Occurrence of β-N-methylamino-l-alanine (BMAA) and Isomers in Aquatic Environments and Aquatic Food Sources for Humans
Abstract
1. Introduction
2. Results
2.1. Presence of BMAA and Isomers in Waters and Phytoplankton Samples
2.1.1. Lakes and Reservoirs
2.1.2. Lagoon, Brackish and Marine Environments
2.2. Contamination of Aquatic Food Sources for Humans
2.2.1. Contamination of Freshwater Organisms
2.2.2. Contamination of Marine Organisms
3. Discussion
4. Conclusions
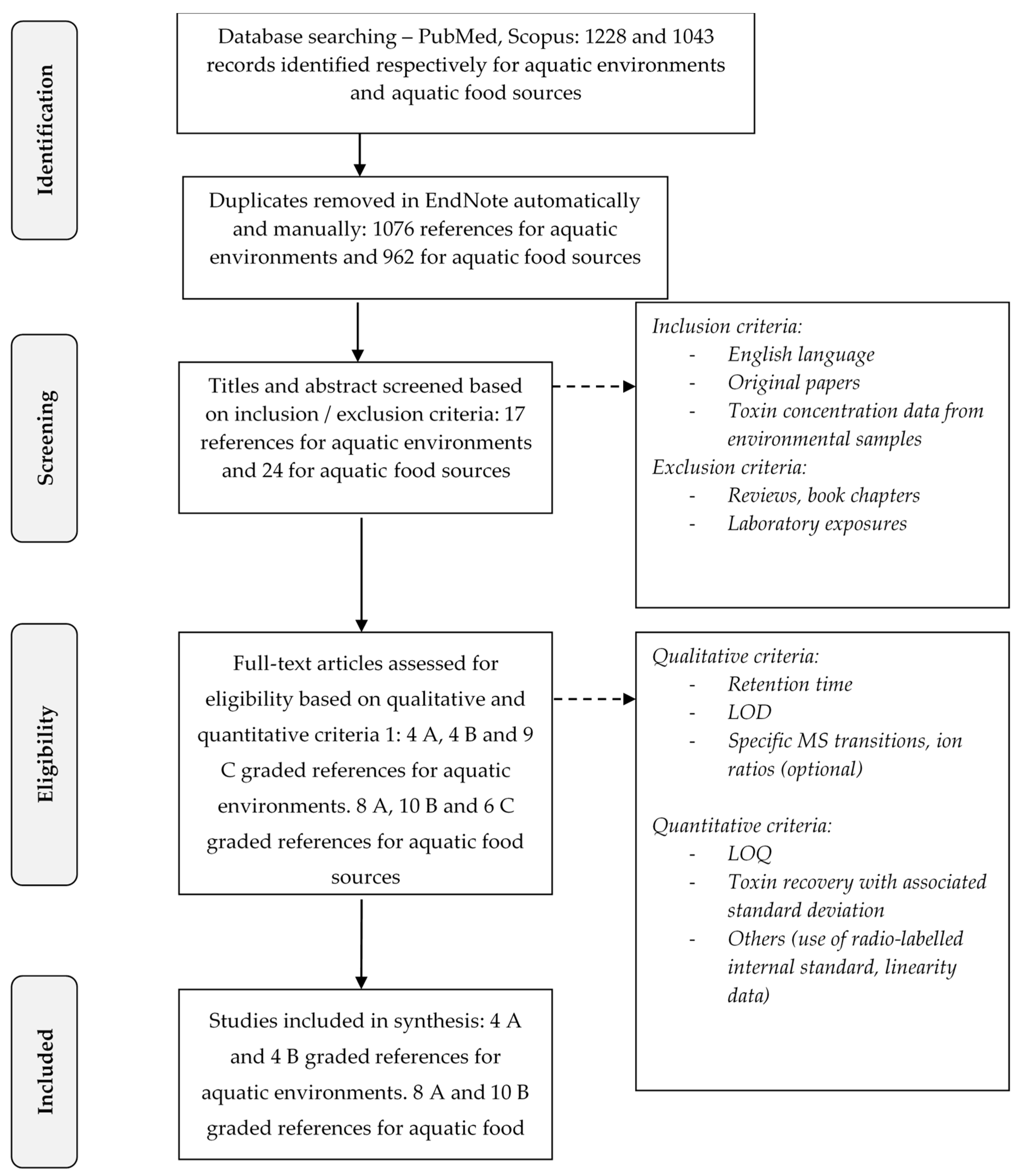
5. Materials and Methods: Methodology for the Scientific Literature Review
5.1. The Strategy for the Extensive Literature Search
5.2. The Selection Process of Scientific Papers
5.2.1. First Screening of Articles
5.2.2. Second Screening of Articles
- Qualitative criteria:
- -
- The indication of the retention time that makes it possible to discriminate BMAA from its isomers; this criterion is strengthened by the monitoring of specific transitions as mentioned below;
- -
- The indication of the LOD;
- -
- The LC-MS/MS monitoring of specific transitions for BMAA (m/z 119 > 88, 76 or 459 > 258 for BMAA without and after derivatization with AQC) and its isomers (m/z 119 > 101, 74 or 459 > 188 for DAB without and after derivatization with AQC; m/z 459 > 214 for AEG after derivatization with AQC) to better differentiate them from one another. The ion ratio was an optional and additional criterion allowing the identification of the molecules (e.g., 88/102 and 76/102 for BMAA; 101/102 and 74/102 for DAB; or 258/119, 188/119 and 214/119 for BMAA, DAB and AEG after derivatization by AQC);
- Quantitative criteria:
- -
- The indication of the LOQ in the matrix (e.g., water and biological matrix);
- -
- The calculation of the recovery rate during extraction of BMAA or its isomers in the matrix, with the associated standard deviation;
- -
- Other additional and optional criteria improving the reliability of the analytical method such as the use of radio-labeled internal standard, information concerning the linearity, specificity of the method etc.
