In Vitro and In Vivo Evaluation of Lactobacillus delbrueckii subsp. bulgaricus KLDS1.0207 for the Alleviative Effect on Lead Toxicity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Bacterial Strain and Culture
2.3. Estimation of Pb Binding and Pb Tolerance
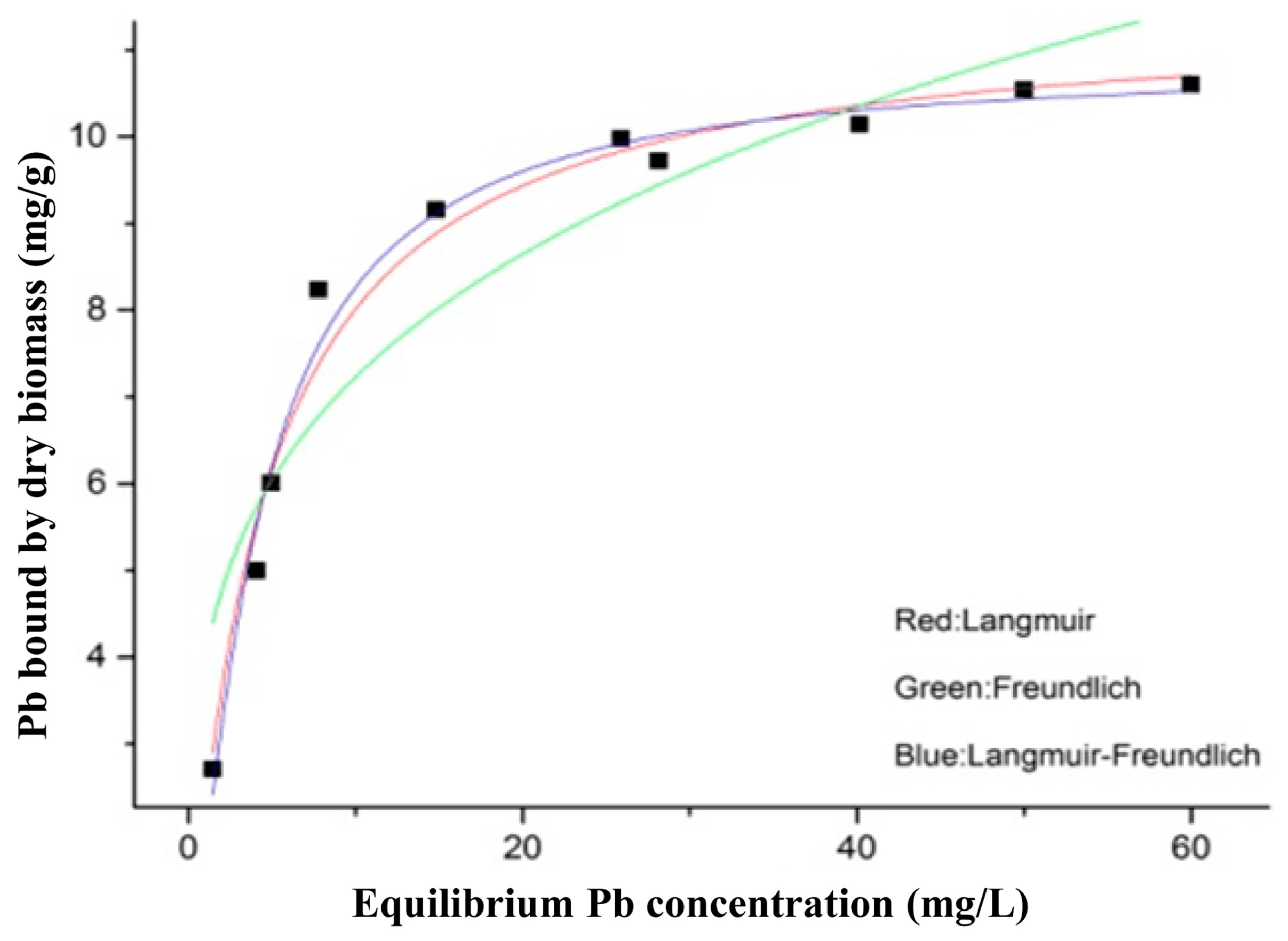
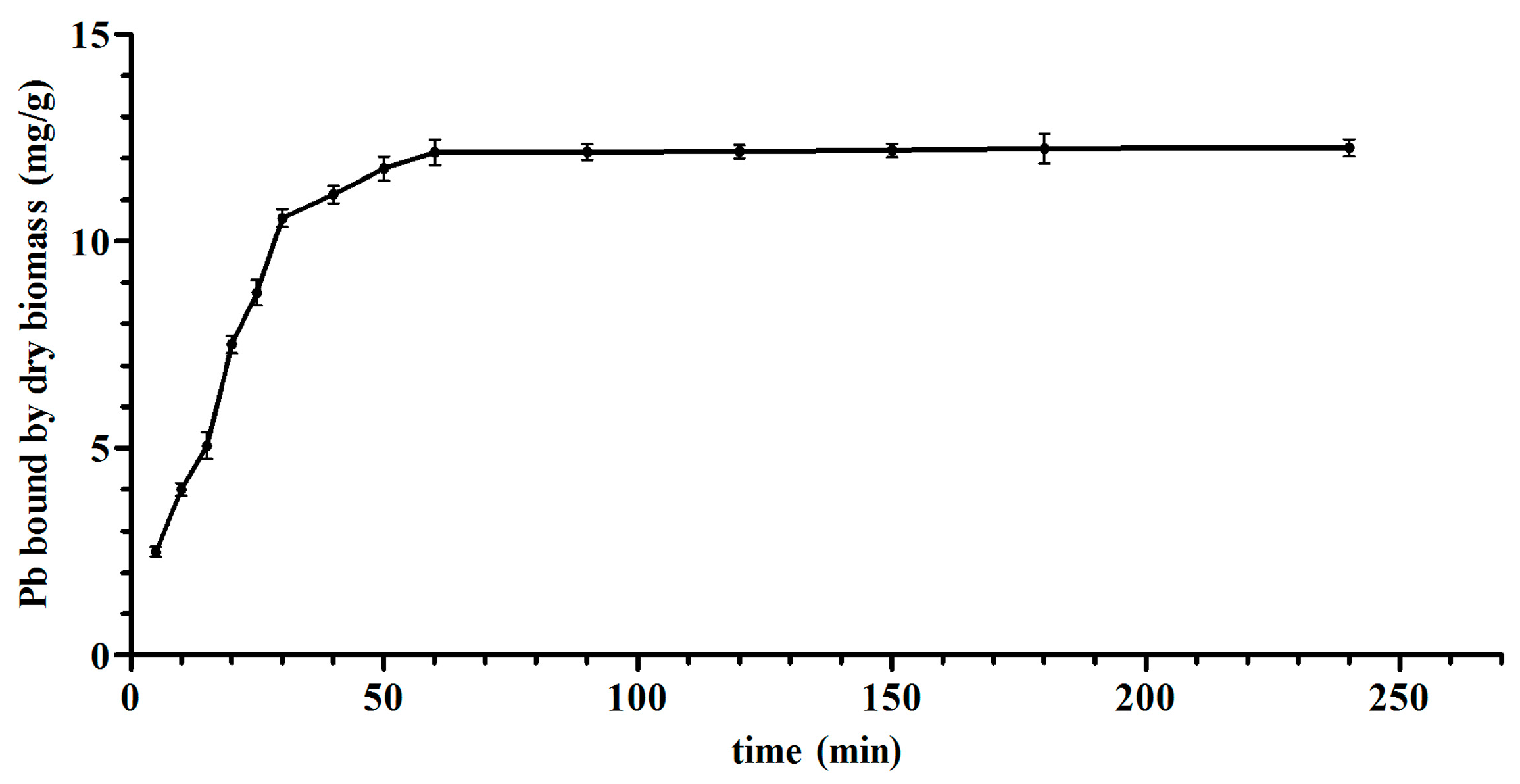
2.4. Equilibrium Isotherm and Kinetic Study
2.5. Scanning Electron Microscopy (SEM) Analysis
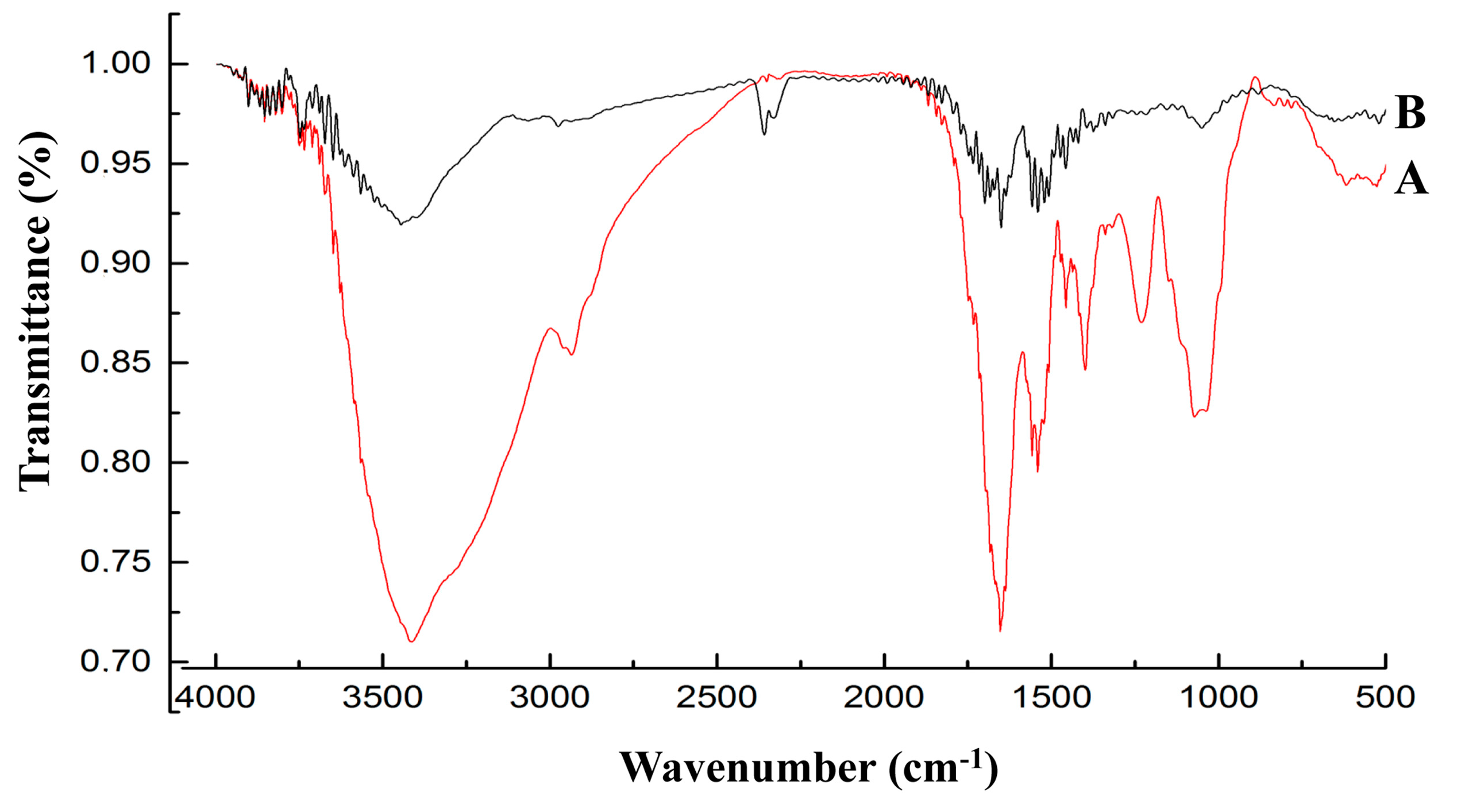
2.6. Fourier Transform Infrared Spectroscopy (FT-IR) Analysis
2.7. In Vivo Protective Potential of L. bulgaricus KLDS1.0207 Against Acute Pb Toxicity
2.7.1. Animals and Experimental Design
2.7.2. Determination of Pb in Blood, Feces and Tissue
2.7.3. Biochemical Assays
2.7.4. Histopathological Studies
2.8. Statistical Analysis
3. Results
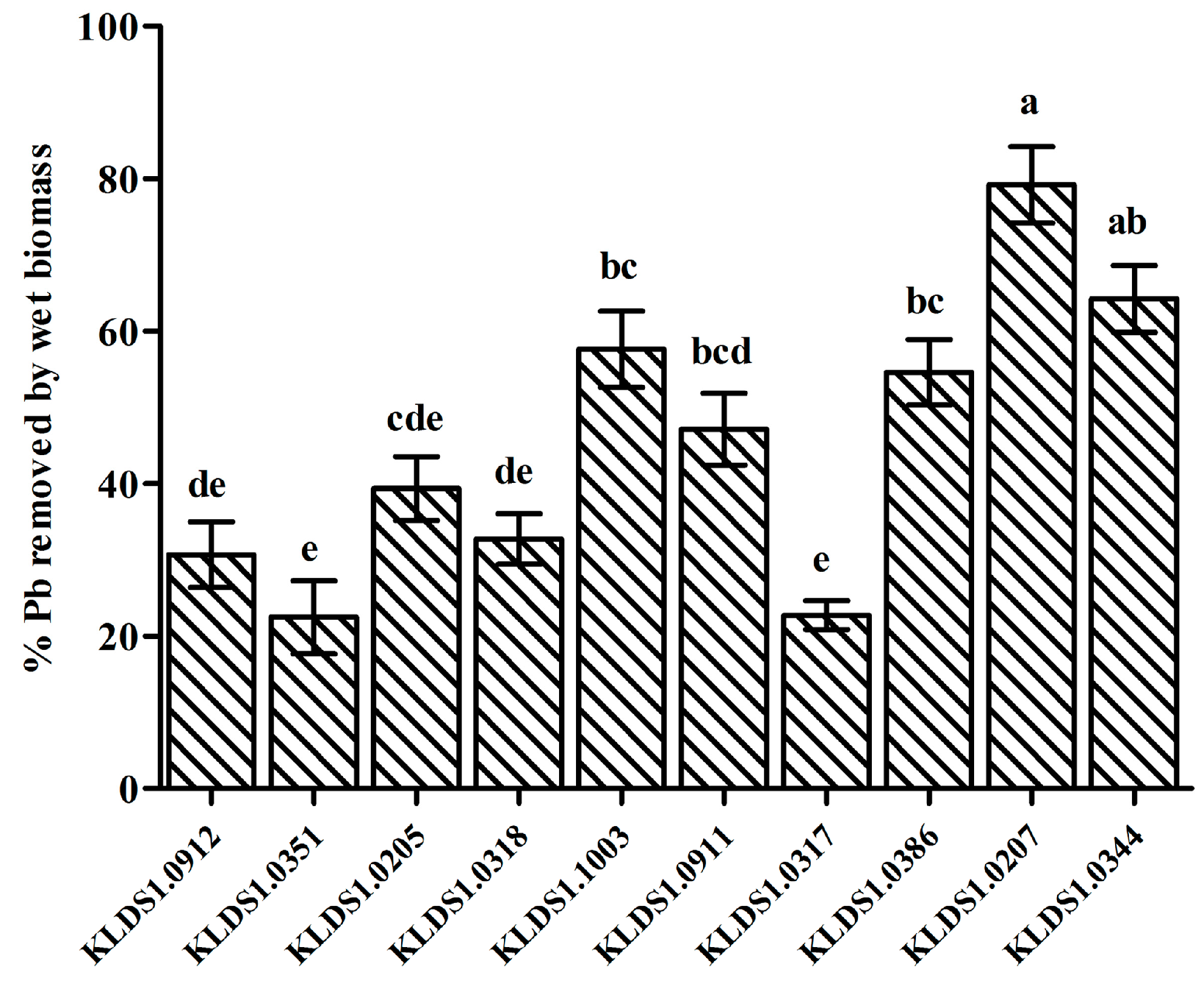
3.1. Pb Biosorption and Pb Tolerance of LAB Strains
3.2. Biosorption Isotherms and Kinetic Models
3.3. Electron Microscopy Analysis
3.4. FT-IR Analysis
3.5. Mortality and Viscera Index Analysis
3.6. Pb Levels in Feces and Tissues
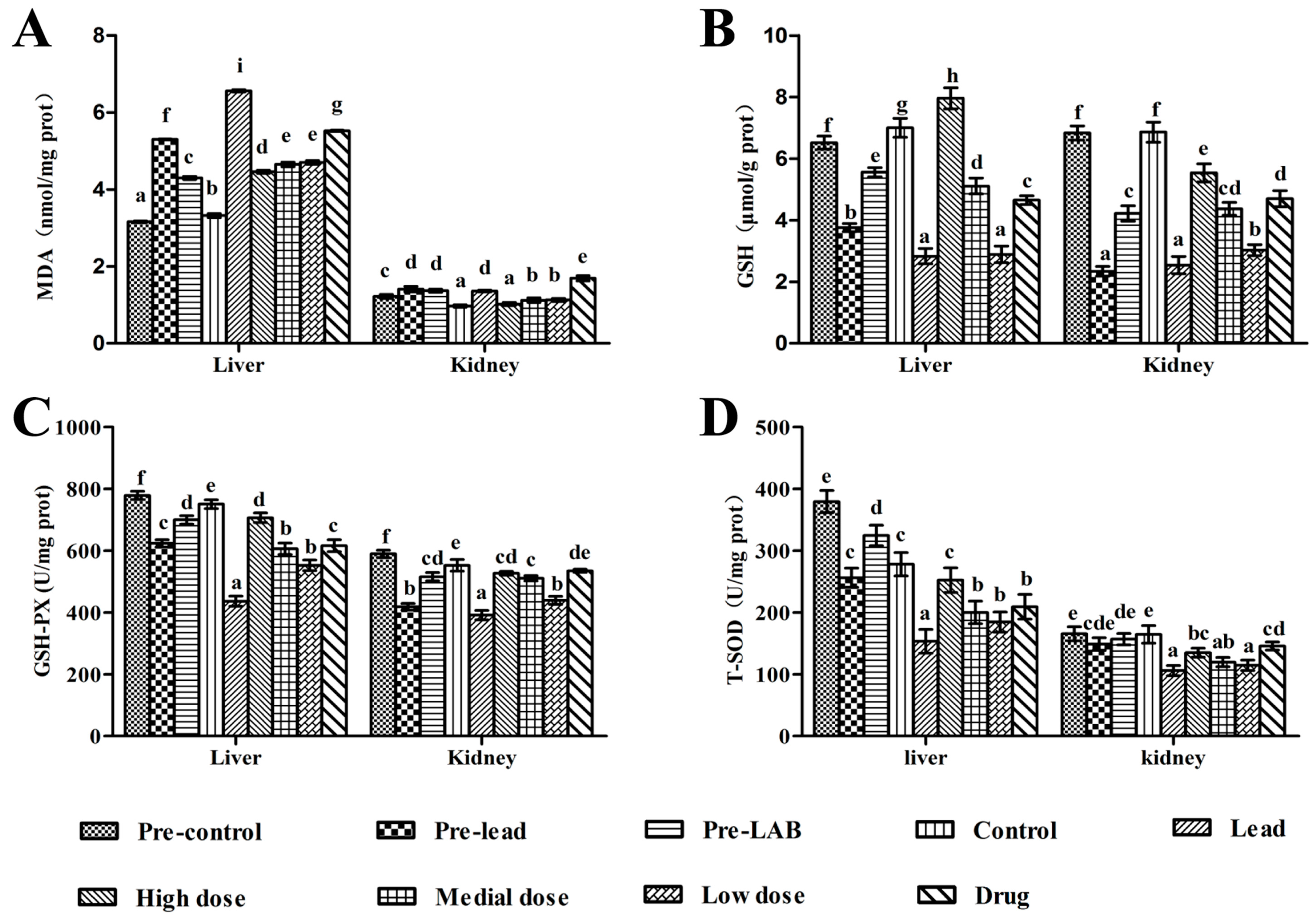
3.7. Activity of Antioxidant Enzymes
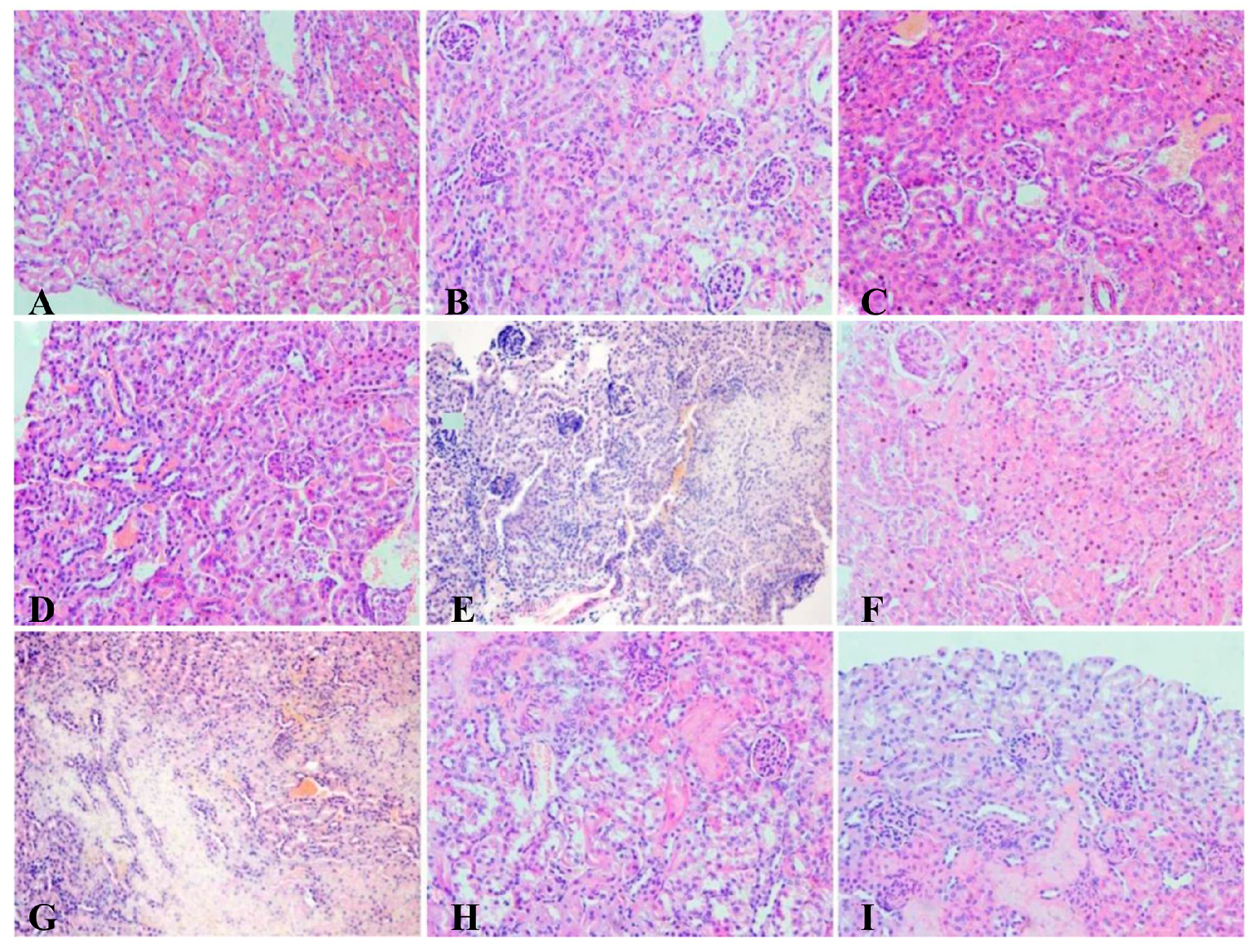
3.8. Histopathology
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fewtrell, L.; Prüss-Üstün, A.; Landrigan, P.; Ayuso-Mateos, J. Estimating the global burden of disease of mild mental retardation and cardiovascular diseases from environmental lead exposure. Environ. Res. 2004, 94, 120–133. [Google Scholar] [CrossRef]
- Okoro, K.I.; Igene, J.O.; Ebabhamiegbebho, P.A.; Evivie, S.E. Lead (Pb) and cadmium (Cd) levels in fresh and smoke-dried grasscutter (Thryonomis swinderianus Temminck) meat. Afr. J. Agric. Res. 2015, 10, 3116–3122. [Google Scholar]
- Mielke, H.W. Research ethics in pediatric environmental health: Lessons from lead. Neurotoxicol. Teratol. 2002, 24, 467–469. [Google Scholar] [CrossRef]
- Tangpong, J.; Satarug, S. Alleviation of lead poisoning in the brain with aqueous leaf extract of the Thunbergia laurifolia (Linn.). Toxicol. Lett. 2010, 198, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Struzyńska, L.; Dabrowska-Bouta, B.; Rafałowska, U. Acute lead toxicity and energy metabolism in rat brain synaptosomes. Acta Neurobiol. Exp. 1996, 57, 275–281. [Google Scholar]
