Natural Occurrence of Nivalenol, Deoxynivalenol, and Deoxynivalenol-3-Glucoside in Polish Winter Wheat
Abstract
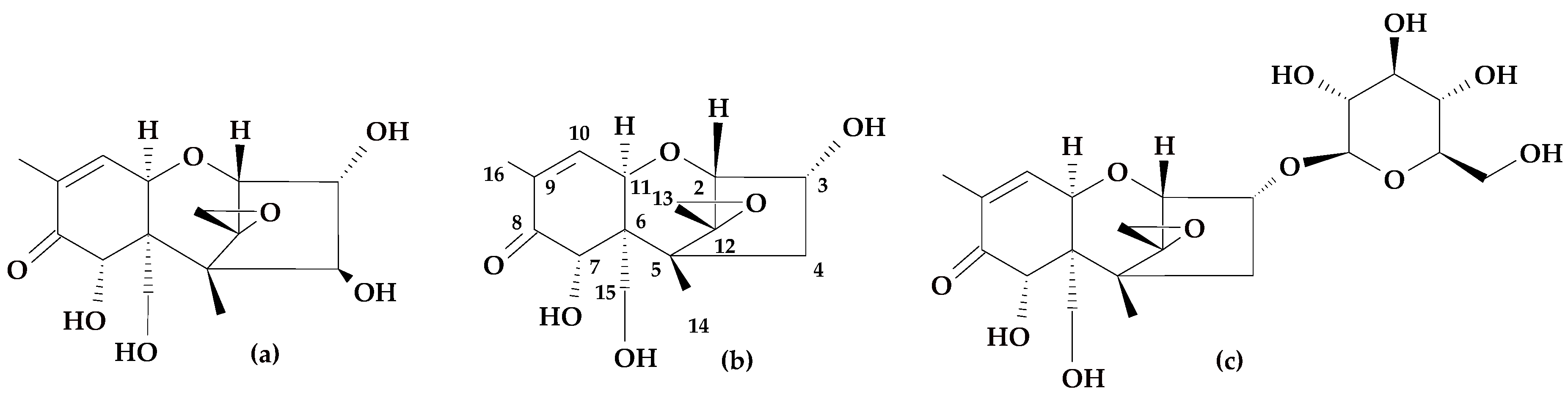
1. Introduction
2. Results
2.1. Sample Processing
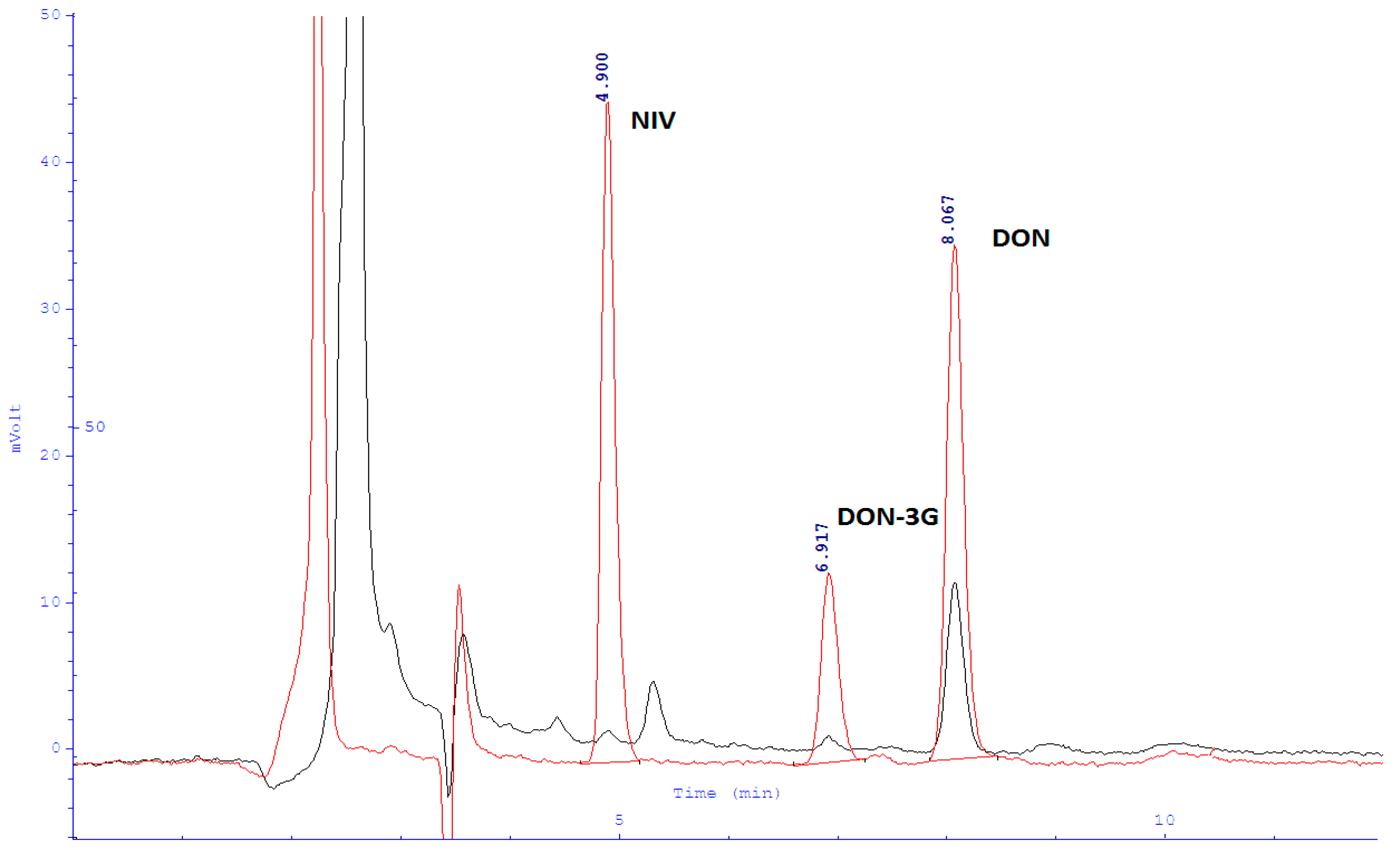
2.2. Validation of the Analytical Method