5.3. Data Reporting
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Spencer, P.S.; Hugon, J.; Ludolph, A.; Nunn, P.B.; Ross, S.M.; Roy, D.N.; Schaumburg, H.H. Discovery and Partial Characterization of Primate Motor-System Toxins. Ciba Found. Symp. 1987, 126, 221–238. [Google Scholar] [PubMed]
- Brownson, D.M.; Mabry, T.J.; Leslie, S.W. The Cycad Neurotoxic Amino Acid, Beta-N-Methylamino-l-Alanine (Bmaa), Elevates Intracellular Calcium Levels in Dissociated Rat Brain Cells. J. Ethnopharmacol. 2002, 82, 159–167. [Google Scholar] [CrossRef]
- Nunn, P.B.; O’Brien, P. The Interaction of β-N-Methylamino-l-Alanine with Bicarbonate: An 1H-NMR Study. FEBS Lett. 1989, 251, 31–35. [Google Scholar] [CrossRef]
- Weiss, J.H.; Christine, C.W.; Choi, D.W. Bicarbonate Dependence of Glutamate Receptor Activation by β-N-Methylamino-l-Alanine: Channel Recording and Study with Related Compounds. Neuron 1989, 3, 321–326. [Google Scholar] [CrossRef]
- Yin, H.Z.; Yu, S.; Hsu, C.I.; Liu, J.; Acab, A.; Wu, R.; Tao, A.; Chiang, B.J.; Weiss, J.H. Intrathecal Infusion of Bmaa Induces Selective Motor Neuron Damage and Astrogliosis in the Ventral Horn of the Spinal Cord. Exp. Neurol. 2014, 261, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Delcourt, N.; Claudepierre, T.; Maignien, T.; Arnich, N.; Mattei, C. Cellular and Molecular Aspects of the β-N-Methylamino-l-Alanine (Bmaa) Mode of Action within the Neurodegenerative Pathway: Facts and Controversy. Toxins 2018, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Lobner, D. Mechanisms of Beta-N-Methylamino-l-Alanine Induced Neurotoxicity. Amyotroph. Lateral Scler. 2009, 10 (Suppl. 2), 56–60. [Google Scholar] [CrossRef] [PubMed]
- Popova, A.A.; Koksharova, O.A. Neurotoxic Non-Proteinogenic Amino Acid Beta-N-Methylamino-l-Alanine and Its Role in Biological Systems. Biochemistry 2016, 81, 794–805. [Google Scholar] [CrossRef] [PubMed]
- Dunlop, R.A.; Cox, P.A.; Banack, S.A.; Rodgers, K.J. The Non-Protein Amino Acid Bmaa Is Misincorporated into Human Proteins in Place of l-Serine Causing Protein Misfolding and Aggregation. PLoS ONE 2013, 8, e75376. [Google Scholar] [CrossRef] [PubMed]
- Van Onselen, R.; Cook, N.A.; Phelan, R.R.; Downing, T.G. Bacteria Do Not Incorporate Beta-N-Methylamino-l-Alanine into Their Proteins. Toxicon 2015, 102, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Van Onselen, R.; Downing, S.; Kemp, G.; Downing, T.G. Investigating β-N-Methylamino-l-Alanine Misincorporation in Human Cell Cultures: A Comparative Study with Known Amino Acid Analogues. Toxins 2017, 9, 400. [Google Scholar] [CrossRef] [PubMed]
- Banack, S.A.; Murch, S.J.; Cox, P.A. Neurotoxic Flying Foxes as Dietary Items for the Chamorro People, Marianas Islands. J. Ethnopharmacol. 2006, 106, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Banack, S.A.; Cox, P.A. Biomagnification of Cycad Neurotoxins in Flying Foxes: Implications for Als-Pdc in Guam. Neurology 2003, 61, 387–389. [Google Scholar] [CrossRef] [PubMed]
- Banack, S.A.; Cox, P.A. Distribution of the Neurotoxic Nonprotein Amino Acid Bmaa in Cycas Micronesica. Bot. J. Linn. Soc. 2003, 143, 165–168. [Google Scholar] [CrossRef]
- Cox, P.A.; Banack, S.A.; Murch, S.J. Biomagnification of Cyanobacterial Neurotoxins and Neurodegenerative Disease among Chamorro People of Guam. Proc. Natl. Acad. Sci. USA 2003, 100, 13380–13383. [Google Scholar] [CrossRef] [PubMed]
- Cox, P.A.; Banack, S.A.; Murch, S.J.; Rasmussen, U.; Tien, G.; Bidigare, R.R.; Metcalf, J.S.; Morrison, L.F.; Codd, G.A.; Bergman, B. Diverse Taxa of Cyanobacteria Produce β-N-Methylamino-l-Alanine, a Neurotoxic Amino Acid. Proc. Natl. Acad. Sci. USA 2005, 102, 5074–5078. [Google Scholar] [CrossRef] [PubMed]
- Banack, S.A.; Metcalf, J.S.; Spacil, Z.; Downing, T.G.; Downing, S.; Long, A.; Nunn, P.B.; Cox, P.A. Distinguishing the Cyanobacterial Neurotoxin Beta-N-Methylamino-l-Alanine (Bmaa) from Other Diamino Acids. Toxicon 2011, 57, 730–738. [Google Scholar] [CrossRef] [PubMed]
- Faassen, E.J. Presence of the Neurotoxin Bmaa in Aquatic Ecosystems: What Do We Really Know? Toxins 2014, 6, 1109–1138. [Google Scholar] [CrossRef] [PubMed]