- Patra, R.; Swarup, D.; Dwivedi, S. Antioxidant effects of α tocopherol, ascorbic acid and L-methionine on lead induced oxidative stress to the liver, kidney and brain in rats. Toxicology 2001, 162, 81–88. [Google Scholar] [CrossRef]
- Khordad, E.; Fazel, A.; Bideskan, A.E. The effect of ascorbic acid and garlic administration on lead-induced apoptosis in rat offspring’s eye retina. Iran. Biomed. J. 2013, 17, 206. [Google Scholar] [PubMed]
- Valverde, M.; Fortoul, T.I.; Díaz-Barriga, F.; Mejía, J.; del Castillo, E.R. Genotoxicity induced in CD-1 mice by inhaled lead: Differential organ response. Mutagenesis 2002, 17, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Sansar, W.; Bouyatas, M.M.; Ahboucha, S.; Gamrani, H. Effects of chronic lead intoxication on rat serotoninergic system and anxiety behavior. Acta Histochem. 2012, 114, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Sansar, W.; Ahboucha, S.; Gamrani, H. Chronic lead intoxication affects glial and neural systems and induces hypoactivity in adult rat. Acta Histochem. 2011, 113, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Goyer, R.A.; Clarkson, T.W. Toxic effects of metals. In The Basic Science of Poisons, 5th ed.; Klaassen, C., Ed.; McGraw-Hill Health Professions Division: New York, NY, USA, 2007; Volume 5, pp. 811–867. [Google Scholar]
- Rentschler, G.; Broberg, K.; Lundh, T.; Skerfving, S. Long-term lead elimination from plasma and whole blood after poisoning. Int. Arch. Occup. Environ. Health 2012, 85, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Ahyayauch, H.; Sansar, W.; Rendón-Ramírez, A.; Goñi, F.M.; Bennouna, M.; Gamrani, H. Effects of chronic and acute lead treatments on the biophysical properties of erythrocyte membranes, and a comparison with model membranes. FEBS Open Bio 2013, 3, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, A.; Sharifi, A.M.; Abdollahi, M.; Najafi, R.; Baeeri, M.; Rayegan, S.; Cheshmehnour, J.; Hassani, S.; Bayrami, Z.; Safa, M. Cerium