Tissue Distribution and Elimination of Ciguatoxins in Tridacna maxima (Tridacnidae, Bivalvia) Fed Gambierdiscus polynesiensis
Abstract
1. Introduction
2. Results
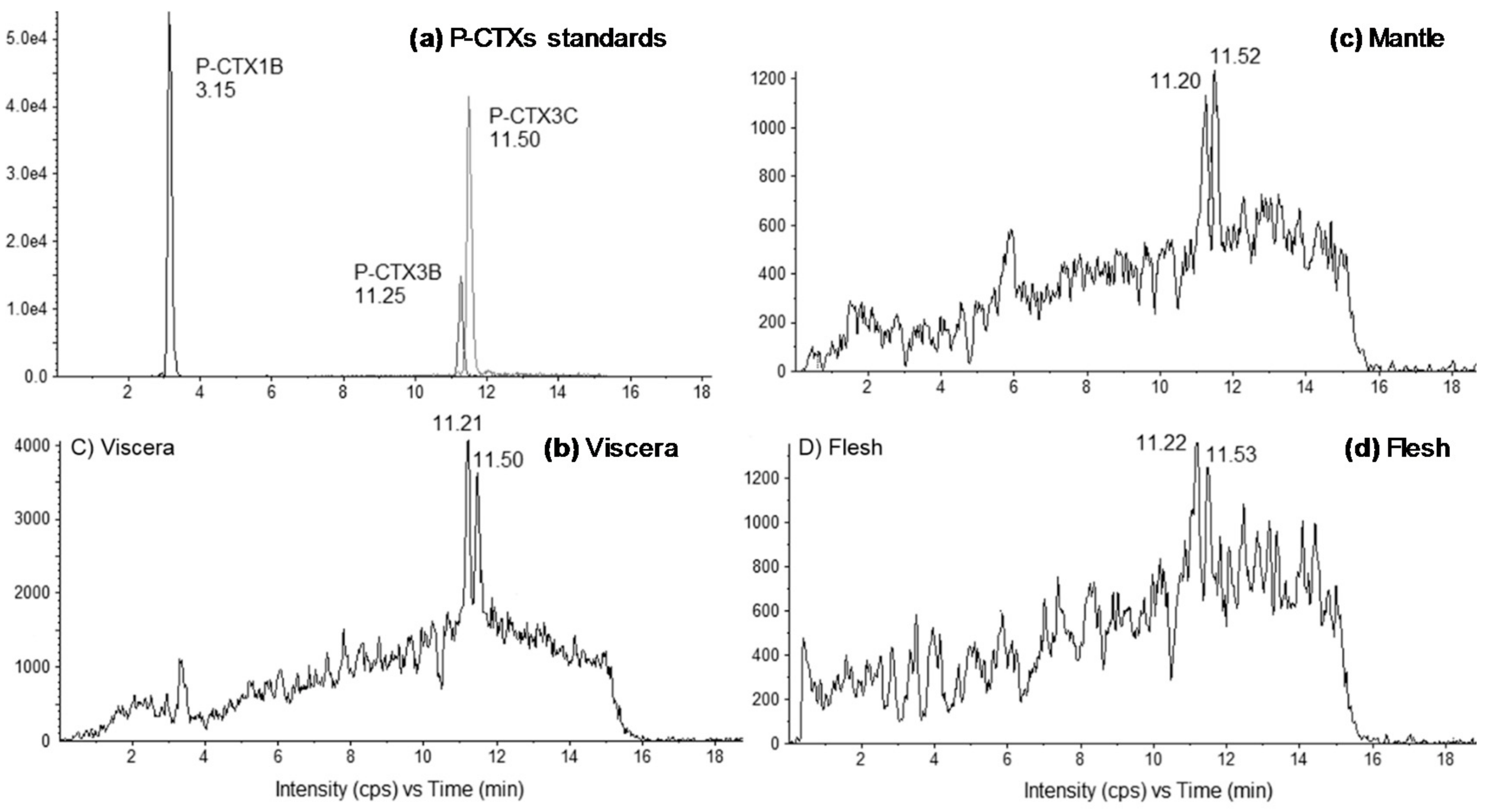
2.1. Tissue Distribution Study
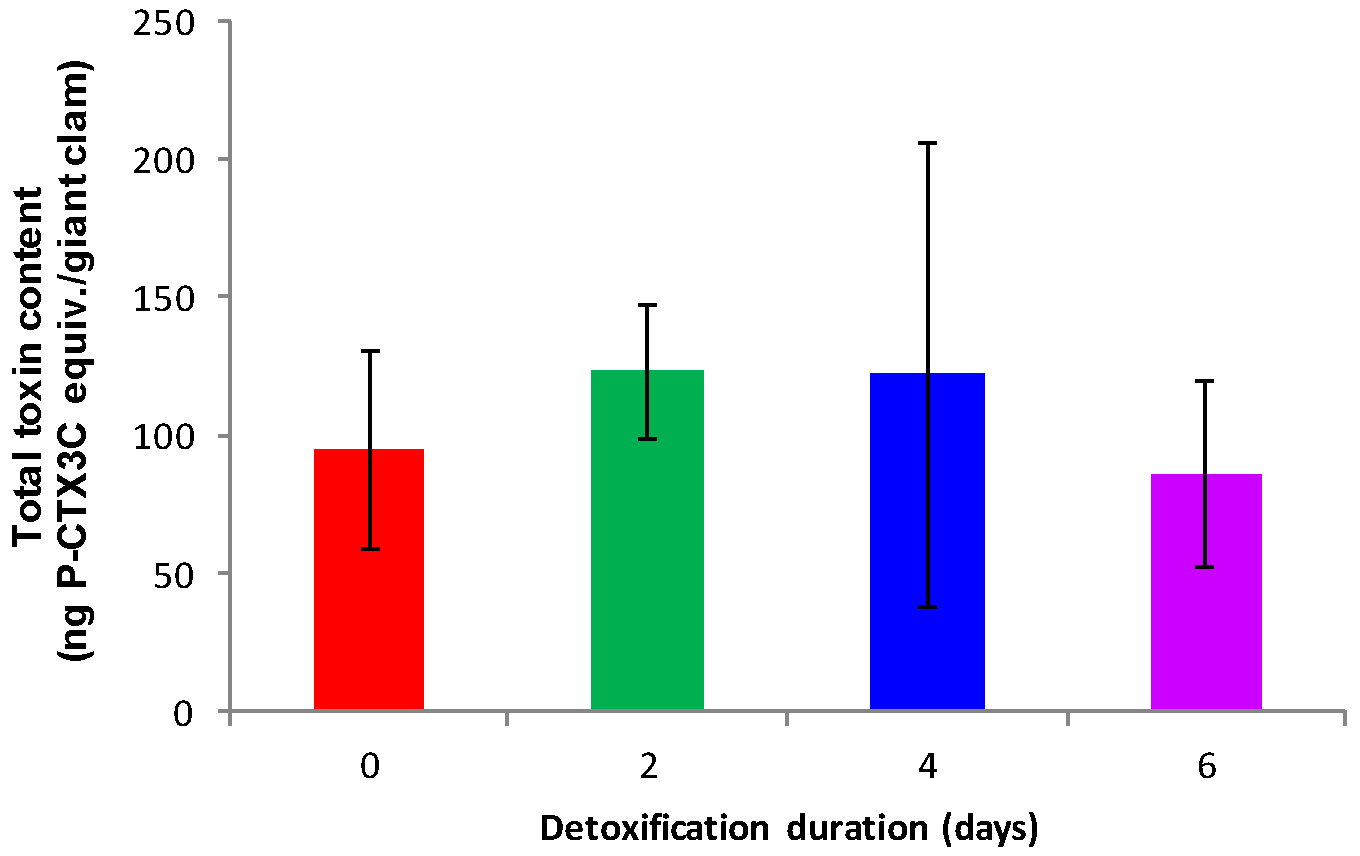
2.2. Detoxification Study
3. Discussion
3.1. Anatomical Distribution of CTXs in Toxic Giant Clams
3.2. Elimination of CTXs from T. maxima
4. Materials and Methods
4.1. Biological Material
4.1.1. Giant Clams
4.1.2. Cultures of Gambierdiscus polynesiensis
4.2. Ex Situ Exposure Experiments
4.2.1. Tissue Distribution Study
4.2.2. Detoxification Study
4.3. Toxin Extration
4.4. Neuroblastoma Cell-Based Assay (CBA-N2a)
4.5. Liquid Chromatography Coupled with Tandem Mass Spectrometry (LC-MS/MS)
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Bell, J.D.; Kronen, M.; Vunisea, A.; Nash, W.J.; Keeble, G.; Demmke, A.; Pontifex, S.; Andréfouët, S. Planning the use of fish for food security in the Pacific. Mar. Policy 2009, 33, 64–76. [Google Scholar] [CrossRef]
- Charlton, K.E.; Russell, J.; Gorman, E.; Hanich, Q.; Delisle, A.; Campbell, B.; Bell, J. Fish, food security and health in Pacific Island countries and territories: A systematic literature review. BMC Public Health 2016, 16. [Google Scholar] [CrossRef] [PubMed]
- Van Wynsberge, S.; Andréfouët, S.; Gaertner-Mazouni, N.; Wabnitz, C.C.C.; Gilbert, A.; Remoissenet, G.; Payri, C.; Fauvelot, C. Drivers of density for the exploited giant clam Tridacna maxima: A meta-analysis. Fish Fish. 2016, 17, 567–584. [Google Scholar] [CrossRef]
- Gilbert, A.; Andréfouët, S.; Yan, L.; Remoissenet, G. The giant clam Tridacna maxima communities of three French Polynesia islands: Comparison of their population sizes and structures at early stages of their exploitation. ICES J. Mar. Sci. 2006, 63, 1573–1589. [Google Scholar] [CrossRef]
- Direction des Ressources Marines et Minières de Polynésie Française. Available online: http://www.peche.pf/spip.php?rubrique213 (accessed on 27 February 2018).
