Consistency of Eating Rate, Oral Processing Behaviours and Energy Intake across Meals
Abstract
1. Introduction
- Determine the consistency of eating behaviours (defined as eating rate, oral processing, and energy intake) within individuals.
- Describe the relationship between eating rate, oral processing behaviours, and energy intake across multiple meals.
- Test whether eating rate at one meal can be used to predict eating rate and energy intake at subsequent meals.
2. Materials and Methods
2.1. Participants
2.2. Test Meals
2.3. Oral Processing Behaviours
2.4. General Procedures
2.5. Analysis
3. Results
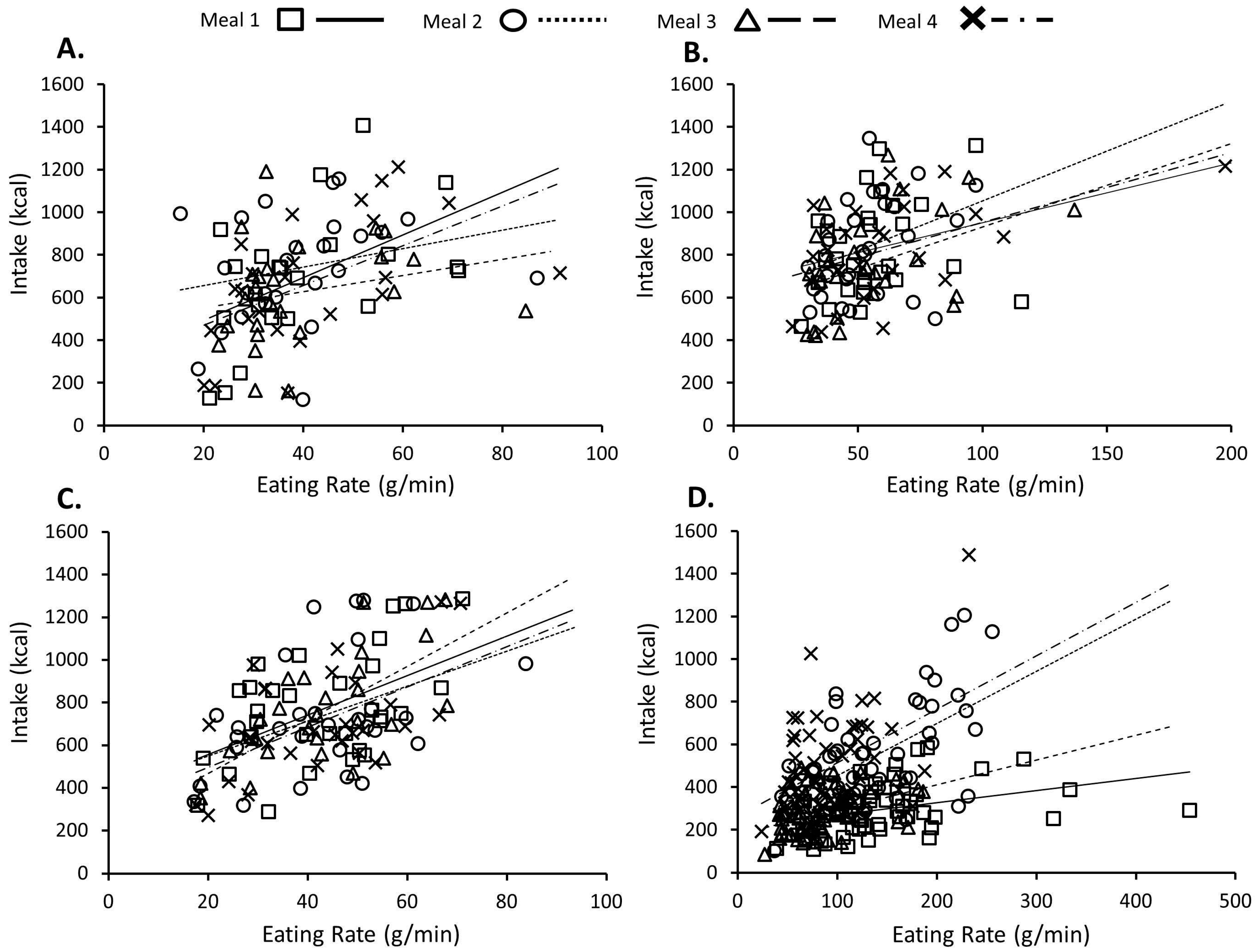
3.1. Consistency of Eating Rate, Oral Processing, and Energy Intake within Individuals
3.2. Relationship between Eating Rate, Oral Processing, and Intake over Multiple Meals
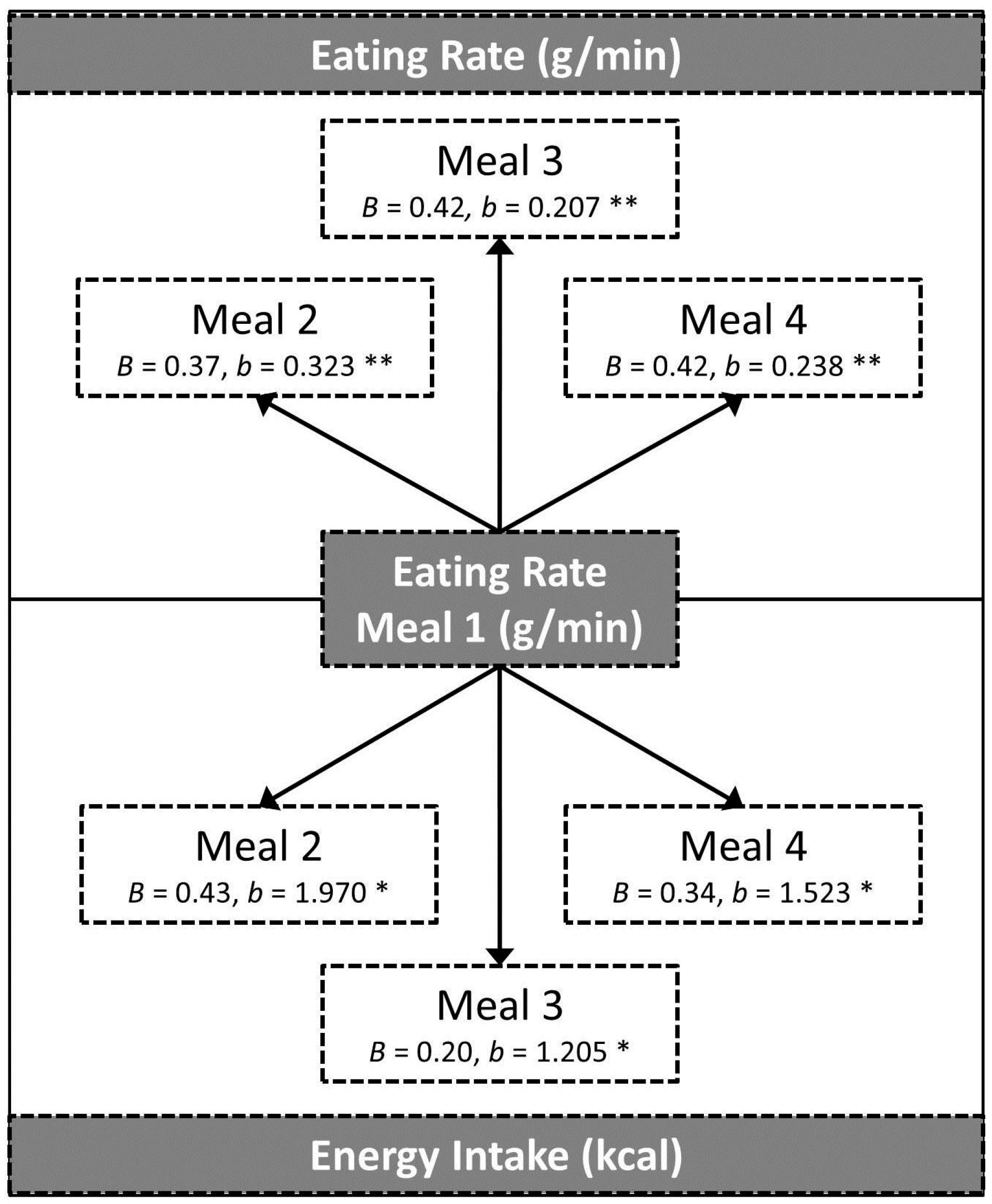
3.3. Eating Rate at One Meal as a Predictor of Eating Behaviours at Subsequent Meals
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Robinson, E.; Almiron-Roig, E.; Rutters, F.; de Graaf, C.; Forde, C.G.; Smith, C.T.; Nolan, S.J.; Jebb, S.A. A systematic review and meta-analysis examining the effect of eating rate on energy intake and hunger. Am. J. Clin. Nutr. 2014, 100, 123–151. [Google Scholar] [CrossRef] [PubMed]
- Ohkuma, T.; Hirakawa, Y.; Nakamura, U.; Kiyohara, Y.; Kitazono, T.; Ninomiya, T. Association between eating rate and obesity: A systematic review and meta-analysis. Int. J. Obes. 2015, 39, 1589–1596. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Haruyama, Y.; Muto, T.; Yamazaki, T. Association Between Eating Speed and Metabolic Syndrome in a Three-Year Population-Based Cohort Study. J. Epidemiol. 2015, 25, 332–336. [Google Scholar] [CrossRef] [PubMed]
- Ford, A.L.; Bergh, C.; Södersten, P.; Sabin, M.A.; Hollinghurst, S.; Hunt, L.P.; Shield, J.P.H. Treatment of childhood obesity by retraining eating behaviour: Randomised controlled trial. BMJ 2010, 340, b5388. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, N.; Hu, L.Z.; Li, Z.; Li, R.; Li, C.; Wang, S. Improvement in chewing activity reduces energy intake in one meal and modulates plasma gut hormone concentrations in obese and lean young Chinese men. Am. J. Clin. Nutr. 2011, 94, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Scisco, J.L.; Muth, E.R.; Dong, Y.J.; Hoover, A.W. Slowing Bite-Rate Reduces Energy Intake: An Application of the Bite Counter Device. J. Am. Diet. Assoc. 2011, 111, 1231–1235. [Google Scholar] [CrossRef] [PubMed]
