Chemopreventive Potential of Ethanolic Extracts of Luobuma Leaves (Apocynum venetum L.) in Androgen Insensitive Prostate Cancer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
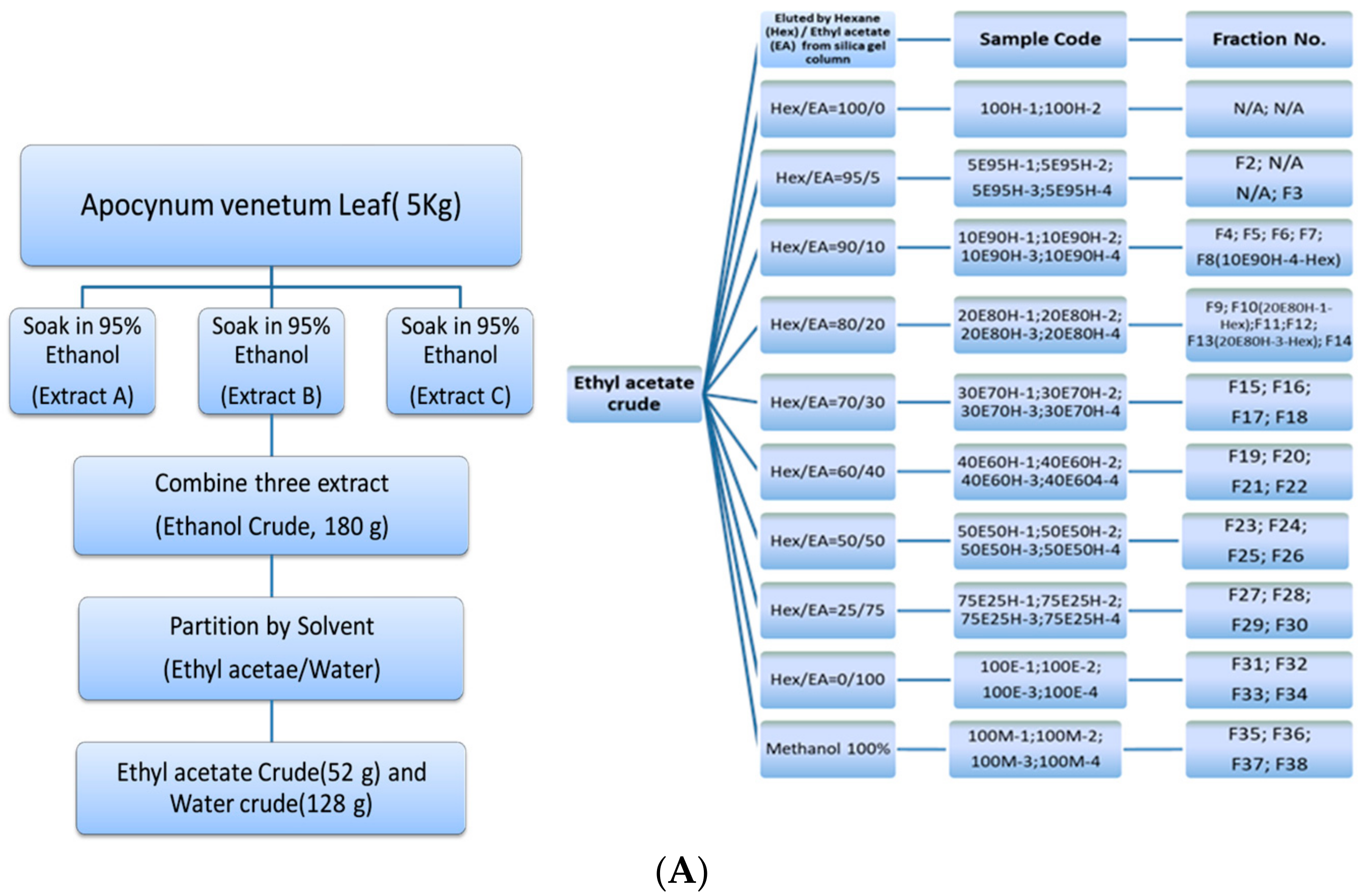
2.2. Extraction
2.3. LC-MS Analysis
2.4. NMR Analysis
2.5. UPLC Analysis
2.6. The Total Phenolic, Flavonoid, Triterpene Contents and Antioxidant Properties
2.7. Cell Culture and Proliferation Assay
2.8. Cell Cycle Analysis
2.9. Assay of Caspase Activity
2.10. Western Blot Analysis
2.11. Statistical Analysis
3. Results
3.1. Extraction, Fractionation, and Cytotoxicity Induced by AVL Ethanolic Extracts
3.2. Identification and Analysis of Bioactive Compounds
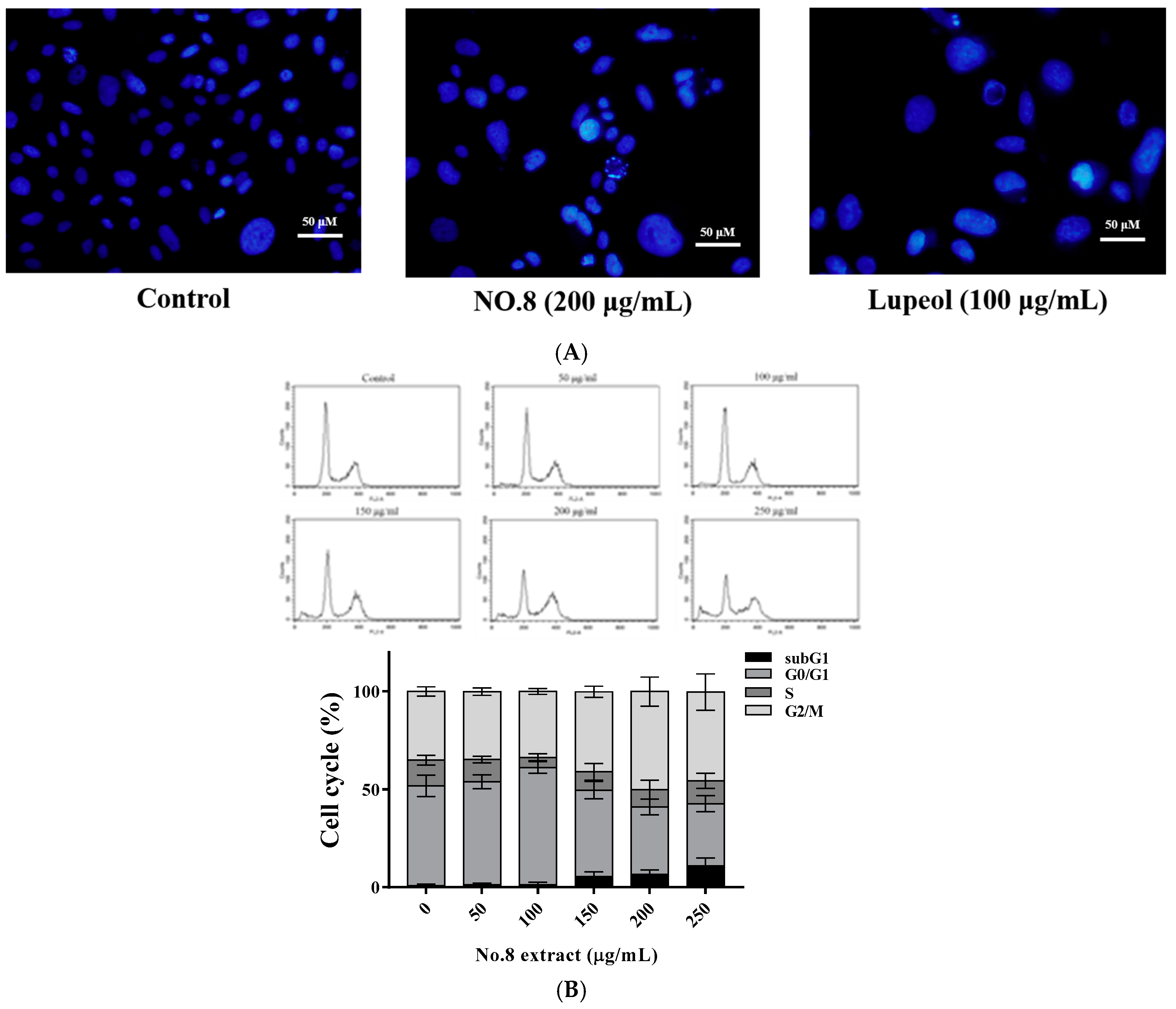
3.3. AVL Fraction No. 8 Inhibited Cell Proliferation by G2/M Arrest in PC3 Cells
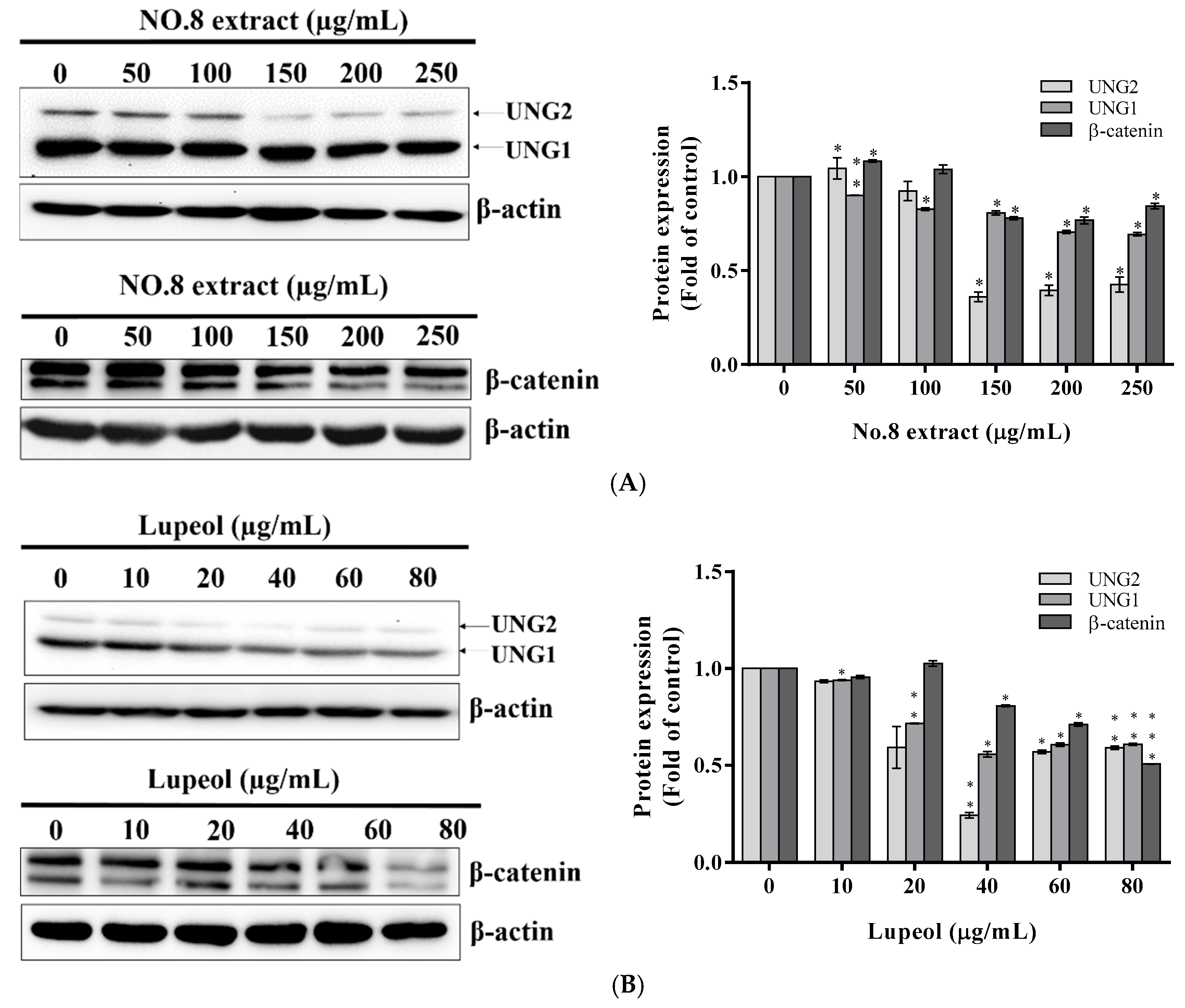
3.4. AVL F8 Impaired DNA Repair System through Downregulation of the Expression of Uracil-DNA Glycosylase Leading to the Promotion of G2/M Arrest
3.5. Reduced β-Catenin Expression Involved in AVL F8-Induced G2/M Arrest
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xie, W.Y.; Zhang, X.Y.; Wang, T.; Hu, J.J. Botany, traditional uses, phytochemistry and pharmacology of Apocynum venetum L. (Luobuma): A review. J. Ethnopharmacol. 2012, 141, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Irie, K.; Sato, T.; Tanaka, I.; Nakajima, J.; Kawaguchi, M.; Himi, T. Cardiotonic effect of Apocynum venetum L. Extracts on isolated guinea pig atrium. J. Nat. Med. 2009, 63, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.N.; Song, L.H.; Gu, Z.L.; Chen, B.Q.; Zhang, K.P.; Li, H.Z.; Peng, Y.K. An experimental observation on the diuretic effect of an extract of Luobuma (Apocynum venetum) leaves. Zhong Yao Tong Bao 1988, 13, 44–46. [Google Scholar] [PubMed]
- Kamata, K.; Seo, S.; Nakajima, J.I. Constituents from leaves of Apocynum venetum L. J. Nat. Med. 2008, 62, 160–163. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.W.; Yokozawa, T.; Hattori, M.; Kadota, S.; Namba, T. Effects of aqueous extracts of Apocynum venetum leaves on spontaneously hypertensive, renal hypertensive and NaCl-fed-hypertensive rats. J. Ethnopharmacol. 2000, 72, 53–59. [Google Scholar] [CrossRef]