- Dickey, R.W.; Plakas, S.M. Ciguatera: A public health perspective. Toxicon 2010, 56, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M.A.; Fernandez, M.; Backer, L.C.; Dickey, R.W.; Bernstein, J.; Schrank, K.; Kibler, S.; Stephan, W.; Gribble, M.O.; Bienfang, P.; et al. An updated review of ciguatera fish poisoning: Clinical, epidemiological, environmental, and public health management. Mar. Drugs 2017, 15, 72. [Google Scholar] [CrossRef] [PubMed]
- Bagnis, R. A propos de quelques cas d’intoxications par des mollusques du genre “bénitier” dans une île de la Société. Bull. Soc. Pathol. Exot. 1967, 60, 580–592. [Google Scholar]
- Bagnis, R. Situation de l’endémicité ciguatérique aux îles Gambier. Cahiers Pacifique 1974, 18, 585–600. [Google Scholar]
- Rongo, T.; van Woesik, R. Socioeconomic consequences of ciguatera poisoning in Rarotonga, southern Cook Islands. Harmful Algae 2012, 20, 92–100. [Google Scholar] [CrossRef]
- Laurent, D.; Kerbrat, A.S.; Darius, H.T.; Rossi, F.; Yeeting, B.; Haddad, M.; Golubic, S.; Pauillac, S.; Chinain, M. Ciguatera Shellfish Poisoniong (CSP): A new ecotoxicological phenomenon from cyanobacteria to humans via giant clams. In Food Chains: New Research; Jensen, M.A., Muller, D.W., Eds.; Nova Science Publishers, Inc.: New York, NY, USA, 2012; pp. 1–44. ISBN 9781613243770. [Google Scholar]
- Gatti, C.; Lonati, D.; Darius, H.T.; Zancan, A.; Roué, M.; Schicchi, A.; Locatelli, C.A.; Chinain, M. Tectus niloticus (Tegulidae, Gastropod) as a Novel Vector of Ciguatera Poisoning: Clinical Characterization and Follow-Up of a Mass Poisoning Event in Nuku Hiva Island (French Polynesia). Toxins 2018, 10, 102. [Google Scholar] [CrossRef] [PubMed]
- Darius, H.T.; Roué, M.; Sibat, M.; Viallon, J.; Gatti, C.M.I.; Vandersea, M.W.; Tester, P.A.; Litaker, R.W.; Amzil, Z.; Hess, P.; et al. Tectus niloticus (Tegulidae, Gastropod) as a Novel Vector of Ciguatera Poisoning: Detection of Pacific Ciguatoxins in Toxic Samples from Nuku Hiva Island (French Polynesia). Toxins 2018, 10, 2. [Google Scholar] [CrossRef] [PubMed]