- Smit, H.J.; Kemsley, E.K.; Tapp, H.S.; Henry, J.K. Does prolonged chewing reduce food intake? Fletcherism revisited. Appetite 2011, 57, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Zandian, M.; Loakimidis, L.; Bergh, C.; Brodin, U.; Södersten, P. Decelerated and linear eaters: Effect of eating rate on food intake and satiety. Physiol. Behav. 2009, 96, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, K.; Sato, S.; Ohira, T.; Maeda, K.; Noda, H.; Kubota, Y.; Nishimura, S.; Kitamura, A.; Kiyama, M.; Okada, T.; et al. The joint impact on being overweight of self reported behaviours of eating quickly and eating until full: Cross sectional survey. BMJ 2008, 337, a2002. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, R.; Tamakoshi, K.; Yatsuya, H.; Murata, C.; Sekiya, A.; Wada, K.; Zhang, H.M.; Matsushita, K.; Sugiura, K.; Takefuji, S.; et al. Eating fast leads to obesity: Findings based on self-administered questionnaires among middle-aged Japanese men and women. J. Epidemiol. 2006, 16, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, S.; Katagiri, A.; Tsuji, T.; Shimoda, T.; Amano, K. Self-reported rate of eating correlates with body mass index in 18-y-old Japanese women. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 1405–1410. [Google Scholar] [CrossRef] [PubMed]
- Tanihara, S.; Imatoh, T.; Miyazaki, M.; Babazono, A.; Momose, Y.; Baba, M.; Uryu, Y.; Une, H. Retrospective longitudinal study on the relationship between 8-year weight change and current eating speed. Appetite 2011, 57, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Bellisle, F.; Le Magnen, J. The structure of meals in humans: Eating and drinking patterns in lean and obese subjects. Physiol. Behav. 1981, 27, 649–658. [Google Scholar] [CrossRef]
- Laessle, R.G.; Lehrke, S.; Dückers, S. Laboratory eating behavior in obesity. Appetite 2007, 49, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Wagner, M.; Hewitt, M.I. Oral satiety in the obese and nonobese. J. Am. Diet. Assoc. 1975, 67, 344–346. [Google Scholar] [PubMed]
- Kaplan, D.L. Eating style of obese and nonobese males. Psychosom. Med. 1980, 42, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, T.A. Rate of intake, bites, and chews-the interpretation of lean-obese differences. Neurosci. Biobehav. Rev. 2000, 24, 229–237. [Google Scholar] [CrossRef]