- Stoner, G.D.; Mukhtar, H. Polyphenols as cancer chemopreventive agents. J. Cell. Biochem. 1995, 59, 169–180. [Google Scholar] [CrossRef]
- Holund, B. Latent prostatic cancer in a consecutive autopsy series. Scand. J. Urol. Nephrol. 1980, 14, 29–35. [Google Scholar] [CrossRef]
- Feldman, B.J.; Feldman, D. The development of androgen-independent prostate cancer. Nat. Rev. Cancer 2001, 1, 34–45. [Google Scholar] [CrossRef]
- Parimi, V.; Goyal, R.; Poropatich, K.; Yang, X.J. Neuroendocrine differentiation of prostate cancer: A review. Am. J. Clin. Exp. Urol. 2014, 2, 273–285. [Google Scholar] [PubMed]
- Behrens, J.; Jerchow, B.A.; Wurtele, M.; Grimm, J.; Asbrand, C.; Wirtz, R.; Kuhl, M.; Wedlich, D.; Birchmeier, W. Functional interaction of an axin homolog, conductin, with beta-catenin, APC, and GSK3 beta. Science 1998, 280, 596–599. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.M.; Kato, Y.; Zhang, Z.H.; Do, V.M.; Yankner, B.A.; He, X. Beta-Trcp couples beta-catenin phosphorylation-degradation and regulates Xenopus axis formation. Proc. Natl. Acad. Sci. USA 1999, 96, 6273–6278. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.M.; Ono, K.; Satoh, S.; Ishiguro, H.; Fujita, M.; Miwa, N.; Tanaka, T.; Tsunoda, T.; Yang, K.C.; Nakamura, Y.; et al. Identification of AF17 as a downstream gene of the beta-catenin/T-cell factor pathway and its involvement in colorectal carcinogenesis. Cancer Res. 2001, 61, 6345–6349. [Google Scholar] [PubMed]
- Saleem, M.; Murtaza, I.; Tarapore, R.S.; Suh, Y.; Adhami, V.M.; Johnson, J.J.; Siddiqui, I.A.; Khan, N.; Asim, M.; Bin Hafeez, B.; et al. Lupeol inhibits proliferation of human prostate cancer cells by targeting beta-catenin signaling. Carcinogenesis 2009, 30, 808–817. [Google Scholar] [CrossRef] [PubMed]
- Pulukuri, S.M.K.; Knost, J.A.; Estes, N.; Rao, J.S. Small interfering RNA-directed knockdown of uracil DNA glycosylase induces apoptosis and sensitizes human prostate cancer cells to genotoxic stress. Mol. Cancer Res. 2009, 7, 1285–1293. [Google Scholar] [CrossRef] [PubMed]