- Kruger, T.; Monch, B.; Oppenhauser, S.; Luckas, B. LC-MS/MS Determination of the Isomeric Neurotoxins Bmaa (Beta-N-Methylamino-l-Alanine) and Dab (2,4-Diaminobutyric Acid) in Cyanobacteria and Seeds of Cycas Revoluta and Lathyrus Latifolius. Toxicon 2010, 55, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Aigret, B.; de Borggraeve, W.M.; Spacil, Z.; Ilag, L.L. Selective LC-MS/MS Method for the Identification of Bmaa from Its Isomers in Biological Samples. Anal. Bioanal. Chem. 2012, 403, 1719–1730. [Google Scholar] [CrossRef] [PubMed]
- Faassen, E.J.; Antoniou, M.G.; Beekman-Lukassen, W.; Blahova, L.; Chernova, E.; Christophoridis, C.; Combes, A.; Edwards, C.; Fastner, J.; Harmsen, J.; et al. A Collaborative Evaluation of LC-MS/MS Based Methods for Bmaa Analysis: Soluble Bound Bmaa Found to Be an Important Fraction. Mar. Drugs 2016, 14. [Google Scholar] [CrossRef] [PubMed]
- Faassen, E.J.; Gillissen, F.; Lurling, M. A Comparative Study on Three Analytical Methods for the Determination of the Neurotoxin Bmaa in Cyanobacteria. PLoS ONE 2012, 7, e36667. [Google Scholar] [CrossRef] [PubMed]
- Reveillon, D.; Abadie, E.; Sechet, V.; Brient, L.; Savar, V.; Bardouil, M.; Hess, P.; Amzil, Z. Beta-N-Methylamino-l-Alanine: LC-MS/MS Optimization, Screening of Cyanobacterial Strains and Occurrence in Shellfish from Thau, a French Mediterranean Lagoon. Mar. Drugs 2014, 12, 5441–5467. [Google Scholar] [CrossRef] [PubMed]
- Reveillon, D.; Abadie, E.; Sechet, V.; Masseret, E.; Hess, P.; Amzil, Z. Beta-N-Methylamino-l-Alanine (Bmaa) and Isomers: Distribution in Different Food Web Compartments of Thau Lagoon, French Mediterranean Sea. Mar. Environ. Res. 2015, 110, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Rosen, J.; Hellenas, K.E. Determination of the Neurotoxin Bmaa (Beta-N-Methylamino-l-Alanine) in Cycad Seed and Cyanobacteria by LC-MS/MS (Liquid Chromatography Tandem Mass Spectrometry). Analyst 2008, 133, 1785–1789. [Google Scholar] [CrossRef] [PubMed]
- Berntzon, L.; Erasmie, S.; Celepli, N.; Eriksson, J.; Rasmussen, U.; Bergman, B. Bmaa Inhibits Nitrogen Fixation in the Cyanobacterium Nostoc sp. PCC 7120. Mar. Drugs 2013, 11, 3091–3108. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Johnston, E.; Aberg, K.M.; Nilsson, U.; Ilag, L.L. Strategy for Quantifying Trace Levels of Bmaa in Cyanobacteria by LC/MS/MS. Anal. Bioanal. Chem. 2013, 405, 1283–1292. [Google Scholar] [CrossRef] [PubMed]
- Spacil, Z.; Eriksson, J.; Jonasson, S.; Rasmussen, U.; Ilag, L.L.; Bergman, B. Analytical Protocol for Identification of Bmaa and Dab in Biological Samples. Analyst 2010, 135, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Scott, L.L.; Downing, S.; Phelan, R.R.; Downing, T.G. Environmental Modulation of Microcystin and β-N-Methylamino-l-Alanine as a Function of Nitrogen Availability. Toxicon 2014, 87, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Reveillon, D.; Sechet, V.; Hess, P.; Amzil, Z. Production of Bmaa and Dab by Diatoms (Phaeodactylum tricornutum, Chaetoceros sp., Chaetoceros calcitrans and, Thalassiosira pseudonana) and Bacteria Isolated from a Diatom Culture. Harmful Algae 2016, 58, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W.; Huisman, J. Climate: Blooms Like It Hot. Science 2008, 320, 57–58. [Google Scholar] [CrossRef] [PubMed]
- Al-Sammak, M.A.; Hoagland, K.D.; Cassada, D.; Snow, D.D. Co-Occurrence of the Cyanotoxins Bmaa, Daba and Anatoxin-a in Nebraska Reservoirs, Fish, and Aquatic Plants. Toxins 2014, 6, 488–508. [Google Scholar] [CrossRef] [PubMed]
- Esterhuizen-Londt, M.; Downing, T.G. Solid Phase Extraction of β-N-Methylamino-l-Alanine (Bmaa) from South African Water Supplies. Water SA 2011, 37, 523–528. [Google Scholar] [CrossRef]