- Spiegel, T.A.; Kaplan, J.M.; Tomassini, A.; Stellar, E. Bite size, ingestion rate, and meal size in lean and obese women. Appetite 1993, 21, 131–145. [Google Scholar] [CrossRef]
- Warner, K.E.; Balagura, S. Intrameal eating patterns of obese and nonobese humans. J. Comp. Physiol. Psychol. 1975, 89, 778–783. [Google Scholar] [CrossRef] [PubMed]
- Fogel, A.; Goh, A.T.; Fries, L.R.; Sadananthan, S.A.; Velan, S.S.; Michael, N.; Tint, M.T.; Fortier, M.V.; Fortier, M.J.; Toh, J.Y.; et al. A description of an ‘obesogenic’ eating style that promotes higher energy intake and is associated with greater adiposity in 4.5year-old children: Results from the GUSTO cohort. Physiol. Behav. 2017, 176, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Guy-Grand, B.; Lehnert, V.; Doassans, M.; Bellisle, F. Type of test-meal affects palatability and eating style in humans. Appetite 1994, 22, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Hill, S.W. Eating responses of humans during dinner meals. J. Comp. Physiol. Psychol. 1974, 86, 652–657. [Google Scholar] [CrossRef] [PubMed]
- Barkeling, B.; Rossner, S.; Sjoberg, A. Methodological studies on single meal food intake characteristics in normal weight and obese men and women. Int. J. Obes. Relat. Metab. Disord. 1995, 19, 284–290. [Google Scholar] [PubMed]
- Hubel, R.; Laessle, R.G.; Lehrke, S.; Jass, J. Laboratory measurement of cumulative food intake in humans: Results on reliability. Appetite 2006, 46, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Sadoul, B.C.; Schuring, E.A.H.; Mela, D.J.; Peters, H.P.F. The relationship between appetite scores and subsequent energy intake: An analysis based on 23 randomized controlled studies. Appetite 2014, 83, 153–159. [Google Scholar] [CrossRef] [PubMed]
- McCrickerd, K.; Forde, C.G. Sensory influences on food intake control: Moving beyond palatability. Obes. Rev. 2016, 17, 18–29. [Google Scholar] [CrossRef] [PubMed]
- McCrickerd, K.; Lim, C.M.H.; Leong, C.; Chia, E.M.; Forde, C.G. Texture-Based Differences in Eating Rate Reduce the Impact of Increased Energy Density and Large Portions on Meal Size in Adults. J. Nutr. 2017, 147, 1208–1217. [Google Scholar] [CrossRef] [PubMed]
