteren, P.; De Meulenaer, B.; et al. Human exposure to mycotoxins and their masked forms through cereal-based foods in Belgium. Toxicol. Lett. 2013, 218, 281–292. [Google Scholar] [CrossRef] [Green Version]
- Sekiyama, B.L.; Ribeiro, A.B.; Machinski, P.A.; Machinski, M. Aflatoxins, ochratoxins and zearalenone in maize-based food products. Braz. J. Microbiol. 2005, 36, 289–294. [Google Scholar] [CrossRef]
- IARC. Some naturally occurring substances: Food ITEMs and constituents, heterocyclic aromatic amines and mycotoxins. In IARC Monographs on the Evaluation of Carcinogenic Risks to Human; IARC: Lyon, France, 1993; Volume 56, pp. 245–395. ISBN 92-832-1256-8. [Google Scholar]
- Logrieco, A.; Bottalico, A.; Mulè, G.; Moretti, A.; Perrone, G. Epidemiology of toxigenic fungi and their associated mycotoxins for some Mediterranean crops. Eur. J. Plant Pathol. 2003, 109, 645–667. [Google Scholar] [CrossRef]
- Balazs, E.; Schepers, J.S. The Mycotoxin Threat to Food Safety. Int. J. Food Microbiol. 2007, 119, 1–2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- EFSA (European Food Safety Authority). Evaluation of the increase of risk for public health related to a possible temporary derogation from the maximum level of deoxynivalenol, zearalenone and fumonisins for maize and maize products. EFSA J. 2014, 12, 3699. [Google Scholar] [CrossRef]
- Blandino, M.; Scarpino, V.; Giordano, D.; Sulyok, M.; Krska, R.; Vanara, F.; Reyneri, A. Impact of sowing time, hybrid and environmental condition of maize by emerging mycotoxins and fungal metabolites. Ital. J. Agron. 2017, 12, 928. [Google Scholar] [CrossRef]
- Battilani, P.; Toscano, P.; Van der Fels-Klerx, H.J.; Moretti, A.; Camardo Leggieri, M.; Brera, C.; Rortais, A.; Goumperis, T.; Robinson, T. Aflatoxin B1 contamination in maize in Europe increases due to climate change. Sci. Rep. 2016, 6, 24328. [Google Scholar] [CrossRef] [Green Version]
- Munkvold, G.P. Mycotoxins in corn: Occurrence, impacts, and management. In Corn Chemistry and Technology, 2nd ed.; White, P., Johnson, L., Eds.; American Association of Cereal Chemist: St. Paul, MI, USA, 2003; pp. 811–881. [Google Scholar]
- Folcher, L.; Jarry, M.; Weissenberger, A.; Gérault, F.; Eychenne, N.; Delos, M.; Regnault-Roger, C. Comparative activity of agrochemical treatments on mycotoxin level with regard to corn borers and Fusarium mycoflora in maize (Zea mays L.) fields. Crop Prot. 2009, 28, 302–308. [Google Scholar] [CrossRef]
- Mesterhazy, A.; Lemmens, M.; Reid, L.M. Breeding for resistance to ear rots caused by Fusarium spp. in maize—A review. Plant Breed. 2012, 131, 1–19. [Google Scholar] [CrossRef]
- Blandino, M.; Reyneri, A.; Vanara, F.; Ferrero, C. Control of mycotoxins in corn from harvesting to processing operation. In Proceedings of the International Quality Grains Conference, Indianapolis, Indiana, IN, USA, 19–22 July 2004. [Google Scholar]
- Abdallah, M.F.; Ameye, M.; De Saeger, S.; Audenaert, K.; Haesaert, G. Biological control of mycotoxigenic fungi and their toxins: An update for the pre-harvest approach. In Fungi and Mycotoxins—Their Occurrence, Impact on Health and the Economy as Well as Pre- and Postharvest Management Strategies; Berka Njobeh, P., Ed.; IntechOpen: London, UK, 2018. [Google Scholar]
- Blandino, M.; Reyneri, A. Effect of fungicide and foliar fertilizer application to winter wheat at anthesis on flag leaf senescence, grain yield, flour bread-making quality and DON contamination. Eur. J. Agron. 2009, 30, 275–282. [Google Scholar] [CrossRef]
