Phenotypic Variation in Mojave Rattlesnake (Crotalus scutulatus) Venom Is Driven by Four Toxin Families
Abstract
1. Introduction
2. Results
2.1. Venom Gland Transcriptomes of C. scutulatus
2.2. Toxin Diversity in C. scutulatus
2.3. Myotoxin a Diversity
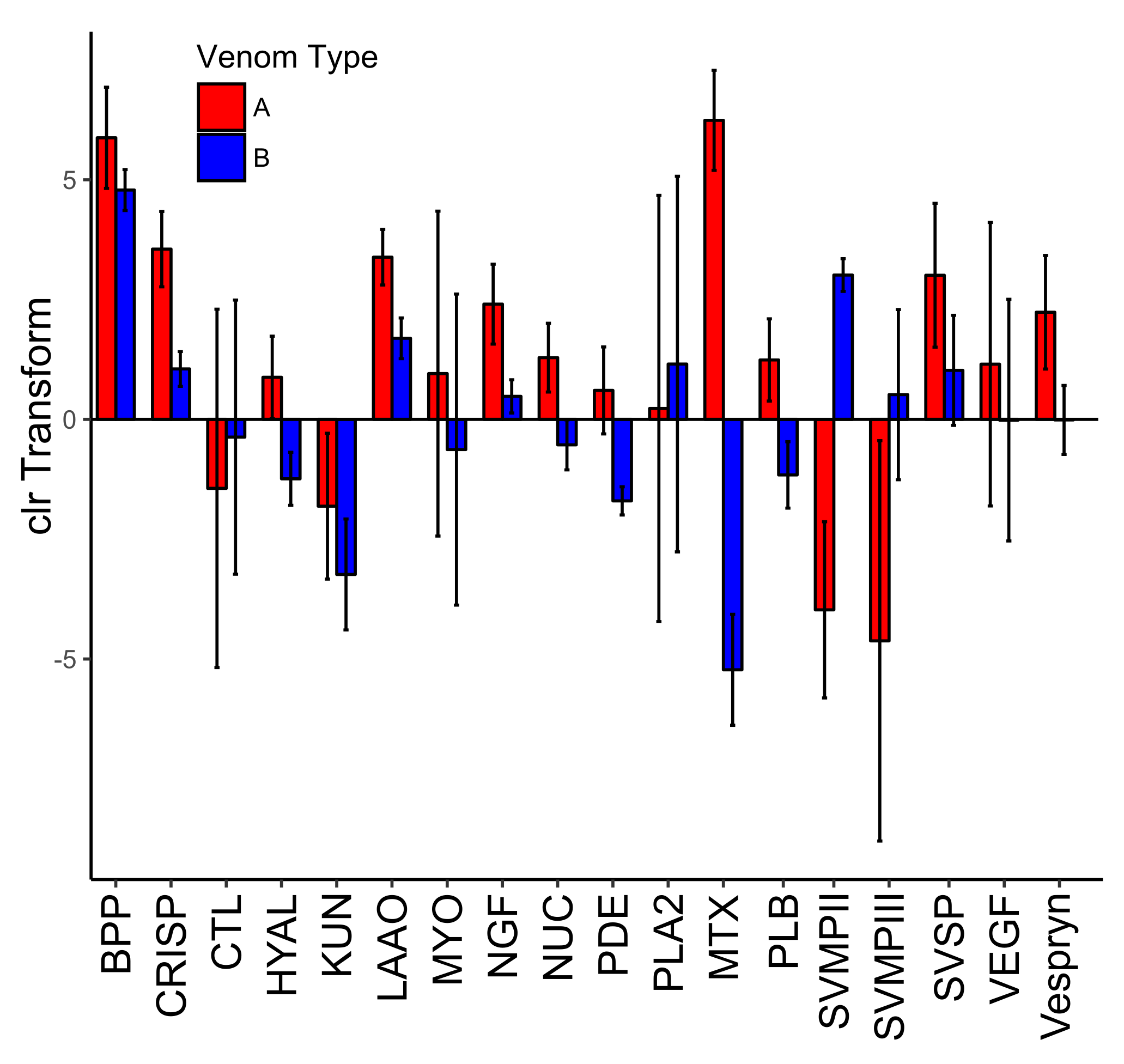
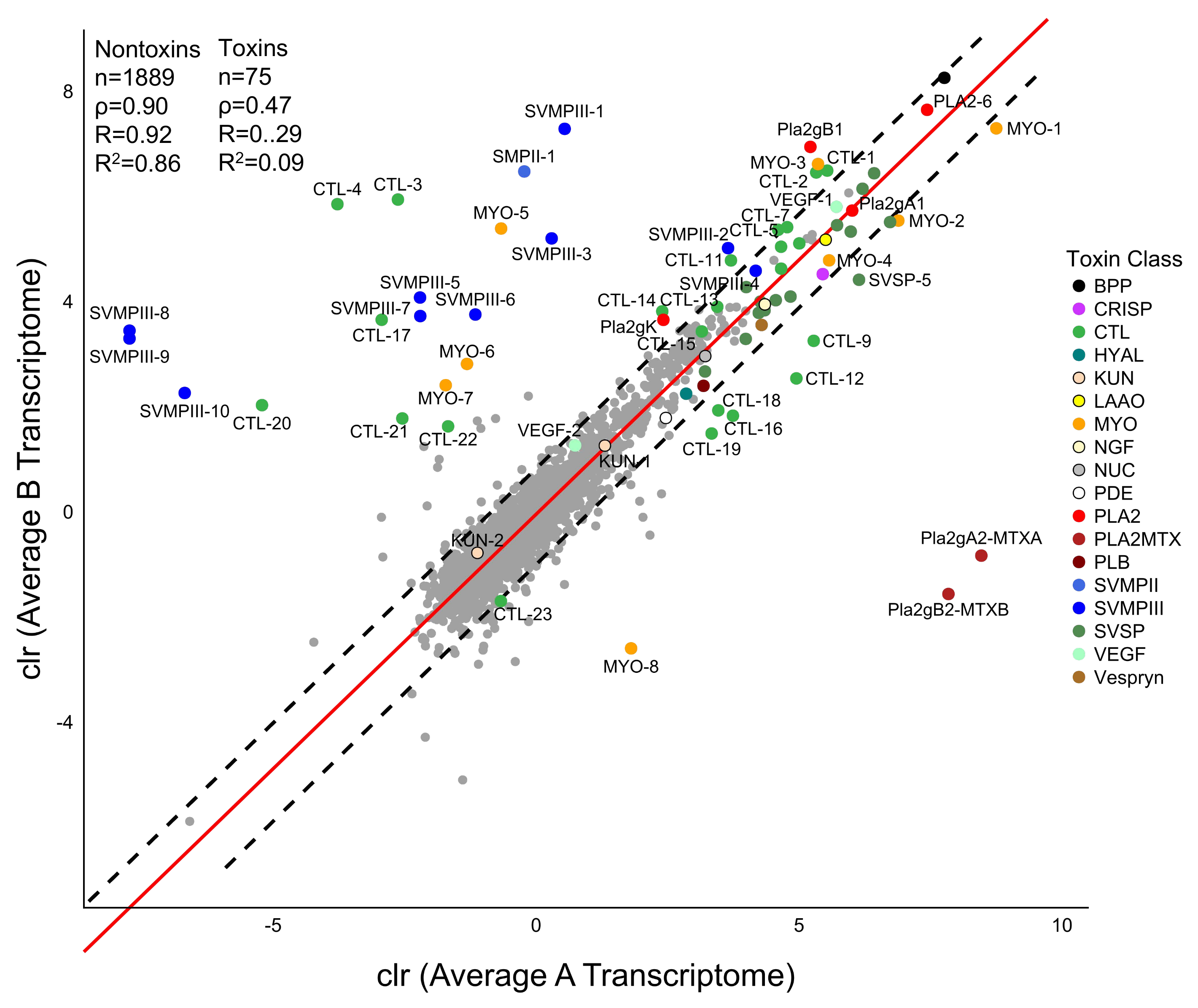
2.4. Expression Differences in Type A and Type B C. scutulatus
2.5. PLA Diversity
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Ethics Statement
5.2. Sample Collection
5.3. Venom Type Determination
5.4. Venom Gland Transcriptome Sequencing
5.5. Transcriptome Assembly
5.6. Expression Analysis
5.7. PLA Diversity
5.8. Data Availability
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ARCC | Advanced Research Computing Center |
| BPP | Bradykinin potentiating peptide |
| clr | centered log-ratio |
| CLP | Christopher L. Parkinson field number |
| CRISP | Cysteine-rich secretory protein |
| CTL | C-type lectin |
| FSU | Florida State University |
| HYAL | Hyaluronidase |
| HPC | High Performance Cluster |
| KUN | Kunitz peptide |
| LAAO | L-amino-acid oxidase |
| LD | Lethal dose |
| MDPI | Multidisciplinary Digital Publishing Institute |
| MTX | Mojave toxin |
| MTXA | Acidic () subunit of Mojave toxin |
| MTXB | Basic () subunit of Mojave toxin |
| MYO | Myotoxin-a |
| NAHR | Non-allelic homologous recombination |
| NGF | Nerve growth factor |
| NUC | 5 nucleotidase |
| PDE | Phosphodiesterase |
| PLA | Phospholipase A |
| PLB | Phospholipase B |
| RP-HPLC | Reverse-phased high performance liquid chromatography |
| SVMP | Snake venom metalloproteinase |
| SVSP | Snake venom serine protease |
| TPM | Transcripts per million reads |
| VEGF | Vascular endothelial growth factor |
| UCF | University of Central Florida |
References
- Kussell, E.; Leibler, S. Phenotypic diversity, population growth, and information in fluctuating environments. Science 2005, 309, 2075–2078. [Google Scholar] [CrossRef] [PubMed]
- Gallego-Romero, I.; Ruvinsky, I.; Gilad, Y. Comparative studies of gene expression and the evolution of gene regulation. Nat. Rev. Genet. 2012, 13, 505. [Google Scholar] [CrossRef] [PubMed]
- Savolainen, O.; Lascoux, M.; Merilä, J. Ecological genomics of local adaptation. Nat. Rev. Genet. 2013, 14, 807. [Google Scholar] [CrossRef] [PubMed]
- Margres, M.J.; Wray, K.P.; Hassinger, A.T.B.; Ward, M.J.; Mcgivern, J.J.; Moriarty-Lemmon, E.; Lemmon, A.R.; Rokyta, D.R. Quantity, not quality: Rapid adaptation in a polygenic trait proceeded exclusively through expression differentiation. Mol. Biol. Evol. 2017, 34, 3099–3110. [Google Scholar] [CrossRef] [PubMed]
- Casewell, N.R.; Wüster, W.; Vonk, F.J.; Harrison, R.A.; Fry, B.G. Complex cocktails: The evolutionary novelty of venoms. Trends Ecol. Evol. 2013, 28, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Margres, M.J.; McGivern, J.J.; Seavy, M.; Wray, K.P.; Facente, J.; Rokyta, D.R. Contrasting modes and tempos of venom expression evolution in two snake species. Genetics 2015, 199, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Dowell, N.L.; Giorgianni, M.W.; Kassner, V.A.; Selegue, J.E.; Sanchez, E.E.; Carroll, S.B. The deep origin and recent loss of venom toxin genes in rattlesnakes. Curr. Biol. 2016, 26, 2434–2445. [Google Scholar] [CrossRef] [PubMed]
- Margres, M.J.; Aronow, K.; Loyacano, J.; Rokyta, D.R. The venom-gland transcriptome of the Eastern Coral Snake (Micrurus fulvius) reveals high venom complexity in the intragenomic evolution of venoms. BMC Genom. 2013, 14, 531. [Google Scholar] [CrossRef] [PubMed]
- Whittington, A.C.; Mason, A.J.; Rokyta, D.R. A single mutation unlocks cascading exaptations in the origin of a potent pitviper neurotoxin. Mol. Biol. Evol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Casewell, N.R.; Wagstaff, S.C.; Wüster, W.; Cook, D.A.N.; Bolton, F.M.S.; King, S.I.; Pla, D.; Sanz, L.; Calvete, J.J.; Harrison, R.A. Medically important differences in snake venom composition are dictated by distinct postgenomic mechanisms. Proc. Natl. Acad. Sci. USA 2014, 111, 9205–9210. [Google Scholar] [CrossRef] [PubMed]