- Faassen, E.J.; Gillissen, F.; Zweers, H.A.J.; Lurling, M. Determination of the Neurotoxins Bmaa (β-N-Methylamino-l-Alanine) and Dab (α-,γ-Diaminobutyric Acid) by LC-MSMS in Dutch Urban Waters with Cyanobacterial Blooms. Amyotroph. Lateral Scler. 2009, 10 (Suppl. 2), 79–84. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, J.S.; Banack, S.A.; Lindsay, J.; Morrison, L.F.; Cox, P.A.; Codd, G.A. Co-Occurrence of Beta-N-Methylamino-l-Alanine, a Neurotoxic Amino Acid with Other Cyanobacterial Toxins in British Waterbodies, 1990-2004. Environ. Microbiol. 2008, 10, 702–708. [Google Scholar] [CrossRef] [PubMed]
- Roy-Lachapelle, A.; Solliec, M.; Sauvé, S. Determination of Bmaa and Three Alkaloid Cyanotoxins in Lake Water Using Dansyl Chloride Derivatization and High-Resolution Mass Spectrometry. Anal. Bioanal. Chem. 2015, 407, 5487–5501. [Google Scholar] [CrossRef] [PubMed]
- Caller, T.A.; Doolin, J.W.; Haney, J.F.; Murby, A.J.; West, K.G.; Farrar, H.E.; Ball, A.; Harris, B.T.; Stommel, E.W. A Cluster of Amyotrophic Lateral Sclerosis in New Hampshire: A Possible Role for Toxic Cyanobacteria Blooms. Amyotroph. Lateral Scler. 2009, 10 (Suppl. 2), 101–108. [Google Scholar] [CrossRef] [PubMed]
- Torbick, N.; Hession, S.; Stommel, E.; Caller, T. Mapping Amyotrophic Lateral Sclerosis Lake Risk Factors across Northern New England. Int. J. Health Geogr. 2014, 13. [Google Scholar] [CrossRef] [PubMed]
- Brand, L.E.; Pablo, J.; Compton, A.; Hammerschlag, N.; Mash, D.C. Cyanobacterial Blooms and the Occurrence of the Neurotoxin Beta-N-Methylamino-l-Alanine (Bmaa) in South Florida Aquatic Food Webs. Harmful Algae 2010, 9, 620–635. [Google Scholar] [CrossRef] [PubMed]
- Jonasson, S.; Eriksson, J.; Berntzon, L.; Spacil, Z.; Ilag, L.L.; Ronnevi, L.O.; Rasmussen, U.; Bergman, B. Transfer of a Cyanobacterial Neurotoxin within a Temperate Aquatic Ecosystem Suggests Pathways for Human Exposure. Proc. Natl. Acad. Sci. USA 2010, 107, 9252–9257. [Google Scholar] [CrossRef] [PubMed]
- Masseret, E.; Banack, S.; Boumédiène, F.; Abadie, E.; Brient, L.; Pernet, F.; Juntas-Morales, R.; Pageot, N.; Metcalf, J.; Cox, P.; et al. Dietary Bmaa Exposure in an Amyotrophic Lateral Sclerosis Cluster from Southern France. PLoS ONE 2013, 8, e83406. [Google Scholar] [CrossRef] [PubMed]
- Reveillon, D.; Sechet, V.; Hess, P.; Amzil, Z. Systematic Detection of Bmaa (Beta-N-Methylamino-l-Alanine) and Dab (2,4-Diaminobutyric Acid) in Mollusks Collected in Shellfish Production Areas Along the French Coasts. Toxicon 2016, 110, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Banack, S.A.; Metcalf, J.S.; Bradley, W.G.; Cox, P.A. Detection of Cyanobacterial Neurotoxin Beta-N-Methylamino-l-Alanine within Shellfish in the Diet of an Als Patient in Florida. Toxicon 2014, 90, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Field, N.C.; Metcalf, J.S.; Caller, T.A.; Banack, S.A.; Cox, P.A.; Stommel, E.W. Linking β-Methylamino-l-Alanine Exposure to Sporadic Amyotrophic Lateral Sclerosis in Annapolis, MD. Toxicon 2013, 70, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Chernoff, N.; Hill, D.J.; Diggs, D.L.; Faison, B.D.; Francis, B.M.; Lang, J.R.; Larue, M.M.; Le, T.T.; Loftin, K.A.; Lugo, J.N.; et al. A Critical Review of the Postulated Role of the Non-Essential Amino Acid, β-N-Methylamino-l-Alanine, in Neurodegenerative Disease in Humans. J. Toxicol. Environ. Health Part B Crit. Rev. 2017, 20, 183–229. [Google Scholar] [CrossRef] [PubMed]
- Agence Nationale de Sécurité Sanitaire de L’alimentation, de L’environnement et du Travail (Anses). Données Relatives À La Toxicité Aiguë Et Chronique De La Bêta-Méthylamino-l-Alanine (Bmaa). In AVIS de L’Anses—Rapport D’expertise Collective; Anses: Maisons-Alfort, France, 2017; p. 166. [Google Scholar]
- Jonnalagadda, S.R.; Goyal, P.; Huffman, M.D. Automating data extraction in systematic reviews: A systematic review. Syst. Rev. 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Lage, S.; Annadotter, H.; Rasmussen, U.; Rydberg, S. Biotransfer of β-N-Methylamino-l-Alanine (Bmaa) in a Eutrophicated Freshwater Lake. Mar. Drugs 2015, 13, 1185–1201. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.; Qiu, J.; Fan, L.; Li, A. Effects of Growth Conditions on the Production of Neurotoxin 2,4-Diaminobutyric Acid (Dab) in Microcystis Aeruginosa and Its Universal Presence in Diverse Cyanobacteria Isolated from Freshwater in China. Environ. Sci. Pollut. Res. 2015, 22, 5943–5951. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Kiselova, N.; Rosen, J.; Ilag, L.L. Quantification of Neurotoxin Bmaa (Beta-N-Methylamino-l-Alanine) in Seafood from Swedish Markets. Sci. Rep. 2014, 4, 6931. [Google Scholar] [CrossRef] [PubMed]