and yttrium oxide nanoparticles against lead-induced oxidative stress and apoptosis in rat hippocampus. Biol. Trace Element Res. 2015, 164, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Flora, G.J.; Seth, P.K. Beneficial effects of S-Adenosyl-L-Methionine on aminolevulinic acid dehydratase, glutathione, and lipid peroxidation during acute lead—Ethanol administration in mice. Alcohol 1999, 18, 103–108. [Google Scholar] [CrossRef]
- Gründlingh, J. Martindale: The complete drug reference 38th edition. J. Forensic Legal Med. 2014, 28, 54. [Google Scholar] [CrossRef]
- Bradberry, S.; Vale, A. A comparison of sodium calcium edetate (edetate calcium disodium) and succimer (DMSA) in the treatment of inorganic lead poisoning. Clin. Toxicol. 2009, 47, 841–858. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.; Mathur, R.; Samim, M.; Lomash, V.; Kushwaha, P.; Pathak, U.; Babbar, A.K.; Flora, S.J.S.; Mishra, A.K.; Kaushik, M.P. Nanoencapsulation of DMSA monoester for better therapeutic efficacy of the chelating agent against arsenic toxicity. Nanomedicine 2014, 9, 465–481. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Q.; Yin, R.; Yu, L.; Wang, G.; Tian, F.; Yu, R.; Zhao, J.; Liu, X.; Chen, Y.Q.; Zhang, H. Screening of lactic acid bacteria with potential protective effects against cadmium toxicity. Food Control 2015, 54, 23–30. [Google Scholar] [CrossRef]
- Milanowski, M.; Pomastowski, P.; Railean-Plugaru, V.; Rafińska, K.; Ligor, T.; Buszewski, B. Biosorption of silver cations onto Lactococcus lactis and Lactobacillus casei isolated from dairy products. PLoS ONE 2017, 12, e0174521. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.-J.; Lim, J.-M.; Gu, S.; Lee, W.-K.; Oh, E.; Lee, S.-M.; Oh, B.-T. Potential use of lactic acid bacteria Leuconostoc mesenteroides as a probiotic for the removal of pb (II) toxicity. J. Microbiol. 2017, 55, 296–303. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Q.; Wang, G.; Zhao, J.; Liu, X.; Tian, F.; Zhang, H.; Chen, W. Protective effects of Lactobacillus plantarum CCFM8610 against acute cadmium toxicity in mice. Appl. Environ. Microbiol. 