- Rokyta, D.R.; Margres, M.J.; Calvin, K. Post-transcriptional mechanisms contribute little to phenotypic variation in snake venoms. G3 Genes Genomes Genet. 2015, 5, 2375–2382. [Google Scholar] [CrossRef] [PubMed]
- Glenn, J.; Straight, R. Mojave Rattlesnake (Crotalus scutulatus scutulatus) venom: variation in toxicity with geographical origin. Toxicon 1978, 16, 81–84. [Google Scholar] [CrossRef]
- Glenn, J.L.; Straight, R.C. The rattlesnakes and their venom yield and lethal toxicity. In Rattlesnake Venoms: Their Actions and Treatment; Tu, A.T., Ed.; Marcel Dekker, Inc.: New York, NY, USA, 1982; pp. 3–119. [Google Scholar]
- Weinstein, S.A.; Minton, S.A.; Wilde, C.E. The distribution among ophidian venoms of a toxin isolated from the venom of the Mojave Rattlesnake (Crotalus scutulatus scutulatus). Toxicon 1985, 23, 825–844. [Google Scholar] [CrossRef]
- Mackessy, S.P. Venom composition in rattlesnakes: Trends and biological significance. In The Biology of Rattlesnakes; Hayes, W.K., Beaman, K.R., Cardwell, M.D., Bush, S.P., Eds.; Loma Linda University Press: Loma Linda, CA, USA, 2008; pp. 495–510. [Google Scholar]
- Doley, R.; Kini, R.M. Protein complexes in snake venom. Cell. Mol. Life Sci. 2009, 66, 2851–2871. [Google Scholar] [CrossRef] [PubMed]
- Doley, R.; Zhou, X.; Kini, R. Snake venom phospholipase A2 enzymes. In Handbook of Venoms and Toxins of Reptiles; Mackessy, S.P., Ed.; CRC Press: Boca Raton, FL, USA, 2009; pp. 173–205. [Google Scholar]
- Wooldridge, B.; Pineda, G.; Banuelas-Ornelas, J.; Dagda, R.; Gasanov, S.; Rael, E.; Lieb, C. Mojave Rattlesnakes (Crotalus scutulatus scutulatus) lacking the acidic subunit DNA sequence lack Mojave toxin in their venom. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2001, 130, 169–179. [Google Scholar] [CrossRef]
- Zancolli, G.; Baker, T.; Barlow, A.; Bradley, R.; Calvete, J.; Carter, K.; de Jager, K.; Owens, J.; Price, J.; Sanz, L.; et al. Is hybridization a source of adaptive venom variation in rattlesnakes? A test, Using a Crotalus scutulatus x viridis hybrid zone in southwestern New Mexico. Toxins 2016, 8, 188. [Google Scholar] [CrossRef] [PubMed]
- Borja, M.; Neri-Castro, E.; Castañeda-Gaytán, G.; Strickland, J.; Parkinson, C.; Castañeda-Gaytán, J.; Ponce-López, R.; Lomonte, B.; Olvera-Rodríguez, A.; Alagón, A.; et al. Biological and proteolytic variation in the venom of Crotalus scutulatus scutulatus from Mexico. Toxins 2018, 10, 35. [Google Scholar] [CrossRef] [PubMed]
- Bieber, A.L.; Tu, T.; Tu, A.T. Studies of an acidic cardiotoxin isolated from the venom of Mojave Rattlesnake (Crotalus scutulatus). Biochim. Biophys. Acta Protein Struct. 1975, 400, 178–188. [Google Scholar] [CrossRef]
- Cate, R.L.; Bieber, A.L. Purification and characterization of Mojave (Crotalus scutulatus scutulatus) toxin and its subunits. Arch. Biochem. Biophys. 1978, 189, 397–408. [Google Scholar] [CrossRef]
- Gopalakrishnakone, P.; Hawgood, B.J.; Holbrooke, S.E.; Marsh, N.A.; Santana De Sa, S.; Tu, A.T. Sites of action of Mojave toxin isolated from the venom of the Mojave Rattlesnake. Br. J. Pharmacol. 1980, 69, 421–431. [Google Scholar] [CrossRef] [PubMed]
- John, T.R.; Smith, L.A.; Kaiser, I.I. Genomic sequences encoding the acidic and basic subunits of Mojave toxin: Unusually high sequence identity of non-coding regions. Gene 1994, 139, 229–234. [Google Scholar] [CrossRef]
- Calvete, J.J.; Pérez, A.; Lomonte, B.; Sánchez, E.E.; Sanz, L. Snake venomics of Crotalus tigris: The minimalist toxin arsenal of the deadliest Neartic rattlesnake venom. Evolutionary clues for generating a pan-specific antivenom against crotalid type II venoms. J. Proteome Res. 2012, 11, 1382–1390. [Google Scholar] [CrossRef] [PubMed]
- Uetz, P. The Reptile Database. Available online: http://www.reptile-database.org/ (accessed on 24 January 2018).