- De Curtis, F.; Haidukowsky, M.; Moretti, A.; Castoria, R.; Lima, G.; Pascale, P. Occurence of Fusarium ear rot and fumonisin contamination of maize in Molise and effects of synthetic fungicides. J. Plant Pathol. 2008, 90, 371–376. [Google Scholar]
- Wang, J.X.; Zhou, M.G. Methods for monitoring resistance of Gibberella zeae to carbendazim. Acta Phytophylacica Sin. 2002, 29, 73–77. [Google Scholar]
- Chen, C.J.; Wang, J.X.; Luo, Q.Q.; Yuan, S.K.; Zhou, M.G. Characterization and fitness of carbendazim-resistant strains of Fusarium graminearum (wheat scab). Pest Manag. Sci. 2007, 63, 1201–1207. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhou, M.G. Characterization of Fusarium graminearum isolates resistant to both carbendazim and a new fungicide JS399-19. Phytopathology 2009, 99, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Liu, X.; Li, B.; Ma, Z. Characterization of steroldemethylation inhibitor-resistant isolates of Fusarium asiaticum and F. graminearum collected from wheat in China. Phytopathology 2009, 99, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Mazzoni, E.; Scandolara, A.; Giorni, P.; Pietri, A.; Battilani, P. Field control of Fusarium ear rot, Ostrinia nubilalis (Hübner) and fumonisins in maize kernels. Pest Manag. Sci. 2011, 67, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Edwards, S.G.; Pirgozliev, S.R.; Hare, M.C.; Jenkinson, P. Quantification of trichothecene-producing Fusarium species in harvest grain by competitive PCR to determine the efficacy of fungicides against Fusarium head blight of winter wheat. Appl. Environ. Microbiol. 2001, 67, 1575–1580. [Google Scholar] [CrossRef] [PubMed]
- Brent, K.J.; Hollomon, D.W. Fungicide Resistance: The Assessment of Risk. In Global Crop Protection Federation. (FRAC Monograph No. 2.); The Fungicide Resistance Action Committee Brussels: Brussels, Belgium, 2007. [Google Scholar]
- Hewitt, G. New modes of action of fungicides. Pestic. Outlook 2000, 11, 28–32. [Google Scholar] [CrossRef]
- Doohan, F.M.; Nicholson, P.; Parry, D.W. Efficacy of the fungicides prochloraz and pyrimethanil against Fusarium culmorum ear blight of wheat. In Proceedings of the International Conference: Pests & Diseases, Brighton, UK, 18–21 November 1996; pp. 409–410. [Google Scholar]
- Mateo, E.M.; Valle-Algarra, F.M.; Mateo, R.; Jimenez, M.; Magan, N. Effect of fenopropimorph, prochloraz and tebuconazole on growth and production of T-2 and HT-2 toxins by Fusarium langsethiae in oat-based medium. Int. J. Food Microbiol. 2011, 151, 289–298. [Google Scholar] [CrossRef]
- Mullenborn, C.; Steiner, U.; Ludwig, M.; Oerke, E.C. Effect of fungicides on the complex of Fusarium species and saprophytic fungi colonizing wheat kernels. Eur. J. Plant Pathol. 2008, 120, 157–166. [Google Scholar] [CrossRef]
- Audenaert, K.; Callewaert, E.; Höfte, M.; De Saeger, S.; Haesaert, G. Hydrogen peroxide induced by the fungicide prothioconazole triggers deoxynivalenol (DON) production by Fusarium graminearum. BMC Microbiol. 2010, 10, 112. [Google Scholar] [CrossRef]
- Formenti, S.; Magan, N.; Pietri, A.; Battilani, P. In Vitro impact on growth, fumonisins and aflatoxins production by Fusarium verticillioides and Aspergillus flavus using anti-fungal compounds and a biological control agent. Phytopathol. Mediterr. 2012, 51, 247–256. [Google Scholar] [CrossRef]