2013, 79, 1508–1515. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Q.; Wang, G.; Zhao, J.; Liu, X.; Narbad, A.; Chen, Y.Q.; Zhang, H.; Tian, F.; Chen, W. Protective effects of Lactobacillus plantarum CCFM8610 against chronic cadmium toxicity in mice: Intestinal sequestration is not the only route of protection. Appl. Environ. Microbiol. 2014, 14, 00762. [Google Scholar]
- Tian, F.; Zhai, Q.; Zhao, J.; Liu, X.; Wang, G.; Zhang, H.; Zhang, H.; Chen, W. Lactobacillus plantarum CCFM8661 alleviates lead toxicity in mice. Biol. Trace Element Res. 2012, 150, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Topcu, A.; Bulat, T. Removal of cadmium and lead from aqueous solution by Enterococcus faecium strains. J. Food Sci. 2010, 75, T13–T17. [Google Scholar] [CrossRef] [PubMed]
- Abou-Shanab, R.; Van Berkum, P.; Angle, J. Heavy metal resistance and genotypic analysis of metal resistance genes in gram-positive and gram-negative bacteria present in Ni-rich serpentine soil and in the rhizosphere of alyssum murale. Chemosphere 2007, 68, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Chakravarty, R.; Banerjee, P.C. Mechanism of cadmium binding on the cell wall of an acidophilic bacterium. Bioresour. Technol. 2012, 108, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraghavan, K.; Padmesh, T.; Palanivelu, K.; Velan, M. Biosorption of nickel (II) ions onto sargassum wightii: Application of two-parameter and three-parameter isotherm models. J. Hazard. Mater. 2006, 133, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Das, S.K.; Chakravarty, R.; Chakrabarti, A.; Ghosh, S.; Guha, A.K. Interaction of malathion, an organophosphorus pesticide with Rhizopus oryzae biomass. J. Hazard. Mater. 2010, 174, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Chakravarty, R.; Banerjee, P.C. Morphological changes in an acidophilic bacterium induced by heavy metals. Extremophiles 2008, 12, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Azar, A.; Trochimowicz, H.; Maxfield, M. Review of Lead Studies in Animals Carried Out at Haskell Laboratory: Two-Year Feeding Study and Response to Hemorrhage Study. In Environmental Health Aspects of Lead: Proceedings of an International Symposium; Commission of the European Communities: Luxembourg, 1972; pp. 199–210. [Google Scholar]