- Rokyta, D.R.; Wray, K.P.; Margres, M.J. The genesis of an exceptionally lethal venom in the Timber Rattlesnake (Crotalus horridus) revealed through comparative venom-gland transcriptomics. BMC Genom. 2013, 14, 394. [Google Scholar] [CrossRef] [PubMed]
- Rokyta, D.R.; Wray, K.P.; McGivern, J.J.; Margres, M.J. The transcriptomic and proteomic basis for the evolution of a novel venom phenotype within the Timber Rattlesnake (Crotalus horridus). Toxicon 2015, 98, 34–48. [Google Scholar] [CrossRef] [PubMed]
- Minton, S.A. Observations on the amphibians and reptiles of the Big Bend region of Texas. Southwest. Nat. 1958, 3, 28–54. [Google Scholar] [CrossRef]
- Martinez, M.; Rael, E.D.; Maddux, N.L. Isolation of a hemorrhagic toxin from Mojave Rattlesnake (Crotalus scutulatus scutulatus) venom. Toxicon 1990, 28, 685–694. [Google Scholar] [CrossRef]
- Wilkinson, J.A.; Glenn, J.L.; Straight, R.C.; Sites, J.W. Distribution and genetic variation in venom A and B populations of the Mojave Rattlesnake (Crotalus scutulatus scutulatus) in Arizona. Herpetologica 1991, 47, 54–68. [Google Scholar]
- Borja, M.; Castañeda, G.; Espinosa, J.; Neri, E.; Carbajal, A.; Clement, H.; García, O.; Alagon, A. Mojave Rattlesnake Crotalus scutulatus scutulatus with Type B Venom from Mexico. Copeia 2014, 2014, 7–13. [Google Scholar] [CrossRef]
- Dobson, J.; Yang, D.; Op Den Brouw, B.; Cochran, C.; Huynh, T.; Kurrupu, S.; Sánchez, E.E.; Massey, D.J.; Baumann, K.; Jackson, T.N.W.; et al. Rattling the border wall: Pathophysiological implications of functional and proteomic venom variation between Mexican and US subspecies of the desert rattlesnake Crotalus scutulatus. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Schield, D.R.; Card, D.C.; Adams, R.H.; Corbin, A.; Jezkova, T.; Hales, N.; Meik, J.M.; Spencer, C.L.; Smith, L.; Campillo-Garcia, G.; et al. Cryptic genetic diversity, population structure, and gene flow in the Mojave Rattlesnake (Crotalus scutulatus). Mol. Phylogenet. Evol. in review.
- Glenn, J.L.; Straight, R.C. Intergradation of two different venom populations of the Mojave Rattlesnake (Crotalus scutulatus scutulatus) in Arizona. Toxicon 1989, 27, 411–418. [Google Scholar] [CrossRef]
- Sánchez, E.E.; Galán, J.A.; Powell, R.L.; Reyes, S.R.; Soto, J.G.; Russell, W.K.; Russell, D.H.; Pérez, J.C. Disintegrin, hemorrhagic, and proteolytic activities of Mohave Rattlesnake, Crotalus scutulatus scutulatus venoms lacking Mojave toxin. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2005, 141, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Githens, T.; George, I. Comparative studies on the venoms of certain rattlesnakes. Bull. Antivenin Inst. Am. 1931, 5, 31–34. [Google Scholar]
- Hardy, D.L. Envenomation by the Mojave Rattlesnake (Crotalus scutulatus scutulatus) in southern Arizona, U.S.A. Toxicon 1983, 21, 111–118. [Google Scholar] [CrossRef]
- Massey, D.J.; Calvete, J.J.; Sánchez, E.E.; Sanz, L.; Richards, K.; Curtis, R.; Boesen, K. Venom variability and envenoming severity outcomes of the Crotalus scutulatus scutulatus (Mojave Rattlesnake) from Southern Arizona. J. Proteom. 2012, 75, 2576–2587. [Google Scholar] [CrossRef] [PubMed]
- Glenn, J.; Straight, R.; Wolt, T. Regional variation in the presence of canebrake toxin in Crotalus horridus venom. Comp. Biochem. Physiol. Part C Pharmacol. Toxicol. Endocrinol. 1994, 107, 337–346. [Google Scholar] [CrossRef]
- Schrider, D.R.; Gout, J.F.; Hahn, M.W. Very few RNA and DNA sequence differences in the human transcriptome. PLoS ONE 2011, 6, e25842. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [PubMed]
- Dagda, R.K.; Gasanov, S.; De La Oiii, Y.; Rael, E.D.; Lieb, C.S. Genetic basis for variation of metalloproteinase-associated biochemical activity in venom of the Mojave Rattlesnake (Crotalus scutulatus scutulatus). Biochem. Res. Int. 2013, 2013, 251474. [Google Scholar] [CrossRef] [PubMed]