- Krokan, H.E.; Drablos, F.; Slupphaug, G. Uracil in DNA—Occurrence, consequences and repair. Oncogene 2002, 21, 8935–8948. [Google Scholar] [CrossRef]
- Wu, L.C.; Hsu, H.W.; Chen, Y.C.; Chiu, C.C.; Lin, Y.I.; Ho, J.A.A. Antioxidant and antiproliferative activities of red pitaya. Food Chem. 2006, 95, 319–327. [Google Scholar] [CrossRef]
- Liu, M.; Li, X.Q.; Weber, C.; Lee, C.Y.; Brown, J.; Liu, R.H. Antioxidant and anti proliferative activities of raspberries. J. Agric. Food Chem. 2002, 50, 2926–2930. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhu, L.; Tan, J.; Zhou, X.; Xiao, L.; Yang, X.; Wang, B. Glucosidase inhibitory activity and antioxidant activity of flavonoid compound and triterpenoid compound from Agrimonia Pilosa Ledeb. BMC Complement. Altern. Med. 2014, 14, 12. [Google Scholar] [CrossRef] [PubMed]
- Prakash, C.V.S.; Prakash, I. Isolation and structural characterization of lupane triterpenes from polypodium vulgare. Res. J. Pharm. Sci. 2012, 1, 23–27. [Google Scholar]
- Saleem, M.; Kweon, M.H.; Yun, J.M.; Adhami, V.M.; Khan, N.; Syed, D.N.; Mukhtar, H. A novel dietary triterpene Lupeol induces fas-mediated apoptotic death of androgen-sensitive prostate cancer cells and inhibits tumor growth in a xenograft model. Cancer Res. 2005, 65, 11203–11213. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Nigam, N.; Kalra, N.; Shukla, Y. Regulation of signaling pathways involved in Lupeol induced inhibition of proliferation and induction of apoptosis in human prostate cancer cells. Mol. Carcinog. 2008, 47, 916–924. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, S.; Mandal, C.; Sangwan, R.; Mandal, C. Coupling G2/M arrest to the Wnt/beta-catenin pathway restrains pancreatic adenocarcinoma. Endocr. Relat. Cancer 2014, 21, 113–125. [Google Scholar] [CrossRef]
