Fiber Patterns in Young Adults Living in Different Environments (USA, Spain, and Tunisia). Anthropometric and Lifestyle Characteristics
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Participants
2.3. General Data
2.4. Food Consumption Assessment
2.5. Statistical Analysis
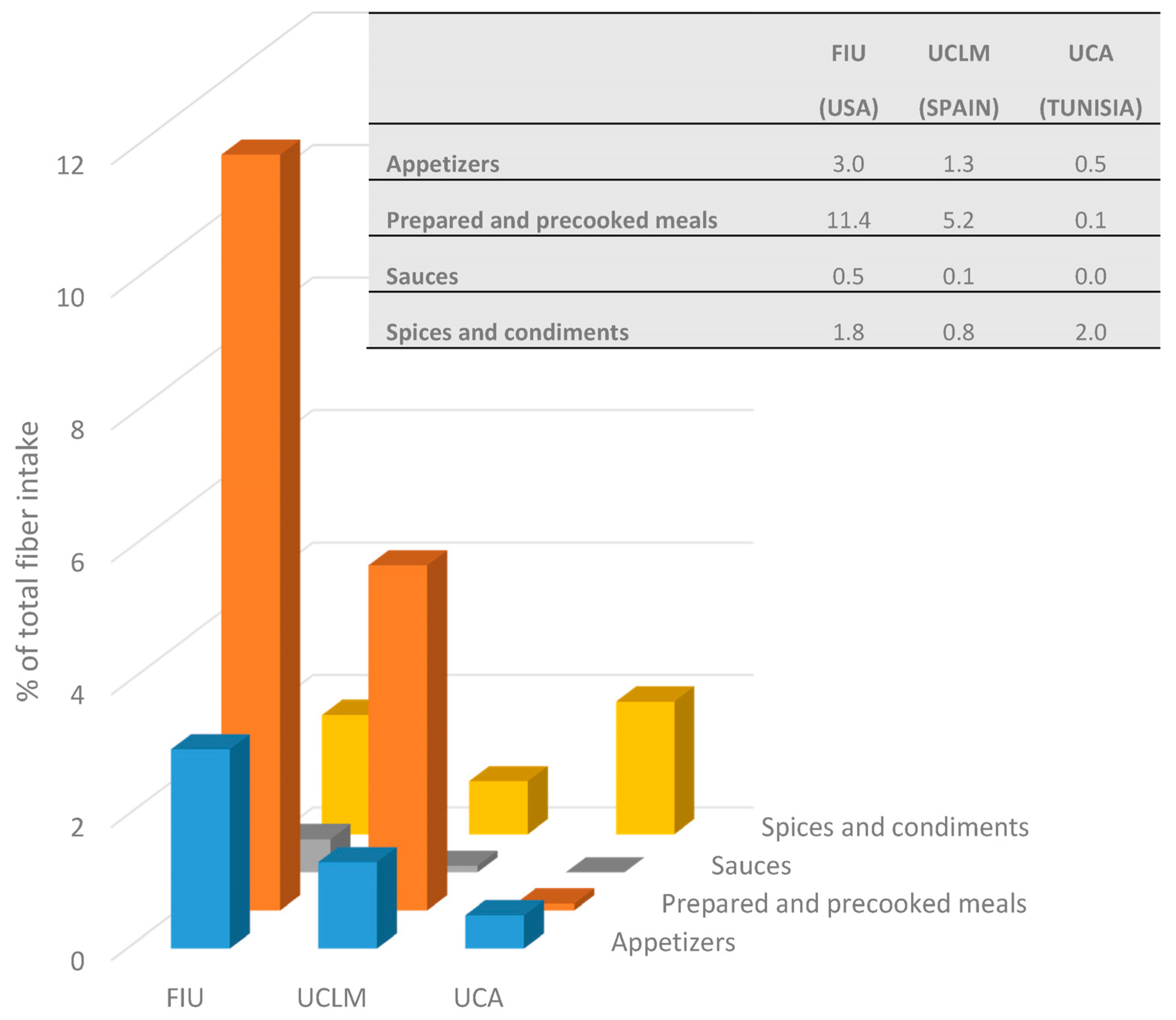
3. Results
4. Discussion
4.1. First Level
4.2. Second Level
4.3. Third Level
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Burkitt, D.P.; Walker, A.R.; Painter, N.S. Effect of dietary fibre on stools and the transit-times, and its role in the causation of disease. Lancet 1972, 2, 1408–1412. [Google Scholar] [CrossRef]
- Trowell, H. Why a new term for dietary fiber? Am. J. Clin. Nutr. 1977, 30, 1003–1004. [Google Scholar] [PubMed]
- Slavin, J.L. Dietary fiber: Classification, chemical analyses, and food sources. J. Am. Diet. Assoc. 1987, 87, 1164–1171. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.W.; Smith, B.M.; Gustafson, N.J. Health benefits and practical aspects of high-fiber diets. Am. J. Clin. Nutr. 1994, 59, 1242S–1247S. [Google Scholar] [PubMed]
- Marlett, J.A.; McBurney, M.I.; Slavin, J.L. Position of the American Dietetic Association: Health implications of dietary fiber. J. Am. Diet. Assoc. 2002, 102, 993–1000. [Google Scholar] [CrossRef]
- Bonnema, A.L.; Kolberg, L.W.; Thomas, W.; Slavin, J.L. Gastrointestinal tolerance of chicory inulin products. J. Am. Diet. Assoc. 2010, 110, 865–868. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wang, H.P.; Zhou, L.; Xu, C.F. Effect of dietary fiber on constipation: A Meta Analysis. World J. Gastroenterol. 2012, 18, 7378–7383. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Bella, F.; Godos, J.; Sciacca, S.; Del Rio, D.; Ray, S.; Galvano, F.; Giovannucci, E.L. Possible role of diet in cancer: Systematic review and multiple meta-analyses of dietary patterns, lifestyle factors, and cancer risk. Nutr. Rev. 2017, 75, 405–419. [Google Scholar] [CrossRef]
- Meyer, K.A.; Kushi, L.H.; Jacobs, D.R., Jr.; Slavin, J.; Sellers, T.A.; Folsom, A.R. Carbohydrates, dietary fiber, and incident type 2 diabetes in older women. Am. J. Clin. Nutr. 2000, 71, 921–930. [Google Scholar] [PubMed]
- Ye, E.Q.; Chacko, S.A.; Chou, E.L.; Kugizaki, M.; Liu, S. Greater whole-grain intake is associated with lower risk of type 2 diabetes, cardiovascular disease, and weight gain. J. Nutr. 2012, 142, 1304–1413. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.S.; Qi, L.; Fahey, G.C., Jr.; Klurfeld, D.M. Consumption of cereal fiber, mixtures of whole grains and bran, and whole grains and risk reduction in type 2 diabetes, obesity, and cardiovascular disease. Am. J. Clin. Nutr. 2013, 98, 594–619. [Google Scholar] [CrossRef] [PubMed]
- Buil-Cosiales, P.; Zazpe, I.; Toledo, E.; Corella, D.; Salas-Salvadó, J.; Diez-Espino, J.; Ros, E.; Fernandez-Creuet Navajas, J.; Santos-Lozano, J.M.; Arós, F.; et al. Fiber intake and all-cause mortality in the Prevención con Dieta Mediterránea (PREDIMED) study. Am. J. Clin. Nutr. 2014, 100, 1498–1507. [Google Scholar] [CrossRef] [PubMed]
- Allman-Farinelli, M.; Partridge, S.R.; Roy, R. Weight-Related Dietary Behaviors in Young Adults. Curr. Obes. Rep. 2016, 5, 23–29. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Diet, Nutrition, and the Prevention of Chronic Diseases; Report of a WHO Study Group; WHO Technical Report Series 797; WHO Library Cataloguing-in-Publication Data: Geneva, Switzerland, 1990; Available online: http://www.who.int/nutrition/publications/obesity/WHO_TRS_797/en/ (accessed on 3 June 2017).
- Nishida, C.; Uauy, R.; Kumanyika, S.; Shetty, P. The joint WHO/FAO expert consultation on diet, nutrition and the prevention of chronic diseases: Process, product and policy implications. Public Health Nutr. 2004, 7, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Freedman, M.R.; Fulgoni, V.L., III. Canned vegetables and fruit consumption is associated with changes in nutrient intake and higher diet quality in children and adults: National Health and Nutrition Examination Survey 2001–2010. J. Acad. Nutr. Diet. 2016, 116, 940–948. [Google Scholar] [CrossRef] [PubMed]
- Howlett, J.F.; Betteridge, V.A.; Champ, M.; Craig, S.A.; Meheust, A.; Jones, J.M. The definition of dietary fiber—Discussions at the Ninth Vahouny Fiber Symposium: Building scientific agreement. Food Nutr. Res. 2010, 54, 5750. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.M. Dietary fiber future directions: Integrating new definitions and findings to inform nutrition research and communication. Adv. Nutr. 2013, 4, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Fuller, S.; Beck, E.; Salman, H.; Tapsell, L. New horizons for the study of dietary fiber and health: A review. Plant Foods Hum. Nutr. 2016, 71, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine of the National Academies (IoMa). Dietary, functional and total fiber. In Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; The National Academies Press: Washington, DC, USA, 2005; pp. 339–421. Available online: https://www.nap.edu/read/10490/chapter/1 (accessed on 3 June 2017).
