The Association between Vitamin D Insufficiency and Nonalcoholic Fatty Liver Disease: A Population-Based Study
Abstract
1. Introduction
2. Materials and Methods
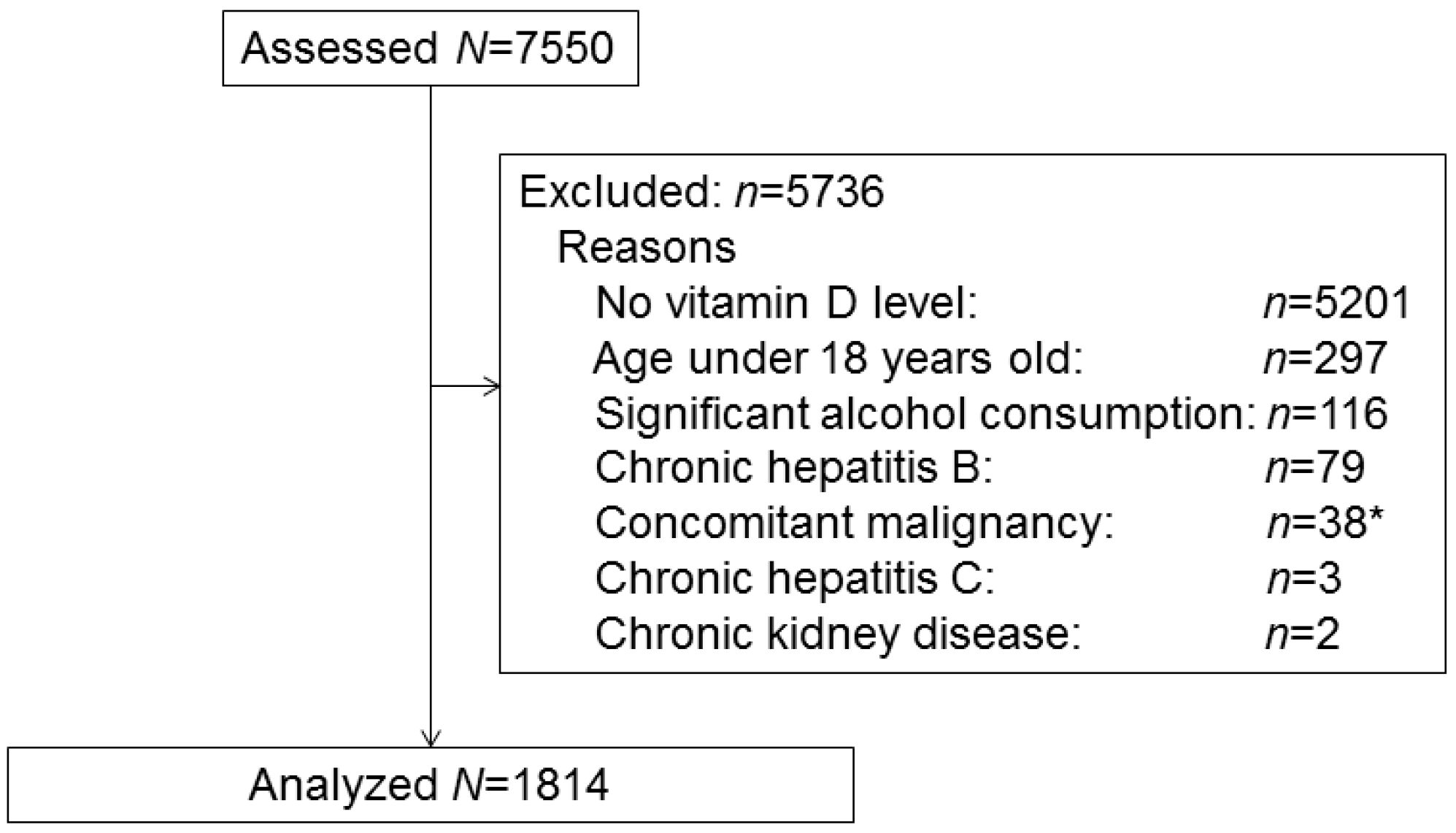
2.1. Participants
2.2. Serum 25(OH)D Measurement
2.3. Definition of NAFLD and Significant Fibrosis
2.4. Statistical Analysis
3. Results
3.1. Characteristics of the Study Participants
3.2. Factors Associated with NAFLD
3.3. Subgroup Analyses
3.3.1. Participants without Obesity
3.3.2. Participants of Male Gender
3.3.3. Participants without Metabolic Syndrome
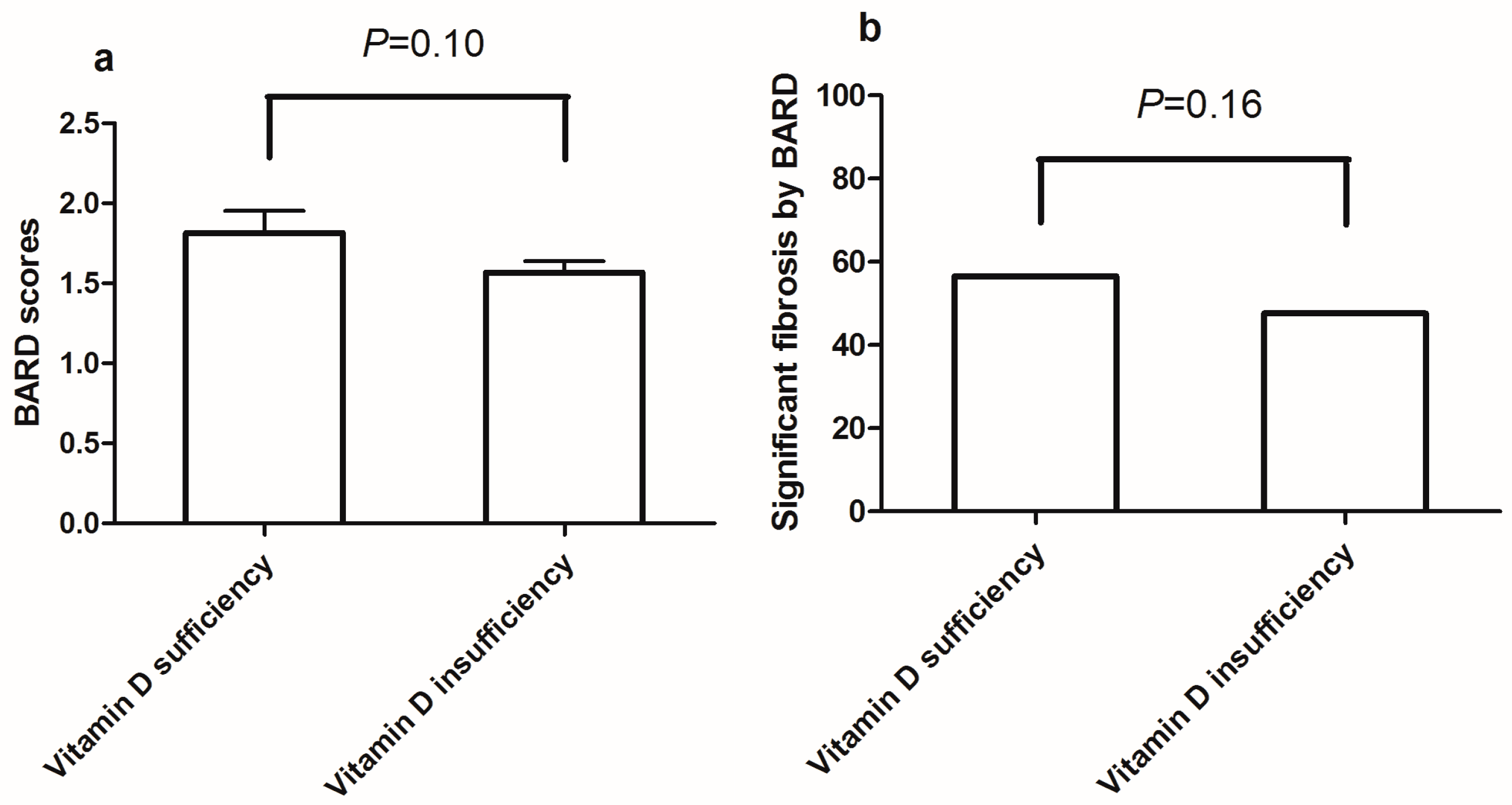
3.4. Association of Vitamin D Insufficiency with Significant Fibrosis in Participants with NAFLD
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Theodoratou, E.; Tzoulaki, I.; Zgaga, L.; Ioannidis, J.P. Vitamin D and multiple health outcomes: Umbrella review of systematic reviews and meta-analyses of observational studies and randomised trials. BMJ 2014, 348, g2035. [Google Scholar] [CrossRef] [PubMed]
- Barchetta, I.; Carotti, S.; Labbadia, G.; Gentilucci, U.V.; Muda, A.O.; Angelico, F.; Silecchia, G.; Leonetti, F.; Fraioli, A.; Picardi, A.; et al. Liver vitamin D receptor, cyp2r1, and cyp27a1 expression: Relationship with liver histology and vitamin D3 levels in patients with nonalcoholic steatohepatitis or hepatitis c virus. Hepatology 2012, 56, 2180–2187. [Google Scholar] [CrossRef] [PubMed]
- Ding, N.; Yu, R.T.; Subramaniam, N.; Sherman, M.H.; Wilson, C.; Rao, R.; Leblanc, M.; Coulter, S.; He, M.; Scott, C.; et al. A vitamin D receptor/smad genomic circuit gates hepatic fibrotic response. Cell 2013, 153, 601–613. [Google Scholar] [CrossRef] [PubMed]
- Putz-Bankuti, C.; Pilz, S.; Stojakovic, T.; Scharnagl, H.; Pieber, T.R.; Trauner, M.; Obermayer-Pietsch, B.; Stauber, R.E. Association of 25-hydroxyvitamin d levels with liver dysfunction and mortality in chronic liver disease. Liver Int. 2012, 32, 845–851. [Google Scholar] [CrossRef] [PubMed]