- Dagda, R.K.; Gasanov, S.E.; Zhang, B.; Welch, W.; Rael, E.D. Molecular models of the Mojave Rattlesnake (Crotalus scutulatus scutulatus) venom metalloproteinases reveal a structural basis for differences in hemorrhagic activities. J. Biol. Phys. 2014, 40, 193–216. [Google Scholar] [CrossRef] [PubMed]
- Arlinghaus, F.T.; Eble, J.A. C-type lectin-like proteins from snake venoms. Toxicon 2012, 60, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Tseng, K.H. Effect of crotamine, a toxin of South American rattlesnake venom, on the sodium channel of murine skeletal muscle. Br. J. Pharmacol. 1978, 63, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Durban, J.; Pérez, A.; Sanz, L.; Gómez, A.; Bonilla, F.; Rodríguez, S.; Chacón, D.; Sasa, M.; Angulo, Y.; Gutiérrez, J.M.; et al. Integrated “omics” profiling indicates that miRNAs are modulators of the ontogenetic venom composition shift in the Central American rattlesnake, Crotalus simus simus. BMC Genom. 2013, 14, 234. [Google Scholar] [CrossRef] [PubMed]
- Lynch, V.J. Inventing an arsenal: Adaptive evolution and neofunctionalization of snake venom phospholipase A2 genes. BMC Evol. Biol. 2007, 7, 2. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rokyta, D.R.; Lemmon, A.R.; Margres, M.J.; Aronow, K. The venom-gland transcriptome of the Eastern Diamondback Rattlesnake (Crotalus adamanteus). BMC Genom. 2012, 13, 312. [Google Scholar] [CrossRef] [PubMed]
- Rotenberg, D.; Bamberger, E.S.; Kochva, E. Studies on ribonucleic acid synthesis in the venom glands of Vipera palaestinae (Ophidia, Reptilia). Biochem. J. 1971, 121, 609–612. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Margres, M.J.; McGivern, J.J.; Wray, K.P.; Seavy, M.; Calvin, K.; Rokyta, D.R. Linking the transcriptome and proteome to characterize the venom of the Eastern Diamondback Rattlesnake (Crotalus adamanteus). J. Proteom. 2014, 96, 145–158. [Google Scholar] [CrossRef] [PubMed]
- Rokyta, D.R.; Wray, K.P.; Lemmon, A.R.; Moriarty-Lemmon, E.; Caudle, S.B. A high-throughput venom-gland transcriptome for the Eastern Diamondback Rattlesnake (Crotalus adamanteus) and evidence for pervasive positive selection across toxin classes. Toxicon 2011, 57, 657–671. [Google Scholar] [CrossRef] [PubMed]
- Rokyta, D.R.; Margres, M.J.; Ward, M.J.; Sanchez, E.E. The genetics of venom ontogeny in the Eastern Diamondback Rattlesnake (Crotalus adamanteus). PeerJ 2017, 5, e3249. [Google Scholar] [CrossRef] [PubMed]
- Rokyta, D.R.; Ward, M.J. Venom-gland transcriptomics and venom proteomics of the Black-back Scorpion (Hadrurus spadix) reveal detectability challenges and an unexplored realm of animal toxin diversity. Toxicon Off. J. Int. Soc. Toxinol. 2017, 128, 23–37. [Google Scholar] [CrossRef] [PubMed]
- Ward, M.J.; Ellsworth, S.A.; Rokyta, D.R. Venom-gland transcriptomics and venom proteomics of the Hentz Striped Scorpion (Centruroides hentzi; Buthidae) reveal high toxin diversity in a harmless member of a lethal family. Toxicon 2018, 142, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Mortazavi, A.; Williams, B.A.; McCue, K.; Schaeffer, L.; Wold, B. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat. Methods 2008, 5, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Kobert, K.; Flouri, T.; Stamatakis, A. PEAR: A fast and accurate Illumina Paired-End reAd mergeR. Bioinformatics 2014, 30, 614–620. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Godzik, A. Cd-hit: A fast program for clustering and comparing large sets of protein or nucleotide sequences. Bioinformatics 2006, 22, 1658–1659. [Google Scholar] [CrossRef] [PubMed]
- Bendtsen, J.D.; Nielsen, H.; Van Heijne, G.; Brunak, S. Improved prediction of signal peptides: SignalP 3.0. J. Mol. Biol. 2004, 340, 783–795. [Google Scholar] [CrossRef] [PubMed]
- Petersen, T.N.; Brunak, S.; von Heijne, G.; Nielsen, H. SignalP 4.0: Discriminating signal peptides from transmembrane regions. Nat. Methods 2011, 8, 785–786. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Trapnell, C.; Pop, M.; Salzberg, S.L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Res. 2009, 10, R25. [Google Scholar] [CrossRef] [PubMed]