- Slavin, J.L.; Savarino, V.; Parades-Diaz, A.; Fotopoulos, G.A. Review of the role of soluble fiber in health with specific reference to wheat dextrin. J. Int. Med. Res. 2009, 37, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Lattimer, J.M.; Haub, M.D. Effects of dietary fiber and its components on metabolic health. Nutrients 2010, 2, 1266–1289. [Google Scholar] [CrossRef] [PubMed]
- Brownawell, A.M.; Caers, W.; Gibson, G.R.; Kendall, C.W.; Lewis, K.D.; Ringel, Y.; Slavin, J.L. Prebiotics and the health benefits of fiber: Current regulatory status, future research, and goals. J. Nutr. 2012, 142, 962–974. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.E.; Lenoir-Wijnkoop, I.; Salminen, S.; Merenstein, D.J.; Gibson, G.R.; Petschow, B.W.; Nieuwdorp, M.; Tancredi, D.J.; Cifelli, C.J.; Jacques, P.; et al. Probiotics and prebiotics: Prospects for public health and nutritional recommendations. Ann. N. Y. Acad. Sci. 2014, 1309, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Dahl, W.J.; Lockert, E.A.; Cammer, A.L.; Whiting, S.J. Effects of flax fiber on laxation and glycemic response in healthy volunteers. J. Med. Food 2005, 8, 508–511. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J.; Green, H. Dietary fibre and satiety. Nutr. Bull. 2007, 32, S32–S42. [Google Scholar] [CrossRef]
- Wahlqvist, M.L. Food structure is critical for optimal health. Food Funct. 2016, 7, 1245–1250. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Health benefits of fruit and vegetables are from additive and synergistic combinations of phytochemicals. Am. J. Clin. Nutr. 2003, 78, 517S–520S. [Google Scholar] [PubMed]
- Okarter, N.; Liu, R.H. Health benefits of whole grain phytochemicals. Crit. Rev. Food Sci. Nutr. 2010, 50, 193–208. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.M. Dietary fibre’s Co-Passengers: Is it the fiber or the co-passengers? In Dietary Fiber: New Frontiers for Food and Health; Wageningen Academic Publishers: Wageningen, The Netherlands, 2010; pp. 365–378. [Google Scholar] [CrossRef]
- Saura-Calixto, F. Dietary fiber as a carrier of dietary antioxidants: An essential physiological function. J. Agric. Food Chem. 2011, 59, 43–49. [Google Scholar] [CrossRef] [PubMed]
- De Vries, J.; Birkett, A.; Hulshof, T.; Verbeke, K.; Gibes, K. Effects of cereal, fruit and vegetable fibers on human fecal weight and transit time: A comprehensive review of intervention trials. Nutrients 2016, 8, 130. [Google Scholar] [CrossRef] [PubMed]
- Buil-Cosiales, P.; Martinez-Gonzalez, M.A.; Ruiz-Canela, M.; Díez-Espino, J.; García-Arellano, A.; Toledo, E. Consumption of fruit or fiber-fruit decreases the risk of cardiovascular disease in a Mediterranean young cohort. Nutrients 2017, 9, 295. [Google Scholar] [CrossRef] [PubMed]
- González-Rodríguez, L.; Perea Sánchez, J.M.; Aranceta-Bartrina, J.A.; González-Gross, M.; Serra-Majem, L.; Varela-Moreiras, G.; Ortega, R.M. Intake and Dietary Food Sources of Fibre in Spain: Differences with Regard to the Prevalence of Excess Body Weight and Abdominal Obesity in Adults of the ANIBES Study. Nutrients 2017, 9, 326. [Google Scholar] [CrossRef] [PubMed]
- Howarth, N.C.; Huang, T.T.K.; Roberts, S.B.; McCrory, M.A. Dietary Fiber and Fat Are Associated with Excess Weight in Young and Middle-Aged US Adults. J. Am. Diet. Assoc. 2005, 105, 1365–1372. [Google Scholar] [CrossRef] [PubMed]
- Myhre, J.; Løken, E.; Wandel, M.; Andersen, L. The contribution of snacks to dietary intake and their association with eating location among Norwegian adults—Results from a cross-sectional dietary survey. BMC Public Health 2015, 15, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bielemann, R.M.; Motta, J.V.; Minten, G.C.; Horta, B.L.; Gigante, D.P. Consumption of ultra-processed foods and their impact on the diet of young adults. Rev. Saude Publica 2015, 49, 28. [Google Scholar] [CrossRef] [PubMed]
- El Ansari, W.; Stock, C.; Mikolajczyk, R.T. Relationships between food consumption and living arrangements among university students in four European countries—A cross-sectional study. Nutr. J. 2012, 11, 28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- García-Meseguer, M.J.; Cervera Burriel, F.; Vico García, C.; Serrano-Urrea, R. Adherence to Mediterranean diet in a Spanish university population. Appetite 2014, 78, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.; Harker, M.; Harker, D.; Reinhard, K. Younth transition to University in Germany and Australia. An empirical investigation of healthy eating behaviour. J. Youth Stud. 2010, 13, 353–367. [Google Scholar] [CrossRef]
- Erlich, R.; Yngve, A.; Wahlqvist, M.L. Cooking as a healthy behaviour. Public Health Nutr. 2012, 15, 1139–1140. [Google Scholar] [CrossRef] [PubMed]
- Szczuko, M.; Gutowska, I.; Seidler, T. Nutrition and nourishment status of Polish students in comparison with students from other countries. Rocz. Panstw. Zakl. Hig. 2015, 66, 261–268. [Google Scholar] [PubMed]
- Ludwig, D.S.; Pereira, M.A.; Kroenke, C.H.; Hilner, J.E.; Van Horn, L.; Slattery, M.L.; Jacobs, D.R., Jr. Dietary fiber, weight gain, and cardiovascular disease risk factors in young adults. JAMA 1999, 282, 1539–1546. [Google Scholar] [CrossRef] [PubMed]
- Farvid, M.S.; Eliassen, A.H.; Cho, E.; Liao, X.; Chen, W.Y.; Willett, W.C. Dietary fiber intake in young adults and breast cancer risk. Pediatrics 2016, 137, e20151226. [Google Scholar] [CrossRef] [PubMed]
- Chourdakis, M.; Tzellos, T.; Papazisis, G.; Toulis, K.; Kouvelas, D. Eating habits, health attitudes and obesity indices among medical students in northern Greece. Appetite 2010, 55, 722–725. [Google Scholar] [CrossRef] [PubMed]
- Parminder, K.G.; Charajeet, K.H. Intake of nutrients, phytin P, polyphenolic compounds, oxalates and dietary fibre by university campus residents. Ecol. Food Nutr. 1995, 34, 11–17. [Google Scholar] [CrossRef]
- Nicklas, T.; Nicklas, T.A.; Farris, R.P.; Myers, L.; Berenson, G.S. Dietary fiber intake of children and young adults: The Bogalusa Heart Study. J. Am. Diet. Assoc. 1995, 95, 209–214. [Google Scholar] [CrossRef]
- Lagerpusch, M.; Enderle, J.; Later, W.; Eggeling, B.; Pape, D.; Muller, M.J.; Bosy-Westphal, A. Impact of glycaemic index and dietary fibre on insulin sensitivity during the refeeding phase of a weight cycle in young healthy men. Br. J. Nutr. 2013, 109, 1606–1616. [Google Scholar] [CrossRef] [PubMed]
- The World Bank. New Country Classifications by Income Level. Available online: https://blogs.worldbank.org/opendata/new-country-classifications-2016 (accessed on 3 June 2017).
- Mokhtar, N.; El Ati, J.; Chabir, R.; Bour, A.; Elkari, K.; Schlossman, N.P.; Caballero, B.; Aguenaou, H. Diet culture and obesity in northern Africa. J. Nutr. 2001, 131, 887S–892S. [Google Scholar] [PubMed]
- World Health Organization (WHO), Regional Office for the Eastern Mediterranean. Promoting a Healthy Diet for the WHO Eastern Mediterranean Region: User-Friendly Guide; WHO: Cairo, Egypt, 2012; Available online: http://applications.emro.who.int/dsaf/emropub_2011_1274.pdf?ua=1 (accessed on 3 June 2017).