- Barchetta, I.; Angelico, F.; Del Ben, M.; Baroni, M.G.; Pozzilli, P.; Morini, S.; Cavallo, M.G. Strong association between non alcoholic fatty liver disease (nafld) and low 25(oh) vitamin D levels in an adult population with normal serum liver enzymes. BMC Med. 2011, 9, 85. [Google Scholar] [CrossRef] [PubMed]
- Chung, G.E.; Kim, D.; Kwak, M.S.; Yang, J.I.; Yim, J.Y.; Lim, S.H.; Itani, M. The serum vitamin D level is inversely correlated with nonalcoholic fatty liver disease. Clin. Mol. Hepatol. 2016, 22, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Eliades, M.; Spyrou, E.; Agrawal, N.; Lazo, M.; Brancati, F.L.; Potter, J.J.; Koteish, A.A.; Clark, J.M.; Guallar, E.; Hernaez, R. Meta-analysis: Vitamin D and non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2013, 38, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Zhai, H.L.; Wang, N.J.; Han, B.; Li, Q.; Chen, Y.; Zhu, C.F.; Chen, Y.C.; Xia, F.Z.; Cang, Z.; Zhu, C.X.; et al. Low vitamin D levels and non-alcoholic fatty liver disease, evidence for their independent association in men in east china: A cross-sectional study (survey on prevalence in east china for metabolic diseases and risk factors (spect-china)). Br. J. Nutr. 2016, 115, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Nelson, J.E.; Roth, C.L.; Wilson, L.A.; Yates, K.P.; Aouizerat, B.; Morgan-Stevenson, V.; Whalen, E.; Hoofnagle, A.; Mason, M.; Gersuk, V.; et al. Vitamin D deficiency is associated with increased risk of non-alcoholic steatohepatitis in adults with non-alcoholic fatty liver disease: Possible role for mapk and nf-kappab? Am. J. Gastroenterol. 2016, 111, 852–863. [Google Scholar] [CrossRef] [PubMed]