- Palarea-Albaladejo, J.; Martín-Fernández, J.A. Software description zCompositions—R package for multivariate imputation of left-censored data under a compositional approach. Chemom. Intell. Lab. Syst. 2015, 143, 85–96. [Google Scholar] [CrossRef]
- Aitchison, J. The Statistical Analysis of Compositional Data; Chapman and Hall: London, UK, 1986; Volume 44, pp. 139–177. [Google Scholar]
- Anders, S.; Huber, W. Differential expression analysis for sequence count data. Genome Biol. 2010, 11, R106. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [PubMed]
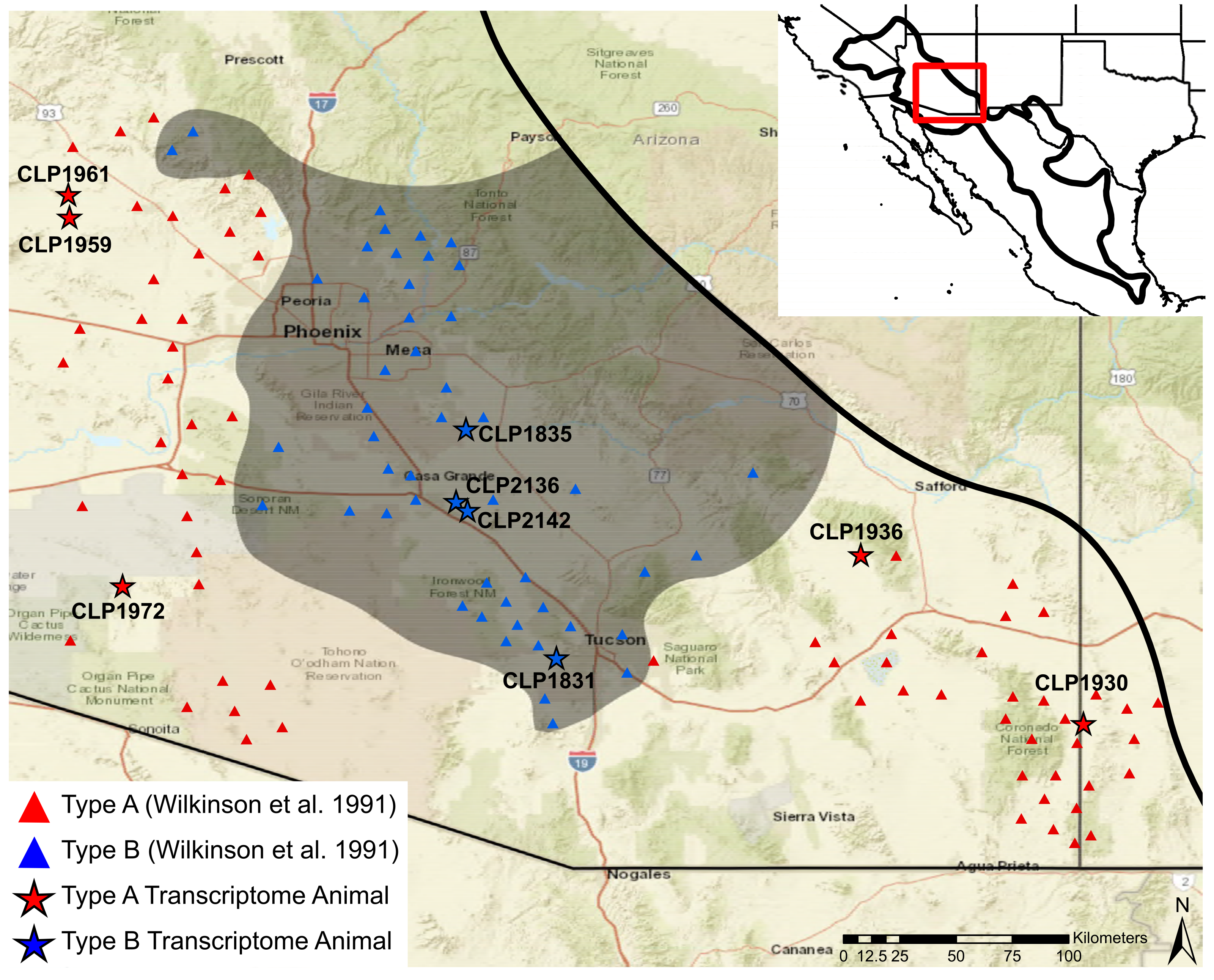
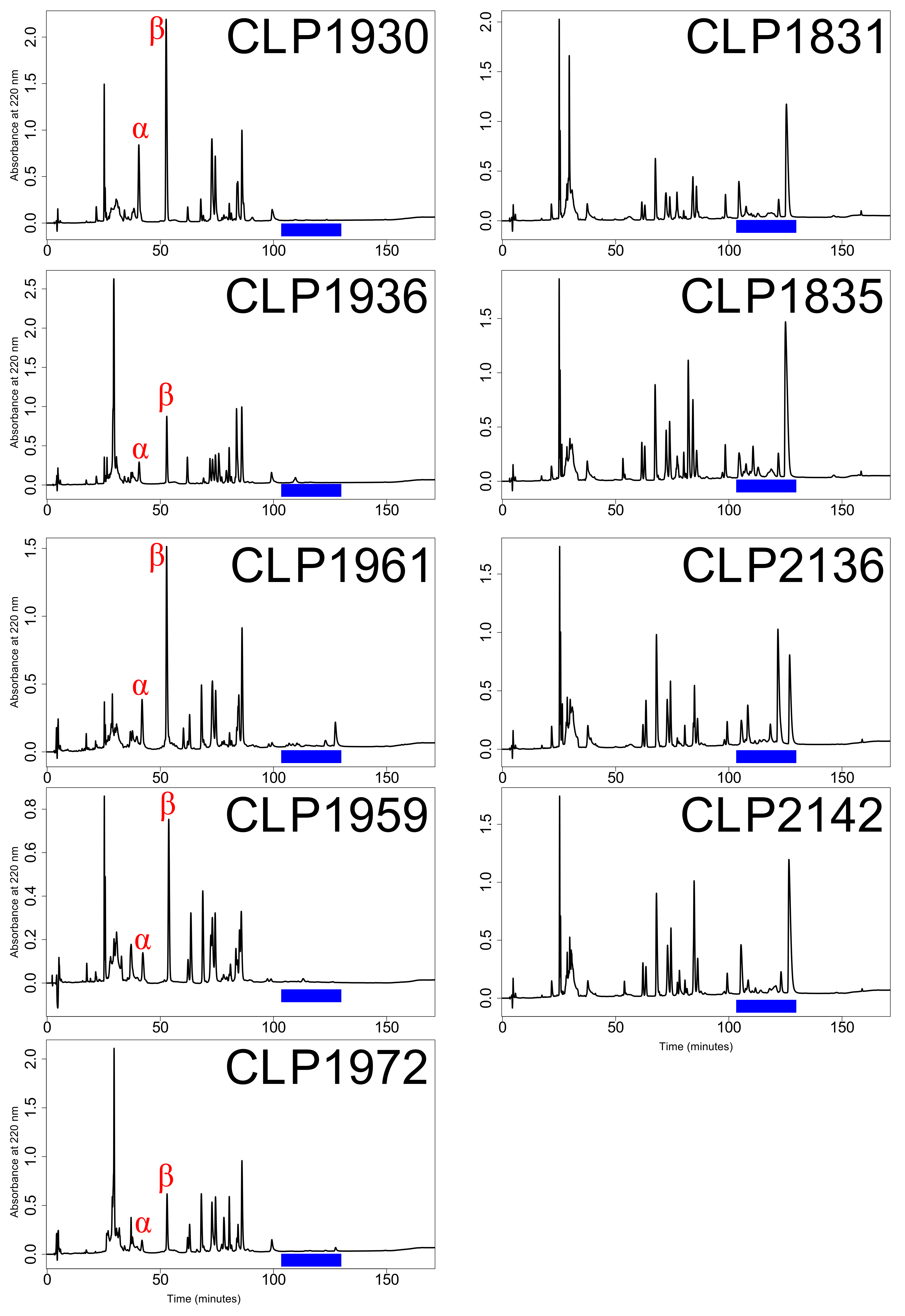
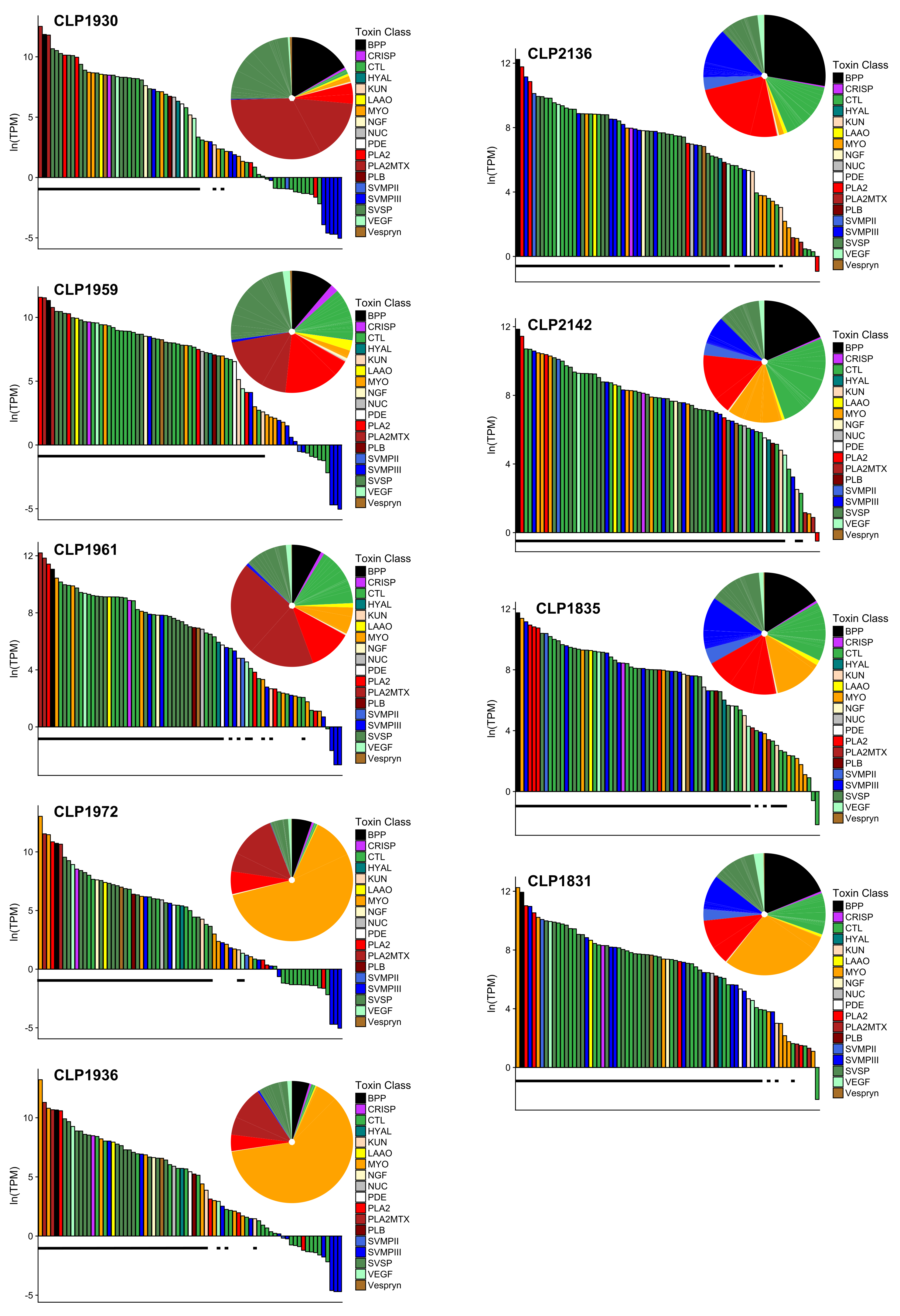
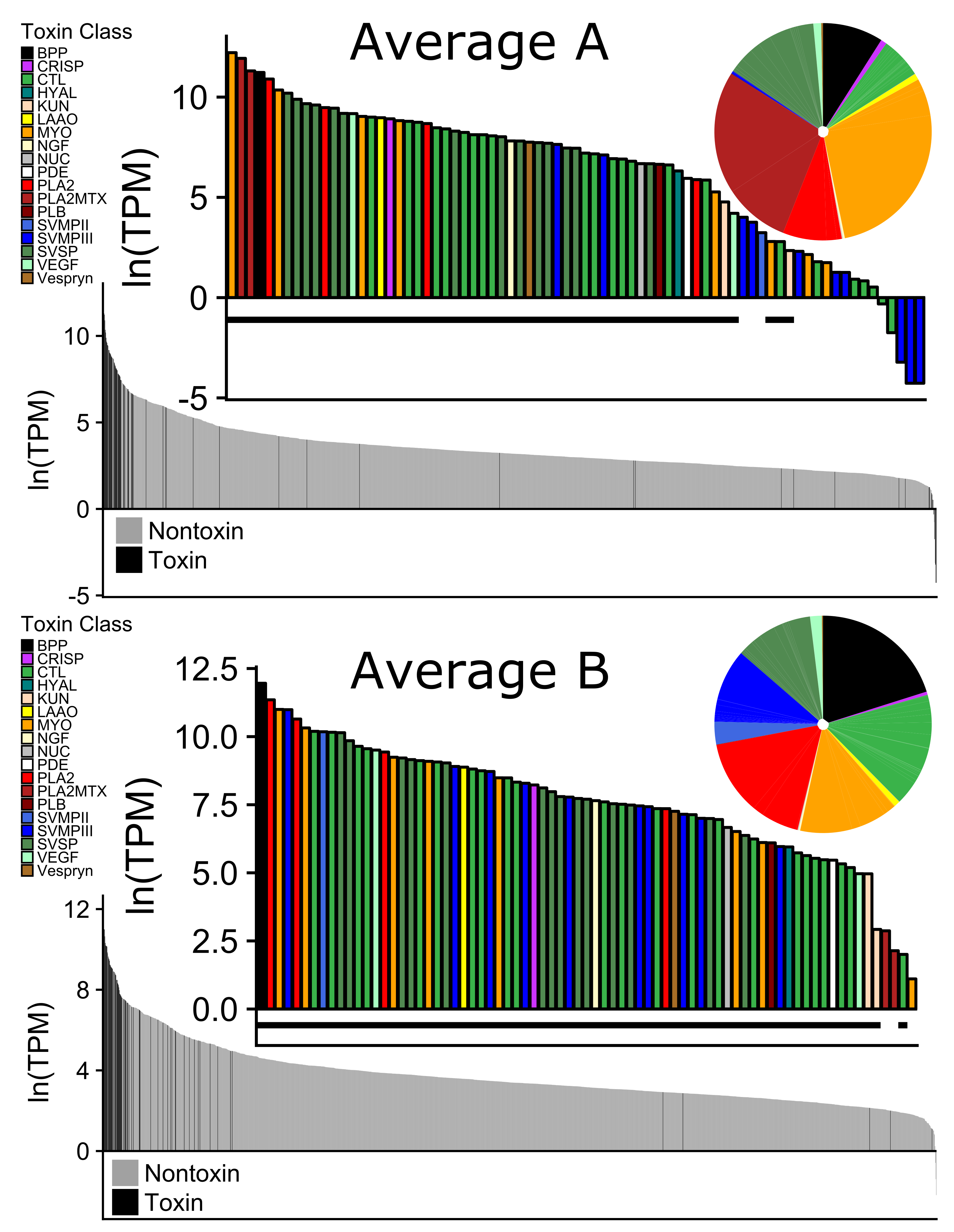
| Specimen | Museum | Venom | Sex | SVL | Mass | State | County | Sequencing | Read | Merged | BioSample |
|---|---|---|---|---|---|---|---|---|---|---|---|
| ID | ID | Type | (mm) | (g) | Platform | Pairs | Reads | Accession | |||
| CLP1930 | ASNHC14997 | A | F | 724 | 195 | NM | Hidalgo | MiSeq | 15,649,085 | 13,083,925 | SAMN08596271 |
| CLP1936 | ASU36035 | A | F | 441 | 48 | AZ | Graham | MiSeq | 20,835,668 | 18,182,033 | SAMN08596272 |
| CLP1959 | ASU36061 | A | M | 730 | 204 | AZ | Yavapai | MiSeq | 20,577,779 | 17,091,930 | SAMN08596273 |
| CLP1961 | ASU36062 | A | M | 564 | 90 | AZ | Yavapai | HiSeq | 11,929,639 | 10,061,262 | SAMN08596274 |
| CLP1972 | ASU36092 | A | M | 635 | 126 | AZ | Pima | MiSeq | 13,168,704 | 11,521,499 | SAMN08596275 |
| CLP1831 | ASU36089 | B | F | 795 | 344 | AZ | Pima | HiSeq | 15,448,552 | 13,526,047 | SAMN08596267 |
| CLP1835 | ASU36102 | B | F | 685 | 146 | AZ | Pinal | HiSeq | 16,271,477 | 14,210,557 | SAMN08596269 |
| CLP2136 | ASU36103 | B | M | 1030 | 627 | AZ | Pinal | MiSeq | 7,771,613 | 6,864,270 | SAMN08596277 |
| CLP2142 | ASU36104 | B | M | 775 | 262 | AZ | Pinal | MiSeq | 10,039,268 | 8,893,097 | SAMN08596278 |
| Toxin | Type A | Type B | |||||||
|---|---|---|---|---|---|---|---|---|---|
| CLP1930 | CLP1936 | CLP1959 | CLP1961 | CLP1972 | CLP1831 | CLP1835 | CLP2136 | CLP2142 | |
| BPP-1 | 140,915.97 | 42,576.13 | 83,149.6 | 63,491.95 | 45,783.43 | 152,393.13 | 126,252.15 | 206,001.59 | 140,706.64 |
| CRISP-1 | 4835.29 | 4825.49 | 15,386.6 | 7108.63 | 5075.55 | 4047.71 | 4625.53 | 2879.87 | 3386.38 |
| CTL-1 | 0.00 | 3.65 | 14,629.20 | 25,897.00 | 0.00 | 17,494.71 | 26,507.89 | 18,545.42 | 44,489.80 |
| CTL-2 | 0.00 | 1.98 | 12,439.65 | 20,441.04 | 0.00 | 18,825.37 | 22,039.29 | 18,679.67 | 43,789.90 |
| CTL-3 | 1.12 | 0.00 | 0.31 | 10.11 | 0.00 | 12,805.18 | 13,046.93 | 13,965.96 | 21,899.71 |
| CTL-4 | 0.00 | 0.45 | 0.00 | 3.20 | 0.00 | 12,605.98 | 15,574.61 | 11,584.92 | 16,750.81 |
| CTL-5 | 0.41 | 2.52 | 7965.07 | 10,951.63 | 0.00 | 4218.11 | 11,546.02 | 10,057.03 | 10,667.89 |
| CTL-6 | 0.40 | 1.26 | 11,399.06 | 12,437.39 | 0.00 | 3461.60 | 3578.61 | 9394.12 | 10,374.16 |
| CTL-7 | 0.38 | 1.44 | 7423.12 | 8508.27 | 0.00 | 4653.94 | 10,065.04 | 9327.24 | 10,662.63 |
| CTL-8 | 22.39 | 8.78 | 7498.38 | 9084.97 | 232.76 | 2273.74 | 4494.79 | 6799.15 | 11,587.90 |
| CTL-9 | 3859.43 | 4539.36 | 9932.82 | 10,002.06 | 3012.54 | 1561.25 | 2.48 | 1.52 | 2634.56 |
| CTL-10 | 10.63 | 4.93 | 7658.06 | 9102.48 | 84.69 | 2499.90 | 3293.65 | 6962.90 | 3823.74 |
| CTL-11 | 0.00 | 0.47 | 2744.48 | 3752.74 | 0.00 | 2814.96 | 6914.87 | 4523.03 | 5131.21 |
| CTL-12 | 3604.05 | 3074.72 | 6797.37 | 6941.60 | 2123.74 | 1975.31 | 14.78 | 24.72 | 40.48 |
| CTL-13 | 0.00 | 0.41 | 2379.11 | 2630.23 | 0.00 | 947.76 | 3008.94 | 1948.93 | 2122.28 |
| CTL-14 | 0.88 | 171.20 | 883.68 | 555.05 | 147.92 | 637.66 | 1994.47 | 2157.31 | 2572.39 |
| CTL-15 | 995.02 | 292.59 | 809.42 | 726.11 | 910.53 | 1168.87 | 748.84 | 1791.77 | 1309.00 |
| CTL-16 | 1246.38 | 418.83 | 2132.33 | 2473.36 | 472.09 | 433.29 | 0.55 | 234.84 | 342.16 |
| CTL-17 | 0.00 | 0.00 | 0.41 | 8.06 | 0.00 | 4.34 | 10.41 | 51.78 | 6202.50 |
| CTL-18 | 325.45 | 305.49 | 4026.88 | 247.89 | 201.87 | 279.93 | 274.45 | 36.75 | 527.21 |
| CTL-19 | 28.46 | 1045.78 | 2968.50 | 60.46 | 395.23 | 49.87 | 213.36 | 285.33 | 172.05 |
