Responses of Oat Grains to Fusarium poae and F. langsethiae Infections and Mycotoxin Contaminations
Abstract
1. Introduction
2. Results
2.1. Outcome of the FP and FL Inoculations
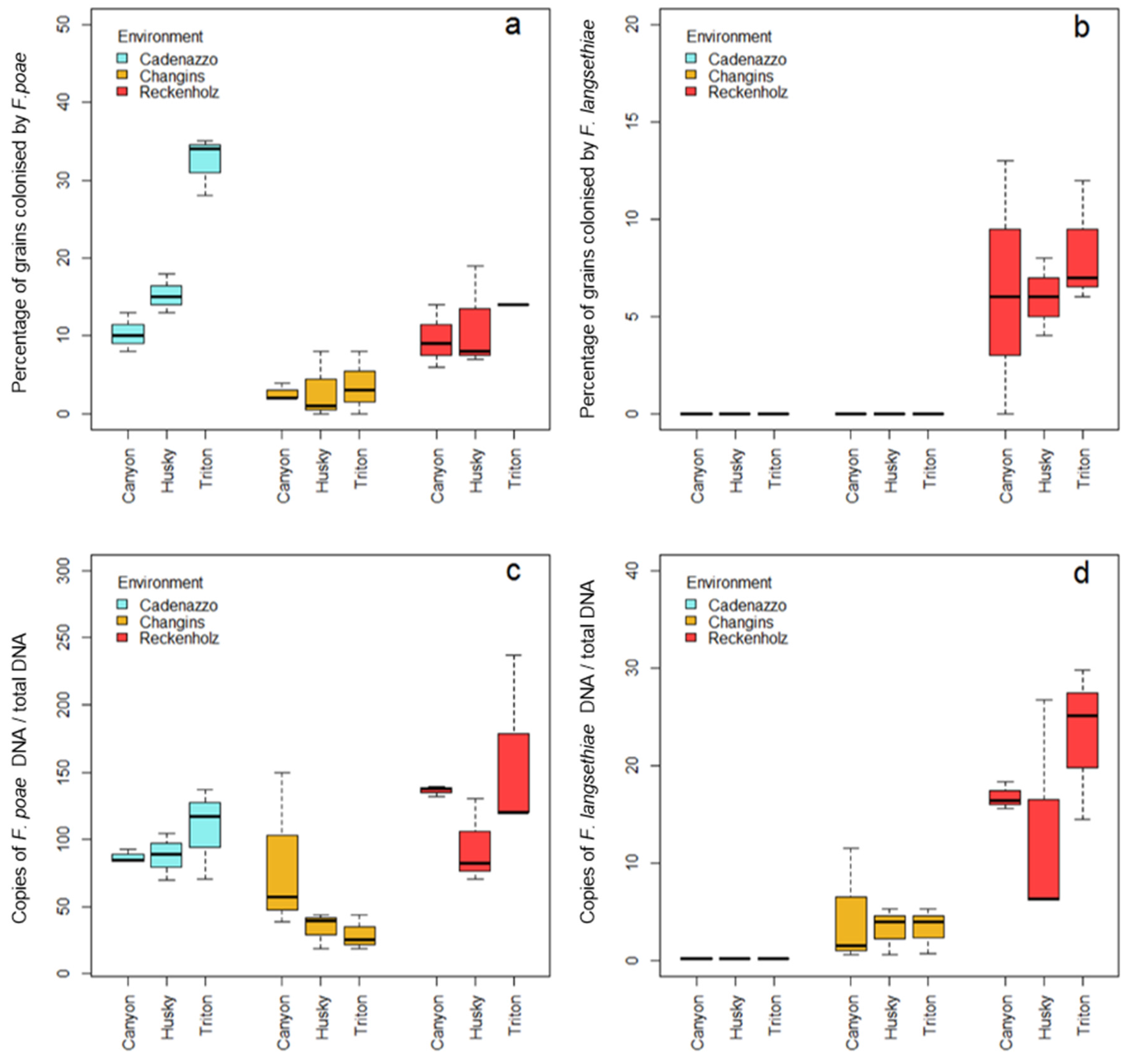
2.2. Incidence and Severity of Grain Infection with FP and FL
2.3. Incidence and Severity of Grain Infection with FP and FL
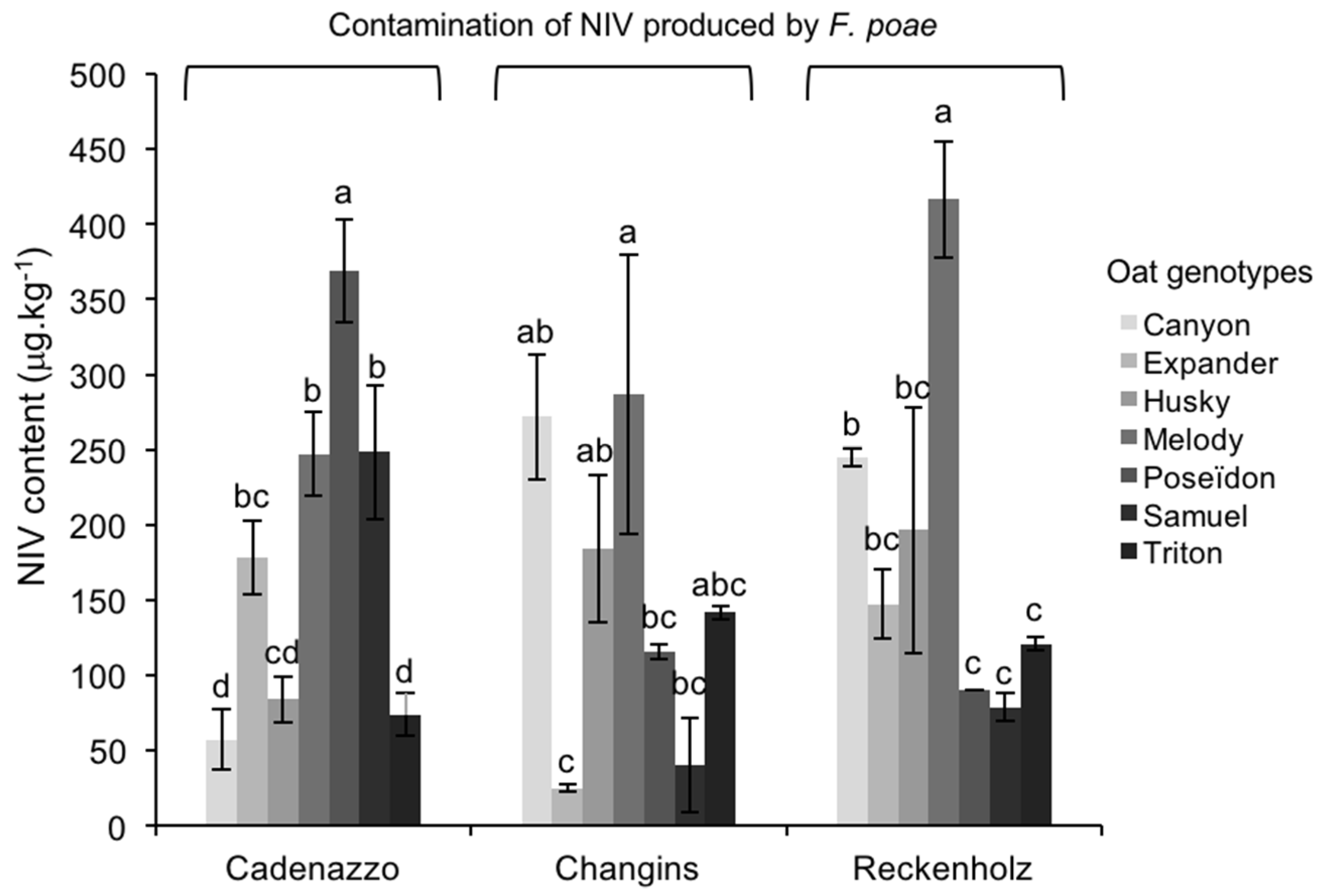
2.3.1. NIV Contamination Caused by FP
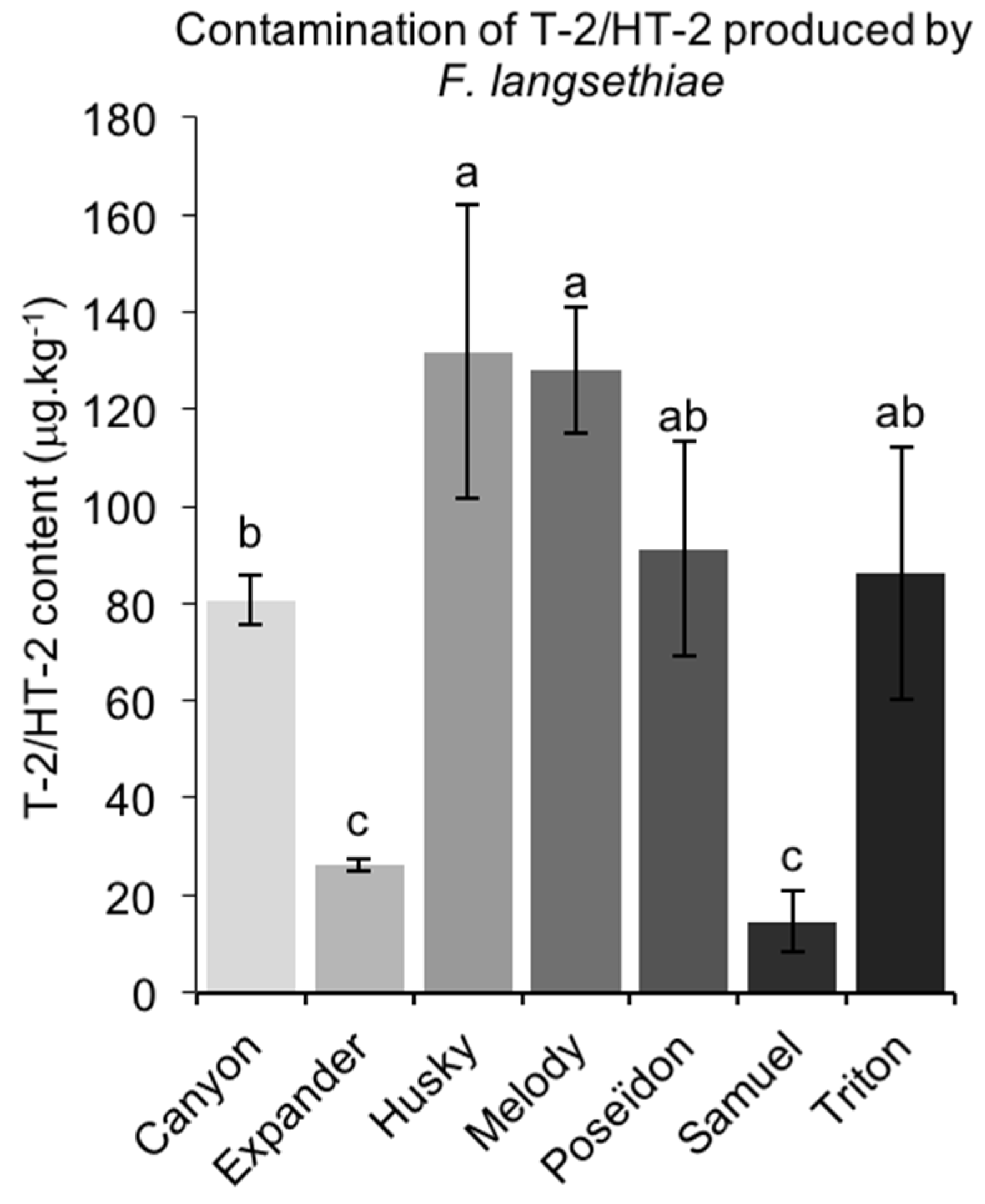
2.3.2. T-2/HT-2 Contamination Caused by FL
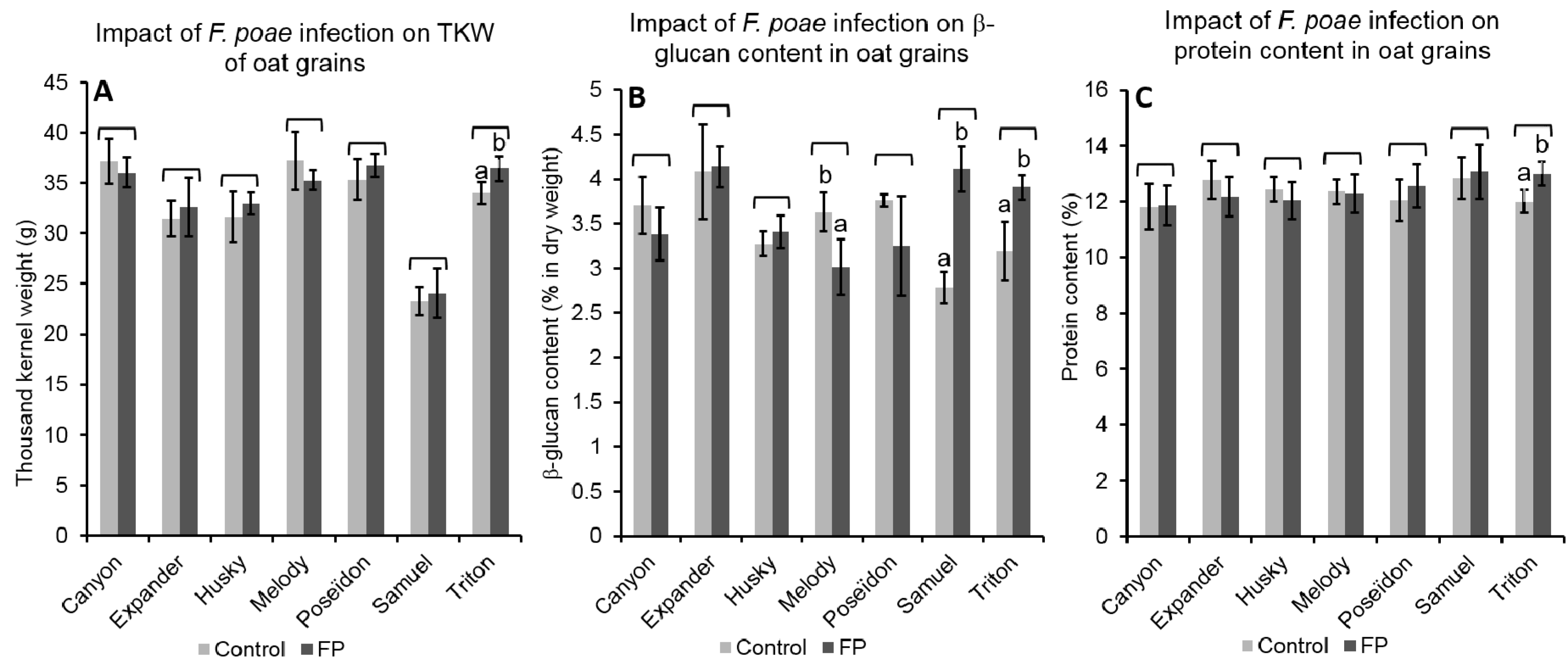
2.4. Impact of the Infection on Grain Properties
2.4.1. Effect of FP Infection on Grain Properties
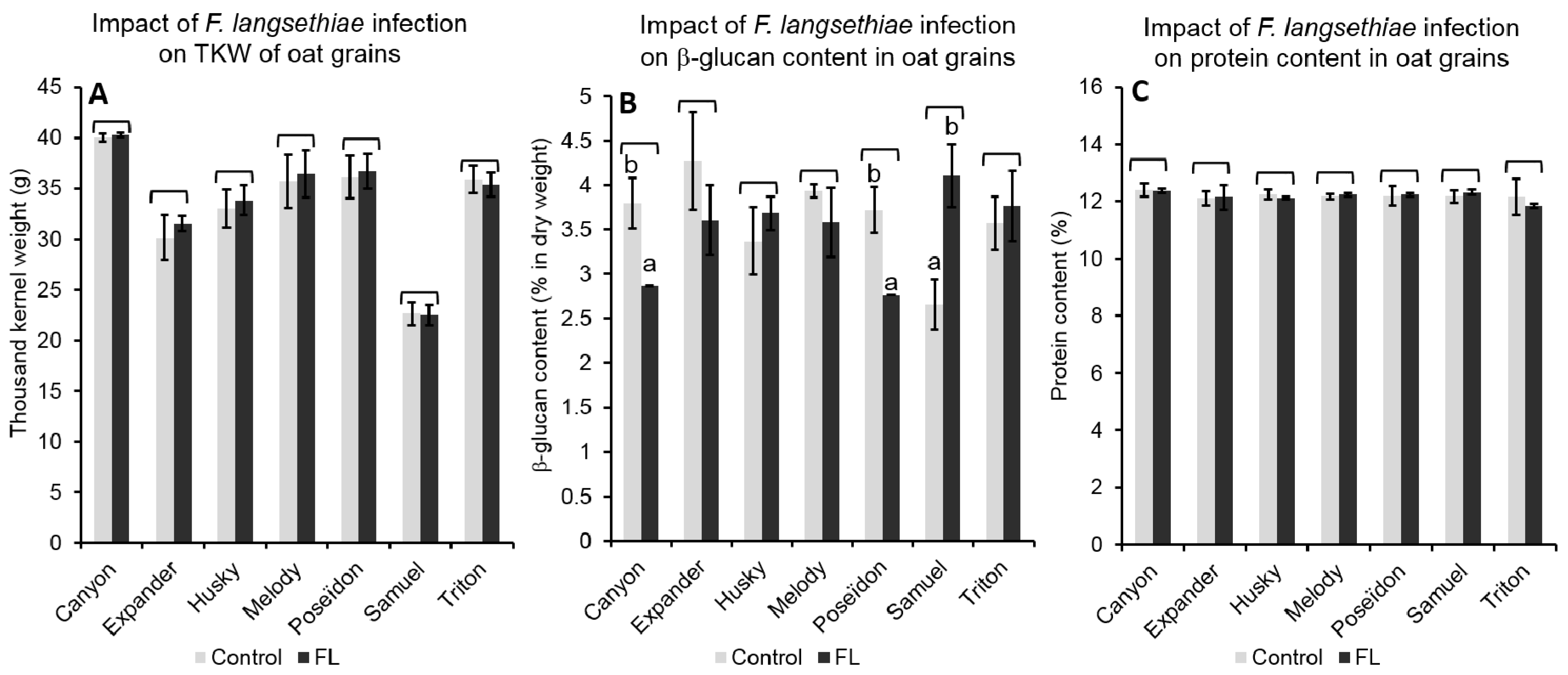
2.4.2. Effect of FL Infection on Grain Properties
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Fungal Material
4.3. Field Experiments and Artificial Inoculations
4.4. Harvest and Sample Collection
4.5. Analyses of Grain Infection
4.5.1. Proportion of Grains Infected by F. poae and F. langsethiae
4.5.2. Quantification of Fungal DNA
4.6. Mycotoxin Analysis
4.7. Analyses of Grain Properties