- Dasarathy, J.; Periyalwar, P.; Allampati, S.; Bhinder, V.; Hawkins, C.; Brandt, P.; Khiyami, A.; McCullough, A.J.; Dasarathy, S. Hypovitaminosis D is associated with increased whole body fat mass and greater severity of non-alcoholic fatty liver disease. Liver Int. 2014, 34, e118–e127. [Google Scholar] [CrossRef] [PubMed]
- Bril, F.; Maximos, M.; Portillo-Sanchez, P.; Biernacki, D.; Lomonaco, R.; Subbarayan, S.; Correa, M.; Lo, M.; Suman, A.; Cusi, K. Relationship of vitamin D with insulin resistance and disease severity in non-alcoholic steatohepatitis. J. Hepatol. 2015, 62, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Anty, R.; Hastier, A.; Canivet, C.M.; Patouraux, S.; Schneck, A.S.; Ferrari-Panaia, P.; Ben-Amor, I.; Saint-Paul, M.C.; Gugenheim, J.; Gual, P.; et al. Severe vitamin D deficiency is not associated with liver damage in morbidly obese patients. Obes. Surg. 2016, 26, 2138–2143. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.J.; Wang, B.W.; Zhang, C.; Xia, M.Z.; Chen, Y.H.; Hu, C.Q.; Wang, H.; Chen, X.; Xu, D.X. Vitamin D deficiency attenuates high-fat diet-induced hyperinsulinemia and hepatic lipid accumulation in male mice. Endocrinology 2015, 156, 2103–2113. [Google Scholar] [CrossRef] [PubMed]
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of the national cholesterol education program (ncep) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (adult treatment panel iii). JAMA 2001, 285, 2486–2497. [Google Scholar]
- Casanovas, T.; Jane, L.; Herdman, M.; Casado, A.; Garcia, B.; Prat, B.; Fabregat, J. Assessing outcomes in liver disease patients: Reliability and validity of the spanish version of the liver disease quality of life questionnaire (LDQOL 1.0). Value Health 2010, 13, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Catharine Ross, A.; Taylor, C.L.; Yaktine, A.L.; del Valle, H.B. Dietary Reference Intakes for Calcium and Vitamin D. Available online: http://books.nap.edu/openbook.php?record_id=13050 (accessed on 13 July 2017).