- Flora, S.J.; Pande, M.; Mehta, A. Beneficial effect of combined administration of some naturally occurring antioxidants (vitamins) and thiol chelators in the treatment of chronic lead intoxication. Chem. Biol. Interact. 2003, 145, 267–280. [Google Scholar] [CrossRef]
- Monachese, M.; Burton, J.P.; Reid, G. Bioremediation and tolerance of humans to heavy metals through microbial processes: A potential role for probiotics? Appl. Environ. Microbiol. 2012, 78, 6397–6404. [Google Scholar] [CrossRef] [PubMed]
- Passot, S.; Gautier, J.; Jamme, F.; Cenard, S.; Dumas, P.; Fonseca, F. Understanding the cryotolerance of lactic acid bacteria using combined synchrotron infrared and fluorescence microscopies. Analyst 2015, 140, 5920–5928. [Google Scholar] [CrossRef] [PubMed]
- Schär-Zammaretti, P.; Ubbink, J. The cell wall of lactic acid bacteria: Surface constituents and macromolecular conformations. Biophys. J. 2003, 85, 4076–4092. [Google Scholar] [CrossRef]
- Mishra, V.; Balomajumder, C.; Agarwal, V.K. Dynamic, mechanistic, and thermodynamic modeling of Zn (II) ion biosorption onto zinc sequestering bacterium VMSDCM. CLEAN Soil Air Water 2013, 41, 883–889. [Google Scholar] [CrossRef]
- Kazy, S.K.; D’souza, S.; Sar, P. Uranium and thorium sequestration by a Pseudomonas sp.: Mechanism and chemical characterization. J. Hazard. Mater. 2009, 163, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Dong, F.; Yan, X.; Zeng, W.; Hou, L.; Pang, X. Biosorption of uranium by saccharomyces cerevisiae and surface interactions under culture conditions. Bioresour. Technol. 2010, 101, 8573–8580. [Google Scholar] [CrossRef] [PubMed]
- Ren, G.; Jin, Y.; Zhang, C.; Gu, H.; Qu, J. Characteristics of Bacillus sp. PZ-1 and its biosorption to pb (II). Ecotoxicol. Environ. Saf. 2015, 117, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Qu, J.; Li, Y.; Gu, H.; Yan, L.; Sun, X. Isolation, identification and pb (ii) biosorption characterization of a lead-resistant strain. Acta Sci. Circumst. 2013, 33, 2248–2255. [Google Scholar]