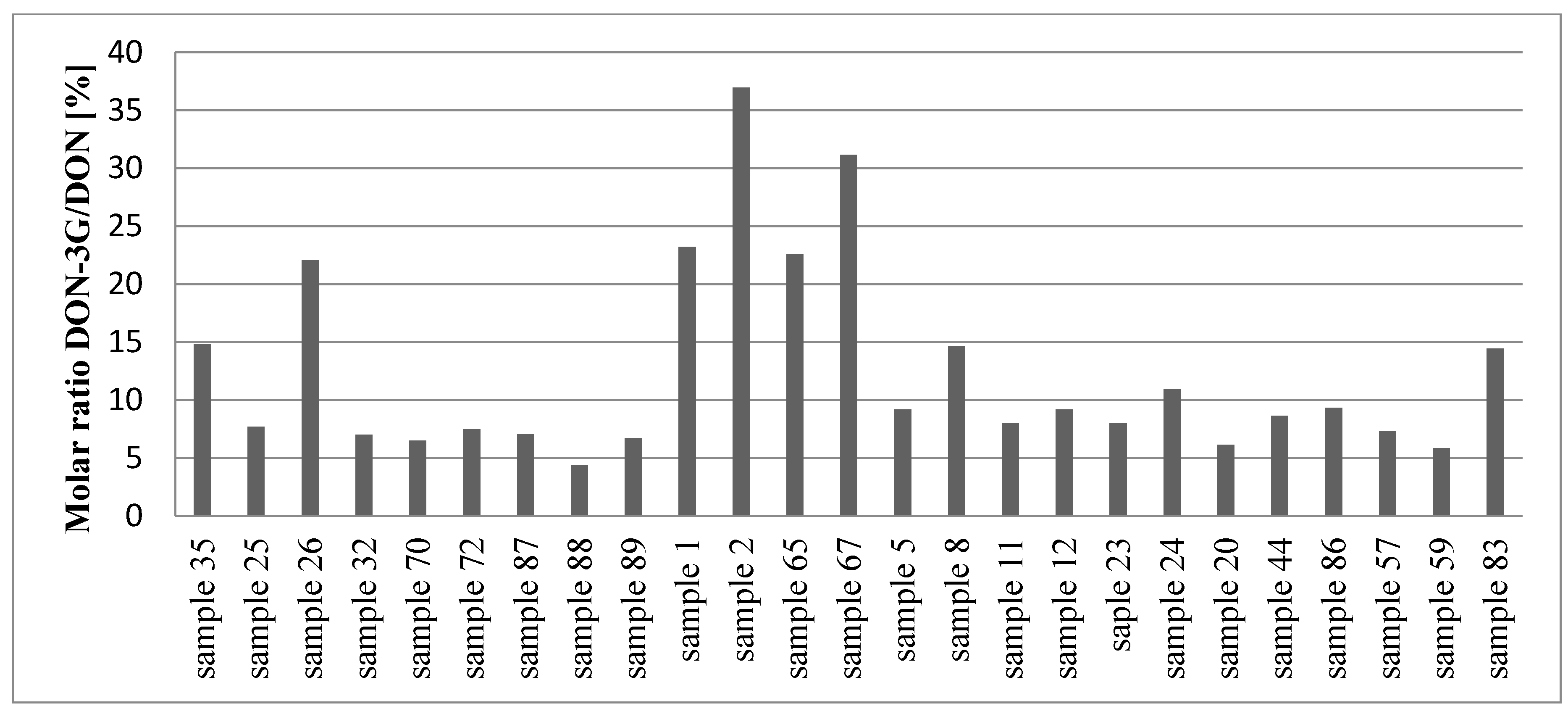
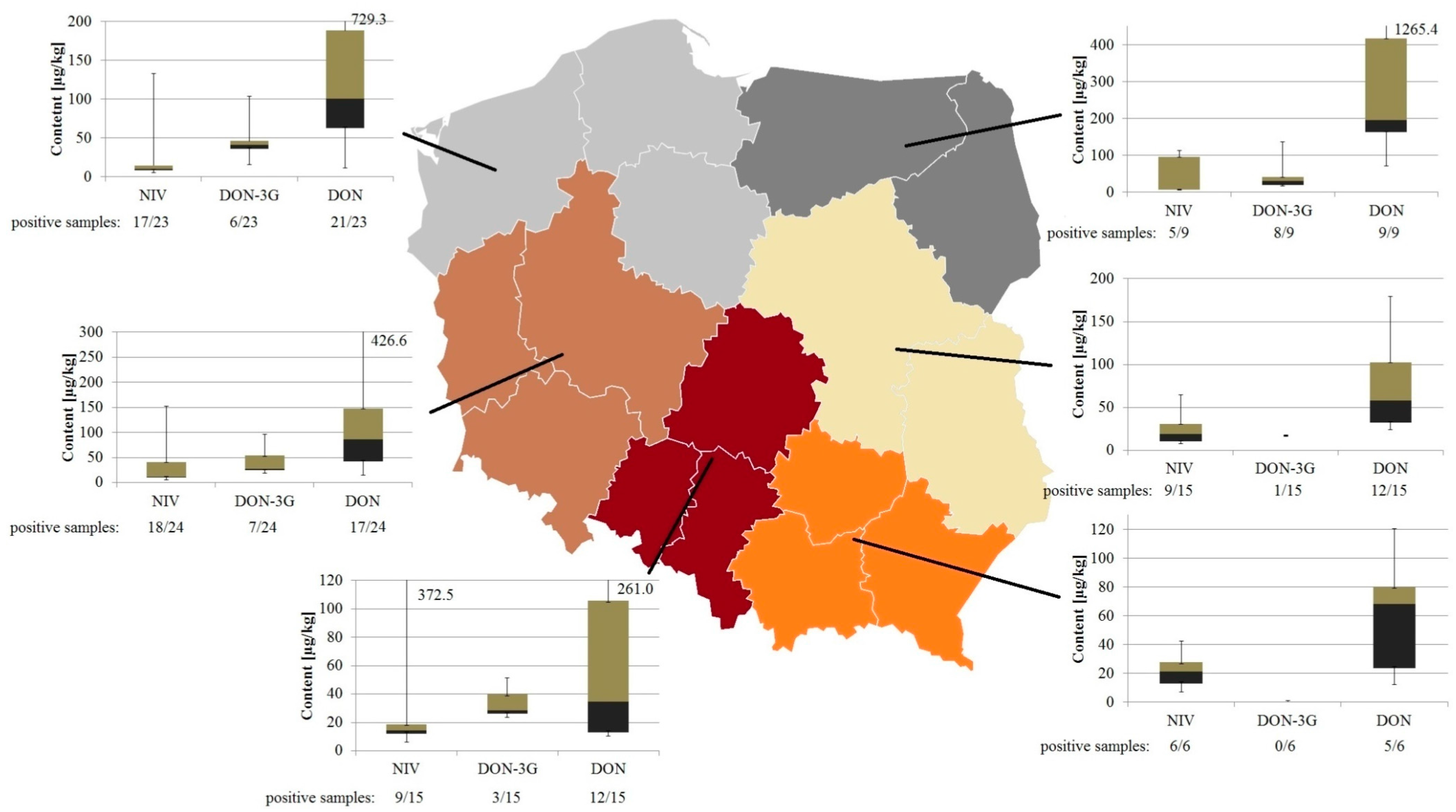
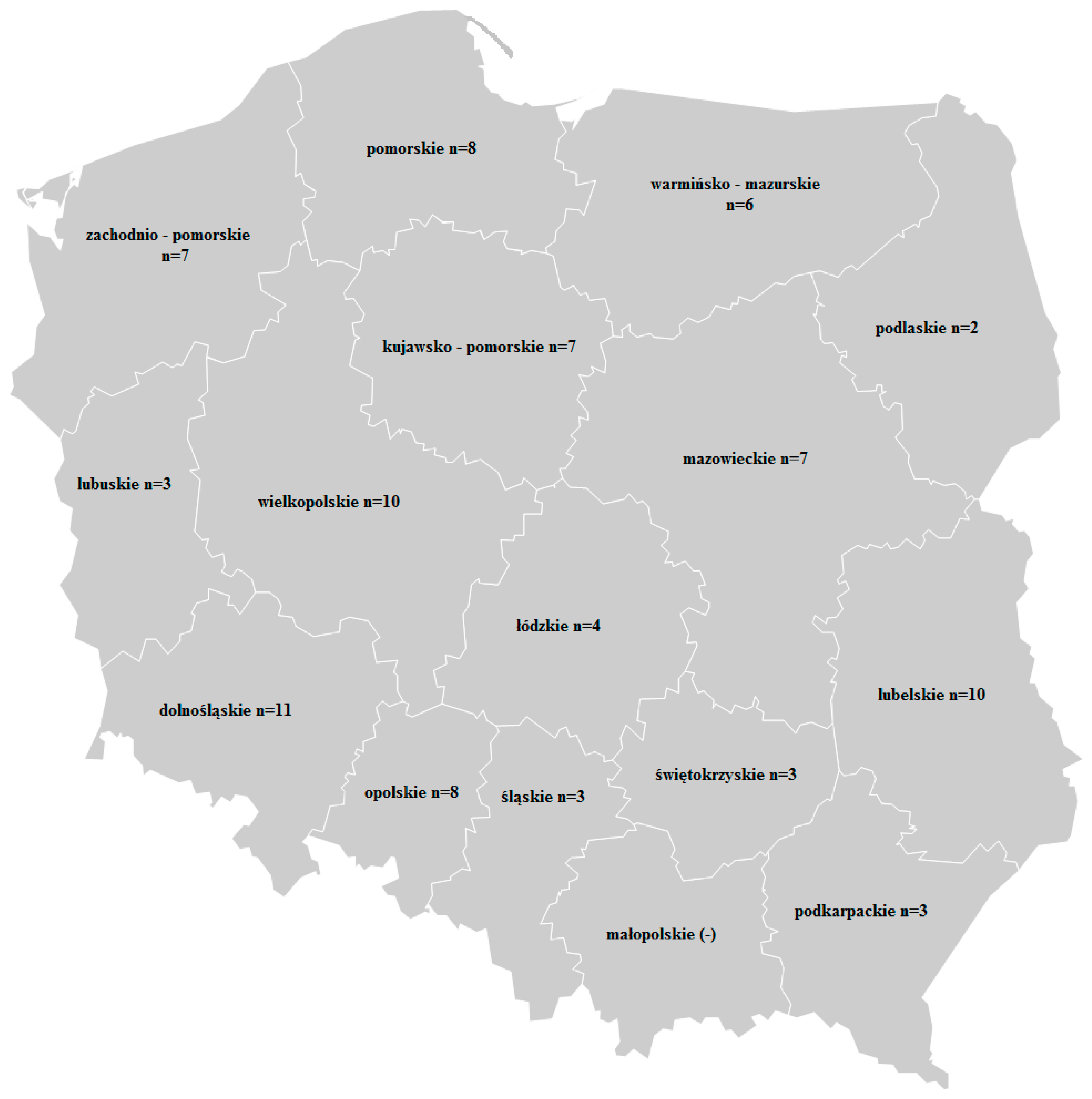
2.3. Occurrence of Mycotoxins in Wheat Grain
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Reagents/Standards/Columns
5.2. Samples
5.3. HPLC-UV
Acknowledgments
Author Contributions
Conflicts of Interest
References
- CSO. Central Statistical Office. Concise Statistical Yearbook of Poland. 2017. Available online: https://danepubliczne.gov.pl/dataset/5c9f136c-025d-4b82-b03c-d8d7148dfe09/resource/cd90dfe3-1665-4a4d-b034-25e0ead1b389/download/malyrocznikstatystycznypolski2017.pdf (accessed on 4 January 2018).
- Duveiller, E.; Singh, P.K.; Mezzalama, M.; Singh, R.P.; Dababat, A.A. Wheat Diseases and Pests: A Guide for Field Identification; CIMMYT: Mexico City, Mexico, 2012; Available online: http://repository.cimmyt.org/xmlui/handle/10883/1115 (accessed on 12 December 2017).