- Banack, S.A.; Caller, T.; Henegan, P.; Haney, J.; Murby, A.; Metcalf, J.S.; Powell, J.; Cox, P.A.; Stommel, E. Detection of Cyanotoxins, Beta-N-Methylamino-l-Alanine and Microcystins, from a Lake Surrounded by Cases of Amyotrophic Lateral Sclerosis. Toxins 2015, 7, 322–336. [Google Scholar] [CrossRef] [PubMed]
- Christensen, S.J.; Hemscheidt, T.K.; Trapido-Rosenthal, H.; Laws, E.A.; Bidigare, R.R. Detection and Quantification of β-Methylamino-l-Alanine in Aquatic Invertebrates. Limnol. Oceanogr. Methods 2012, 10, 891–898. [Google Scholar] [CrossRef]
- Salomonsson, M.L.; Hansson, A.; Bondesson, U. Development and in-House Validation of a Method for Quantification of Bmaa in Mussels Using Dansyl Chloride Derivatization and Ultra Performance Liquid Chromatography Tandem Mass Spectrometry. Anal. Methods 2013, 5, 4865–4874. [Google Scholar] [CrossRef]
- Salomonsson, M.L.; Fredriksson, E.; Alfjorden, A.; Hedeland, M.; Bondesson, U. Seafood Sold in Sweden Contains Bmaa: A Study of Free and Total Concentrations with Uhplc-Ms/Ms and Dansyl Chloride Derivatization. Toxicol. Rep. 2015, 2, 1473–1481. [Google Scholar] [CrossRef] [PubMed]
- Mondo, K.; Glover, W.B.; Murch, S.J.; Liu, G.; Cai, Y.; Davis, D.A.; Mash, D.C. Environmental Neurotoxins Beta-N-Methylamino-l-Alanine (Bmaa) and Mercury in Shark Cartilage Dietary Supplements. Food Chem. Toxicol. 2014, 70, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Lage, S.; Costa, P.R.; Moita, T.; Eriksson, J.; Rasmussen, U.; Rydberg, S.J. Bmaa in Shellfish from Two Portuguese Transitional Water Bodies Suggests the Marine Dinoflagellate Gymnodinium Catenatum as a Potential Bmaa Source. Aquat. Toxicol. 2014, 152, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Beach, D.G.; Kerrin, E.S.; Quilliam, M.A. Selective Quantitation of the Neurotoxin Bmaa by Use of Hydrophilic-Interaction Liquid Chromatography-Differential Mobility Spectrometry-Tandem Mass Spectrometry (Hilic-Dms-MS/MS). Anal. Bioanal. Chem. 2015, 407, 8397–8409. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Song, J.; Hu, Y.; Deng, L.; Ding, L.; Li, M. New Typical Vector of Neurotoxin β-N-Methylamino-l-Alanine (Bmaa) in the Marine Benthic Ecosystem. Mar. Drugs 2016, 14. [Google Scholar] [CrossRef] [PubMed]
- Hammerschlag, N.; Davis, D.A.; Mondo, K.; Seely, M.S.; Murch, S.J.; Glover, W.B.; Divoll, T.; Evers, D.C.; Mash, D.C. Cyanobacterial Neurotoxin Bmaa and Mercury in Sharks. Toxins 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Mondo, K.; Hammerschlag, N.; Basile, M.; Pablo, J.; Banack, S.A.; Mash, D.C. Cyanobacterial Neurotoxin β-N-Methylamino-l-Alanine (Bmaa) in Shark Fins. Mar. Drugs 2012, 10, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Andrys, R.; Zurita, J.; Zguna, N.; Verschueren, K.; de Borggraeve, W.M.; Ilag, L.L. Improved Detection of Beta-N-Methylamino-l-Alanine Using N-Hydroxysuccinimide Ester of N-Butylnicotinic Acid for the Localization of Bmaa in Blue Mussels (Mytilus Edulis). Anal. Bioanal. Chem. 2015, 407, 3743–3750. [Google Scholar] [CrossRef] [PubMed]
- Pip, E.; Munford, K.; Bowman, L. Seasonal nearshore occurrence of the neurotoxin β-N-methylamino-l-alanine (BMAA) in lake Winnipeg, Canada. Environ. Pollut. 2016, 5. [Google Scholar] [CrossRef]
- Baptista, M.S.; Vasconcelos, R.G.; Ferreira, P.C.; Almeida, C.M.; Vasconcelos, V.M. Assessment of the Non-Protein Amino Acid Bmaa in Mediterranean Mussel Mytilus galloprovincialis after Feeding with Estuarine Cyanobacteria. Environ. Sci. Pollut. Res. Int. 2015, 22, 12501–12510. [Google Scholar] [CrossRef] [PubMed]