4.8. Experimental Design and Statistical Analyses
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Clear, R.M.; Patrick, S.K.; Platford, R.G.; Desjardins, M.; Clear, R.M. Occurrence and distribution of Fusarium species in barley and oat seed from Manitoba in 1993 and 1994. Can. Plant Pathol. 1996, 18, 409–414. [Google Scholar] [CrossRef]
- McCallum, B.; Tekauz, A.; Ames, N.; Fetch, J.M.; Campbell, D.; Platford, G.; Savard, M. Fusarium head blight in oat. In Proceedings of the Canadian Workshop on Fusarium Head Blight/Colloque Canadien Sur La Fusariose, Winnipeg, MB, Canada, 28 November 1999. [Google Scholar]
- Yan, W.; Fregeau-Reid, J.; Rioux, S.; Pageau, D.; Xue, A.; Martin, R.; Fedak, G.; Lajeunesse, J.; Savard, M. Response of oat genotypes to Fusarium Head Blight in Eastern Canada. Crop Sci. 2010, 50, 134–142. [Google Scholar] [CrossRef]
- Müller, H.M.; Reimann, J.; Schumacher, U.; Schwadorf, K. Natural occurrence of Fusarium toxins in oats harvested during five years in an area of Southwest Germany. Food Addit. Contam. Part A 1998, 15, 801–806. [Google Scholar] [CrossRef] [PubMed]