- McCrickerd, K.; Salleh, N.B.; Forde, C.G. Removing energy from a beverage influences later food intake more than the same energy addition. Appetite 2016, 105, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Salleh, N.B.; Henry, J.; Forde, C.G. Effects of aspartame-, monk fruit-, stevia- and sucrose-sweetened beverages on postprandial glucose, insulin and energy intake. Int. J. Obes. 2017, 41, 450–457. [Google Scholar] [CrossRef] [PubMed]
- De Lauzon, B.; Romon, M.; Deschamps, V.; Lafay, L.; Borys, J.-M.; Karlsson, J.; Ducimetière, P.; Charles, M.A. Fleurbaix Laventie Ville Sante (FLVS) Study Group The Three-Factor Eating Questionnaire-R18 is able to distinguish among different eating patterns in a general population. J. Nutr. 2004, 134, 2372–2380. [Google Scholar] [PubMed]
- Forde, C.G.; Leong, C.; Chia-Ming, E.; McCrickerd, K. Fast or slow-foods? Describing natural variations in oral processing characteristics across a wide range of Asian foods. Food Funct. 2017, 8, 595–606. [Google Scholar] [CrossRef] [PubMed]
- Lausberg, H.; Sloetjes, H. Coding gestural behavior with the NEUROGES-ELAN system. Behav. Res. Methods Instrum. Comput. 2009, 41, 841–849. [Google Scholar] [CrossRef] [PubMed]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Forde, C.G.; van Kuijk, N.; Thaler, T.; de Graaf, C.; Martin, N. Oral processing characteristics of solid savoury meal components, and relationship with food composition, sensory attributes and expected satiation. Appetite 2013, 60, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Ello-Martin, J.A.; Roe, L.S.; Ledikwe, J.H.; Beach, A.M.; Rolls, B.J. Dietary energy density in the treatment of obesity: A year-long trial comparing 2 weight-loss diets. Am. J. Clin. Nutr. 2007, 85, 1465–1477. [Google Scholar] [PubMed]
- Raynor, H.A.; Van Walleghen, E.L.; Bachman, J.L.; Looney, S.M.; Phelan, S.; Wing, R.R. Dietary energy density and successful weight loss maintenance. Eat. Behav. 2011, 12, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Saquib, N.; Natarajan, L.; Rock, C.L.; Flatt, S.W.; Madlensky, L.; Kealey, S.; Pierce, J.P. The Impact of a Long-Term Reduction in Dietary Energy Density on Body Weight within a Randomized Diet Trial. Nutr. Cancer 2008, 60, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Bolhuis, D.P.; Forde, C.G.; Cheng, Y.J.; Xu, H.H.; Martin, N.; de Graaf, C. Slow Food: Sustained Impact of Harder Foods on the Reduction in Energy Intake over the Course of the Day. PLoS ONE 2014, 9, e93370. [Google Scholar] [CrossRef] [PubMed]