- Tian, F.; Xiao, Y.; Li, X.; Zhai, Q.; Wang, G.; Zhang, Q.; Zhang, H.; Chen, W. Protective effects of Lactobacillus plantarum CCFM8246 against copper toxicity in mice. PLoS ONE 2015, 10, e0143318. [Google Scholar] [CrossRef] [PubMed]
- Patra, R.; Swarup, D. Effect of lead on erythrocytic antioxidant defence, lipid peroxide level and thiol groups in calves. Res. Vet. Sci. 2000, 68, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Jomova, K.; Valko, M. Advances in metal-induced oxidative stress and human disease. Toxicology 2011, 283, 65–87. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Morris, H.; Cronin, M. Metals, toxicity and oxidative stress. Curr. Med. Chem. 2005, 12, 1161–1208. [Google Scholar] [CrossRef] [PubMed]
- Odewabi, A.O.; Ekor, M. Levels of heavy and essential trace metals and their correlation with antioxidant and health status in individuals occupationally exposed to municipal solid wastes. Toxicol. Ind. Health 2016. [Google Scholar] [CrossRef] [PubMed]
- Vijayavel, K.; Balasubramanian, M.P. Fluctuations of biochemical constituents and marker enzymes as a consequence of naphthalene toxicity in the edible estuarine crab Scylla serrata. Ecotoxicol. Environ. Saf. 2006, 63, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Agrahari, S.; Gopal, K. Retracted: Fluctuations of certain biochemical constituents and markers enzymes as a consequence of monocrotophos toxicity in the edible freshwater fish, channa punctatus. Pestic. Biochem. Physiol. 2009, 94, 5–9. [Google Scholar] [CrossRef]
- Klobukowski, J.; Modzelewska-Kapitula, M.; Kornacki, K. Calcium bioavailability from diets based on white cheese containing probiotics or synbiotics in short-time study in rats. Pak. J. Nutr. 2009, 8, 933–936. [Google Scholar] [CrossRef]
- Kruger, M.C.; Fear, A.; Chua, W.-H.; Plimmer, G.G.; Schollum, L.M. The effect of Lactobacillus rhamnosus HN001 on mineral absorption and bone health in growing male and ovariectomised female rats. Dairy Sci. Technol. 2009, 89, 219–231. [Google Scholar] [CrossRef]