- Šliková, S.; Šrobárová, A.; Šudyová, V.; Polišenská, I.; Gregová, E.; Miháli, D. Response of oat cultivars to Fusarium infection with a view to their suitability for food use. Biologia 2010, 65, 609–614. [Google Scholar] [CrossRef]
- Bjørnstad, Å.; Skinnes, H. Resistance to Fusarium infection in oats (Avena Sativa L.). Cereal Res. Commun. 2008, 36 (Suppl. 6), 57–62. [Google Scholar] [CrossRef]
- Paavanen-Huhtala, S.; Jestoi, M.; Parikka, P.; Hietaniemi, V.; Gagkaeva, T.; Sarlin, T.; Haikara, A.; Laaksonen, S.; Rizzo, A.; Yli-Mattila, T. Real-time PCR detection and quantification of Fusarium poae, F. graminearum, F. sporotrichioides and F. langsethiae in cereal grains in Finland and Russia. Arch. Phytopathol. Plant Prot. 2007, 41, 243–260. [Google Scholar]
- Fredlund, E.; Gidlund, A.; Sulyok, M.; Börjesson, T.; Kriska, R.; Olsen, M.; Lindblad, M. Deoxynivalenol and other selected Fusarium toxins in Swedish oats—Occurrence and correlation to specific Fusarium species. Int. J. Food Microbiol. 