- Darius, H.T.; Roué, M.; Sibat, M.; Viallon, J.; Gatti, C.; Vandersea, M.; Tester, P.A.; Litaker, R.W.; Amzil, Z.; Hess, P.; et al. Toxicological investigations on the sea urchin Tripneustes gratilla (Toxopneustidae, Echinoid) from Anaho Bay (Nuku Hiva, French Polynesia): Evidence for the presence of Pacific ciguatoxins. Mar. Drugs 2018, 16, 122. [Google Scholar] [CrossRef] [PubMed]
- Laurent, D.; Kerbrat, A.S.; Darius, H.T.; Girard, E.; Golubic, S.; Benoit, E.; Sauviat, M.P.; Chinain, M.; Molgo, J.; Pauillac, S. Are cyanobacteria involved in Ciguatera Fish Poisoning-like outbreaks in New Caledonia? Harmful Algae 2008, 7, 827–838. [Google Scholar] [CrossRef]
- Pawlowiez, R.; Darius, H.T.; Cruchet, P.; Rossi, F.; Caillaud, A.; Laurent, D.; Chinain, M. Evaluation of seafood toxicity in the Australes archipelago (French Polynesia) using the neuroblastoma cell-based assay. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2013, 30, 567–586. [Google Scholar] [CrossRef] [PubMed]
- Roué, M.; Darius, H.T.; Picot, S.; Ung, A.; Viallon, J.; Gaertner-Mazouni, N.; Sibat, M.; Amzil, Z.; Chinain, M. Evidence of the bioaccumulation of ciguatoxins in giant clams (Tridacna maxima) exposed to Gambierdiscus spp. cells. Harmful Algae 2016, 57, 78–87. [Google Scholar] [CrossRef]
- Bricelj, V.M.; Shumway, S.E. Paralytic Shellfish Toxins in Bivalve Molluscs: Occurrence, Transfer Kinetics, and Biotransformation. Rev. Fish. Sci. 1998, 6, 315–383. [Google Scholar] [CrossRef]
- Lewis, R.J.; Sellin, M.; Street, R.; Holmes, M.H.; Gillepsie, N.C. Excretion of ciguatoxin from moray eels (Muraenidae) of the central Pacific. In Proceedings of the Third International Conference on Ciguatera Fish Poisoning; Tosteson, T.R., Ed.; Polysciences Publications: Quebec, QC, Canada, 1992; pp. 131–143. [Google Scholar]
- Chateau-Degat, M.L.; Huin-Blondey, M.O.; Chinain, M.; Darius, T.; Legrand, A.M.; Ngoc, L.N.; Laudon, F.; Chansin, R.; Dewailly, E. Prevalence of chronic symptoms of ciguatera disease in French Polynesian adults. Am. J. Trop. Med. Hyg. 2007, 77, 842–846. [Google Scholar] [PubMed]