- Downing, S.; Contardo-Jara, V.; Pflugmacher, S.; Downing, T.G. The Fate of the Cyanobacterial Toxin β-N-Methylamino-l-Alanine in Freshwater Mussels. Ecotoxicol. Environ. Saf. 2014, 101, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Lepoutre, A.; Milliote, N.; Bonnard, M.; Rioult, D.; Geffard, A.; Faassen, E.J.; Lance, E. Genotoxic and Cytotoxic Effects on Immune Cells of the Freshwater Bivalve Dreissena Polymorpha Exposed to the Environmental Neurotoxin BMAA. Toxins 2018. submitted. [Google Scholar]
- Glover, W.B.; Mash, D.C.; Murch, S.J. The Natural Non-Protein Amino Acid N-β-Methylamino-l-Alanine (BMAA) Is Incorporated into Protein During Synthesis. Amino Acids 2014, 46, 2553–2559. [Google Scholar] [CrossRef] [PubMed]
- Rosen, J.; Westerberg, E.; Hellenas, K.E.; Salomonsson, M.L. A New Method for Analysis of Underivatized Free Beta-Methylamino-Alanine: Validation and Method Comparison. Toxicon 2016, 121, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Ibelings, B.W.; Chorus, I. Accumulation of Cyanobacterial Toxins in Freshwater “Seafood” and Its Consequences for Public Health: A Review. Environ. Pollut. 2007, 150, 177–192. [Google Scholar] [CrossRef] [PubMed]
- Lance, E.; Josso, C.; Dietrich, D.; Ernst, B.; Paty, C.; Senger, F.; Bormans, M.; Gérard, C. Histopathology and Microcystin Distribution in Lymnaea Stagnalis (Gastropoda) Following Toxic Cyanobacterial or Dissolved Microcystin-Lr Exposure. Aquat. Toxicol. 2010, 98, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Perkins, H.R.; Cummins, C.S. Chemical Structure of Bacterial Cell Walls. Nature 1964, 201, 1105–1107. [Google Scholar] [CrossRef] [PubMed]
- Groth, I.; Schumann, P.; Weiss, N.; Martin, K.; Rainey, F.A. Agrococcus jenensis gen. nov., sp. nov., a New Genus of Actinomycetes with Diaminobutyric Acid in the Cell Wall. Int. J. Syst. Evol. Microbiol. 1996, 46, 234–239. [Google Scholar] [CrossRef] [PubMed]
- McGorum, B.C.; Pirie, R.S.; Glendinning, L.; McLachlan, G.; Metcalf, J.S.; Banack, S.A.; Cox, P.A.; Codd, G.A. Grazing Livestock Are Exposed to Terrestrial Cyanobacteria. Vet. Res. 2015, 46. [Google Scholar] [CrossRef] [PubMed]
- Regueiro, J.; Negreira, N.; Carreira-Casais, A.; Pérez-Lamela, C.; Simal-Gándara, J. Dietary Exposure and Neurotoxicity of the Environmental Free and Bound Toxin β-N-Methylamino-l-Alanine. Food Res. Int. 2017, 100, 1–13. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Application of Systematic Review Methodology to Food and Feed Safety Assessments to Support Decision Making. EFSA J. 2010, 8, 1637. [Google Scholar] [CrossRef]
- Souci, S.W.; Fachmann, W.; Kraut, H. Food Composition and Nutrition Tables, 5th Revised and Completed Edition; Medpharm: Stuttgart, Germany, 1994. [Google Scholar]
| Type of Sample | Type and Number of Samples | Origins | Concentrations of BMAA and Isomers, Reported in µg g−1 DW of Phytoplankton Biomass or in µg L−1 of Water Sample | LOD | LOQ | Reference | Score | ||
|---|---|---|---|---|---|---|---|---|---|
| BMAA | AEG | DAB | |||||||
| Water | Lake water (n = 12) | Canada | Fbmaa (1) ND-0.3 μg L−1 | fAEG (1) ND-0.08 μg L−1 | fDAB (1) ND-0.04 μg L−1 | in µg L−1 BMAA 0.008 AEG 0.009 DAB 0.007 | in µg L−1 BMAA 0.02 AEG 0.03 DAB 0.02 | [36] | A |
| Finjasjön Lake | Sweden | tBMAA ND-6 ng g−1 DW | – | – | BMAA (2) 0.8 µg L−1 | BMAA (2) 0.8 µg L−1 | [48] | B | |
| Lake water (n = 3) | China | fBMAA ND pbBMAA ND | – | fDAB 0.4–3.8 ng g−1 WW pbDAB ND | BMAA 2 pg DAB 5 pg | – | [49] | B | |
| Cyanobacteria | Cyanobacteria | NA | fBMAA ND tBMAA ND | – | – | In µg g−1 DW fBMAA 0.2–1 tBMAA 2.5–20 | – | [21] | A |
| Lagoon periphyton | France | tsBMAA 1.3–4.3 μg g−1 DW | tsAEG ND-0.8 μg g−1 | tsDAB 1.3–4.8 μg g−1 DW fDAB: mean 0.49 μg g−1 DW | 0.23 µg g−1 DW | – | [24] | A | |
| Lagoon seston | France | Micro sbBMAA mean 0.49 μg g−1 DW Zoo: sbBMAA mean 0.63 μg g−1 DW | Micro ND Zoo ND | Micro sbDAB mean 0.69 μg g−1 DW Zoo sbDAB mean 0.92 μg g−1 DW | 0.23 µg g−1 DW | – | [24] | A | |
| Baltic sea blooms (n = 4) | Sweden Finland | fBMAA ND pbBMAA ND | – | – | In µg g−1 fBMAA 1 tBMAA 4 | – | [25] | A | |
| Cyanobacterial scums, urban water bodies (n = 21) | Netherlands | fBMAA ND-42 μg g−1 DW (9/21) pbBMAA ND | – | tDAB ND-4 μg g−1 DW (2/21) pbDAB ND | – | – | [34] | B | |