- Agencia Española de Seguridad Alimentaria y Nutrición (AESAN). Encuesta Nacional de Ingesta Dietética de la Población Española. (2009–2010) (ENIDE). 2012. Available online: http://www.tappers.es/media/wysiwyg/valoracion_nutricional_enide_macronutrientes.pdf (accessed on 3 June 2017).
- U.S. Department of Agriculture; U.S. Department of Health and Human Services. Dietary Guidelines for Americans, 7th ed.; U.S. Government Printing Office: Washington, DC, USA, 2010. Available online: https://health.gov/dietaryguidelines/dga2010/dietaryguidelines2010.pdf (accessed on 3 June 2017).
- Hoy, M.K.; Goldman, J.D. Fiber Intake of the U.S. Population: What We Eat in America, NHANES 2009–2010. Food Surveys Research Group Dietary Data Brief No. 12; U.S. Department of Agriculture, 2014. Available online: https://www.ars.usda.gov/ARSUserFiles/80400530/pdf/DBrief/12_fiber_intake_0910.pdf (accessed on 3 June 2017).
- Aounallah-Skhiri, H.; Traissac, P.; El Ati, J.; Eymard-Duvernay, S.; Landais, E.; Achour, N.; Delpeuch, F.; Ben Romdhane, H.; Maire, B. Nutrition transition among adolescents of a south-Mediterranean country: Dietary patterns, association with socio-economic factors, overweight and blood pressure. A cross-sectional study in Tunisia. Nutr. J. 2011, 10, 38. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, E.; Ávila, J.M.; Valero, T.; Del Pozo, S.; Rodriguez, P.; Aranceta-Bartrina, J.; Gil, Á.; González-Gross, M.; Ortega, R.M.; Serra-Majem, L.; et al. Macronutrient distribution and dietary sources in the Spanish population: Findings from the ANIBES study. Nutrients 2016, 8, 177. [Google Scholar] [CrossRef] [PubMed]
- McGill, C.R.; Devareddy, L. Ten-year trends in fiber and whole grain intakes and food sources for the United States population: National Health and Nutrition Examination Survey 2001–2010. Nutrients 2015, 7, 1119–1130. [Google Scholar] [CrossRef] [PubMed]
- Willet, W. Issues in Analysis and Presentation of Dietary Data. Nutritional Epidemiology, 3rd ed.; Oxford University Press: New York, NY, USA, 2013. [Google Scholar]
- World Health Organization (WHO). Obesity: Preventing and Managing the Global Epidemic; Report of a WHO Consultation; WHO Technical Report Series 894; WHO Library Cataloguing-in-Publication Data: Geneva, Switzerland, 2000; Available online: http://www.who.int/nutrition/publications/obesity/WHO_TRS_894/en/ (accessed on 3 June 2017).
- Gómez, C.; Loria, V.; Lourenço, T. Guía Visual de Alimentos y Raciones; EDIMSA (Editores Médicos, S.A.): Madrid, España, 2007. [Google Scholar]
- WebMD Health Corporation. The WebMD Portion Size Plate. Available online: http://www.webmd.com/diet/healthtool-portion-size-plate (accessed on 15 May 2017).
- Institute of Medicine of the National Academies (IoMb). Physical activity. In Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; The National Academies Press: Washington, DC, USA, 2005; pp. 880–935. Available online: https://www.nap.edu/read/10490/chapter/12 (accessed on 15 May 2017).
- Institut National de Nutrition & de Technologie Alimentaire (INNTA). Table de Composition des Aliments Tunisiens. Available online: http://www.institutdenutrition.rns.tn/ (accessed on 15 May 2017).
- Moreiras, O.; Carbajal, A.; Cabrera, L.; Cuadrado, C. Tablas de Composición de Alimentos. Guía de Prácticas, 17th ed.; Ediciones Pirámide: Madrid, España, 2015. [Google Scholar]
- U.S. Department of Agriculture. USDA Food Composition Databases. Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 15 May 2017).
- Vellas, B.; Guigoz, Y.; Garry, P.J.; Nourhashemi, F.; Bennahum, D.; Lauque, S.; Albarede, J.L. The Mini Nutritional Assessment (MNA) and its use in grading the nutritional state of elderly patients. Nutrition 1999, 15, 116–122. [Google Scholar] [CrossRef]
- Tur, J.A.; Serra-Majem, L.; Romaguera, D.; Pons, A. Does the diet of the Balearic population, a Mediterranean type diet, still provide adequate antioxidant nutrient intakes? Eur. J. Nutr. 2005, 44, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Wang, Z.; Zhu, J.; Murad, A.L.; Prokop, L.J.; Murad, M.H. Nut consumption and risk of cancer and type 2 diabetes: A systematic review and meta-analysis. Nutr. Rev. 2015, 73, 409–425. [Google Scholar] [CrossRef] [PubMed]
- Omran, A.R. The epidemiologic transition: A theory of the epidemiology of population change. Milbank Q. 2005, 83, 731–757. [Google Scholar] [CrossRef] [PubMed]
- Popkin, B.M.; Adair, L.S.; Ng, S.W. Global nutrition transition and the pandemic of obesity in developing countries. Nutr. Rev. 2012, 70, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Dahl, W.J.; Stewart, M.L. Position of the Academy of Nutrition and Dietetics: Health implications of dietary fiber. J. Acad. Nutr. Diet. 2015, 115, 1861–1870. [Google Scholar] [CrossRef] [PubMed]
- Wardle, J.; Haase, A.M.; Steptoe, A. Body image and weight control in young adults: International comparisons in university students from 22 countries. Inter. J. obes. 2006, 30, 644–651. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 22, 1417–1435. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Knight, R. Dietary effects on human gut microbiome diversity. Br. J. Nutr. 2015, 113, S1–S5. [Google Scholar] [CrossRef] [PubMed]
- Cervera Burriel, F.; Serrano Urrea, R.; Daouas, T.; Delicado Soria, A.; García Meseguer, M.J. Food habits and nutritional assessment in a Tunisian university population. Nutr. Hosp. 2014, 30, 1350–1358. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Colditz, G.A.; Cotterchio, M.; Boucher, B.A.; Kreiger, N. Adolescent dietary fiber, vegetable fat, vegetable protein, and nut intakes and breast cancer risk. Breast Cancer Res. Treat. 2014, 145, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Wolk, A. Dietary fiber intake is inversely associated with stroke incidence in healthy Swedish adults. J. Nutr. 2014, 144, 1952–1955. [Google Scholar] [CrossRef] [PubMed]
- Mrabet, A.; Rodríguez-Arcos, R.; Guillén-Bejarano, R.; Chaira, N.; Ferchichi, A.; Jiménez-Araujo, A. Dietary fiber from Tunisian common date cultivars (Phoenix dactylifera L.): Chemical composition, functional properties, and antioxidant capacity. J. Agric. Food Chem. 2012, 60, 3658–3664. [Google Scholar] [CrossRef] [PubMed]
- Eurostat. GDP at Regional Level. Available online: http://ec.europa.eu/eurostat/statistics-explained/index.php/GDP_at_regional_level/es (accessed on 25 May 2017).
- Food Agriculture Organization (FAO). Tunisia Case Study. Prepared for FAO as part of the State of the World’s Forests 2016 (SOFO). 2015. Available online: http://www.fao.org/3/a-c0185e.pdf (accessed on 25 May 2017).
- World Health Organization (WHO) / Food Agriculture Organization (FAO). Global and regional food consumption patterns and trends. In Diet, Nutrition and the Prevention of Chronic Diseases; Report of a Joint WHO/FAO Expert Consultation; WHO Technical Report Series 916; WHO Library Cataloguing-in-Publication Data: Geneva, Switzerland, 2003; pp. 13–29. Available online: ftp://ftp.fao.org/docrep/fao/005/ac911e/ac911e01.pdf (accessed on 25 May 2017).
- Florence, M.D.; Asbridge, M.; Veugelers, P.J. Diet quality and academic performance. J. Sch. Health 2008, 78, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Food Agriculture Organization (FAO). Profil Nutritionnel de la Tunisie. Division de l’Alimentation et de la Nutrition. 2005. Available online: http://www.fao.org/docrep/017/ap850f/ap850f.pdf (accessed on 25 May 2017).