- Chen, C.Y.; Chou, H.N. Accumulation and depuration of paralytic shellfish poisoning toxins by purple clam Hiatula rostrata Lighttoot. Toxicon 2001, 39, 1029–1034. [Google Scholar] [CrossRef]
- Blanco, J.; Acosta, C.P.; Bermúdez de la Puente, M.; Salgado, C. Depuration and anatomical distribution of the amnesic shellfish poisoning (ASP) toxin domoic acid in the king scallop Pecten maximus. Aquat. Toxicol. 2002, 60, 111–121. [Google Scholar] [CrossRef]
- Kwong, R.W.M.; Wang, W.X.; Lam, P.K.S.; Yu, P.K.N. The uptake, distribution and elimination of paralytic shellfish toxins in mussels and fish exposed to toxic dinoflagellates. Aquat. Toxicol. 2006, 80, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Blanco, J.; Mariño, C.; Martín, H.; Acosta, C.P. Anatomical distribution of diarrhetic shellfish poisoning (DSP) toxins in the mussel Mytilus galloprovincialis. Toxicon 2007, 50, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Lassus, P.; Amzil, Z.; Baron, R.; Séchet, V.; Barillé, L.; Abadie, E.; Bardouil, M.; Sibat, M.; Truquet, P.; Bérard, J.B.; et al. Modelling the accumulation of PSP toxins in Thau Lagoon oysters (Crassostrea gigas) from trials using mixed cultures of Alexandrium catenella and Thalassiosira weissflogii. Aquat. Living Resour. 2007, 20, 59–67. [Google Scholar] [CrossRef]
- Murray, S.A.; O’Connor, W.A.; Alvin, A.; Mihali, T.K.; Kalaitzis, J.; Neilan, B.A. Differential accumulation of paralytic shellfish toxins from Alexandrium minutum in the pearl oyster, Pinctada imbricata. Toxicon 2009, 54, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Matsushima, R.; Uchida, H.; Nagai, S.; Watanabe, R.; Kamio, M.; Nagai, H.; Kaneniwa, M.; Suzuki, T. Assimilation, accumulation, and metabolism of dinophysistoxins (DTXs) and pectenotoxins (PTXs) in the several tissues of Japanese scallop Patinopecten yessoensis. Toxins 2015, 7, 5141–5154. [Google Scholar] [CrossRef] [PubMed]
- García, C.; Pérez, F.; Contreras, C.; Figueroa, D.; Barriga, A.; López-Rivera, A.; Araneda, O.F.; Contreras, H.R. Saxitoxins and okadaic acid group: Accumulation and distribution in invertebrate marine vectors from Southern Chile. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2015, 32, 984–1002. [Google Scholar] [CrossRef] [PubMed]