- Simsek, S.; Burgess, K.; Whitney, K.L.; Gu, Y.; Qian, S.Y. Analysis of Deoxynivalenol and Deoxynivalenol-3-glucoside in wheat. Food Control 2012, 26, 287–292. [Google Scholar] [CrossRef]
- Lenc, L.; Czecholiński, G.; Wyczling, D.; Turów, T.; Kaźmierczak, A. Fusarium head blight (FHB) and Fusarium spp. on grain of spring wheat cultivars grown in Poland. J. Plant Protect. Res. 2015, 55, 266–267. [Google Scholar] [CrossRef]
- Tanaka, K.; Kobayashi, H.; Nagata, T.; Manabe, M. Natural occurrence of trichothecenes on lodged and water-damaged domestic rice in Japan. Food Hyg. Saf. Sci. 2004, 45, 63–66. [Google Scholar] [CrossRef]
- Schollenberger, M.; Müller, H.M.; Rüfle, M.; Suchy, S.; Planck, S.; Drochner, W. Survey of Fusarium toxins in foodstuffs of plant origin marketed in Germany. Int. J. Food Microbiol. 2005, 97, 317–326. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer (IARC). Some Naturally Occurring Substances: Food Items and Constituents, Heterocyclic Aromatic Amines and Mycotoxins. Monograph on the Evaluation of Carcinogenic Risks to Humans; World Health Organization, International Agency for Research on Cancer: Lyon, France, 1993; pp. 397–444. [Google Scholar]
- EFSA. Scientific Opinion on risks for animal and public health related to the presence of nivalenol in food and feed. EFSA J. 2013, 11, 119. [Google Scholar]
- Rocha, O.; Ansari, K.; Doohan, F.M. Effects of trichothecene mycotoxins on eukaryotic cells: A review. Food Addit. Contam. 2005, 22, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, A.E. Chapter 1. Trichothecenes. In Fusarium Mycotoxins-Chemistry, Genetics, and Biology; American Phytopathological Society Press: St. Paul, MN, USA, 2007; pp. 13–64. [Google Scholar]
- McCormick, S. The role of DON in pathogenicity. In Fusarium Head Blight of Wheat and Barley; Leonard, K.J., Bushnell, W.R., Eds.; American Phytopathological Society Press: St. Paul, MN, USA, 2003; pp. 35–43. [Google Scholar]
- Rychlik, M.; Humpf, H.; Marko, D.; Dänicke, S.; Mally, A.; Berthiller, F.; Klaffke, H.; Lorenz, N. Proposal of a comprehensive definition of modified and other forms of mycotoxins including “masked” mycotoxins. Mycotoxin Res. 2014, 30, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, B. Fates of deoxynivalenol and deoxynivalenol-3-glucoside during bread and noodle processing. Food Control 2015, 50, 754–757. [Google Scholar] [CrossRef]
- Shin, S.; Torres-Acosta, J.A.; Heinen, S.J.; McCormick, S.; Lemmens, M.; Paris, M.P.; Berthiller, F.; Adam, G.; Meuhlbauer, G.J. Transgenic Arabidopsis thaliana expressing a barley UDP-glucosyltransferase exhibit resistance to the mycotoxin deoxynivalenol. J. Exp. Bot. 2012, 63, 4731–4740. [Google Scholar] [CrossRef] [PubMed]
- Schweiger, W.; Boddu, J.; Shin, S.; Poppenberger, B.; Berthiller, F.; Lemmens, M.; Muehlbauer, G.J.; Adam, G. Validation of a candidate deoxynivalenol-inactivating UDP-glucosyltransferase from barley by heterologous expression in yeast. Mol. Plant Microbe Interact. 2010, 23, 977–986. [Google Scholar] [CrossRef] [PubMed]
- Berthiller, F.; Dall’Asta, C.; Schuhmacher, R.; Lemmens, M.; Adam, G.; Krska, R. Masked mycotoxins: Determination of a deoxynivalenol glucoside in artificially and naturally contaminated wheat by liquid chromatography-tandem mass spectrometry. J. Agric. Food Chem. 2005, 53, 3421–3425. [Google Scholar] [CrossRef] [PubMed]