- Chourdakis, M.; Tzellos, T.; Pourzitaki, C.; Toulis, K.A.; Papazisis, G.; Kouvelas, D. Evaluation of dietary habits and assessment of cardiovascular disease risk factors among Greek university students. Appetite 2011, 57, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; van der A, D.L.; Boshuizen, H.C.; Forouhi, N.G.; Wareham, N.J.; Halkjaer, J.; Tjønneland, A.; Overvad, K.; Jakobsen, M.U.; Boeing, H.; et al. Dietary fiber and subsequent changes in body weight and waist circumference in European men and women. Am. J. Clin. Nutr. 2010, 91, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Ben Slama, F.; Jebali, N.; Chemli, R.; Ben Rayana, C.; Achour, A.; Najar, M.F.; Achour, N.; Belhadj, O. Dietary fiber in the diets of urban Tunisian women: Association of fiber intake with BMI, waist circumference and blood chemistry: Preliminary study. Clin. Res. Hepatol. Gastroenterol. 2011, 35, 750–754. [Google Scholar] [CrossRef] [PubMed]
- Sawada, N.; Iwasaki, M.; Yamaji, T.; Shimazu, T.; Sasazuki, S.; Inoue, M.; Tsugane, S.; Japan Public Health Center–based Prospective Study Group. Fiber intake and risk of subsequent prostate cancer in Japanese men. Am. J. Clin. Nutr. 2015, 101, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Gordon, T.; Fisher, M.; Rifkind, B.M. Some difficulties inherent in the interpretation of dietary data from free-living populations. Am. J. Clin. Nutr. 1984, 39, 152–156. [Google Scholar] [PubMed]
- Lesser, L.I.; Mazza, M.C.; Lucan, S.C. Nutrition myths and healthy dietary advice in clinical practice. Am. Fam. Physician 2015, 91, 634–638. [Google Scholar] [PubMed]
- Shau, J.P.; Chen, P.H.; Chan, C.F.; Hsu, Y.; Wu, T.C.; James, F.E.; Pan, W.H. Fast foods—Are they a risk factor for functional gastrointestinal disorders? Asia Pac. J. Clin. Nutr. 2016, 25, 393–401. [Google Scholar] [CrossRef] [PubMed]
- US Food & Drug Administration. CFR—Code of Federal Regulations Title 21. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=172.120 (accessed on 3 June 2017).
- Trude, A.C.; Kharmats, A.Y.; Hurley, K.M.; Anderson Steeves, E.; Talegawkar, S.A.; Gittelsohn, J. Household, psychosocial, and individual-level factors associated with fruit, vegetable, and fiber intake among low-income urban African American youth. BMC Public Health 2016, 16, 872. [Google Scholar] [CrossRef] [PubMed]
- Allman-Farinelli, M.A.; Chey, T.; Bauman, A.E.; Gill, T.; James, W.P. Age, period and birth cohort effects on prevalence of overweight and obesity in Australian adults from 1990 to 2000. Eur. J Clin. Nutr. 2008, 62, 898–907. [Google Scholar] [CrossRef] [PubMed]
- McKeown-Eyssen, G.E.; Yeung, K.S.; Bright-See, E. Assessment of past diet in epidemiologic studies. Am. J. Epidemiol. 1986, 124, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Weitkunat, K.; Schumann, S.; Nickel, D.; Hornemann, S.; Petzke, K.J.; Schulze, M.B.; Pfeiffer, A.F.; Klaus, S. Odd-chain fatty acids as a biomarker for dietary fiber intake: A novel pathway for endogenous production from propionate. Am. J. Clin. Nutr. 2017, 105, 1544–1551. [Google Scholar] [CrossRef] [PubMed]
- Kranz, S.; Dodd, K.W.; Juan, W.Y.; Johnson, L.K.; Jahns, L. Whole grains contribute only a small proportion of dietary fiber to the U.S. diet. Nutrients 2017, 9, 153. [Google Scholar] [CrossRef] [PubMed]
| Total Sample (n = 730) | Men (n = 239) | Women (n = 491) | p | |
|---|---|---|---|---|
| Population (%) | 100 | 32.7 | 67.3 | - |
| Age (years) | p = 0.705 † | |||
| Mean + SD (95% CI) | 21.2 ± 2.8 (21.0–21.4) | 21.2 ± 2.9 (20.9–21.6) | 21.2 ± 2.7 (21.0–21.5) | |
| Median (IR) | 21 (4) | 20 (4) | 21 (3) | |
| Weight (Kg) | p < 0.001 †,* | |||
| Mean + SD (95% CI) | 65.0 ± 13.3 (64.0–65.9) | 75.7 ± 12.3 (74.1–77.2) | 59.8 ± 10.3 (58.9–60.7) | |
| Median (IR) | 63 (17.0) | 74.0 (16.0) | 59.0 (12.0) | |
| BMI (Kg/m2) | p < 0.001 †,* | |||
| Mean + SD (95% CI) | 22.9 ± 13.3 (22.6–23.2) | 23.7 ± 4.1 (23.2–24.3) | 22.4 ± 4.0 (22.1–22.8) | |
| Median (IR) | 22.3 (4.4) | 23.3 (4.3) | 21.8 (4.2) | |
| BMI-categories (%) | ||||
| Underweight | 8.2 | 6.7 | 9.0 | p = 0.011 §,* |
| Normal range | 69.6 | 64.4 | 72.1 | |
| Overweight | 15.9 | 19.2 | 14.3 | |
| Obese | 6.3 | 9.6 | 4.7 | |
| BMI-2-categories (%) | ||||
| Underw. + normal rg. | 77.8 | 71.1 | 81.1 | p = 0.002 §,* |