- Olmeda, D.; Castel, S.; Vilaro, S.; Cano, A. Beta-catenin regulation during the cell cycle: Implications in G2/M and apoptosis. Mol. Biol. Cell 2003, 14, 2844–2860. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Kalra, N.; Shukla, Y. Induction of apoptosis by Lupeol and mango extract in mouse prostate and LNCap cells. Nutr. Cancer 2008, 60, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, H.L.; Staerk, D.; Christensen, J.; Hviid, L.; Hagerstrand, H.; Jaroszewski, J.W. In vitro Plasmodium falciparum drug sensitivity assay: Inhibition of parasite growth by incorporation of stomatocytogenic amphiphiles into the erythrocyte membrane. Antimicrob. Agents Chemother. 2002, 46, 1441–1446. [Google Scholar] [CrossRef] [PubMed]
- Lento, W.; Ito, T.; Zhao, C.; Harris, J.R.; Huang, W.; Jiang, C.; Owzar, K.; Piryani, S.; Racioppi, L.; Chao, N.; et al. Loss of beta-catenin triggers oxidative stress and impairs hematopoietic regeneration. Genes Dev. 2014, 28, 995–1004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, X.; Nguyen, T.A.; Appella, E.; Donehower, L.A. Homeostatic regulation of base excision repair by a p53-induced phosphatase: Linking stress response pathways with DNA repair proteins. Cell Cycle 2004, 3, 1363–1366. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Bocangel, D.; Nannenga, B.; Yamaguchi, H.; Appella, E.; Donehower, L.A. The p53-induced oncogenic phosphatase PPM1D interacts with uracil DNA glycosylase and suppresses base excision repair. Mol. Cell 2004, 15, 621–634. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Yu, T.; Shi, J.; Chen, X.; Zhang, W.; Lin, T.; Liu, Z.; Wang, Y.; Zeng, Z.; Wang, C.; et al. Thymine DNA glycosylase is a positive regulator of wnt signaling in colorectal cancer. J. Biol. Chem. 2014, 289, 8881–8890. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, A.L.; Schar, P. DNA glycosylases: In DNA repair and beyond. Chromosoma 2012, 121, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Saleem, M.; Murtaza, I.; Witkowsky, O.; Kohl, A.M.; Maddodi, N. Lupeol triterpene, a novel diet-based microtubule targeting agent: Disrupts survivin/cFLIP activation in prostate cancer cells. Biochem. Biophys. Res. Commun. 2009, 388, 576–582. [Google Scholar] [CrossRef] [PubMed]