- Kameneva, P.A.; Krasheninina, E.A.; Slobodskova, V.V.; Kukla, S.P.; Orlova, T.Y. Accumulation and tissue distribution of dinophysitoxin-1 and dinophysitoxin-3 in the mussel Crenomytilus grayanus feeding on the benthic dinoflagellate Prorocentrum foraminosum. Mar. Drugs 2017, 15, 330. [Google Scholar] [CrossRef] [PubMed]
- Jauffrais, T.; Marcaillou, C.; Herrenknecht, C.; Truquet, P.; Séchet, V.; Nicolau, E.; Tillmann, U.; Hess, P. Azaspiracid accumulation, detoxification and biotransformation in blue mussels (Mytilus edulis) experimentally fed Azadinium spinosum. Toxicon 2012, 60, 582–595. [Google Scholar] [CrossRef] [PubMed]
- Drum, A.S.; Siebens, T.L.; Crecelius, E.A.; Elston, R.A. Domoic acid in the Pacific razor clam Siliqua patula (Dixon, 1789). J. Shellfish Res. 1993, 12, 443–450. [Google Scholar]
- Wekell, J.C.; Gauglitz, E.J.; Bamett, H.J.; Hatfield, C.L.; Simons, D.; Ayres, D. Occurrence of domoic acid in washington state razor clams (Siliqua patula) during 1991–1993. Nat. Toxins 1994, 2, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Sagou, R.; Amanhir, R.; Taleb, H.; Vale, P.; Blaghen, M.; Loutfi, M. Comparative study on differential accumulation of PSP toxins between cockle (Acanthocardia tuberculatum) and sweet clam (Callista chione). Toxicon 2005, 46, 612–618. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Scientific Opinion on marine biotoxins in shellfish-Emerging toxins: Ciguatoxin group. EFSA J. 2010, 8, 1627. [Google Scholar] [CrossRef]
- Hossen, V.; Soliño, L.; Leroy, P.; David, E.; Velge, P.; Dragacci, S.; Krys, S.; Flores Quintana, H.; Diogène, J. Contribution to the risk characterization of ciguatoxins: LOAEL estimated from eight ciguatera fish poisoning events in Guadeloupe (French West Indies). Environ. Res. 2015, 143, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Anonymous. Domoic Acid in the King Scallop, Pecten Maximus; Unpublished Report Prepared for the EU ASP Working Group; UK National Reference Laboratory for Marine Biotoxins: Belfast, UK, 2001. [Google Scholar]