2013, 167, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Bernhoft, A.; Torp, M.; Clasen, P.; Løes, A.; Kristoffersen, A.B. Influence of agronomic and climatic factors on Fusarium infestation and mycotoxin contamination of cereals in Norway. Food Addit. Contam. Part A 2012, 29, 1129–1140. [Google Scholar] [CrossRef] [PubMed]
- Edwards, S.G.; Barrier-Guillot, P.; Clasen, P.E.; Hietaniemi, V.; Petterson, H. Emerging Issues of HT-2 and T-2 Toxins in European cereal production. World Mycotoxin J. 2009, 2, 173–179. [Google Scholar] [CrossRef]
- Scudamore, K.; Baillie, H.; Patel, S.; Edwards, S.G. The occurrence and fate of Fusarium mycotoxins during the commercial processing of oats in the UK. Food Addit. Contam. Part A 2007, 24, 1374–1385. [Google Scholar] [CrossRef] [PubMed]
- Van der Fels-Klerx, H.; Stratakou, I. T-2 toxin and HT-2 toxin in grain and grain-based commodities in Europe: Occurrence, factors affecting occurrence, co-occurrence and toxicological effects. World Mycotoxin J. 2010, 3, 349–367. [Google Scholar] [CrossRef]
- Schöneberg, T.; Jenny, E.; Wettstein, F.E.; Bucheli, T.D.; Mascher, F.; Bertossa, M.; Vogelgsang, S. Occurrence of Fusarium species and mycotoxins in Swiss oats—Impact of cropping factors. Eur. J. Agron. 2018, 92, 123–132. [Google Scholar] [CrossRef]
- European Food Safety Authority. Scientific Opinion on risks for animal and public health related to the presence of nivalenol in food and feed. EFSA J. 2013, 11, 3262. [Google Scholar]
- Pitt, J. Mycotoxins—Deoxynivalenol and other trichothecenes. In Encyclopedia of Food Safety; Motarjemi, Y., Moy, G., Todd, E., Eds.; Academic Press: New York, NY, USA, 2014; Volume 2, pp. 295–299. [Google Scholar]
- Yagen, B.; Joffe, Z. Screening of toxic isolates of Fusarium poae and Fusarium sporotrichioides involved in causing alimentary toxic. Appl. Environ. Microbiol. 1976, 3, 423–427. [Google Scholar]
- European Commission, Commission Recommendation of 27 March 2013 on the Presence of T-2 and HT-2 Toxin in Cereals and Cereal Products 2013/165/EC. 2013. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2013:091:0012:0015:EN:PDF (accessed on 20 December 2017).