| Cyanobacteria Baltic Sea, Askö Island | Sweden | tBMAA 2.3–15 ng g−1 DW | – | – | 70 fmol | – | [40] | B | |
| Type of Sample | Species | Origins | Concentrations (mg kg−1 FW) (1) (Number of Positive Samples/Number of Samples) | LOD | LOQ | Reference | ||
|---|---|---|---|---|---|---|---|---|
| BMAA | AEG | DAB | ||||||
| Bivalves | Mussels Mytilus galloprovincialis | Thau Lagoon, Mediterranean sea, France | fBMAA ND-0.2 (16/34) tsBMAA max 1.65, mean 0.68 (34/34) | fAEG ND-0.05 (5/34) tsAEG max 0.2 (31/34) | fDAB ND-1.05 (34/34) tsDAB max 1.8, mean 1.22 (34/34) | – | 0.15 DW | [24] |
| Mussels Mytilus galloprovincialis and Mytilus edulis, Oysters Crassostrea gigas | Channel, Atlantic, Mediterranean Sea, France | tsBMAA 0.07–1.13 (74/74) tsBMAA 0.03–0.41(23/23) | ND ND | Mussels and oysters: tsDAB 0.2–4.84 (97/97) | – | BMAA: 0.45 DW DAB: 0.15 DW | [42] | |
| Mussels Mytilus sp Oysters Ostrea edulis, Crassostrea gigas | North Atlantic, Sweden west coast, Greece, France | tBMAA 0.08–0.9 (6/6) tBMAA 0.1–0.66 (4/4) | <0.01 (2) | <0.01 (2) | [50] | |||
| Oysters Crassostrea virginica | Louisiana Mississipi | tBMAA 1.5–8 (12/12) tBMAA 1.2–1.7 (3/3) | 0.5 (3) | 1.7 (3) | [52] | |||
| Mussels Mytilus galloprovincialis | Thau Lagoon, Mediterranean Sea, France | fBMAA < 0.34 (4/11) tsBMAA 0.64–2.45 (11/11) | fAEG < 0.08 (3/11) tsAEG 0.1–0.2 (11/11) | fDAB 0.08-1.2 (11/11) tsDAB 0.6–1.6 (11/11) | – | 0.15 DW | [23] | |
| Oysters Crassostrea gigas | fBMAA < 0.08 (1/8) tsBMAA 0.5–1.8 (8/8) | fAEG ND (0/8) tsAEG 0.1–0.3 (8/8) | fDAB 0.03–0.6 (8/8) tsDAB 0.6–1.5 (8/8) | – | 0.15 DW | [23] | ||
| Mussels | Western coast of Sweden | tBMAA 0.27–1.6 (4/4) | 0.15 | [53] | ||||
| Mussels Mytilus edulis Mytilus edulis platensis Perna Canaliculus Scallops Placopecten magellanicus | Scandinavia South America Australia US | fBMAA ND (0/6) tBMAA 0.28–0.59 (6/6) fBMAA ND-0.38 (5/12) tBMAA 1.69–7.08 (12/12) fBMAA ND-0.38 (1/3) tBMAA 0.55–1.14 (3/3) fBMAA 0.18–0.46 (3/3) tBMAA 1.12–1.46 (3/3) | 0.10 (4) | 0.15 (4) | [54] | |||
| Crustaceans | Shrimps Caridea sp Crayfish Astacus leptodactylus | North Atlantic, Sweden Turkey, Sweden | tBMAA 0.11–0.46 (6/6) tBMAA ND (0/6) | <0.01 (2) | <0.01 (2) | [50] | ||
| Blue crabs Callinectes sapidus | Florida | tBMAA 1.08–3.02 (5/5) | 0.5 (3) | 1.7 (3) | [52] | |||
| Crabs Cancer pagarus Portunus haani Crayfish Procambrus claarki Shrimps Pandalus borealis | Ireland, North Atlantic Vietnam China Greenland, North Atlantic | tBMAA detected, NQ (1/1) tBMAA ND (0/1) tBMAA ND (0/1) tBMAA ND (0/3) | 0.10 (3) | 0.15 (3) | [54] | |||
| Fish | Plaice Pleuronectes platessa, Herring Clupea harengus Char Salvelinus alpinus Salmon Salmo salar Cod Gadus morhua Perch Perca fluviatilis | North Atlantic Baltic Sea Baltic Sea Sweden Norway Norway North Atlantic Sweden | tBMAA 0.01–0.02 (3/3) tBMAA ND-0.02 (1/3) tBMAA ND-0.01 (1/3) tBMAA ND (0/4) tBMAA ND (0/4) tBMAA ND (0/4) | <0.01 (2) | <0.01 (2) | [50] | ||
| Atlantic salmon Salmo salar Sea bass Dicentrarchus labrax Sea bream Sparus aurata Whitefish Coregonus sp, Pike perch Sander lucioperca, Sea trout Salmo truttae | Norway Italia Greece Sweden Sweden Baltic Sea Sweden Bothnain Sea | tBMAA ND (0/1) tBMAA ND (0/1) tBMAA ND (0/1) tBMAA ND (0/1) tBMAA ND (0/1) tBMAA ND (0/2) | 0.10 (4) | 0.15 (4) | [54] | |||
| Shark cartilage powder, variety of shark species not identified | Commercial food supplements, from 7 manufacturers | (In mg kg−1 DW) tBMAA (5) 74.8–352.2 (15/16) | (In mg kg−1 DW) tAEG (5) 1298.4–1765.1 (16/16) | (In mg kg−1 DW) etDAB (5) 69.2–1483.4 (16/16) | (in pg L−1) BMAA 1.1 AEG 1.2 DAB 0.8 | [55] | ||
| Type of Sample | Species, Number of Sample | Origins | Concentrations (mg kg−1 FW) (1) | LOD | LOQ | Reference | ||
|---|---|---|---|---|---|---|---|---|
| BMAA | AEG | DAB | ||||||
| Bivalves | Mussels Mytilus edulis Oyster Ostrea edulis | Baltic Sea | tBMAA 0.02–0.03 (3/3) tBMAA ND-0.02 (3/3) | – | – | 70 fmol | – | [40] |