| CTL-20 | 0.00 | 0.00 | 0.00 | 0.87 | 0.00 | 0.00 | 0.00 | 1.60 | 1233.72 |
| CTL-21 | 0.40 | 0.00 | 0.53 | 11.61 | 0.00 | 59.06 | 27.62 | 511.53 | 361.34 |
| CTL-22 | 0.00 | 0.00 | 0.38 | 29.57 | 0.00 | 51.73 | 54.86 | 280.15 | 438.68 |
| CTL-23 | 3.52 | 9.49 | 15.25 | 7.90 | 45.56 | 5.09 | 13.42 | 1.34 | 9.87 |
| HYAL-1 | 557.54 | 304.13 | 1309.25 | 379.62 | 221.07 | 468.14 | 408.71 | 436.41 | 222.96 |
| KUN-1 | 178.35 | 48.25 | 168.92 | 123.70 | 71.34 | 107.63 | 147.09 | 195.49 | 122.11 |
| KUN-2 | 14.96 | 4.32 | 13.36 | 14.48 | 5.19 | 20.25 | 20.77 | 20.98 | 12.38 |
| LAAO-1 | 5188.99 | 2817.21 | 20,588.57 | 9120.95 | 1626.36 | 5792.61 | 10,578.38 | 6868.00 | 5503.11 |
| MYO-1 | 1220.73 | 549,957.26 | 1072.89 | 27.84 | 457,306.12 | 209,616.39 | 44.93 | 44.14 | 29,489.77 |
| MYO-2 | 6119.03 | 49,381.65 | 2467.88 | 3325.84 | 95,421.49 | 27,536.10 | 2903.51 | 6987.97 | 3925.07 |
| MYO-3 | 0.00 | 0.00 | 0.00 | 34,019.55 | 0.00 | 0.00 | 87,549.28 | 30.87 | 33,694.44 |
| MYO-4 | 5843.47 | 3702.71 | 12,274.75 | 19,693.00 | 497.74 | 1611.10 | 10,899.95 | 2894.17 | 4002.29 |
| MYO-5 | 0.00 | 82.14 | 0.00 | 0.00 | 0.00 | 44.63 | 0.00 | 43.19 | 35,485.73 |
| MYO-6 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 2706.33 |
| MYO-7 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 1805.60 |
| MYO-8 | 3.83 | 960.47 | 8.14 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| NGF-1 | 2021.69 | 761.53 | 5039.75 | 2527.51 | 2023.71 | 1592.10 | 2240.15 | 2459.36 | 2108.54 |
| NUC-1 | 779.38 | 366.04 | 1526.31 | 945.96 | 368.05 | 610.32 | 960.93 | 1068.46 | 502.67 |
| PDE-1 | 445.18 | 229.67 | 691.74 | 312.13 | 236.13 | 209.66 | 277.73 | 208.12 | 249.61 |
| Pla2gA1 | 25,306.99 | 39,764.45 | 61.41 | 46.36 | 2.19 | 4.48 | 50,036.23 | 0.00 | 0.61 |
| Pla2gB1 | 3.44 | 7.16 | 29,405.58 | 14.34 | 1.43 | 37,548.76 | 46,634.43 | 51,949.04 | 31,992.11 |
| Pla2gK | 0.00 | 0.30 | 1793.61 | 3.04 | 0.00 | 1356.66 | 2971.25 | 1123.48 | 807.59 |
| Pla2gA2-MTXA | 274,390.23 | 80,733.96 | 102,588.14 | 199,107.20 | 102,677.33 | 4.93 | 65.65 | 0.00 | 0.00 |
| Pla2gB2-MTXB | 133,822.75 | 43,410.96 | 48,028.34 | 138,470.84 | 42,571.19 | 3.73 | 30.28 | 0.00 | 0.00 |
| PLA-6 | 21,306.42 | 22.86 | 106,639.29 | 90,911.79 | 51,893.58 | 60,760.91 | 56,428.98 | 129,974.00 | 93,064.76 |
| PLB-1 | 837.02 | 189.30 | 1170.66 | 1059.23 | 597.70 | 509.37 | 745.22 | 346.61 | 182.83 |
| SVMPII-1 | 0.00 | 0.84 | 0.60 | 122.86 | 3.30 | 23,801.48 | 32,375.22 | 24,762.53 | 24,369.30 |
| SVMPIII-1 | 0.00 | 1.20 | 1.83 | 262.28 | 9.58 | 58,107.40 | 69,839.79 | 69,573.48 | 39,410.80 |
| SVMPIII-2 | 8.60 | 3074.27 | 61.30 | 2524.10 | 479.43 | 3581.65 | 14,465.14 | 2364.36 | 4089.81 |
| SVMPIII-3 | 0.01 | 0.79 | 1.32 | 206.34 | 6.17 | 6920.71 | 8968.12 | 7129.26 | 6456.84 |
| SVMPIII-4 | 1497.61 | 1018.71 | 4893.89 | 2711.96 | 276.26 | 3637.08 | 4729.55 | 5060.59 | 2488.36 |
| SVMPIII-5 | 6.65 | 4.39 | 4.51 | 2.02 | 0.00 | 1293.10 | 2689.88 | 3646.35 | 1942.60 |
| SVMPIII-6 | 19.52 | 12.50 | 6.89 | 9.20 | 2.20 | 274.19 | 3250.08 | 2739.21 | 663.84 |
| SVMPIII-7 | 0.02 | 0.17 | 0.57 | 16.36 | 0.53 | 643.82 | 2557.44 | 2534.21 | 994.18 |
| SVMPIII-8 | 0.00 | 0.00 | 0.00 | 0.07 | 0.00 | 44.33 | 50.02 | 4977.26 | 25.70 |
| SVMPIII-9 | 0.00 | 0.00 | 0.00 | 0.07 | 0.00 | 279.91 | 2000.74 | 1017.78 | 1092.48 |
| SVMPIII-10 | 0.00 | 0.01 | 0.00 | 0.19 | 0.00 | 181.26 | 751.33 | 220.24 | 404.67 |
| SVSP-1 | 25,158.51 | 15,910.07 | 30,344.55 | 17,038.01 | 10,432.56 | 21,732.91 | 32,652.46 | 20,711.71 | 26,686.11 |
| SVSP-2 | 42,979.80 | 20,190.56 | 35,325.26 | 21,389.97 | 14,037.74 | 16,208.71 | 10,861.87 | 6624.20 | 6521.70 |
| SVSP-3 | 24,402.09 | 7206.88 | 34,812.15 | 9121.20 | 3774.14 | 19,942.19 | 20,070.68 | 20,385.25 | 15,388.82 |
| SVSP-4 | 29,001.28 | 5094.40 | 18,056.16 | 9305.84 | 1916.43 | 8654.88 | 9431.62 | 7042.42 | 8412.05 |
| SVSP-5 | 37,090.47 | 5414.68 | 21,446.17 | 8874.54 | 1346.38 | 1863.68 | 5619.86 | 2373.32 | 3563.03 |
| SVSP-6 | 11,830.88 | 7170.10 | 15,765.87 | 9603.42 | 4485.31 | 8500.16 | 12,274.97 | 6642.73 | 10,481.74 |