- Bozic, I.; Reiter, J.G.; Allen, B.; Antal, T.; Chatterjee, K.; Shah, P.; Moon, Y.S.; Yaqubie, A.; Kelly, N.; Le, D.T.; et al. Evolutionary dynamics of cancer in response to targeted combination therapy. Elife 2013, 2, e00747. [Google Scholar] [CrossRef]
- Duthie, S.J.; Johnson, W.; Dobson, V.L. The effect of dietary flavonoids on DNA damage (strand breaks and oxidised pyrimdines) and growth in human cells. Mutat. Res.-Genet. Toxicol. Environ. Mutagen. 1997, 390, 141–151. [Google Scholar] [CrossRef]
- Yang, F.Q.; Liu, M.; Li, W.; Che, J.P.; Wang, G.C.; Zheng, J.H. Combination of quercetin and hyperoside inhibits prostate cancer cell growth and metastasis via regulation of microRNA-21. Mol. Med. Rep. 2015, 11, 1085–1092. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.S.; Lu, J.J.; Guo, J.J.; Li, Y.B.; Tan, W.; Dang, Y.Y.; Zhong, Z.F.; Xu, Z.T.; Chen, X.P.; Wang, Y.T. Ganoderic acid DM, a natural triterpenoid, induces DNA damage, G1 cell cycle arrest and apoptosis in human breast cancer cells. Fitoterapia 2012, 83, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.H.; Deng, C.X.; Sporn, M.B.; Liby, K.T. Cddo-imidazolide induces DNA damage, G2/M arrest and apoptosis in BRCA1-mutated breast cancer cells. Cancer Prev. Res. 2011, 4, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Shiono, J.; Shimizu, K.; Kukita, A.; Kukita, T.; Kondo, R. Ganoderic acid dm: Anti-androgenic osteoclastogenesis inhibitor. Bioorg. Med. Chem. Lett. 2009, 19, 2154–2157. [Google Scholar] [CrossRef] [PubMed]
- Scholtysek, C.; Krukiewicz, A.A.; Alonso, J.L.; Sharma, K.P.; Sharma, P.C.; Goldmann, W.H. Characterizing components of the saw palmetto berry extract (SPBE) on prostate cancer cell growth and traction. Biochem. Biophys. Res. Commun. 2009, 379, 795–798. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.; Dixit, S.; Ali, D.; Alqahtani, S.M.; Alkahtani, S.; Alarifi, S. Isolation and evaluation of anticancer efficacy of stigmasterol in a mouse model of DMBA-induced skin carcinoma. Drug Des. Dev. Ther. 2015, 9, 2793–2800. [Google Scholar] [CrossRef] [PubMed]