- Zlatevska, N.; Dubelaar, C.; Holden, S.S. Sizing up the Effect of Portion Size on Consumption: A Meta-Analytic Review. J. Mark. 2014, 78, 140–154. [Google Scholar] [CrossRef]

| N = 146 | Study 1 | Study 2 | Study 3 | Study 4 | p 3 | |
|---|---|---|---|---|---|---|
| Male (n = 26) | Male (n = 30) | Male (n = 34) | Male (n = 27) | Female (n = 29) | ||
| Age (years) | 27 ± 5 a (21–37) | 28 ± 5 a (21–41) | 29 ± 7 a (21–48) | 25 ± 3 a,b (21–36) | 23 ± 3 b (21–39) | 0.001 |
| BMI (kg/m2) | 23 ± 3 (18–29) | 22 ± 2 a (19–25) | 22 ± 2 a (18–25) | 23 ± 3 a (18–28) | 21 ± 3 a (16–29) | 0.043 |
| Fat Mass (%) 1 | 21 ± 7 b (7–34) | 18 ± 8 b,c (3–33) | 16 ± 4 c (8–24) | 17 ± 5 b,c (9–27) | 29 ± 6 a (18–43) | 0.001 |
| Dietary Restraint (0–100%) 2 | 36 ± 21 a,b (0–81) | 33 ± 25 b (0–81) | 35 ± 23 a,b (0–95) | 52 ± 21 a (11–100) | 46 ± 27 a,b (6–95) | 0.012 |
| Eating Behaviour | Study 1 | Study 2 | Study 3 | Study 4 | Combined |
|---|---|---|---|---|---|
| Intake (kcal) | 0.97 [0.94, 0.98] | 0.94 [0.90, 0.97] | 0.96 [0.92, 0.98] | 0.88 [0.82, 0.92] | 0.95 [0.94, 0.96] |
| Eating rate (g/min) | 0.95 [0.90, 0.98] | 0.87 [0.75, 0.94] | 0.95 [0.91, 0.98] | 0.77 [0.65, 0.86] | 0.87 [0.83, 0.91] |
| Bite size (g) | 0.81 [0.62, 0.92] | 0.84 [0.70, 0.92] | 0.95 [0.91, 0.98] | 0.76 [0.63, 0.85] | 0.80 [0.74, 0.85] |
| Oral exposure per bite (s) | 0.82 [0.64, 0.92] | 0.97 [0.94, 0.99] | 0.94 [0.88, 0.97] | 0.83 [0.74, 0.89] | 0.93 [0.74, 0.89] |
| Chews per gram (chews/g) | 0.95 [0.96, 0.98] | 0.96 [0.92, 0.98] | 0.96 [0.93, 0.98] | 0.85 [0.77, 0.90] | 0.96 [0.95, 0.97] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McCrickerd, K.; Forde, C.G. Consistency of Eating Rate, Oral Processing Behaviours and Energy Intake across Meals. Nutrients 2017, 9, 891. https://doi.org/10.3390/nu9080891
McCrickerd K, Forde CG. Consistency of Eating Rate, Oral Processing Behaviours and Energy Intake across Meals. Nutrients. 2017; 9(8):891. https://doi.org/10.3390/nu9080891
Chicago/Turabian StyleMcCrickerd, Keri, and Ciaran G. Forde. 2017. "Consistency of Eating Rate, Oral Processing Behaviours and Energy Intake across Meals" Nutrients 9, no. 8: 891. https://doi.org/10.3390/nu9080891
APA StyleMcCrickerd, K., & Forde, C. G. (2017). Consistency of Eating Rate, Oral Processing Behaviours and Energy Intake across Meals. Nutrients, 9(8), 891. https://doi.org/10.3390/nu9080891