| Overweight + obese | 22.2 | 28.9 | 18.9 | |
| Total fiber intake (g) | ||||
| Mean + SD (95% CI) | 17.8 ± 9.6 (17.1–18.5) | 17.2 ± 8.4 (16.2–18.3) | 18.1 ± 10.1 (17.2–19.0) | p = 0.303 † |
| Median (IR) | 15.6 (8.6) | 14.9 (8.3) | 15.7 (9.0) | |
| Energy intake (Kcal/day) | ||||
| Mean + SD | 1971.9 ± 553.0 | 2070.2 ± 604.9 | 1924.1 ± 519.9 | p = 0.001 ‡,* |
| (95% CI) | (1931.8–2012.1) | (1993.1–2147.3) | (1878.0–1970.2) | |
| Median (IR) | 1932.5 (747.9) | 2041.0 (897.6) | 1889.0 (730.0) | |
| Weight-loss diet (%) | 14.0 | 14.6 | 13.6 | p = 0.715 § |
| Smoking habits (%) | ||||
| Non-smoker | 88.1 | 83.7 | 90.2 | p = 0.029 §,* |
| ≤5 cigarettes per day | 6.3 | 7.9 | 5.5 | |
| >5 cigarettes per day | 5.6 | 8.4 | 4.3 | |
| Physical activity (%) | ||||
| Sedentary | 9.0 | 7.5 | 9.8 | p < 0.001 §,* |
| Low active | 27.9 | 28.5 | 27.7 | |
| Active | 40.3 | 32.6 | 44.0 | |
| Very active | 22.7 | 31.4 | 18.5 | |
| UCLM (SPAIN) | ||||
| Total Sample (n = 272) | Men (n = 120) | Women (n = 152) | p | |
| Population (%) | 100 | 44.12 | 55.88 | - |
| BMI (Kg/m2) | p = 0.041 †,* | |||
| Mean + SD (95% CI) | 22.4 ± 3.3 (22.0–22.8) | 22.8 ± 3.4 (22.2–23.4) | 22.1 ± 3.2 (21.5–22.6) | |
| Median (IR) | 22.0 (4.1) | 22.7 (4.2) | 21.6 (3.8) | |
| BMI-categories (%) | ||||
| Underweight | 8.5 | 10 | 7.2 | p = 0.297 §§ |
| Normal range | 73.2 | 67.5 | 77.7 | |
| Overweight | 14.7 | 17.5 | 12.5 | |
| Obese | 3.7 | 5.0 | 2.6 | |
| BMI-2-categories (%) | p = 0.119 § | |||
| Underw. + normal rg. | 81.7 | 77.5 | 84.9 | |
| Overweight + obese | 18.4 | 22.5 | 15.1 | |
| Total fiber intake (g) | ||||
| Mean + SD (95% CI) | 15.7 ± 6.6 (14.9–16.5) | 16.4 ± 6.3 (15.3–17.5) | 15.1 ± 6.9 (14.0–16.2) | p = 0.047 †,* |
| Median (IR) | 14.4 (7.4) | 14.7 (8.4) | 13.9 (7.0) | |
| Energy intake (Kcal/day) | ||||
| Mean + SD | 1980.5 ± 519.5 | 2123.4 ± 548.8 | 1867.7 ± 466.8 | p < 0.001 ‡,* |
| (95% CI) | (1918.5–2042.6) | (2024.2–2222.6) | (1792.9–1942.5) | |
| Median (IR) | 1944.2 (643.5) | 2067.6 (729.3) | 1829.9 (652.0) | |
| UCA (TUNISIA) | ||||
| Total Sample (n = 132) | Men (n = 46) | Women (n = 86) | p | |
| Population (%) | 100 | 34.84 | 65.15 | |
| BMI (Kg/m2) | p = 0.001 †,* | |||
| Mean + SD (95% CI) | 21.4 ± 2.8 (20.9–21.8) | 22.3 ± 2.8 (21.5–23.2) | 20.8 ± 2.7 (20.2–21.4) | |
| Median (IR) | 21.1 (3.6) | 22.2 (3.7) | 20.6 (4.0) | |
| BMI-categories (%) | ||||
| Underweight | 15.9 | 6.5 | 20.9 | p = 0.059 §§ |
| Normal range | 75.0 | 78.3 | 73.2 | |
| Overweight | 7.6 | 13.0 | 4.7 | |
| Obese | 1.5 | 2.2 | 1.2 | |
| BMI-2-categories (%) | p = 0.073 § | |||
| Underw. + normal rg. | 90.9 | 84.8 | 94.1 | |
| Overweight + obese | 9.1 | 15.2 | 5.9 | |
| Total fiber intake (g) | ||||
| Mean + SD (95% CI) | 15.0 ± 4.8 (14.1–15.8) | 14.6 ± 5.6 (13.0–16.3) | 15.1 ± 4.3 (14.2–16.1) | p = 0.352 † |
| Median (IR) | 14.5 (5.4) | 14.0 (5.7) | 14.8 (5.4) | |
| Energy intake (Kcal/day) | ||||
| Mean + SD | 1843.4 ± 563.1 | 1841.2 ± 670.5 | 1844.5 ± 500.6 | p = 0.567 † |
| (95% CI) | (1746.4–1940.3) | (1642.1–2040.4) | (1737.2–1951.9) | |
| Median (IR) | 1769.5 (667.0) | 1672.5 (786.0) | 1801.0 (646.3) | |
| FIU (USA) | ||||
| Total Sample (n = 326) | Men (n = 73) | Women (n = 253) | p | |
| Population (%) | 100 | 22.39 | 77.61 | - |
| BMI (Kg/m2) | p < 0.001 †,* | |||
| Mean + SD (95% CI) | 23.9 ± 4.8 (23.4–24.4) | 26.2 ± 4.9 (25.1–27.4) | 23.2 ± 4.6 (22.6–23.8) | |
| Median (IR) | 23.1 (5.1) | 24.9 (5.9) | 22.5 (4.9) | |
| BMI-categories (%) | ||||
| Underweight | 4.9 | 1.4 | 5.9 | p = 0.001 §§,* |
| Normal range | 64.4 | 50.7 | 68.4 | |
| Overweight | 20.2 | 26.0 | 18.6 | |
| Obese | 10.4 | 21.9 | 7.1 | |
| BMI-2-categories (%) | p < 0.001 §,* | |||
| Underw. + normal rg. | 69.3 | 52.1 | 74.3 | |
| Overweight + obese | 30.6 | 47.9 | 25.7 | |
| Total fiber intake (g) | ||||
| Mean + SD (95% CI) | 20.8 ± 12.0 (17.5–22.1) | 20.2 ± 11.6 (17.5–22.9) | 20.9 ± 12.1 (19.4–22.4) | p = 0.393 † |
| Median (IR) | 18.1 (12.8) | 17.3 (14.8) | 18.1 (12.5) | |
| Energy intake (Kcal/day) | ||||
| Mean + SD | 2016.8 ± 596.6 | 2127.0 ± 623.8 | 1985.0 ± 550.2 | p = 0.060 ‡ |
| (95% CI) | (1954.7–2078.9) | (1981.5–2272.6) | (1916.9–2053.1) | |
| Median (IR) | 1993.0 (841.5) | 2211.0 (947.5) | 1947.0 (808.0) | |