- Tekauz, A.; Mitchell Fetch, J.; Rossnagel, B.G.; Savard, M. Progress in assessing the impact of Fusarium head blight on oat in western Canada and screening of avena germplasm for resistance. Cereal Res. Commun. 2008, 36 (Suppl. 6), 49–56. [Google Scholar] [CrossRef]
- Tekauz, A.; McCallum, B.; Ames, N.; Mitchell Fetch, J. Fusarium Head Blight of Oat—Current Status in Western Canada. Can. Plant Pathol. 2004, 479, 473–479. [Google Scholar]
- Langevin, F.; Eudes, F.; Comeau, A. Effect of trichothecenes produced by Fusarium graminearum during Fusarium head blight development in six cereal species’. Eur. J. Plant Pathol. 2004, 110, 735–746. [Google Scholar] [CrossRef]
- Xue, A.G.; Yuanhong, C.; Marchand, G.; Guo, W.; Ren, C.; Savard, M.; McElroy, A.R.B. Timing of inoculation and Fusarium species affect the severity of Fusarium head blight on oat’. Can. J. Plant Sci. 2014, 95, 517–524. [Google Scholar] [CrossRef]
- Kibler, K. Epidemiology of Fusarium Langsethiae in Oats and the Possible role of Health Promoting Compounds in Control of Fusarium Head Blight. MSc Thesis, University of Hohenheim, Stuttgart, Germany, 2016. [Google Scholar]
- Divon, H.H.; Razzaghian, J.; Udnes-Aamot, H.; Klemsdal, S.S. Fusarium langsethiae (Torp and Nirenberg), investigation of alternative infection routes in oats. Eur. J. Plant Pathol. 2012, 132, 147–161. [Google Scholar] [CrossRef]
- Tekle, S.; Dill-Macky, R.; Skinnes, H.; Tronsmo, A.M.; Bjørnstad, Å. Infection process of Fusarium graminearum in Oats (Avena Sativa L.). Eur. J. Plant Pathol. 2012, 132, 431–442. [Google Scholar] [CrossRef]
- Mielniczuk, E.; Kiecana, I.; Perkowski, J. Susceptibility of oat genotypes to Fusarium crookwellense Burgess, Nelson and Toussoun infection and mycotoxin accumulation in kernels. Biologia 2004, 59, 809–816. [Google Scholar]
- Bjørnstad, Å.; He, X.; Tekle, S.; Klos, K.; Huang, H.F.; Tinker, N.A.; Helge, D. Genetic variation and associations involving Fusarium head blight and deoxynivalenol accumulation in cultivated oat (Avena Sativa L.). Plant Breed. 2017, 136, 1–17. [Google Scholar] [CrossRef]
- He, X.; Skinnes, H.; Oliver, R.E.; Jackson, E.W.; Bjørnstad, Å. Linkage Mapping and identification of QTL affecting deoxynivalenol (DON) content (Fusarium Resistance) in oats (Avena Sativa L.). Theor. Appl. Genet. 2013, 126, 2655–2670. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.D.; Young, J.C.; Sampson, D.R. Deoxynivalenol and Fusarium head blight resistance in spring cereals. J. Phytopathol. 1985, 113, 359–367. [Google Scholar] [CrossRef]
- Gagkaeva, T.; Gavrilova, O.P.; Yli-Mattila, T.; Loskutov, I.G. Sources of resistance to Fusarium head blight in VIR Oat Collection. Euphytica 2013, 195, 355–364. [Google Scholar] [CrossRef]
- Mesterházy, A. Types and components of resistance to Fusarium head blight of wheat. Plant Breed. 1995, 114, 377–386. [Google Scholar] [CrossRef]
- Schroeder, H.W.; Christensen, J.J. Factors affecting resistance of wheat to scab caused by Gibberella zeae. Phytopathology 1963, 53, 831–838. [Google Scholar]
- Vogelgsang, S.; Sulyok, M.; Hecker, A.; Jenny, E.; Kriska, R.; Schuhmacher, R.; Forrer, H.R. Toxigenicity and pathogenicity of Fusarium poae and Fusarium avenaceum on wheat. Eur. J. Plant Pathol. 2008, 122, 265–276. [Google Scholar] [CrossRef]
- Federal Office of Meteorology and Climatology. Available online: http://www.meteoswiss.admin.ch (accessed on 20 December 2107).
- Torp, M.; Nirenberg, H.I. Fusarium langsethiae Sp. Nov. on Cereals in Europe. Int. J. Food Microbiol. 2004, 95, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Imathiu, S.; Edwards, S.G.; Ray, R.V.; Back, M.A. Fusarium langsethiae—A HT-2 and T-2 Toxins Producer that Needs More Attention. J. Phytopathol. 2012, 161, 1–10. [Google Scholar] [CrossRef]
- Gagkaeva, T.; Gavrilova, O.P.; Yli-Mattila, T.; Loskutov, I.G. Evaluation of oat germplasm for resistance to Fusarium head blight. Plant Breed. Seed Sci. 2011, 64, 15–22. [Google Scholar] [CrossRef]
- Tekle, S.; Skinnes, H.; Bjørnstad, Å. The germination problem of oat seed lots affected by Fusarium head blight. Eur. J. Plant Pathol. 2013, 135, 147–158. [Google Scholar] [CrossRef]
- Hope, R.; Aldred, D.; Magan, N. Comparison of environmental profiles for growth and deoxynivalenol production by Fusarium culmorum and F. graminearum on wheat grain. Lett. Appl. Microbiol. 2005, 40, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Medina, A.; Magan, N. Comparisons of water activity and temperature impacts on growth of Fusarium langsethiae strains from Northern Europe on oat-based Media’. Int. J. Food Microbiol. 2010, 142, 365–369. [Google Scholar] [CrossRef] [PubMed]
- Meng-Reiterer, J.; Bueschl, C.; Rechthaler, J.; Berthiller, F.; Lemmens, M.; Schuhmacher, R. Metabolism of HT-2 Toxin and T-2 Toxin in Oats. Toxins 2016, 8, 364. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.; Schöneberg, T.; Vogelgsang, S.; Morisoli, R.; Bertossa, M.; Mauch-Mani, B.; Mascher, F. Resistance against Fusarium graminearum and the relationship to β-glucan in barley grains. Eur. J. Plant Pathol. under review.