- Cirlini, M.; Generotti, S.; Dall’Erta, A.; Lancioni, P.; Ferrazzano, G.; Massi, A.; Galaverna, G.; Dall’Asta, C. Durum Wheat (Triticum Durum Desf.) Lines Show Different Abilities to Form Masked Mycotoxins under Greenhouse Conditions. Toxins 2013, 6, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Kostelanska, M.; Hajslova, J.; Zachariasova, M.; Malachova, A.; Kalachova, K.; Poustka, J.; Fiala, J.; Scott, P.M.; Berthiller, F.; Krska, R. Occurrence of deoxynivalenol and its major conjugate, deoxynivalenol-3-glucoside, in beer and some brewing intermediates. J. Agric. Food Chem. 2009, 57, 3187–3194. [Google Scholar] [CrossRef] [PubMed]
- Poppenberger, B.; Berthiller, F.; Lucyshyn, D.; Sieberer, T.; Schuhmacher, R.; Krska, R.; Kuchler, K.; Glössl, J.; Luschnig, C.; Adam, G. Detoxification of the Fusarium mycotoxin deoxynivalenol by a UDP-glucosyltransferase from Arabidopsis thaliana. J. Biol. Chem. 2003, 278, 47905–47914. [Google Scholar] [CrossRef] [PubMed]
- Lemmens, M.; Scholz, U.; Berthiller, F.; Dall’Asta, C.; Koutnik, A.; Schuhmacher, R.; Adam, G.; Buerstmayr, H.; Mesterházy, A.; Krska, R.; et al. The ability to detoxify the mycotoxin deoxynivalenol colocalizes with a major quantitative trait locus for Fusarium head blight resistance in wheat. Mol. Plant Microbe Interact. 2005, 18, 1318–1324. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Sanghyun, S.; Heinen, S.; Dill-Macky, R.; Berthiller, F.; Nersesian, N.; Clemente, T.; McCormick, S.; Muehlbauer, G.J. Transgenic wheat expressing a barley UDP-glucosyltransferase detoxifies deoxynivalenol and provides high levels of resistance to Fusarium graminearum. Mol. Plant Microbe Interact. 2015, 28, 1237–1246. [Google Scholar] [CrossRef] [PubMed]
- De Nijs, M.; Van den Top, H.J.; Portier, L.; Oegema, G.; Kramer, E.; Van Egmond, H.P.; Hoogenboom, L.A.P. Digestibility and absorption of deoxynivalenol-3-ß-glucoside in in vitro models. World Mycotoxin J. 2012, 5, 319–324. [Google Scholar] [CrossRef]
- Berthiller, F.; Krska, R.; Domig, K.J.; Kneifel, W.; Juge, N.; Schuhmacher, R.; Adam, G. Hydrolytic fate of deoxynivalenol-3-glucoside during digestion. Toxicol. Lett. 2011, 206, 264–267. [Google Scholar] [CrossRef] [PubMed]
- Pierron, A.; Mimoun, S.; Murate, L.S.; Loiseau, N.; Lippi, Y.; Bracarense, A.P.; Liaubet, L.; Schatzmayr, G.; Berthiller, F.; Moll, W.D.; et al. Intestinal toxicity of the masked mycotoxin deoxynivalenol-3-β-d-glucoside. Arch. Toxicol. 2016, 90, 2037–2046. [Google Scholar] [CrossRef] [PubMed]
- Trombete, F.; Barros, A.; Vieira, M.; Saldanha, T.; Venâncio, A.; Fraga, M. Simultaneous Determination of Deoxynivalenol, Deoxynivalenol-3-Glucoside and Nivalenol in Wheat Grains by HPLC-PDA with Immunoaffinity Column Cleanup. Food Anal. Methods 2016, 9, 2579–2586. [Google Scholar] [CrossRef]
- Geng, Z.; Yang, D.; Zhou, M.; Zhang, P.; Wang, D.; Liu, F.; Zhu, Y.; Zhang, M. Determination of deoxynivalenol-3-glucoside in cereals by hydrophilic interaction chromatography with ultraviolet detection. Food Anal. Methods 2014, 7, 1139–1146. [Google Scholar] [CrossRef]
- Yoshinari, T.; Sakuda, S.; Furihata, K.; Furusawa, H.; Ohnishi, T.; Sugita-Konishi, Y.; Ishizaki, N.; Terajima, J. Structural determination of a nivalenol glucoside and development of an analytical method for the simultaneous determination of nivalenol and deoxynivalenol, and their glucosides, in wheat. J. Agric. Food Chem. 2014, 62, 1174–1180. [Google Scholar] [CrossRef] [PubMed]