- Klix, M.B.; Verreet, J.A.; Beyer, M. Comparison of the declining triazole sensitivity of Gibberella zeae and increased sensitivity achieved by advances in triazole fungicide development. Crop Prot. 2007, 26, 683–690. [Google Scholar] [CrossRef]
- Avenot, H.F.; Michailides, T.J. Progress in understanding molecular mechanisms and evolution of resistance to succinate dehydrogenase inhibiting (SDHI) fungicides in phytopathogenic fungi. Crop Prot. 2010, 29, 643–651. [Google Scholar] [CrossRef]
- Zhang, C.Q.; Liu, Y.H.; Ma, X.Y.; Feng, Z.; Ma, Z.H. Characterization of sensitivity of Rhizoctonia solani, causing rice sheath blight, to mepronil and boscalid. Crop Prot. 2009, 28, 381–386. [Google Scholar] [CrossRef]
- Ishii, H.; Miyamoto, T.; Ushio, S.; Kakishima, M. Lack of cross-resistance to a novel succinate dehydrogenase inhibitor, fluopyram, in highly boscalid-resistant isolates of Corynespora cassiicola and Podosphaer axanthii. Pest Manag. Sci. 2011, 67, 474–482. [Google Scholar] [CrossRef] [PubMed]
- De Miccolis Angelini, R.M.; Masiello, M.; Rotolo, C.; Pollastro, S.; Faretra, F. Molecular characterization and detection of resistance to succinate dehydrogenase inhibitor fungicides in Botryotinia fuckeliana (Botrytis cinerea). Pest Manag. Sci. 2014, 70, 1884–1893. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, K.; Ward, T.J.; Geiser, D.M.; Kistler, H.C.; Aoki, T. Genealogical concordance between mating type locus and seven other nuclear genes supports formal recognition of nine phylogenetically distinct species within the Fusarium graminearum clade. Fungal Genet. Biol. 2004, 41, 600–623. [Google Scholar] [CrossRef]
- Hou, Y.P.; Mao, X.W.; Wang, J.X.; Zhan, S.W.; Zhou, M.G. Sensitivity of Fusarium asiaticum to a novel succinate dehydrogenase inhibitor fungicide pydiflumetofen. Crop Prot. 2017, 96, 237–244. [Google Scholar] [CrossRef]
- Marques, L.N.; Pizzutti, I.R.; Balardin, R.S.; Dos Santos, I.D.; Dias, J.V.; Stefanello, M.T.; Serafini, P.T. Occurrence of mycotoxins in wheat grains exposed to fungicides on Fusarium head blight control in southern Brazil. J. Environ. Sci. Health B 2017, 52, 244–250. [Google Scholar] [CrossRef]
- Dubos, T.; Pasquali, M.; Pogoda, F.; Casanova, A.; Hoffmann, L.; Beyer, M. Differences between the succinate dehydrogenase sequences of isopyrazam sensitive Zymoseptoria tritici and insensitive Fusarium graminearum strains. Pestic. Biochem. Phys. 2013, 105, 28–35. [Google Scholar] [CrossRef]
- Dubos, T.; Pasquali, M.; Pogoda, F.; Hoffmann, L.; Beyer, M. Evidence for natural resistance towards trifloxystrobin in Fusarium graminearum. Eur. J. Plant Pathol. 2011, 130, 239–248. [Google Scholar] [CrossRef]
- O’Neill, T.M. Evaluation of fungicides against fusarium wilt (Fusarium oxysporum f.sp. cyclaminis) of cyclamen. Tests Agrochem. Cultiv. 1995, 16, 20–21. [Google Scholar]
- Munkvold, G.P.; O’Mara, J.K. Laboratory and Growth Chamber Evaluation of Fungicidal Seed Treatments for Maize Seedling Blight Caused by Fusarium Species. Plant Dis. 2002, 86, 143–150. [Google Scholar] [CrossRef]
- Broders, K.D.; Lipps, P.E.; Paul, P.A.; Dorrance, A.E. Evaluation of Fusarium graminearum associated with corn and soybean seed and seedling disease in Ohio. Plant Dis. 2007, 91, 1155–1160. [Google Scholar] [CrossRef]