- Schwarz, P.B.; Jones, B.L.; Steffenson, B.J. Enzymes associated with Fusarium infection of barley. J. Am. Soc. Brew. Chem. 2002, 60, 130–134. [Google Scholar]
- Miller, S.S.; Fulcher, R.G.; Sen, A.; Arnason, J.T. Oat endosperm cell walls. Isolation, composition, and comparison with other tissues. Cereal Chem. 1995, 72, 421–427. [Google Scholar]
- Martinelli, J.A.; Chaves, M.S.; Graichen, F.A.S.; Federizzi, L.C.; Dresch, L.F. Impact of Fusarium head blight in reducing the weight of oat grains. J. Agric. Sci. 2014, 6, 188–198. [Google Scholar] [CrossRef]
- Gavrilova, O.P.; Gagkaeva, T.; Burkin, A.; Kononenko, G.; Loskutov, I.G. Susceptibility of oat germplasm to Fusarium infection and mycotoxin accumulation in grains. In Proceedings of the 8th International Oat Conference, Minneapolis, MN, USA, 28 June–2 July 2008; pp. 7–16. [Google Scholar]
- Loskutov, I.G.; Blinova, E.; Gavrilova, O.P.; Gagkaeva, T. The valuable characteristics and resistance to Fusarium disease of oat genotypes. Russ. J. Genet. 2017, 7, 290–298. [Google Scholar] [CrossRef]
- IBPGR. Oat descriptors. In International Board for Plant Genetic Resources; IBPGR: Rome, Italy, 1985. [Google Scholar]
- Centraal Bureau voor Schimmelcultures CBS. Available online: http://www.cbs.knaw.nl/fusarium/ (accessed on 20 December 2017).
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual; Blackwell Publishing: Oxford, UK, 2006; ISBN 13 978-0-8138-1919-8. [Google Scholar]
- Edwards, S.G.; Imathiu, S.M.; Ray, R.V.; Back, M.; Hare, M.C. Molecular studies to identify the Fusarium species responsible for HT-2 and T-2 mycotoxins in UK oats. Int. J. Food Microbiol. 2012, 156, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.; Simpson, D.; Chandler, E.; Jennings, P.; Nicholson, P. Development of PCR assays for the detection and differentiation of Fusarium sporotrichioides and Fusarium langsethiae. FEMS Microbiol. Lett. 2004, 233, 69–76. [Google Scholar] [CrossRef] [PubMed]
- International Association for Cereal Science and Technology (ICC). ICC Standard Method No. 166. Determination of ß-Glucan in Barley, Oat and Rye. 1998. Available online: https://www.icc.or.at/standard_methods/166 (accessed on 20 December 2017).
- International Association for Cereal Science and Technology (ICC). ICC Standard Method No. 105/2. Determination of Crude Protein in Cereals and Cereal Products for Food and for Feed. 1980. Available online: https://www.icc.or.at/standard_methods/105.2 (accessed on 20 December 2017).
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, 2013. Available online: http://www.R-project.org/ (accessed on 20 December 2017).
- De Mendiburu, F. Agricolae: Statistical Procedures for Agricultural Research, Version 1.2-4. 2015. Available online: http://tarwi.lamolina.edu.pe/~fmendiburu (accessed on 20 December 2017).