| Cockles Cerastoderma edule samples of 30 individuals | Ria de Aveiro Ria Formosa Portugal | pbBMAA 0.018–0.081 (9/9) pbBMAA ND-0.1 (5/10) | – | – | 0.05 ng | 0.05 ng | [56] | |
| Mussels Mytilus edulis | Canada | tBMAA 0.19–0.24 (9/9) | NQ | NQ | 20 ng g−1 DW | – | [57] | |
| Mollusks (29 species) 68 samples Mussel Mytilus coruscus Razor clam Solen strictus Gastropod Neverita didyma Other marine species: Mytilus galloprovincialis, Crassostrea sp., Perna viridis, Antigona lamellaris, Atrina pectinata, Meretrix lusoria, Periglypta petechialis, Chlamys farreri, Mactra chinensis, Ruditapes philippinarum, Sinonovacula constrita, Tegillarca granosa, Haliotis discus hannai, Turritella bacillum, Natica maculosa, Batillaria zonalis, Moerella iridescens, Scapharca subcrenata, Mactra chinensis, Volutharpa ampullacea, Neptunea cumingii, Arca inflata, Merceneria merceneria, Rapana venosa, Argopecten irradians, Mimachlamys nobilis, Gafrarium tumidum | Sampling in aquaculture zones and markets from 10 cities along the Chinese coast, and in situ sampling of gastropods | fBMAA (5/68) in: fBMAA 0.45 (1/2) fBMAA 0.66 (1/1) fBMAA 0.99–3.97 (3/5) For all samples pbBMAA ND (0/68) | For all samples fAEG and pbAEG ND | For all samples pbDAB ND fDAB (53/68), in 23 marine species 0.05–2.65 | BMAA: 0.31 AEG: 0.10 DAB: 0.013 | – | [58] | |
| Crustaceans | Lobster Panulirus sp muscle tail | Florida | Boiled flesh: tBMAA (2) 0.70–6.94 (4/4) Fresh flesh: fBMAA ND-0.08 (1/2) pbBMAA 0.42–2.20 (1/2) | – | Boiled flesh: tDAB (2) 1.10–8.33 (4/4) Fresh flesh: fDAB 0.17–0.21 (2/2) pbDAB 0.01–0.08 (2/2) | BMAA: 48 fmol DAB: 26 fmol | BMAA: 0.48 fmol DAB: 0.26 fmol | [43] |
| Blue crab Callinectes sapidus meat of claws | East Atlantic | tsBMAA ND-24.8 (2/3) pbBMAA ND-10.80 (1/3) | tDAB 11.57–15.67 (3/3) pbDAB ND-2.91 (1/3) | – | – | [44] | ||
| Fish | Smelt Osmerus eperlanus Turbot Scophthalmus maximus Herring Clupea harengus Common whitefish Coregonus laveratus Pike-perch Sander lucioperca, fourhorn sculpin Triglopsis quadricornis, salmon Salmo salar | Baltic Sea | tBMAA in muscles 0.003–0.05 (3/3) 0.001–0.003 (2/3) 0.002 (1/3) 0.007–0.014 (2/3) ND (0/3 for all species) | – | – | 70 fmol | – | [40] |
| Bream Abramis brama Perch Perca fluviatilis Pike Esox Lucius Pike-perch Sander lucioperca Roach Rutilus rutilus Ruffe Gymnocephalus cernua Tench Tinca tinca | Sweden, freshwater lake | tBMAA in muscles 0.00002 ± 0.00006 (9/32) 0.00002 (1/29) 0.00001 (1/22) 0.0003–0.00013 (3/29) 0.00004 (1/24) 0.0008 ± 0.0008 (4/15) 0.0014 (1/15) | – | – | 0.8 ng mL−1 | – | [48] | |
| Carp Cyprinus carpio | New Hampshire | f+pbBMAA 0.25 (1/1) in muscles | – | f+pbDAB ND (0/1) | (in fmol) BMAA 48 DAB 26 | (in fmol) BMAA 0.48 DAB 0.26 | [51] | |
| Fin of 1 species, Tiger shark Galeocerdo cuvier evaluated by LC-MS/MS | Atlantic and Pacific | tBMAA 19.2–33.15 (4/4) | – | – | HPLC-FD: 2.7 ng | HPLC: 7.0 ng | [59] | |
| A single fin sample of the hammerhead shark Sphyrna mokarran evaluated by LC-MS/MS | Biscayne Bay Florida Bay | tBMAA identified, NQ | – | – | HPLC-FD: 2.7 ng | HPLC: 7.0 ng | [60] | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lance, E.; Arnich, N.; Maignien, T.; Biré, R. Occurrence of β-N-methylamino-l-alanine (BMAA) and Isomers in Aquatic Environments and Aquatic Food Sources for Humans. Toxins 2018, 10, 83. https://doi.org/10.3390/toxins10020083
Lance E, Arnich N, Maignien T, Biré R. Occurrence of β-N-methylamino-l-alanine (BMAA) and Isomers in Aquatic Environments and Aquatic Food Sources for Humans. Toxins. 2018; 10(2):83. https://doi.org/10.3390/toxins10020083
Chicago/Turabian StyleLance, Emilie, Nathalie Arnich, Thomas Maignien, and Ronel Biré. 2018. "Occurrence of β-N-methylamino-l-alanine (BMAA) and Isomers in Aquatic Environments and Aquatic Food Sources for Humans" Toxins 10, no. 2: 83. https://doi.org/10.3390/toxins10020083
APA StyleLance, E., Arnich, N., Maignien, T., & Biré, R. (2018). Occurrence of β-N-methylamino-l-alanine (BMAA) and Isomers in Aquatic Environments and Aquatic Food Sources for Humans. Toxins, 10(2), 83. https://doi.org/10.3390/toxins10020083