- Von Holtz, R.L.; Fink, C.S.; Awad, A.B. Beta-sitosterol activates the sphingomyelin cycle and induces apoptosis in LNCap human prostate cancer cells. Nutr. Cancer 1998, 32, 8–12. [Google Scholar] [CrossRef] [PubMed]



| Fraction | IC50 (24 h) | Total Flavonoid Content | Total Phenolic Content | Total Triterpenoids | ABTS+ | DPPH |
|---|---|---|---|---|---|---|
| μg/mL | mg Catecin Equivalents/g | mg Gallic Acid Equivalents/g | Oleanolic Acid (%) | μmol Trolox Equivalents/g | μmol Vitamin C Equivalents/g | |
| No. 8 | 99 | 100.3 ± 4.9 | 28.3 ± 1.2 | 51.6 ± 6.3 | 65.2 ± 1.0 | 34.6 ± 0.2 |
| No. 10 | 216 | 64.0 ± 5.5 | 35.5 ± 0.1 | 49.4 ± 2.4 | 180.9 ± 3.0 | 55.1 ± 0.1 |
| No. 13 | 274 | 25.2 ± 0.8 | 12.8 ± 0.2 | 26.3 ± 1.6 | 118.2 ± 1.6 | 77.4 ± 0.4 |
| No. 22 | 240 | 64.0 ± 0.4 | 54.8 ± 0.1 | 79.4 ± 7.5 | 196.1 ± 2.7 | 80.4 ± 0.4 |
| No. 25 | 227 | 86.8 ± 0.5 | 51.9 ± 1.2 | 39.4 ± 3.0 | 251.6 ± 3.6 | 96.5 ± 1.4 |
| Compounds | In AVL * | In Fract. # | In AVL * | In Fract. # | In AVL * | In Fract. # | In AVL * | In Fract. # | In AVL * | In Fract. # |
|---|---|---|---|---|---|---|---|---|---|---|
| Fraction | No. 8 | No. 10 | No. 13 | No. 22 | No. 25 | |||||
| Sterols | ||||||||||
| Sitgmasterol | 5.01 | 4.09 | 10.71 | 25.10 | 0.88 | 1.34 | ||||
| β-sitosterol | 1.66 | 3.89 | 19.10 | 29.07 | ||||||
| Triterpenoid | ||||||||||
| Lupeol | 23.65 | 19.31 | 1.25 | 2.92 | ||||||
| Flavonoids | ||||||||||
| Kaempferol | 0.37 | 0.60 | 0.12 | 0.36 | ||||||
| Isorhamnetin | 0.05 | 0.16 | ||||||||
| Compounds | Molecular Formula | Molecular Weight | Q1 Mass (m/z) |
|---|---|---|---|
| Sterols | |||
| Sitgmasterol | C29H48O | 412.69 | 413 [M + H]+ |
| β-sitosterol | C29H50O | 414.71 | 437 [M + Na]+ |
| Triterpenoid | |||
| Lupeol | C30H50O | 426.72 | 449 [M + Na]+ |
| Flavonoids | |||
| Kaempferol | C15H10O6 | 286.23 | 287 [M + H]+ |
| Isorhamnetin | C16H12O7 | 316.26 | 317 [M + H]+ |
| Quercetin | C15H10O7 | 302.24 | 303 [M + H]+ |
| Compounds | Selected 1H-NMR Data δ (Multiplicity/Hz) | Selected 13C-NMR Data δ |
|---|---|---|
| Sterols | ||