| Temperature (°C) | Precipitation (mm) | Relative Humidity (%) | Evapotranspiration (mm/day) | |
|---|---|---|---|---|
| from 15 June 2015 to 30 June 2015 | ||||
| Cadenazzo | 20.6 | 32.1 | 66.8 | 3.5 |
| Changins | 19.1 | 21.4 | 58.9 | 4.4 |
| Reckenholz | 17.0 | 61.4 | 71.8 | 2.8 |
| from 15 June 2015 to 10 August 2015 | ||||
| Cadenazzo | 23.4 | 129.7 | 67.8 | 3.1 |
| Changins | 22.1 | 95.4 | 56.3 | 4.8 |
| Reckenholz | 20.9 | 114.7 | 65.5 | 3.7 |
| Source of Variation | Sum Square | Mean Square | Significance |
|---|---|---|---|
| Environment | 13,410 | 6705 | |
| Error (a) | 2137 | 534 | |
| Genotype | 188,457 | 31,409 | *** |
| Environment x Genotype | 344,011 | 28,668 | *** |
| Error (b) | 225,612 | 6267 |
| TKW | β-Glucan Content | Protein Content | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Source of Variation | Sum Square | Mean Square | Significance | Sum Square | Mean Square | Significance | Sum Square | Mean Square | Significance |
| Environment E | 4.6 | 2.3 | 0.5 | 0.3 | 6.4 | 3.2 | * | ||
| Error (a) | 25.6 | 6.4 | 0.2 | 0.04 | 1.9 | 0.4 | |||
| Inoculation I | 0.5 | 0.5 | 0.4 | 0.4 | 0.3 | 0.3 | |||
| ExI | 97.8 | 48.9 | *** | 2.3 | 1.4 | *** | 3.6 | 1.8 | * |
| Error (b) | 9.7 | 1.6 | 1.5 | 0.25 | 1.5 | 0.2 | |||
| Genotype G | 2339.3 | 390 | *** | 7.6 | 1.3 | *** | 12.1 | 2.0 | *** |
| ExG | 248.4 | 20.7 | *** | 6.9 | 0.6 | ** | 20.0 | 1.7 | *** |
| IxG | 45.5 | 7.6 | ** | 13.2 | 2.2 | *** | 7.5 | 1.3 | *** |
| ExIx G | 95.7 | 8.0 | *** | 2.7 | 0.2 | 10.3 | 0.9 | *** | |
| Error (c) | 166.3 | 2.3 | 16.9 | 0.2 | 9.3 | 0.1 | |||
| Variety | Breeder | Plant Height (cm) | Panicle Shape | Panicle Erectness | Number of Seeds in a Panicle | Kernel Covering | Lemma Colour | Hairiness of Lemma | Hairiness at Basal Part of the Primary Grain |
|---|---|---|---|---|---|---|---|---|---|
| Canyon | NORDSAAT Saatzucht (DE) | 110 | Equilateral | Drooping | 71 | Covered | Yellow | Glabrous | Slightly pubescent |
| Expander | SZ Edelhof (DE) | 105 | Equilateral | Drooping | 96 | Covered | Yellow | Glabrous | Highly pubescent |
| Husky | NORDSAAT Saatzucht (DE) | 110 | Equilateral | Drooping | 90 | Covered | Yellow | Slightly pubescent | Moderately pubescent |
| Melody | NORDSAAT Saatzucht (DE) | 110 | Unilateral | Drooping | 69 | Covered | White | Slightly pubescent | Moderately pubescent |
| Poseidon | NORDSAAT Saatzucht (DE) | 115 | Equilateral | Drooping | 85 | Covered | Yellow | Slightly pubescent | Moderately pubescent |
| Samuel | Nufarm Deutschland GmbH (DE) | 120 | Equilateral | Drooping | 65 | Naked | / | / | Slightly pubescent |
| Triton | NORDSAAT Saatzucht (DE) | 105 | Equilateral | Semi-erected | 62 | Covered | White | Highly pubescent | Moderately pubescent |
| Species | Strain ID | Origin (Swiss Canton) | Isolation Year | Toxin Produced |
|---|---|---|---|---|
| F. poae | 13013 | Luzern | 2013 | NIV |
| F. poae | 13045 | Thurgau | 2013 | NIV |
| F. poae | 13059 | Vaud | 2013 | NIV |
| F. langsethiae | 13014 | Jura | 2013 | T-2/HT-2 |
| F. langsethiae | 13005 | Schaffhausen | 2013 | T-2/HT-2 |
| F. langsethiae | 14001 | Vaud | 2014 | T-2/HT-2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martin, C.; Schöneberg, T.; Vogelgsang, S.; Mendes Ferreira, C.S.; Morisoli, R.; Bertossa, M.; Bucheli, T.D.; Mauch-Mani, B.; Mascher, F. Responses of Oat Grains to Fusarium poae and F. langsethiae Infections and Mycotoxin Contaminations. Toxins 2018, 10, 47. https://doi.org/10.3390/toxins10010047
Martin C, Schöneberg T, Vogelgsang S, Mendes Ferreira CS, Morisoli R, Bertossa M, Bucheli TD, Mauch-Mani B, Mascher F. Responses of Oat Grains to Fusarium poae and F. langsethiae Infections and Mycotoxin Contaminations. Toxins. 2018; 10(1):47. https://doi.org/10.3390/toxins10010047
Chicago/Turabian StyleMartin, Charlotte, Torsten Schöneberg, Susanne Vogelgsang, Carla Susana Mendes Ferreira, Romina Morisoli, Mario Bertossa, Thomas D. Bucheli, Brigitte Mauch-Mani, and Fabio Mascher. 2018. "Responses of Oat Grains to Fusarium poae and F. langsethiae Infections and Mycotoxin Contaminations" Toxins 10, no. 1: 47. https://doi.org/10.3390/toxins10010047
APA StyleMartin, C., Schöneberg, T., Vogelgsang, S., Mendes Ferreira, C. S., Morisoli, R., Bertossa, M., Bucheli, T. D., Mauch-Mani, B., & Mascher, F. (2018). Responses of Oat Grains to Fusarium poae and F. langsethiae Infections and Mycotoxin Contaminations. Toxins, 10(1), 47. https://doi.org/10.3390/toxins10010047