- Dall’Asta, C.; Dall’Erta, A.; Mantovani, P.; Massi, A.; Galaverna, G. Occurrence of deoxynivalenol and deoxynivalenol-3-glucoside in durum wheat. World Mycotoxin J. 2012, 6, 83–91. [Google Scholar] [CrossRef]
- Dong, F.; Wang, S.; Yu, M.; Sun, Y.; Xu, J.; Shi, J. Natural occurrence of deoxynivalenol and deoxynivalenol-3-glucoside in various wheat cultivars grown in Jiangsu province, China. World Mycotoxin J. 2017, 10, 285–293. [Google Scholar] [CrossRef]
- Palacios, S.A.; Erazo, J.G.; Ciasca, B.; Lattanzio, V.M.; Reynoso, M.M.; Farnochi, M.C.; Torres, A.M. Occurrence of deoxynivalenol and deoxynivalenol-3-glucoside in durum wheat from Argentina. Food Chem. 2017, 230, 728–734. [Google Scholar] [CrossRef] [PubMed]
- Bryła, M.; Waśkiewicz, A.; Podolska, G.; Szymczyk, K.; Jędrzejczak, R.; Damaziak, K. Occurrence of 26 Mycotoxins in the Grain of Cereals Cultivated in Poland. Toxins 2016, 8, 160. [Google Scholar] [CrossRef] [PubMed]
- Calori-Domingues, M.A.; Bernardi, C.M.; Nardin, M.S.; de Souza, G.V.; Dos Santos, F.G.; Stein Mde, A.; Gloria, E.M.; Dias, C.T.; de Camargo, A.C. Co-occurrence and distribution of deoxynivalenol, nivalenol and zearalenone in wheat from Brazil. Food Addit. Contam. Part B Surveill. 2016, 9, 142–151. [Google Scholar] [CrossRef] [PubMed]
- De Boevre, M.; Landschoot, S.; Audenaert, K.; Maene, P.; Di Mavungu, D.; Eeckhout, M.; Haesaert, G.; De Saeger, S. Occurrence and within field variability of Fusarium mycotoxins and their masked forms in maize crops in Belgium. World Mycotoxin J. 2014, 7, 91–102. [Google Scholar] [CrossRef]
- Berthiller, F.; Dall’Asta, C.; Corradini, R.; Marchelli, R.; Sulyok, M.; Krska, R.; Adam, G.; Schuhmaher, R. Occurrence of deoxynivalenol and its 3-β-d-glucoside in wheat and maize. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2009, 26, 507–511. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, P.H.; Nielsen, K.F.; Ghorbani, F.; Spliid, N.H.; Nielsen, G.C.; Jørgensen, L.N. Occurrence of different trichothecenes and deoxynivalenol-3-β-d-glucoside in naturally and artificially contaminated Danish cereal grains and whole maize plants. Mycotoxin Res. 2012, 28, 181–190. [Google Scholar] [CrossRef] [PubMed]
| Compound | Fortification Level (n = 3) | |||||
|---|---|---|---|---|---|---|
| 225 µg/kg | 450 µg/kg | 900 µg/kg | ||||
| R% | RSD% | R% | RSD% | R% | RSD% | |
| NIV | 109.4 | 2.2 | 102.3 | 7.4 | 78.6 | 4.0 |
| DON | 91.8 | 2.0 | 95.0 | 5.0 | 76.9 | 3.4 |
| DON3G | 82.8 | 6.4 | 81.7 | 7.9 | 98.2 | 5.4 |
| Voivodeship | NIV | DON-3G | DON | Voivodeship | NIV | DON-3G | DON | ||
|---|---|---|---|---|---|---|---|---|---|
| Zachodnio-Pomorskie n = 7 | positive samples | 6 (86%) | 1 (14%) | 5 (71%) | Dolnośląskie n = 11 | positive samples | 8 (72%) | 3 (27%) | 10 (91%) |
| mean 1 | 37.8 | 43.3 | 86.5 | mean 1 | 31.1 | 35.5 | 137.0 | ||
| median 1 | 9.4 | 43.3 | 76.3 | median | 12.0 | 25.6 | 88.7 | ||
| Min-Max 1 | 5.2–133.5 | - 2 | 19.5–188.6 | Min-Max | 5.1–152.8 | 24.1–56.9 | 21.6–426.6 | ||
| Pomorskie n = 7 | positive samples | 6 (71%) | 2 (29%) | 7 (100%) | Opolskie n = 8 | positive samples | 5 (63%) | 2 (25%) | 6 (75%) |
| mean 1 | 21.1 | 42.9 | 134.1 | mean 1 | 112.4 | 26.0 | 106.0 | ||
| median | 14.3 | 42.9 | 137.6 | median | 18.8 | 26.0 | 45.7 | ||