| SVSP-7 | 7289.06 | 2382.92 | 5895.10 | 3081.90 | 1513.33 | 4024.78 | 2685.70 | 1659.02 | 1379.27 |
| SVSP-8 | 4813.04 | 2087.12 | 5900.27 | 2021.75 | 407.33 | 2169.99 | 2985.67 | 1994.54 | 1945.35 |
| SVSP-9 | 4048.20 | 724.15 | 1391.55 | 1288.30 | 1231.13 | 3097.60 | 3252.51 | 2154.34 | 3169.89 |
| SVSP-10 | 4038.45 | 1189.72 | 4293.59 | 1766.03 | 84.61 | 1208.22 | 2752.40 | 2430.80 | 2459.42 |
| SVSP-11 | 5777.06 | 1464.77 | 2503.34 | 1599.56 | 962.33 | 2311.84 | 3050.76 | 478.40 | 1664.01 |
| SVSP-12 | 3674.93 | 1456.06 | 3014.74 | 2310.02 | 565.32 | 2209.80 | 1897.49 | 1761.75 | 1281.51 |
| SVSP-13 | 3238.56 | 798.05 | 3159.21 | 1118.98 | 287.65 | 1467.22 | 709.22 | 982.38 | 1198.93 |
| SVSP-14 | 1558.37 | 620.40 | 1082.12 | 667.47 | 38.63 | 762.05 | 289.19 | 599.21 | 693.61 |
| VEGF-1 | 4255.45 | 10,557.63 | 14,271.01 | 11,732.75 | 7493.65 | 20,763.31 | 9620.66 | 12,596.93 | 10,796.52 |
| VEGF-2 | 134.91 | 18.84 | 82.27 | 95.20 | 3.90 | 96.12 | 72.87 | 313.81 | 91.89 |
| Vespryn-1 | 4976.55 | 718.69 | 3827.06 | 1024.92 | 1089.92 | 2106.99 | 2068.47 | 922.31 | 582.28 |
| Toxin | Type A | Type B | |||||||
|---|---|---|---|---|---|---|---|---|---|
| CLP1930 | CLP1936 | CLP1959 | CLP1961 | CLP1972 | CLP1831 | CLP1835 | CLP2136 | CLP2142 | |
| CTL-1 | - | - | + | + | - | + | + | + | + |
| CTL-2 | - | - | + | + | - | + | + | + | + |
| CTL-3 | - | - | - | - | - | + | + | + | + |
| CTL-4 | - | - | - | - | - | + | + | + | + |
| CTL-5 | - | - | + | + | - | + | + | + | + |
| CTL-6 | - | - | + | + | - | + | + | + | + |
| CTL-7 | - | - | + | + | - | + | + | + | + |
| CTL-8 | - | - | + | + | + | + | + | + | + |
| CTL-9 | + | + | + | + | + | + | - | - | + |
| CTL-10 | - | - | + | + | + | + | + | + | + |
| CTL-11 | - | - | + | + | - | + | + | + | + |
| CTL-12 | + | + | + | + | + | + | + | - | - |
| CTL-13 | - | - | + | + | - | + | + | + | + |
| CTL-14 | - | + | + | + | + | + | + | + | + |
| CTL-16 | + | + | + | + | + | + | - | + | + |
| CTL-17 | - | - | - | - | - | - | - | + | + |
| CTL-19 | + | + | + | + | + | - | + | - | + |
| CTL-20 | - | - | - | - | - | - | - | - | + |
| CTL-21 | - | - | - | - | - | + | + | + | + |
| CTL-22 | - | - | - | - | - | + | + | + | + |
| CTL-23 | + | + | + | + | + | + | + | - | + |
| MYO-3 | - | - | - | + | - | - | + | + | + |
| MYO-5 | - | + | - | - | - | + | - | + | + |
| MYO-6 | - | - | - | - | - | - | - | - | + |
| MYO-7 | - | - | - | - | - | - | - | - | + |
| MYO-8 | - | + | + | - | - | - | - | - | - |
| Pla2gA1 | + | + | + | - | - | - | + | - | - |
| Pla2gB1 | - | - | + | - | - | + | + | + | + |
| Pla2gK | - | - | + | - | - | + | + | + | + |
| Pla2gA2-MTXA | + | + | + | + | + | - | - | - | - |
| Pla2gB2-MTXB | + | + | + | + | + | - | - | - | - |
| PLA-6 | + | - | + | + | + | + | + | + | + |
| SVMPII-1 | - | - | - | - | - | + | + | + | + |
| SVMPIII-1 | - | - | - | - | - | + | + | + | + |
| SVMPIII-2 | - | + | + | + | + | + | + | + | + |
| SVMPIII-3 | - | - | - | - | - | + | + | + | + |
| SVMPIII-5 | - | - | - | - | - | + | + | + | + |
| SVMPIII-6 | - | - | - | - | - | + | + | + | + |
| SVMPIII-7 | - | - | - | - | - | + | + | + | + |
| SVMPIII-8 | - | - | - | - | - | - | - | + | - |
| SVMPIII-9 | - | - | - | - | - | + | + | + | + |
| SVMPIII-10 | - | - | - | - | - | + | + | + | + |
| Toxins Present | 42 | 45 | 56 | 53 | 45 | 64 | 66 | 64 | 69 |
| Toxin | UpB | UpA | B to A | AveB to AveA | Log DESeq1 | P DESeq1 | Log DESeq2 | P DESeq2 | |
|---|---|---|---|---|---|---|---|---|---|
| CTL-3 | 20 | 0 | 20 | Up | Up | 12.76 | 6.55 × 10 | 12.73 | 1.11 × 10 |
| CTL-4 | 20 | 0 | 20 | Up | Up | 14.14 | 5.91 × 10 | 14.13 | 1.03 × 10 |
| SVMPII-1 | 20 | 0 | 20 | Up | Up | 9.94 | 1.96 × 10 | 9.97 | N/A |
| SVMPIII-1 | 20 | 0 | 20 | Up | Up | 9.97 | 1.01 × 10 | 10.00 | N/A |
| SVMPIII-10 | 20 | 0 | 20 | Up | Up | 13.17 | 6.48 × 10 | 13.28 | 4.27 × 10 |
| SVMPIII-3 | 20 | 0 | 20 | Up | Up | 7.35 | 2.89 × 10 | 7.38 | N/A |
| SVMPIII-5 | 20 | 0 | 20 | Up | Up | 8.96 | 8.76 × 10 | 8.96 | 4.42 × 10 |