- Munkvold, G.P.; Watrin, C.; Scheller, M.; Zeun, R.; Olaya, G. Benefits of Chemical Seed Treatments on Crop Yield and Quality. In Global Perspectives on the Health of Seeds and Plant Propagation Material. Plant Pathology in the 21st Century; Gullino, M., Munkvold, G., Eds.; Springer: Heidelberg, Germany, 2014; Volume 6, pp. 89–103. [Google Scholar]
- Sooväli, P.; Koppel, M.; Kangor, T. Effectiveness of seed treatment against Fusarium spp. and Cochliobolus sativus of spring barley in different conditions. Agron. Res. 2017, 15, 280–287. [Google Scholar]
- Peters, R.D.; Platt, H.W.; Drake, K.A.; Coffin, R.H.; Moorehead, S.; Clark, M.M.; Al-Mughrabi, K.I.; Howard, R.J. First Report of Fludioxonil-Resistant Isolates of Fusarium spp. Causing Potato Seed-Piece Decay. Plant Dis. 2008, 92, 172. [Google Scholar] [CrossRef]
- Ellis, M.L.; Broders, K.D.; Paul, P.A.; Dorrance, A.E. Infection of Soybean Seed by Fusarium graminearum and Effect of Seed Treatments on Disease Under Controlled Conditions. Plant Dis. 2011, 95, 401–407. [Google Scholar] [CrossRef]
- Miguel, T.A.; Bordini, J.G.; Saito, G.H.; Andrade, C.G.T.J.; Ono, M.A.; Hirooka, E.Y.; Vizoni, E.; Ono, E.Y.S. Effect of fungicide on Fusarium verticillioides mycelia morphology and fumonisin B1 production. Braz. J. Microbiol. 2015, 46, 293–299. [Google Scholar] [CrossRef] [PubMed]
| Commercial Name | Active Ingredient | Active Ingredient Tested (mg L−1) | Chemical Group * | Group Name * | Target Site * | Mode of Action * |
|---|---|---|---|---|---|---|
| Cantus | Boscalid | 500–50–5 | pyridine-carboxamides | SDHI (Succinate dehydrogenase inhibitors) | complex II: succinate-dehydrogenase | Respiration |
| Zulu | Isopyrazam | 200–20–2 | pyrazole-4-carboxamides | |||
| Carnival | Prochloraz | 400–40–4 | Imidazoles | Demethylation Inhibitors SBI Class I | C14-demethylase in sterol biosynthesis | Sterol biosynthesis in membranes |
| Proline | Prothioconazole | 200–20–2 | Triazolinthiones | |||
| Icarus | Tebuconazole | 320–32–3.2 | Triazoles | |||
| Opinion Ecna | Propiconazole | 250–25–2.5 | ||||
| Caramba | Metconazole | 90–9–0.9 | ||||
| Score | Difenoconazole | 250–25–2.5 | ||||
| Celest | Fludioxonil | 50–5–0.5 | phenylpyrroles | PP-fungicides (PhenylPyrroles) | MAP/Histidine—Kinase in osmotic signal transduction | signal transduction |
| Enovit Metil FL | Thiophanate-methyl | 1500–150–15 | Thiophanates | MBC-Fungicides (Methyl Benzimidazole Carbamates) | β-tubuline assembly in mitosis | mitosis and cell division |
| Folpan80 | Folpet | 1200–120–12 | phthalimides | Phthalimides | multisite contact activity | Multisite contact activity |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Masiello, M.; Somma, S.; Ghionna, V.; Logrieco, A.F.; Moretti, A. In Vitro and in Field Response of Different Fungicides against Aspergillus flavus and Fusarium Species Causing Ear Rot Disease of Maize. Toxins 2019, 11, 11. https://doi.org/10.3390/toxins11010011
Masiello M, Somma S, Ghionna V, Logrieco AF, Moretti A. In Vitro and in Field Response of Different Fungicides against Aspergillus flavus and Fusarium Species Causing Ear Rot Disease of Maize. Toxins. 2019; 11(1):11. https://doi.org/10.3390/toxins11010011
Chicago/Turabian StyleMasiello, Mario, Stefania Somma, Veronica Ghionna, Antonio Francesco Logrieco, and Antonio Moretti. 2019. "In Vitro and in Field Response of Different Fungicides against Aspergillus flavus and Fusarium Species Causing Ear Rot Disease of Maize" Toxins 11, no. 1: 11. https://doi.org/10.3390/toxins11010011