- Chinain, M.; Darius, H.T.; Ung, A.; Cruchet, P.; Wang, Z.; Ponton, D.; Laurent, D.; Pauillac, S. Growth and toxin production in the ciguatera-causing dinoflagellate Gambierdiscus polynesiensis (Dinophyceae) in culture. Toxicon 2010, 56, 739–750. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.; Lovatelli, A.; Ababouch, L. Bivalve Depuration: Fundamental and Practical Aspects; FAO Fisheries Technical Paper No 511; Food and Agriculture Organization of the United Nations: Rome, Italy, 2008; ISBN 9789251060063. [Google Scholar]
- Blanco, J.; Bermúdez De La Puente, M.; Arévalo, F.; Salgado, C.; Moroño, Á. Depuration of mussels (Mytilus galloprovincialis) contaminated with domoic acid. Aquat. Living Resour. 2002, 15, 53–60. [Google Scholar] [CrossRef]
- Blanco, J.; Perez-Acosta, C.; Mariño, C.; Muñiz, S.; Martín, H.; Moroño, A.; Correa, J.; Arévalo, F.; Salgado, C. Depuration of domoic acid from different body compartments of the king scallop Pecten maximus grown in raft culture and natural bed. Aquat. Living Resour. 2006, 19, 257–265. [Google Scholar] [CrossRef]
- McFarland, K.; Jean, F.; Soudant, P.; Volety, A.K. Uptake and elimination of brevetoxin in the invasive green mussel, Perna viridis, during natural Karenia brevis blooms in southwest Florida. Toxicon 2015, 97, 46–52. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Suzuki, T.; Ota, H.; Yamasaki, M. Direct evidence of transformation of dinophysistoxin-1 to 7-O-acyl-dinophysistoxin-1 (dinophysistoxin-3) in the scallop Patinopecten yessoensis. Toxicon 1999, 37, 187–198. [Google Scholar] [CrossRef]
- Bauder, A.G.; Cembella, A.D.; Bricelj, V.M.; Quilliam, M.A. Uptake and fate of diarrhetic shellfish poisoning toxins from the dinoflagellate Prorocentrum lima in the bay scallop Argopecten irradians. Mar. Ecol. Prog. Ser. 2001, 213, 39–52. [Google Scholar] [CrossRef]