| Min-Max | 10.2–52.0 | 39.0–46.9 | 11.7–359.2 | Min-Max | 14.3–372.5 | 23.6–28.4 | 12.7–261.0 | ||
| Warmińsko-Mazurskie n = 6 | positive samples | 3 (50%) | 5 (83%) | 6 (100%) | Łódzkie n = 4 | positive samples | 2 (50%) | - 2 | 4 (100%) |
| mean 1 | 71.8 | 50.8 | 432.0 | mean 1 | 9.2 | - 2 | 24.5 | ||
| median | 95.9 | 28.0 | 290.2 | median | 9.2 | - 2 | 12.6 | ||
| Min-Max | 7.0–112.4 | 16.4–137.5 | 71.3–1265.4 | Min-Max | 6.3–12.1 | - 2 | 10.5–62.4 | ||
| Lubuskie n = 3 | positive samples | 3 (100%) | 2 (67%) | 2 (67%) | Śląskie n = 3 | positive samples | 2 (67%) | 1 (33%) | 2 (67%) |
| mean 1 | 21.1 | 22.8 | 49.7 | mean 1 | 10.6 | 51.3 | 136.2 | ||
| median | 11.0 | 22.8 | 49.7 | median | 10.6 | 51.3 | 136.2 | ||
| Min-Max | 10.7–41.7 | 18.8–26.9 | 47.0–52.3 | Min-Max | 7.8–13.4 | - 2 | 42.2–230.1 | ||
| Wielkopolskie n = 10 | positive samples | 7 (70%) | 2 (20%) | 5 (50%) | Świętokrzyskie n = 3 | positive samples | 3 (100%) | - 2 | 3 (100%) |
| mean 1 | 48.1 | 73.1 | 97.9 | mean 1 | 18.3 | - 2 | 74.6 | ||
| median | 17.7 | 73.1 | 100.0 | median | 14.7 | - 2 | 79.9 | ||
| Min-Max | 9.7–150.8 | 50.3–95.9 | 15.0–198.8 | Min-Max | 12.4–27.7 | - 2 | 23.7–120.3 | ||
| Kujawsko-Pomorskie n = 9 | positive samples | 6 (67%) | 3 (33%) | 9 (100%) | Lubelskie n = 10 | positive samples | 4 (40%) | - 2 | 9 (90%) |
| mean 1 | 9.7 | 51.5 | 206.6 | mean 1 | 37.8 | - 2 | 75.0 | ||
| median | 9.3 | 35.1 | 148.9 | median | 38.2 | - 2 | 41.4 | ||
| Min-Max | 5.2–14.9 | 15.8–103.6 | 45.4–729.3 | Min-Max | 10.0–64.8 | - 2 | 27.6–179.5 | ||
| Mazowieckie n = 5 | positive samples | 5 (100%) | 1 (20%) | 3 (60%) | Podkarpackie n = 3 | positive samples | 3 (100%) | - 2 | 2 (67%) |
| mean 1 | 17.8 | 17.0 | 72.5 | mean 1 | 25.4 | - 2 | 40.2 | ||
| median | 18.6 | 17.0 | 73.8 | median | 27.2 | - 2 | 40.2 | ||
| Min-Max | 7.8–30.8 | - 2 | 23.8–119.9 | Min-Max | 6.8–42.2 | - 2 | 12.2–68.1 | ||
| Podlaskie n = 3 | positive samples | 2 (50%) | 3 (100%) | 3 (100%) | TOTAL n = 92 | positive samples | 64 (70%) | 25 (27%) | 76 (83%) |
| mean 1 | 6.7 | 30.1 | 223.1 | mean 1 | 35.0 | 41.9 | 140.2 | ||
| median | 6.7 | 33.3 | 196.4 | median | 13.6 | 33.3 | 83.2 | ||
| Min-Max | 10.2–52.0 | 39.0–46.9 | 11.7–359.2 | Min-Max | 5.1–372.5 | 15.8–137.5 | 10.5–1265.4 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bryła, M.; Ksieniewicz-Woźniak, E.; Waśkiewicz, A.; Szymczyk, K.; Jędrzejczak, R. Natural Occurrence of Nivalenol, Deoxynivalenol, and Deoxynivalenol-3-Glucoside in Polish Winter Wheat. Toxins 2018, 10, 81. https://doi.org/10.3390/toxins10020081
Bryła M, Ksieniewicz-Woźniak E, Waśkiewicz A, Szymczyk K, Jędrzejczak R. Natural Occurrence of Nivalenol, Deoxynivalenol, and Deoxynivalenol-3-Glucoside in Polish Winter Wheat. Toxins. 2018; 10(2):81. https://doi.org/10.3390/toxins10020081
Chicago/Turabian StyleBryła, Marcin, Edyta Ksieniewicz-Woźniak, Agnieszka Waśkiewicz, Krystyna Szymczyk, and Renata Jędrzejczak. 2018. "Natural Occurrence of Nivalenol, Deoxynivalenol, and Deoxynivalenol-3-Glucoside in Polish Winter Wheat" Toxins 10, no. 2: 81. https://doi.org/10.3390/toxins10020081
APA StyleBryła, M., Ksieniewicz-Woźniak, E., Waśkiewicz, A., Szymczyk, K., & Jędrzejczak, R. (2018). Natural Occurrence of Nivalenol, Deoxynivalenol, and Deoxynivalenol-3-Glucoside in Polish Winter Wheat. Toxins, 10(2), 81. https://doi.org/10.3390/toxins10020081