- Jenab, M.; Bueno-de-Mesquita, H.B.; Ferrari, P.; van Duijnhoven, F.J.; Norat, T.; Pischon, T.; Jansen, E.H.; Slimani, N.; Byrnes, G.; Rinaldi, S.; et al. Association between pre-diagnostic circulating vitamin D concentration and risk of colorectal cancer in European populations: A nested case-control study. BMJ 2010, 340, b5500. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Sabour, S.; Sagar, U.N.; Adams, S.; Whellan, D.J. Prevalence of hypovitaminosis D in cardiovascular diseases (from the national health and nutrition examination survey 2001 to 2004). Am. J. Cardiol. 2008, 102, 1540–1544. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, R.; Kunutsor, S.; Vitezova, A.; Oliver-Williams, C.; Chowdhury, S.; Kiefte-de-Jong, J.C.; Khan, H.; Baena, C.P.; Prabhakaran, D.; Hoshen, M.B.; et al. Vitamin D and risk of cause specific death: Systematic review and meta-analysis of observational cohort and randomised intervention studies. BMJ 2014, 348, g1903. [Google Scholar] [CrossRef] [PubMed]
- Schottker, B.; Jorde, R.; Peasey, A.; Thorand, B.; Jansen, E.H.; Groot, L.; Streppel, M.; Gardiner, J.; Ordonez-Mena, J.M.; Perna, L.; et al. Vitamin D and mortality: Meta-analysis of individual participant data from a large consortium of cohort studies from europe and the united states. BMJ 2014, 348, g3656. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, D.; Kim, H.J.; Lee, C.H.; Yang, J.I.; Kim, W.; Kim, Y.J.; Yoon, J.H.; Cho, S.H.; Sung, M.W.; et al. Hepatic steatosis index: A simple screening tool reflecting nonalcoholic fatty liver disease. Dig. Liver Dis. 2010, 42, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.A.; Oliver, D.; Arnold, H.L.; Gogia, S.; Neuschwander-Tetri, B.A. Development and validation of a simple NAFLD clinical scoring system for identifying patients without advanced disease. Gut 2008, 57, 1441–1447. [Google Scholar] [CrossRef] [PubMed]
- Korean Association for the Study of the Liver (KASL). Kasl clinical practice guidelines: Management of alcoholic liver disease. Clin. Mol. Hepatol. 2013, 19, 216–254. [Google Scholar]
- European Association for the Study of the Liver (EASL). Easl clinical practical guidelines: Management of alcoholic liver disease. J. Hepatol. 2012, 57, 399–420. [Google Scholar]
- Chung, M.; Lee, J.; Terasawa, T.; Lau, J.; Trikalinos, T.A. Vitamin D with or without calcium supplementation for prevention of cancer and fractures: An updated meta-analysis for the U.S. Preventive services task force. Ann. Int. Med. 2011, 155, 827–838. [Google Scholar] [CrossRef] [PubMed]
- Forman, J.P.; Giovannucci, E.; Holmes, M.D.; Bischoff-Ferrari, H.A.; Tworoger, S.S.; Willett, W.C.; Curhan, G.C. Plasma 25-hydroxyvitamin d levels and risk of incident hypertension. Hypertension 2007, 49, 1063–1069. [Google Scholar] [CrossRef] [PubMed]
- Balion, C.; Griffith, L.E.; Strifler, L.; Henderson, M.; Patterson, C.; Heckman, G.; Llewellyn, D.J.; Raina, P. Vitamin D, cognition, and dementia: A systematic review and meta-analysis. Neurology 2012, 79, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- Patel, Y.A.; Henao, R.; Moylan, C.A.; Guy, C.D.; Piercy, D.L.; Diehl, A.M.; Abdelmalek, M.F. Vitamin D is not associated with severity in NAFLD: Results of a paired clinical and gene expression profile analysis. Am. J. Gastroenterol. 2016, 111, 1591–1598. [Google Scholar] [CrossRef] [PubMed]