| Groups | Treatment on the Indicated Day(s) for the Following: | |
|---|---|---|
| Prevention groups | 1–14 days | 15 day |
| Pre-control (n = 8) | SM | PW |
| Pre-Pb (n = 8) | SM | Pb |
| Pre-LAB (n = 8) | SM + LAB | Pb |
| Therapy groups | 1 day | 2–15 days |
| Control (n = 8) | PW | SM |
| Pb (n = 8) | Pb | SM |
| High dose (n = 8) | Pb | SM + LAB |
| Medial dose (n = 8) | Pb | SM + LAB |
| Low dose (n = 8) | Pb | SM + LAB |
| Drug (n = 8) | Pb | DMSA(50 mg/kg/day) |
| Strains | Minimum Inhibitory Concentration for Pb (mg/L) |
|---|---|
| L. bulgaricus KLDS1.0207 | >1000 |
| L. bulgaricus KLDS1.9201 | 200 |
| L. helveticus KLDS1.0903 | 350 |
| L. acidophilus KLDS1.1003 | 300 |
| L. plantarum KLDS1.0386 | 150 |
| L. plantarum KLDS1.0344 | 450 |
| L. rhamnosus KLDS1.0205 | 100 |
| L. rhamnosus KLDS1.0911 | 50 |
| L. rhamnosus KLDS1.0912 | 400 |
| L. casei KLDS1.0351 | 200 |
| Groups | Mortality | Liver Body Ratio | Kidney Body Ratio |
|---|---|---|---|
| Pre-control | 0/8 | 3.80 ± 0.25 b | 1.17 ± 0.03 bc |
| Pre-Pb | 2/8 | 3.28 ± 0.22 a | 1.06 ± 0.04 a |
| Pre-LAB | 1/8 | 3.65 ± 0.23 ab | 1.16 ± 0.05 bc |
| Control | 0/8 | 4.95 ± 0.31 d | 1.42 ± 0.06 d |
| Pb | 2/8 | 3.33 ± 0.14 a | 1.11 ± 0.04 ab |
| High dose | 0/8 | 4.47 ± 0.27 c | 1.40 ± 0.03 d |
| Medial dose | 1/8 | 3.43 ± 0.19 ab | 1.31 ± 0.03 c |
| Low dose | 1/8 | 3.37 ± 0.28 ab | 1.21 ± 0.04 c |
| Drug | 0/8 | 3.39 ± 0.15 ab | 1.23 ± 0.07 c |
| Groups | Pb (μg/g) | |
|---|---|---|
| First Week | Second Week | |
| Control | 0.16 ± 0.03 a | 0.13 ± 0.04 a |
| Pb | 23.15 ± 1.35 b | 0.40 ± 0.13 b |
| High dose | 31.15 ± 2.32 c | 0.65 ± 0.14 bc |
| Medial dose | 29.48 ± 1.12 c | 0.52 ± 0.10 bc |
| Low dose | 28.81 ± 1.87 c | 0.41 ± 0.18 b |
| Drug | 42.81 ± 2.43 d | 0.70 ± 0.16 c |
| Groups | Blood (μg/L) | Liver (μg/g) | Kidney (μg/g) |
|---|---|---|---|
| Pre-control | 0.32 ± 0.02 a | 0.15 ± 0.02 a | 0.17 ± 0.03 a |
| Pre-Pb | 383.30 ± 23.12 f | 1.51 ± 0.04 e | 1.72 ± 0.07 g |
| Pre-LAB | 344.03 ± 24.32 e | 1.13 ± 0.09 d | 1.25 ± 0.06 f |
| Control | 0.43 ± 0.05 a | 0.12 ± 0.04 a | 0.16 ± 0.04 a |