| Sitgmasterl | 0.70 (3H, s, H-29), 0.82(3H, d, H-26), 0.84 (3H, d, H-24), 0.85 (3H, d, H-27), 1.03 (3H, s, H-28), 3.53 (1H, m, H-3), 5.02 (1H, dd, H-21), 5.16 (1H, dd, H-20), 5.35 (1H, d, H-6) | 12.0 (CH3, C-18), 12.3 (CH3, C-29), 18.8 (CH3, C-21), 19.0 (CH3, C-27), 19.4 (CH3, C-19), 21.0 (CH3, C-26), 21.1 (CH2, C-11), 24.4 (CH2, C-15), 25.4 (CH2, C-23) 28.9 (CH, C-25), 31.6 (CH2, C-2), 31.9 (CH, C-8), 31.9 (CH2, C-7), 36.5 (C, C-10), 37.2 (CH2, C-1), 39.7 (CH2, C-12), 42.2 (CH2, C-4), 42.3 (C, C-13), 50.1 (CH, C-9), 56.0 (CH, C-17), 56.8 (CH, C-14), 71.8 (CH, C-3), 121.7 (CH, C-6), 129.2 (CH, C-3), 138.3 (CH, C-22), 140.7 (C, C-5) |
| β-sitosterol | 0.68 (3H, s, H-18), 0.83(3H, d, H-27), 0.84 (3H, d, H-26), 0.90 (3H, s, H-29), 0.93 (3H, d, H-21), 1.01 (3H, s, H-19), 3.54 (1H, m, H-3), 5.35 (1H, dd, H-6) | 1.8 (CH3, C-18), 12.0 (CH3, C-29), 18.8 (CH3, C-21), 19.0 (CH3, C-27), 19.4 (CH3, C-19), 19.8 (CH3, C-26), 21.1 (CH2, C-11), 23.1 (CH2, C-28), 24.3 (CH2, C-15), 26.0 (CH2, C-23), 28.2 (CH2, C-16), 29.1 (CH, C-25), 31.6 (CH2, C-2), 31.8 (CH, C-8), 31.9 (CH2, C-7), 33.9 (CH2, C-22), 36.1 (CH, C-20), 36.5 (C, C-10), 37.2 (CH2, C-1), 39.7 (CH2, C-12), 42.3 (CH2, C-4), 42.3 (C, C-13), 45.8 (CH, C-24), 50.1 (CH, C-9), 56.0 (CH, C-17), 56.7 (CH, C-14), 71.8 (CH, C-3), 121.7 (CH, C-6), 140.7 (C, C-5) |
| Triterpenoid | ||
| Lupeol | 0.69 (1H, d, H-5), 0.76 (3H, s, H-23), 0.78 (3H, s, H-28), 0.83 (3H, s, H-25), 0.94 (3H, s, H-27), 0.97 (3H, s, H-26), 1.03 (3H, s, H-24), 1.27 (2H, m, H-21), 1.30 (1H, m, H-9), 1.38 (2H, m, H-7), 1.39 (2H, m, H-6), 1.38 (1H, m, H-18), 1.68 (1H, s, H-30), 2.38 (1H, m, H-19), 3.18 (1H, m, H-3), 4.56 (1H, s, H-29b), 4.68 (1H, s, H-29a) | 14.5 (CH3, C-27), 15.4(CH3, C-24), 16.0 (CH3, C-26), 16.1 (CH3, C-25), 18.0 (CH3, C-28), 18.3 (CH2, C-6), 19.3 (CH3, C-30), 20.9 (CH2, C-11), 25.1 (CH2, C-12), 27.2 (CH2, C-15), 27.4 (CH2, C-2), 28.0 (CH3, C-23), 29.8 (CH2, C-21), 34.3 (CH2, C-7), 35.9 (CH2, C-16), 37.2 (C, C-10), 38.0(CH, C-13), 38.7 (CH2, C-1), 38.9 (C, C-4), 40.0 (CH2, C-22), 40.8 (C, C-8), 42.8 (C, C-14), 43.0 (C, C-17), 48.0 (CH, C-19), 48.3 (CH, C-18), 50.4 (CH, C-9), 55.3 (CH, C-5), 79.0 (CH, C-3), 109.3 (CH2, C-29), 151.0 (C, C-20) |