- Plakas, S.M.; Wang, Z.; El Said, K.R.; Jester, E.L.E.; Granade, H.R.; Flewelling, L.; Scott, P.; Dickey, R.W. Brevetoxin metabolism and elimination in the Eastern oyster (Crassostrea virginica) after controlled exposures to Karenia brevis. Toxicon 2004, 44, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Medhioub, W.; Guéguen, M.; Lassus, P.; Bardouil, M.; Truquet, P.; Sibat, M.; Medhioub, N.; Soudant, P.; Kraiem, M.; Zouher, A. Detoxification enhancement in the gymnodimine-contaminated grooved carpet shell, Ruditapes decussatus (Linné). Harmful Algae 2010, 9, 200–207. [Google Scholar] [CrossRef]
- Silvert, W.L.; Cembella, A.D. Dynamic modelling of phycotoxin kinetics in the blue mussel, Mytilus edulis, with implications for other marine invertebrates. Can. J. Fish. Aquat. Sci. 1995, 52, 521–531. [Google Scholar] [CrossRef]
- Yu, K.N.; Kwong, R.W.M.; Wang, W.-X.; Lam, P.K.S. Biokinetics of paralytic shellfish toxins in the green-lipped mussel, Perna viridis. Mar. Pollut. Bull. 2007, 54, 1068–1071. [Google Scholar] [CrossRef] [PubMed]
- Manger, R.L.; Leja, L.S.; Lee, S.Y.; Hungerford, J.M.; Wekell, M.M. Tetrazolium-based cell bioassay for neurotoxins active on voltage-sensitive sodium channels: Semiautomated assay for saxitoxins, brevetoxins, and ciguatoxins. Anal. Biochem. 1993, 214, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Yogi, K.; Oshiro, N.; Inafuku, Y.; Hirama, M.; Yasumoto, T. Detailed LC-MS/MS Analysis of Ciguatoxins Revealing Distinct Regional and Species Characteristics in Fish and Causative Alga from the Pacific. Anal. Chem. 2011, 83, 8886–8891. [Google Scholar] [CrossRef] [PubMed]

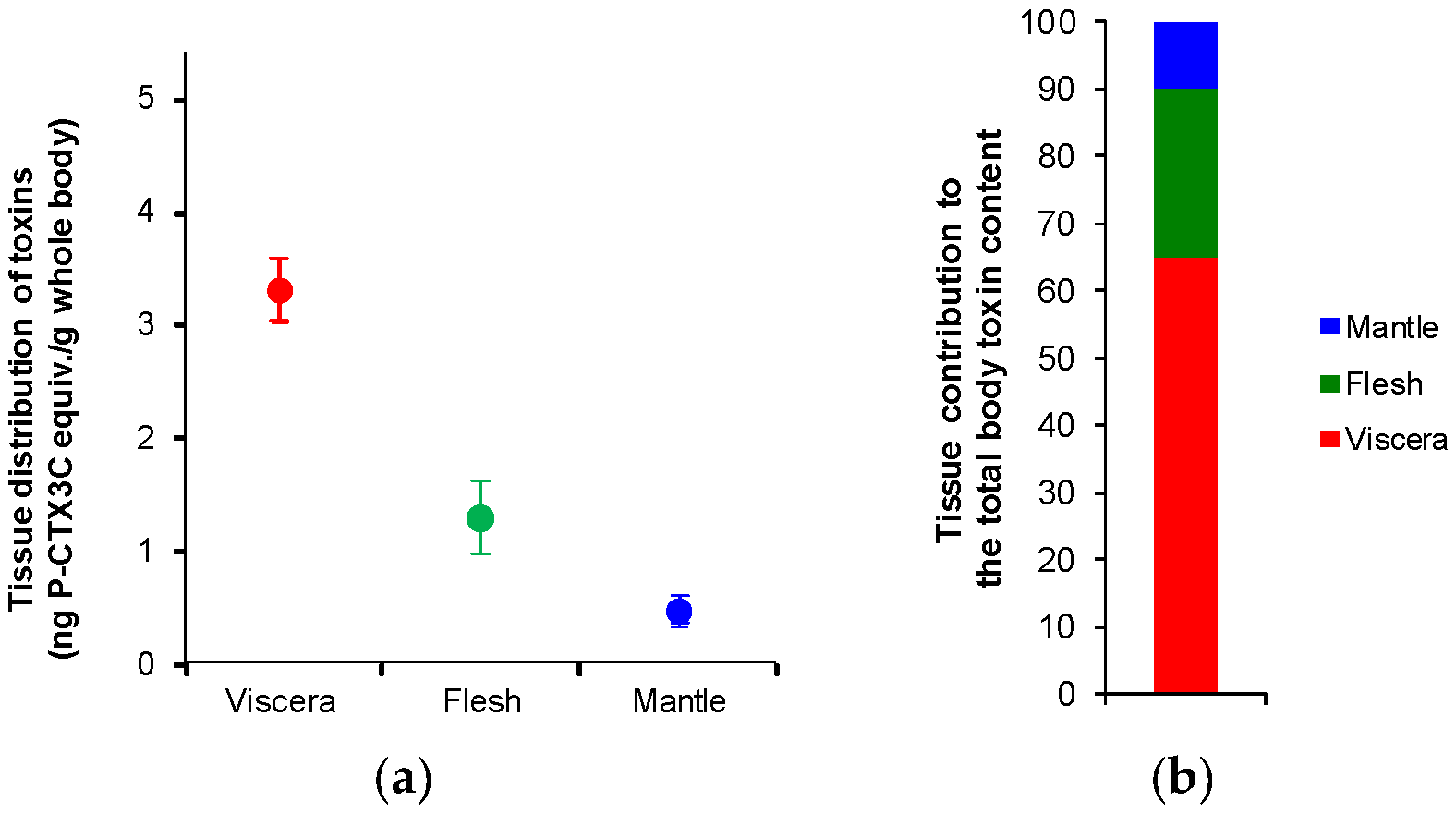
| Tank | Tissue | Absolute Toxicity of Tissue 1 | Tissue Wet Weight [g] (Relative Weight Contribution, %) | Tissue Distribution of Toxins 2 | Tissue Contribution to the Total Body Toxin Content |
|---|---|---|---|---|---|
| 1 | Viscera | 12.2 ± 0.4 | 36 (26%) | 3.2 ± 0.1 | 59% |
| Flesh | 3.0 ± 0.2 | 75 (54%) | 1.6 ± 0.1 | 31% | |
| Mantle | 2.6 ± 0.2 | 28.5 (20%) | 0.5 ± 0.0 | 10% | |
| 2 | Viscera | 13.1 ± 0.8 | 37.5 (25%) | 3.3 ± 0.2 | 71% |
| Flesh | 2.0 ± 0.2 | 72 (48%) | 0.9 ± 0.1 | 20% | |