| UCLM (SPAIN) | UCA (TUNISIA) | FIU (USA) | p | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean + SD | CI 95% | Median | IR | Mean + SD | CI 95% | Median | IR | Mean + SD | CI 95% | Median | IR | ||
| Age (years) | 20.3 ± 2.4 | 20.0–20.5 | 19.0 a | 2 | 19.8 ± 1.4 | 19.6–20.1 | 19.0 a | 1 | 22.6 ± 2.8 | 22.3–22.9 | 22.0 a,b | 3 | p < 0.001 †,* |
| Weight (Kg) | 65.7 ± 12.9 | 64.2–67.3 | 64 | 18.0 | 64.3 ± 11.3 | 62.4–66.3 | 63.0 | 14.0 | 64.6 ± 14.3 | 63.0–66.2 | 61.5 | 18.0 | p = 0.278 † |
| BMI (Kg/m2) | 22.4 ± 3.3 | 22.0–22.8 | 22 c,d | 4.1 | 21.4 ± 2.8 | 20.9–21.8 | 21.1 c,e | 3.6 | 23.9 ± 4.8 | 23.4–24.4 | 23.1 d,e | 5.1 | p < 0.001 †,* |
| Total fiber intake (g) | 15.7 ± 6.6 | 14.9–16.5 | 14.4 f | 7.4 | 15.0 ± 4.8 | 14.1–15.8 | 14.5 g | 5.4 | 20.8 ± 12.0 | 19.5–22.1 | 18.1 f,g | 12.8 | p < 0.001 †,* |
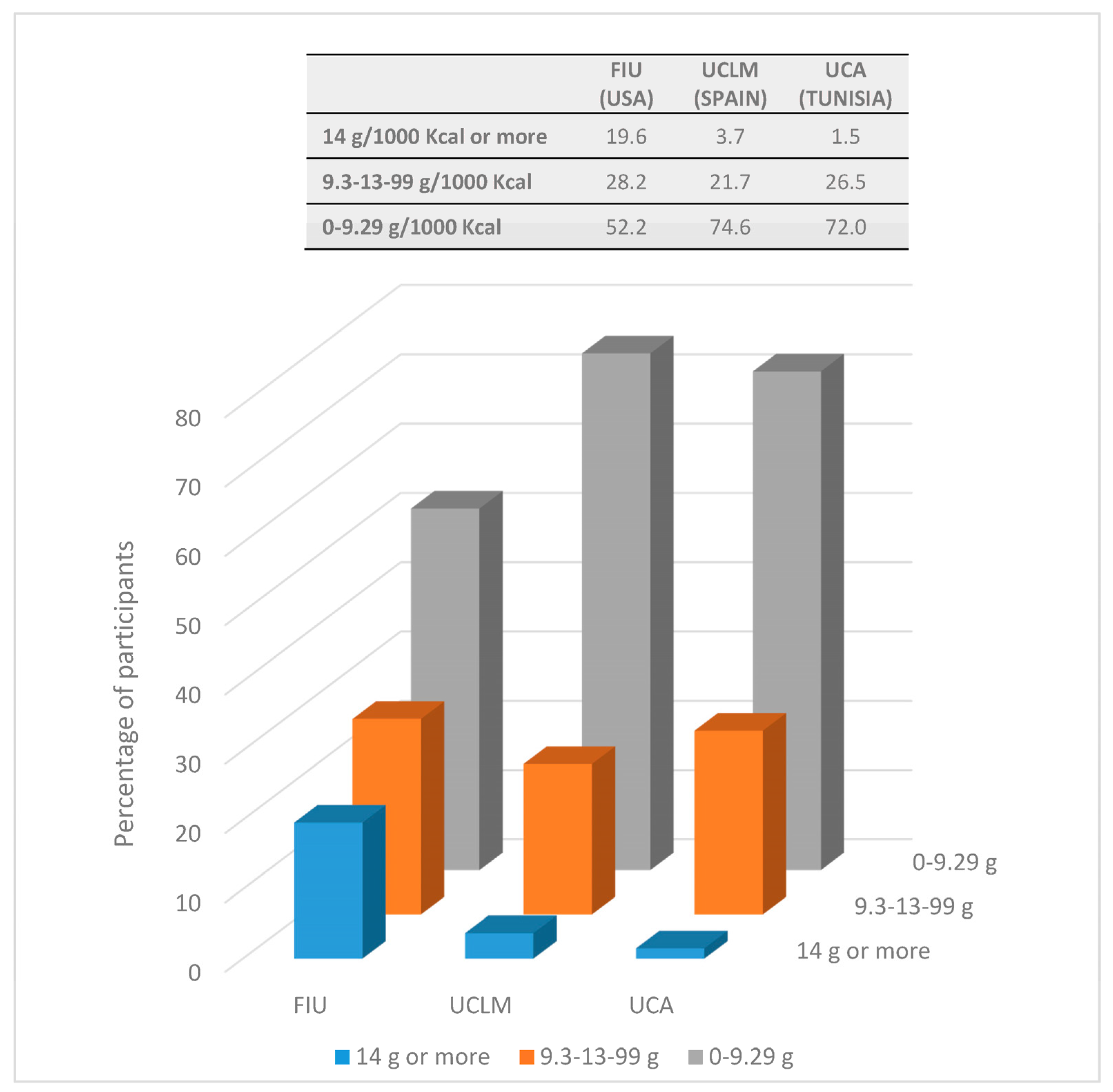
| Fiber intake (g/1000 Kcal) | 8.1 ± 3.2 | 7.7–8.5 | 7.5 h | 3.4 | 8.4 ± 2.2 | 8.0–8.7 | 8.0 i | 2.7 | 10.5 ± 5.6 | 9.9–11.1 | 8.9 h,i | 5.9 | p < 0.001 †,* |
| Energy (Kcal/day) | 1980.5 ± 519.5 | 1918.5–2042.6 | 1944.2 j | 643.5 | 1843.4 ± 563.1 | 1746.4–1940.3 | 1769.5 j,k | 667.0 | 2016.8 ± 596.6 | 1954.7–2078.9 | 1993.0 k | 841.5 | p = 0.002 †,* |
| Weight-loss diet (%) | p < 0.001 §,* | ||||||||||||
| Yes | 5.1 | 12.1 | 22.1 | ||||||||||
| No | 94.9 | 87.9 | 77.9 | ||||||||||
| Smoking habits (%) | p < 0.001 §§,* | ||||||||||||
| Non-smoker | 84.6 | 77.3 | 95.4 | ||||||||||
| ≤5 cigarettes/day | 8.5 | 8.3 | 3.7 | ||||||||||
| >5 cigarettes/day | 7.0 | 14.4 | 0.9 | ||||||||||
| Level of physical activity (%) | p < 0.001 §§,* | ||||||||||||
| Sedentary | 22.1 | 1.5 | 1.2 | ||||||||||
| Low active | 47.4 | 26.5 | 12.3 | ||||||||||
| Active | 20.2 | 43.2 | 55.8 | ||||||||||
| Very active | 10.3 | 28.8 | 30.7 | ||||||||||
| BMI-categories (%) | p < 0.001§§,* | ||||||||||||
| Underweight | 8.5 | 15.9 | 4.9 | ||||||||||
| Normal range | 73.2 | 75.0 | 64.4 | ||||||||||
| Overweight | 14.6 | 7.6 | 20.2 | ||||||||||
| Obese | 3.7 | 1.5 | 10.4 | ||||||||||
| BMI-2-categories (%) | p < 0.001 §,* | ||||||||||||
| Underw + normal | 81.7 | 90.9 | 69.3 | ||||||||||
| Overw. + obese | 18.3 | 9.1 | 30.6 | ||||||||||
| UCLM (SPAIN) | ||||||
| n | Mean + SD | 95% CI | Median | IR | p | |
| Gender | 0.047 †,* | |||||
| Men | 120 | 16.4 ± 6.3 | 15.3–17.5 | 14.7 | 8.4 | |
| Women | 152 | 15.1 ± 6.9 | 14.0–16.2 | 13.9 | 7.0 | |
| Weight-loss diet | 0.691 † | |||||
| Yes | 14 | 15.3 ± 6.6 | 11.5–19.1 | 14.4 | 10.3 | |
| No | 258 | 15.7 ± 6.6 | 14.9–16.5 | 14.4 | 7.3 | |
| BMI-categories | p-trend 0.051 § | |||||
| Underweight | 23 | 14.2 ± 5.6 | 11.8–16.7 | 12.7 | 5.2 | |
| Normal range | 199 | 16.1 ± 6.8 | 15.1–17.0 | 14.8 | 7.5 | |
| Overweight | 40 | 14.8 ± 6.3 | 12.8–16.8 | 13.2 | 8.3 | |
| Obese | 10 | 14.5 ± 5.8 | 10.3–18.7 | 12.1 | 8.7 | |
| BMI-2-categories | 0.190 † | |||||
| Underw. + normal rg. | 222 | 15.9 ± 6.7 | 15.0–16.8 | 14.5 | 7.2 | |
| Overweight + obese | 50 | 14.7 ± 6.1 | 13.0–16.5 | 12.9 | 8.3 | |
| BMI | r = −0.019 (Spearman’s correlation coefficient) | 0.753 | ||||
| Energy intake | r = 0.462 (Spearman’s correlation coefficient) | <0.001 ** | ||||
| UCA (TUNISIA) | ||||||
| n | Mean + SD | 95% CI | Median | IR | p | |
| Gender | 0.352 † | |||||
| Men | 46 | 14.6 ± 5.6 | 13.0–16.3 | 14.0 | 5.7 | |
| Women | 86 | 15.1 ± 4.3 | 14.2–16.1 | 14.8 | 5.4 | |
| Weight-loss diet | 0.734 ‡ | |||||
| Yes | 16 | 14.6 ± 6.2 | 11.3–17.9 | 14.7 | 7.1 | |
| No | 116 | 15.0 ± 4.6 | 14.2–15.9 | 14.5 | 5.3 | |
| BMI-categories | p-trend 0.471 § | |||||
| Underweight | 21 | 15.3 ± 4.7 | 13.2–17.5 | 13.6 | 6.4 | |
| Normal range | 99 | 15.0 ± 4.6 | 14.1–15.9 | 14.9 | 5.3 | |
| Overweight | 10 | 14.1 ± 6.7 | 9.3–18.9 | 13.2 | 8.0 | |
| Obese | 2 | 12.1 ± 2.4 | 0.0–33.7 | 12.1 | - | |
| BMI-2-categories | 0.128 † | |||||
| Underw. + normal rg. | 120 | 15.1 ± 4.6 | 14.3–15.9 | 14.8 | 5.3 | |
| Overweight + obese | 12 | 13.7 ± 6.2 | 9.8–17.7 | 13.0 | 5.8 | |
| BMI | r = −0.053 (Spearman’s correlation coefficient) | 0.543 | ||||
| Energy intake | r = 0.595 (Spearman’s correlation coefficient) | <0.001 ** | ||||
| FIU (USA) | ||||||
| n | Mean + SD | 95% CI | Median | IR | p | |
| Gender | 0.393 † | |||||
| Men | 73 | 20.2 ± 11.6 | 17.5–22.9 | 17.3 | 14.8 | |