| Flavonoids | ||
| Kaempferol | 6.20 (1H, d, H-6), 6.44 (1H, d, H-8), 6.93 (1H, d, H-5′), 8.03 (1H, m, H-6′), 9.39 (1H, s, OH), 10.10 (1H, s, OH), 10.78 (1H, s, OH), 12.47 (1H, s, OH) | 146.9 (C-2), 135.7 (C-3), 175.9 (C-4), 156.3 (C-5), 98.3(C-6), 163.9 (C-7), 93.6 (C-8), 160.8(C-9), 103.1 (C-10), 121.7 (C-1′),115.5 (C-2′), 129.6 (C-3′), 159.3 (C-4′), 115.5 (C-5′), 121.7 (C-6′) |
| Isorhamnetin | 3.84 (3H, s, OCH3), 6.20 (1H, d, H-6), 6.48 (1H, d, H-8), 6.93 (1H, d, H-5′), 7.70 (1H, m, H-6′), 7.75 (1H, d, H-2′), 9.44 (1H, s, OH), 9.75 (1H, s, OH), 10.76 (1H, s, OH), 12.47 (1H, s, OH) | 148.8 (C-2), 135.8 (C-3), 175.9 (C-4), 156.2 (C-5), 98.2(C-6), 163.9 (C-7), 93.6 (C-8), 160.7(C-9), 103.0 (C-10), 121.9 (C-1′),111.7 (C-2′), 146.6 (C-3′), 147.4 (C-4′),115.5 (C-5′), 121.7 (C-6′), 55.8 (OCH3) |
| Quercetin | 6.24 (1H, d, H-6), 6.48 (1H, d, H-8), 6.96 (1H, d, H-5′), 7.63 (1H, m, H-6′), 7.70 (1H, d, H-2′), 12.00 (1H, s, OH) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, S.-P.; Ho, T.-M.; Yang, C.-W.; Chang, Y.-J.; Chen, J.-F.; Shaw, N.-S.; Horng, J.-C.; Hsu, S.-L.; Liao, M.-Y.; Wu, L.-C.; et al. Chemopreventive Potential of Ethanolic Extracts of Luobuma Leaves (Apocynum venetum L.) in Androgen Insensitive Prostate Cancer. Nutrients 2017, 9, 948. https://doi.org/10.3390/nu9090948
Huang S-P, Ho T-M, Yang C-W, Chang Y-J, Chen J-F, Shaw N-S, Horng J-C, Hsu S-L, Liao M-Y, Wu L-C, et al. Chemopreventive Potential of Ethanolic Extracts of Luobuma Leaves (Apocynum venetum L.) in Androgen Insensitive Prostate Cancer. Nutrients. 2017; 9(9):948. https://doi.org/10.3390/nu9090948
Chicago/Turabian StyleHuang, Szu-Ping, Tzu-Ming Ho, Chih-Wen Yang, Ya-Ju Chang, Jie-Fu Chen, Ning-Sing Shaw, Jia-Cherng Horng, Shih-Lan Hsu, Ming-Yuan Liao, Li-Chen Wu, and et al. 2017. "Chemopreventive Potential of Ethanolic Extracts of Luobuma Leaves (Apocynum venetum L.) in Androgen Insensitive Prostate Cancer" Nutrients 9, no. 9: 948. https://doi.org/10.3390/nu9090948