| Mantle | 1.4 ± 0.2 | 39.5 (27%) | 0.4 ± 0.1 | 9% | |
| 3 | Viscera | 13.6 ± 1.4 | 33.5 (26%) | 3.5 ± 0.4 | 64% |
| Flesh | 3.0 ± 0.1 | 58.5 (45%) | 1.4 ± 0.0 | 25% | |
| Mantle | 2.1 ± 0.3 | 38 (29%) | 0.6 ± 0.1 | 11% |
| Days of Detoxification | Giant Clam Individual | Wet Weight [g] | Toxin Content 1 |
|---|---|---|---|
| 0 | 1 | 39.9 | 1.4 ± 0.2 |
| 2 | 57.1 | 1.9 ± 0.1 | |
| 3 | 60.8 | 2.2 ± 0.4 | |
| Average of 1–3 | 53 ± 11 | 1.8 ± 0.4 | |
| 2 | 1 | 39.0 | 3.3 ± 0.4 |
| 2 | 40.8 | 2.3 ± 0.1 | |
| 3 | 67.2 | 1.9 ± 0.4 | |
| Average of 1–3 | 49 ± 16 | 2.5 ± 0.7 | |
| 4 | 1 | 71.0 | 3.3 ± 0.4 |
| 2 | 54.3 | 1.6 ± 0.2 | |
| 3 | 57.6 | 1.1 ± 0.2 | |
| Average of 1–3 | 61 ± 9 | 2.0 ± 1.1 | |
| 6 | 1 | 46.6 | 1.4 ± 0.1 |
| 2 | 43.9 | 2.8 ± 0.8 | |
| 3 | 32.1 | 2.0 ± 0.3 | |
| Average of 1–3 | 41 ± 8 | 2.1 ± 0.7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roué, M.; Darius, H.T.; Ung, A.; Viallon, J.; Sibat, M.; Hess, P.; Amzil, Z.; Chinain, M. Tissue Distribution and Elimination of Ciguatoxins in Tridacna maxima (Tridacnidae, Bivalvia) Fed Gambierdiscus polynesiensis. Toxins 2018, 10, 189. https://doi.org/10.3390/toxins10050189
Roué M, Darius HT, Ung A, Viallon J, Sibat M, Hess P, Amzil Z, Chinain M. Tissue Distribution and Elimination of Ciguatoxins in Tridacna maxima (Tridacnidae, Bivalvia) Fed Gambierdiscus polynesiensis. Toxins. 2018; 10(5):189. https://doi.org/10.3390/toxins10050189
Chicago/Turabian StyleRoué, Mélanie, Hélène Taiana Darius, André Ung, Jérôme Viallon, Manoella Sibat, Philipp Hess, Zouher Amzil, and Mireille Chinain. 2018. "Tissue Distribution and Elimination of Ciguatoxins in Tridacna maxima (Tridacnidae, Bivalvia) Fed Gambierdiscus polynesiensis" Toxins 10, no. 5: 189. https://doi.org/10.3390/toxins10050189
APA StyleRoué, M., Darius, H. T., Ung, A., Viallon, J., Sibat, M., Hess, P., Amzil, Z., & Chinain, M. (2018). Tissue Distribution and Elimination of Ciguatoxins in Tridacna maxima (Tridacnidae, Bivalvia) Fed Gambierdiscus polynesiensis. Toxins, 10(5), 189. https://doi.org/10.3390/toxins10050189