| Women | 253 | 20.9 ± 12.1 | 19.4–22.4 | 18.1 | 12.5 | |
| Weight-loss diet | <0.001 †,* | |||||
| Yes | 72 | 26.8 ± 15.6 | 23.2–30.5 | 22.7 | 17.7 | |
| No | 254 | 19.0 ± 10.2 | 17.8–20.3 | 16.7 | 11.3 | |
| BMI-categories | p-trend 0.322 § | |||||
| Underweight | 16 | 25.6 ± 18.4 | 15.7–35.4 | 20.2 | 14.2 | |
| Normal range | 210 | 21.0 ± 11.7 | 19.4–22.6 | 18.1 | 13.4 | |
| Overweight | 66 | 19.6 ± 12.7 | 16.5–22.7 | 15.3 | 13.0 | |
| Obese | 34 | 19.3 ± 8.5 | 16.4–22.3 | 18.3 | 8.7 | |
| BMI-2-categories | 0.087 † | |||||
| Underw. + normal rg. | 226 | 21.3 ± 12.3 | 19.7–22.9 | 18.5 | 13.3 | |
| Overweight + obese | 100 | 19.5 ± 11.4 | 17.2–21.8 | 15.7 | 11.0 | |
| BMI | r = −0.127 (Spearman’s correlation coefficient) | 0.022 **,(1) | ||||
| Energy intake | r = 0.449 (Spearman’s correlation coefficient) | <0.001 ** | ||||
| UCLM (SPAIN) | UCA (TUNISIA) | FIU (USA) | p † | ||
|---|---|---|---|---|---|
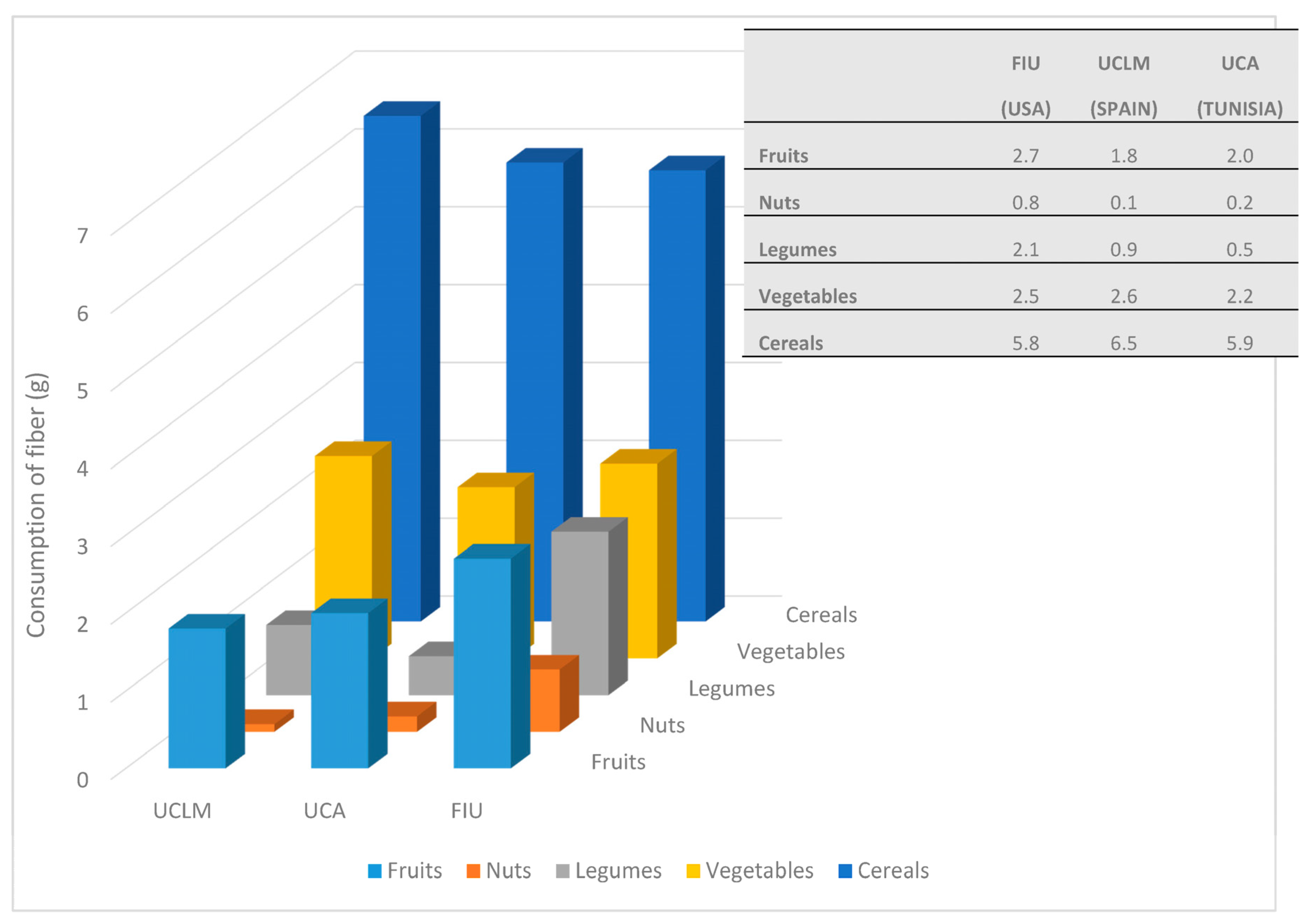
| Total sample | Cereals | 3.3 ± 1.3 (3.1–3.4) | 3.6 ± 1.1 (3.4–3.8) | 3.0 ± 1.5 (2.9–3.2) | p < 0.001 * |
| 3.0 (1.4) a | 3.6 (1.5) a,b | 2.9 (2.1) b | |||
| Legumes | 0.5 ± 1.1 (0.4–0.6) | 0.5 ± 1.1 (0.3–0.6) | 1.2 ± 2.5 (0.9–1.4) | p = 0.001 * | |
| 0.000 (0.3) c | 0.000 (0.4) | 0.000 (1.1) c | |||
| Vegetables | 1.5 ± 1.3 (1.4–1.7) | 1.5 ± 1.0 (1.4–1.7) | 1.7 ± 1.6 (1.6–1.9) | p = 0.360 | |
| 1.2 (1.6) | 1.4 (1.4) | 1.3 (1.7) | |||
| Fruits | 1.1 ± 1.4 (0.9–1.3) | 1.3 ± 1.2 (1.0–1.5) | 2.0 ± 2.4 (1.7–2.2) | p < 0.001 * | |
| 0.6 (1.7) d | 1.0 (1.6) e | 1.3 (2.2) d,e | |||
| Total intake | 8.1 ± 3.2 (7.7–8.5) | 8.4 ± 2.2 (8.0–8.7) | 10.5 ± 5.7 (9.9–11.1) | p < 0.001 * | |
| 7.5 (3.4) f | 8.0 (2.7) g | 8.9 (5.9) f,g | |||
| 0–9.29 g | Cereals | 3.2 ± 1.1 (3.0–3.3) | 3.5 ± 1.1 (3.3–3.7) | 3.0 ± 1.3 (2.8–3.1) | p < 0.001 * |
| 3.0 (1.4) h | 3.5 (1.5) h,i | 2.9 (1.8) i | |||
| Legumes | 0.2 ± 0.7 (0.2–0.3) | 0.2 ± 0.4 (0.1–0.3) | 0.2 ± 0.5 (0.2–0.3) | p = 0.224 | |
| 0.000 (0.00) | 0.000 (0.00) | 0.000 (0.04) | |||
| Vegetables | 1.2 ± 1.0 (1.1–1.4) | 1.3 ± 0.8 (1.1–1.5) | 1.2 ± 0.9 (1.1–1.4) | p = 0.670 | |
| 1.1 (1.3) | 1.2 (1.3) | 1.1 (1.2) | |||
| Fruits | 0.8 ± 1.0 (0.6–0.9) | 1.1 ± 1.0 (0.9–1.3) | 0.9 ± 0.9 (0.8–1.1) | p = 0.004 * | |
| 0.5 (1.1) j,k | 0.9 (1.4) j | 0.7 (1.3) k | |||
| Total intake | 6.7 ± 1.5 (6.5–6.9) | 7.3 ± 1.3 (7.0–7.5) | 6.7 ± 1.5 (6.5–6.9) | p = 0.003 * | |
| 6.7 (2.4) l | 7.4 (1.9) l,m | 6.8 (2.1) m | |||
| 9.30–13.99 g | Cereals | 3.7 ± 1.8 (3.3–4.2) | 3.9 ± 1.1 (3.5–4.3) | 3.1 ± 1.6 (2.8–3.4) | p = 0.007 * |
| 3.4 (1.7) | 3.9 (1.7) n | 3.0 (2.3) n | |||
| Legumes | 1.0 ± 1.4 (0.6–1.3) | 1.2 ± 1.8 (0.6–1.8) | 1.2 ± 1.8 (0.8–1.5) | p = 0.867 | |
| 0.5 (1.7) | 0.5 (1.5) | 0.000 (1.6) | |||
| Vegetables | 2.3 ± 1.7 (1.9–2.7) | 2.2 ± 1.1 (1.8–2.6) | 1.9 ± 1.5 (1.6–2.2) | p = 0.057 | |
| 2.0 (1.4) | 2.1 (1.9) | 1.5 (1.9) | |||
| Fruits | 2.0 ± 1.8 (1.5–2.4) | 1.6 ± 1.4 (1.1–2.1) | 2.5 ± 1.9 (2.1–2.9) | p = 0.027 * | |
| 2.0 (2.7) | 1.3 (2.6) o | 2.3 (2.9) o | |||
| Total intake | 11.1 ± 1.3 (10.8–11.4) | 10.9 ± 1.3 (10.5–11.4) | 11.4 ± 1.2 (11.1–11.6) | p = 0.134 | |
| 10.9 (2.2) | 10.5 (2.1) | 11.3 (2.0) | |||
| ≥14 g | Cereals | 3.5 ± 1.8 (2.2–4.8) | 4.2 ± 0.2 (2.6–5.7) | 3.1 ± 2.1 (2.6–3.6) | p = 0.366 |
| 3.5 (2.7) | 4.2 (-) | 3.1 (2.6) | |||
| Legumes | 2.2 ± 2.7 (0.3–4.1) | 0.2 ± 0.3 (-2.5–3.0) | 3.7 ± 4.2 (2.6–4.7) | p = 0.426 | |
| 1.2 (4.4) | 0.2 (-) | 2.4 (6.5) | |||
| Vegetables | 1.9 ± 1.5 (0.9–3.0) | 2.7 ± 0.6 (-3.03–8.5) | 2.9 ± 2.2 (2.3–3.4) | p = 0.378 | |
| 1.5 (1.9) | 2.7 (-) | 2.5 (2.8) | |||
| Fruits | 3.0 ± 2.4 (1.3–4.8) | 5.0 ± 1.0 (-3.6–13.6) | 4.1 ± 3.8 (3.1–5.0) | p = 0.474 | |
| 2.8 (4.2) | 5.0 (-) | 3.3 (4.0) | |||
| Total intake | 18.2 ± 6.1(13.8–22.5) | 14.7 ± 0.8 (7.3–22.2) | 19.4 ± 5.6 (18.0–20.8) | p = 0.093 | |
| 15.5 (4.4) | 14.7 (-) | 18.0 (5.6) |
| UCLM (SPAIN) | ||||||
| TOTAL | Q1 | Q2 | Q3 | Q4 | p-trend † | |
| Total | 15.7 ± 6.6 (14.9–16.5) | 9.1 ± 1.6 (8.8–9.5) | 12.8 ± 0.9 (12.6–13.0) | 16.5 ± 1.4 (16.1–16.8) | 24.3 ± 6.7 (22.6–25.9) | <0.001 * |
| 14.4 (7.4) | 9.5 (2.2) | 12.9 (1.4) | 16.4 (2.0) | 22.7 (6.9) | ||
| Fruits | 1.8 ± 2.4 (1.6–2.1) | 0.8 ± 1.1 (0.6–1.1) | 1.4 ± 1.8 (0.9–1.8) | 1.8 ± 2.2 (1.2–2.3) | 3.4 ± 3.2 (2.6–4.2) | <0.001 * |