| SVMPIII-6 | 20 | 0 | 20 | Up | Up | 6.86 | 5.44 × 10 | 6.86 | 5.30 × 10 |
| SVMPIII-7 | 20 | 0 | 20 | Up | Up | 8.75 | 6.91 × 10 | 8.79 | 1.00 × 10 |
| SVMPIII-9 | 20 | 0 | 20 | Up | Up | 16.23 | 1.76 × 10 | 15.89 | 2.41 × 10 |
| CTL-21 | 19 | 0 | 19 | Up | Up | 6.65 | 2.60 × 10 | 6.58 | 3.92 × 10 |
| Pla2gB1 | 19 | 0 | 19 | Up | Up | 3.06 | 6.07 × 10 | 3.04 | N/A |
| CTL-22 | 18 | 0 | 18 | Up | Up | - | - | - | - |
| CTL-14 | 16 | 0 | 16 | Up | Up | 2.22 | 9.75 × 10 | - | - |
| Pla2gK | 16 | 0 | 16 | Up | Up | - | - | 2.32 | N/A |
| CTL-1 | 15 | 0 | 15 | Up | Up | - | - | - | - |
| CTL-2 | 15 | 0 | 15 | Up | Up | - | - | - | - |
| CTL-11 | 14 | 0 | 14 | Up | Up | 2.01 | 7.05 × 10 | - | - |
| SVMPIII-2 | 14 | 0 | 14 | Up | Up | - | - | - | - |
| CTL-7 | 12 | 0 | 12 | Up | Up | - | - | - | - |
| CTL-17 | 10 | 0 | 10 | Up | Up | - | - | 10.07 | N/A |
| MYO-5 | 9 | 1 | 8 | Up | Up | - | - | 8.04 | N/A |
| MYO-3 | 9 | 2 | 7 | Up | Up | - | - | - | - |
| CTL-20 | 5 | 0 | 5 | Up | Up | - | - | 11.01 | N/A |
| MYO-6 | 5 | 0 | 5 | Up | Up | - | - | 23.74 | N/A |
| MYO-7 | 5 | 0 | 5 | Up | Up | - | - | 23.49 | N/A |
| SVMPIII-8 | 5 | 0 | 5 | Up | Up | - | - | 15.96 | N/A |
| MYO-2 | 4 | 3 | 1 | Up | Down | - | - | - | - |
| CRISP-1 | 0 | 0 | 0 | No Difference | - | - | - | −1.32 | 3.16 × 10 |
| CTL-19 | 6 | 8 | −2 | Down | Down | - | - | - | - |
| MYO-1 | 7 | 9 | −2 | Down | Down | - | - | - | - |
| PLB-1 | 0 | 2 | −2 | Down | - | - | - | −1.07 | 7.76 × 10 |
| SVSP-2 | 1 | 4 | −3 | Down | Down | −1.77 | 9.50 × 10 | −1.77 | 5.11 × 10 |
| MYO-8 | 0 | 4 | −4 | Down | Down | - | - | −23.22 | N/A |
| SVSP-5 | 1 | 5 | −4 | Down | Down | - | - | −2.32 | 5.29 × 10 |
| CTL-18 | 0 | 5 | −5 | Down | Down | - | - | - | - |
| CTL-23 | 0 | 7 | −7 | Down | Down | - | - | - | - |
| CTL-16 | 0 | 9 | −9 | Down | Down | −2.49 | 5.78 × 10 | - | - |
| CTL-9 | 0 | 11 | −11 | Down | Down | −2.73 | 2.56 × 10 | - | - |
| CTL-12 | 0 | 15 | −15 | Down | Down | −3.36 | 1.14 × 10 | −3.35 | 7.88 × 10 |
| Pla2gA2-MTXA | 0 | 20 | −20 | Down | Down | −13.55 | 8.76 × 10 | −13.51 | 1.18 × 10 |
| Pla2gB2-MTXB | 0 | 20 | −20 | Down | Down | −13.65 | 5.75 × 10 | −13.61 | 2.10 × 10 |
| Specimen | Pla2gA1 | Pla2gB1 | Pla2gK | Pla2gA2-MTXA | Pla2gB2-MTXB | PLA-6 |
|---|---|---|---|---|---|---|
| C. atrox | + | + | + | - | - | - |
| C. atrox | + | + | + | - | - | - |
| C. adamanteus | + | + | - | - | - | - |
| C. scutulatus | + | - | - | + | + | - |
| CLP1930A | 55,212.77 | - | - | 603,261.66 | 294,142.52 | 47,256.79 |
| CLP1936A | 242,536.57 | - | - | 492,462.39 | 264,770.94 | - |
| CLP1959A | 214.27 | 101,885.11 | 6162.67 | 355,273.29 | 166,449.59 | 369,588.5 |
| CLP1961A | - | - | - | 464,139.64 | 323,205.86 | 212,285.6 |
| CLP1972A | - | - | - | 520,518.57 | 216,000.5 | 263,309.5 |
| CLP1831B | - | 370,853.15 | 13,305.52 | - | - | 600,310.86 |
| CLP1835B | 320,034.14 * | 297,244.14 | 18,761.21 | - | - | 58,732.73 |
| CLP2136B | - | 283,162.92 | 6014.01 | - | - | 709,003.82 |
| CLP2142B | - | 253,634.76 | 6285.41 | - | - | 738,317.63 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Strickland, J.L.; Mason, A.J.; Rokyta, D.R.; Parkinson, C.L. Phenotypic Variation in Mojave Rattlesnake (Crotalus scutulatus) Venom Is Driven by Four Toxin Families. Toxins 2018, 10, 135. https://doi.org/10.3390/toxins10040135
Strickland JL, Mason AJ, Rokyta DR, Parkinson CL. Phenotypic Variation in Mojave Rattlesnake (Crotalus scutulatus) Venom Is Driven by Four Toxin Families. Toxins. 2018; 10(4):135. https://doi.org/10.3390/toxins10040135
Chicago/Turabian StyleStrickland, Jason L., Andrew J. Mason, Darin R. Rokyta, and Christopher L. Parkinson. 2018. "Phenotypic Variation in Mojave Rattlesnake (Crotalus scutulatus) Venom Is Driven by Four Toxin Families" Toxins 10, no. 4: 135. https://doi.org/10.3390/toxins10040135
APA StyleStrickland, J. L., Mason, A. J., Rokyta, D. R., & Parkinson, C. L. (2018). Phenotypic Variation in Mojave Rattlesnake (Crotalus scutulatus) Venom Is Driven by Four Toxin Families. Toxins, 10(4), 135. https://doi.org/10.3390/toxins10040135