| Pb | 302.20 ± 25.32 d | 0.46 ± 0.08 c | 0.66 ± 0.08 e |
| High dose | 234.12 ± 10.18 c | 0.35 ± 0.07 bc | 0.45 ± 0.06 bc |
| Medial dose | 248.01 ± 7.54 c | 0.39 ± 0.05 c | 0.54 ± 0.04 cd |
| Low dose | 258.33 ± 8.13 c | 0.43 ± 0.07 c | 0.63 ± 0.05 de |
| Drug | 176.14 ± 6.17 b | 0.27 ± 0.06 b | 0.36 ± 0.08 b |
| Groups | ALT (U/L) | AST (U/L) | AST/ALT |
|---|---|---|---|
| Pre-control | 33.56 ± 2.13 a | 55.87 ± 3.01 a | 1.67 ± 0.02 a |
| Pre-Pb | 40.74 ± 1.76 c | 84.23 ± 3.32 c | 2.07 ± 0.01 b |
| Pre-LAB | 38.67 ± 2.38 bc | 65.89 ± 4.21 b | 1.70 ± 0.00 a |
| Control | 35.39 ± 3.01 ab | 100.23 ± 4.54 d | 2.84 ± 0.11 c |
| Pb | 46.19 ± 2.88 d | 228.12 ± 5.65 h | 4.95 ± 0.19 g |
| High dose | 34.23 ± 1.47 a | 123.33 ± 4.25 e | 3.60 ± 0.03 d |
| Medial dose | 37.87 ± 2.13 abc | 153.33 ± 4.02 f | 4.05 ± 0.12 e |
| Low dose | 40.02 ± 2.42 c | 167.88 ± 5.23 g | 4.20 ± 0.12 ef |
| Drug | 33.87 ± 2.12 a | 146.46 ± 4.37 f | 4.33 ± 0.14 f |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, B.; Jin, D.; Yu, S.; Etareri Evivie, S.; Muhammad, Z.; Huo, G.; Liu, F. In Vitro and In Vivo Evaluation of Lactobacillus delbrueckii subsp. bulgaricus KLDS1.0207 for the Alleviative Effect on Lead Toxicity. Nutrients 2017, 9, 845. https://doi.org/10.3390/nu9080845
Li B, Jin D, Yu S, Etareri Evivie S, Muhammad Z, Huo G, Liu F. In Vitro and In Vivo Evaluation of Lactobacillus delbrueckii subsp. bulgaricus KLDS1.0207 for the Alleviative Effect on Lead Toxicity. Nutrients. 2017; 9(8):845. https://doi.org/10.3390/nu9080845
Chicago/Turabian StyleLi, Bailiang, Da Jin, Shangfu Yu, Smith Etareri Evivie, Zafarullah Muhammad, Guicheng Huo, and Fei Liu. 2017. "In Vitro and In Vivo Evaluation of Lactobacillus delbrueckii subsp. bulgaricus KLDS1.0207 for the Alleviative Effect on Lead Toxicity" Nutrients 9, no. 8: 845. https://doi.org/10.3390/nu9080845
APA StyleLi, B., Jin, D., Yu, S., Etareri Evivie, S., Muhammad, Z., Huo, G., & Liu, F. (2017). In Vitro and In Vivo Evaluation of Lactobacillus delbrueckii subsp. bulgaricus KLDS1.0207 for the Alleviative Effect on Lead Toxicity. Nutrients, 9(8), 845. https://doi.org/10.3390/nu9080845