| 0.9 (2.9) | 0.2 (1.4) | 0.8 (2.1) | 0.7 (3.6) | 2.5 (5.0) | ||
| Nuts | 0.1 ± 0.4 (0.1–0.2) | 0.1 ± 0.4 (0.0–0.2) | 0.2 ± 0.6 (0.0–0.3) | 0.1 ± 0.3 (0.0–0.2) | 0.1 ± 0.3 (0.0–0.2) | 0.168 |
| 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | ||
| Legumes | 0.9 ± 2.2 (0.7–1.2) | 0.2 ± 0.7 (0.0–0.3) | 0.2 ± 0.8 (0.04–0.4) | 0.7 ± 1.4 (0.4–1.1) | 2.6 ± 3.5 (1.8–3.5) | <0.001 * |
| 0.0 (0.8) | 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 1.6 (3.9) | ||
| Vegetables | 2.6 ± 1.9 (2.4–2.8) | 1.7 ± 1.4 (1.4–2.1) | 2.4 ± 1.9 (2.0–2.9) | 3.0 ± 2.0 (2.5–3.5) | 3.1 ± 2.0 (2.6–3.6) | <0.001 * |
| 2.2 (2.6) | 1.4 (2.2) | 2.0 (2.0) | 2.5 (3.1) | 2.6 (2.9) | ||
| Cereals | 6.5 ± 3.2(6.1–6.9) | 4.6 ± 1.7 (4.2–5.0) | 6.1 ± 2.5 (5.5–6.7) | 7.0 ± 2.9 (6.3–7.7) | 8.5 ± 4.0 (7.5–9.4) | <0.001 * |
| 6.0 (3.9) | 4.6 (2.1) | 6.0 (3.4) | 6.4 (4.7) | 8.1 (5.7) | ||
| UCA (TUNISIA) | ||||||
| TOTAL | Q1 | Q2 | Q3 | Q4 | p-trend † | |
| Total | 15.0 ± 4.8 (14.1–15.8) | 9.5 ± 1.8 (8.9–10.2) | 13.3 ± 0.8 (13.0–13.5) | 15.9 ± 0.9 (15.6–16.2) | 21.1 ± 3.8 (19.8–22.5) | <0.001 * |
| 14.5 (5.4) | 9.5 (2.3) | 13.5 (1.5) | 15.7 (1.8) | 20.1 (4.6) | ||
| Fruits | 2.0 ± 2.1 (1.7–2.4) | 0.7 ± 1.1 (0.3–1.1) | 1.9 ± 1.6 (1.3–2.4) | 2.1 ± 1.9 (1.4–2.8) | 3.5 ± 2.5 (2.6–4.4) | <0.001 * |
| 1.6 (3.0) | 0.000 (1.5) | 1.6 (2.7) | 1.7 (2.4) | 2.8 (2.9) | ||
| Nuts | 0.2 ± 0.8 (0.0–0.3) | 0.04 ± 0.2 (0.0–0.1) | 0.2 ± 0.6 (0.0–0.4) | 0.1 ± 0.4 (0.0–0.2) | 0.5 ± 1.5 (0.0–1.0) | 0.351 |
| 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | ||
| Legumes | 0.5 ± 1.1 (0.3–0.7) | 0.1 ± 0.5 (0.0–0.3) | 0.6 ± 1.0 (0.2–0.9) | 0.3 ± 0.7 (0.1–0.6) | 1.0 ± 1.7 (0.4–1.6) | 0.007 * |
| 0.0 (0.7) | 0.0 (0.0) | 0.0 (0.7) | 0.0 (0.3) | 0.0 (1.6) | ||
| Vegetables | 2.2 ± 1.5 (20.–2.5) | 1.5 ± 1.1 (1.1–1.8) | 2.0 ± 1.3 (1.6–2.5) | 2.2 ± 1.3 (1.7–2.7) | 3.3 ± 1.6 (2.7–3.9) | <0.001 * |
| 1.9 (2.2) | 1.2 (1.2) | 1.9 (2.0) | 1.8 (2.0) | 3.2 (2.5) | ||
| Cereals | 5.9 ± 2.2 (5.5–6.3) | 4.9 ± 2.0 (4.2–5.6) | 5.2 ± 1.9 (4.6–5.9) | 6.8 ± 2.3 (5.9–7.6) | 6.8 ± 2.0 (6.1–7.5) | <0.001 * |
| 6.0 (3.0) | 4.6 (3.4) | 5.4 (3.0) | 6.0 (3.2) | 6.8 (2.8) | ||
| FIU (USA) | ||||||
| TOTAL | Q1 | Q2 | Q3 | Q4 | p-trend † | |
| Total | 20.8 ± 12.0 (19.5–22.1) | 10.0 ± 2.2 (9.5–10.5) | 15.3 ± 1.5 (15.0–15.6) | 21.1 ± 2.3 (20.6–21.6) | 36.8 ± 12.8 (34.0–39.6) | <0.001 * |
| 18.1 (12.8) | 10.4 (2.9) | 15.2 (2.3) | 20.5 (4.3) | 32.9 (11.2) | ||
| Fruits | 2.7 ± 3.0 (2.4–3.0) | 1.0 ± 1.2 (0.7–1.3) | 2.1 ± 1.8 (1.7–2.5) | 3.4 ± 3.0 (2.7–4.0) | 4.3 ± 4.0 (3.4–5.2) | <0.001 * |
| 1.8 (3.0) | 0.7 (1.6) | 1.6 (2.2) | 2.5 (3.2) | 3.6 (4.8) | ||
| Nuts | 0.8 ± 1.4 (0.3–0.6) | 0.05 ± 0.3 (0–0.1) | 0.3 ± 0.9 (0.1–0.5) | 0.6 ± 1.6 (0.2–0.9) | 1.0 ± 2.0 (0.5–1.4) | <0.001 * |
| 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.9) | ||
| Legumes | 2.1 ± 4.6 (1.6–2.6) | 0.3 ± 0.7 (0.2–0.5) | 0.6 ± 1.0 (0.3–0.8) | 1.3 ± 2.6 (0.7–1.9) | 6.3 ± 7.3 (4.7–7.9) | <0.001 * |
| 0.000 (1.8) | 0.0 (0.0) | 0.0 (1.1) | 0.0 (1.8) | 3.8 (12.4) | ||
| Vegetables | 2.5 ± 2.1(2.3–2.8) | 1.7 ± 1.4 (1.4–2.00) | 2.6 ± 2.1 (2.1–3.1) | 2.9 ± 1.9 (2.4–3.3) | 3.1 ± 2.6 (2.5–3.6) | <0.001 * |
| 2.0 (2.7) | 1.5 (1.5) | 2.1 (2.8) | 2.7 (3.1) | 2.8 (3.3) | ||
| Cereals | 5.8 ± 3.1 (5.5–6.2) | 4.6 ± 2.0 (4.2–5.1) | 5.6 ± 2.9 (4.9–6.2) | 6.8 ± 3.0 (6.1–7.4) | 6.4 ± 3.7 (5.6–7.2) | <0.001 * |
| 5.3 (4.0) | 4.9 (2.6) | 5.1 (3.6) | 6.6 (4.9) | 6.5 (5.0) | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Meseguer, M.J.; Delicado-Soria, A.; Serrano-Urrea, R. Fiber Patterns in Young Adults Living in Different Environments (USA, Spain, and Tunisia). Anthropometric and Lifestyle Characteristics. Nutrients 2017, 9, 1030. https://doi.org/10.3390/nu9091030
García-Meseguer MJ, Delicado-Soria A, Serrano-Urrea R. Fiber Patterns in Young Adults Living in Different Environments (USA, Spain, and Tunisia). Anthropometric and Lifestyle Characteristics. Nutrients. 2017; 9(9):1030. https://doi.org/10.3390/nu9091030
Chicago/Turabian StyleGarcía-Meseguer, María José, Amalia Delicado-Soria, and Ramón Serrano-Urrea. 2017. "Fiber Patterns in Young Adults Living in Different Environments (USA, Spain, and Tunisia). Anthropometric and Lifestyle Characteristics" Nutrients 9, no. 9: 1030. https://doi.org/10.3390/nu9091030