- Barchetta, I.; Del Ben, M.; Angelico, F.; Di Martino, M.; Fraioli, A.; La Torre, G.; Saulle, R.; Perri, L.; Morini, S.; Tiberti, C.; et al. No effects of oral vitamin D supplementation on non-alcoholic fatty liver disease in patients with type 2 diabetes: A randomized, double-blind, placebo-controlled trial. BMC Med. 2016, 14, 92. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Bouillon, R.; Van Schoor, N.M.; Gielen, E.; Boonen, S.; Mathieu, C.; Vanderschueren, D.; Lips, P. Optimal vitamin D status: A critical analysis on the basis of evidence-based medicine. J. Clin. Endocrinol. Metab. 2013, 98, E1283–E1304. [Google Scholar] [CrossRef] [PubMed]
- Kupferschmidt, K. Uncertain verdict as vitamin D goes on trial. Science 2012, 337, 1476–1478. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Yu, C.; Ma, H.; Xu, L.; Miao, M.; Li, Y. Prevalence and risk factors for the development of nonalcoholic fatty liver disease in a nonobese Chinese population: The Zhejiang Zhenhai study. Am. J. Gastroenterol. 2013, 108, 1299–1304. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.L.; Leung, J.C.; Loong, T.C.; Wong, G.L.; Yeung, D.K.; Chan, R.S.; Chan, H.L.; Chim, A.M.; Woo, J.; Chu, W.C.; et al. Prevalence and severity of nonalcoholic fatty liver disease in non-obese patients: A population study using proton-magnetic resonance spectroscopy. Am. J. Gastroenterol. 2015, 110, 1306–1314. [Google Scholar] [CrossRef] [PubMed]
- Farrell, G.C.; Larter, C.Z. Nonalcoholic fatty liver disease: From steatosis to cirrhosis. Hepatology 2006, 43, S99–S112. [Google Scholar] [CrossRef] [PubMed]
- Sanyal, A.J. Aga technical review on nonalcoholic fatty liver disease. Gastroenterology 2002, 123, 1705–1725. [Google Scholar] [CrossRef] [PubMed]
- Ruhl, C.E.; Everhart, J.E. Determinants of the association of overweight with elevated serum alanine aminotransferase activity in the United States. Gastroenterology 2003, 124, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Stepanova, M.; Negro, F.; Hallaji, S.; Younossi, Y.; Lam, B.; Srishord, M. Nonalcoholic fatty liver disease in lean individuals in the united states. Medicine 2012, 91, 319–327. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Center. Obesity Status in Korea by Statistics (Summary). Available online: http://www.cancer.go.kr/ebook/159/webview/index.html (accessed on 6 May 2017).
- Park, S.H.; Jeon, W.K.; Kim, S.H.; Kim, H.J.; Park, D.I.; Cho, Y.K.; Sung, I.K.; Sohn, C.I.; Keum, D.K.; Kim, B.I. Prevalence and risk factors of non-alcoholic fatty liver disease among korean adults. J. Gastroenterol. Hepatol. 2006, 21, 138–143. [Google Scholar] [CrossRef] [PubMed]
| Variables | NAFLD † (n = 409) | Non-NAFLD (n = 1403) | p-Value |
|---|---|---|---|
| Age, year | 44.8 ± 14.4 | 43.3 ± 15.2 | 0.08 |
| Male gender, n (%) | 232 (56.7) | 625 (44.5) | <0.001 |
| BMI, kg/m2 | 28.1 ± 3.0 | 22.5 ± 2.5 | <0.001 |
| Waist circumference, cm | 91.4 ± 8.5 | 77.4 ± 8.4 | <0.001 |
| Diabetes, n (%) ‡ | 90 (22.7) | 55 (4.0) | <0.001 |
| Hypertension, n (%) § | 159 (39.4) | 247 (17.7) | <0.001 |
| Metabolic syndrome, n (%) || | 234 (57.2) | 231 (16.5) | <0.001 |
| Season of blood draw, n (%) | - | - | - |
| Spring | 106 (25.9) | 359 (25.6) | 0.06 |
| Summer | 90 (22.0) | 393 (28.0) | |
| Autumn | 109 (26.7) | 360 (25.7) | |
| Winter | 104 (25.4) | 291 (20.7) | |
| AST, IU/L | 25.7 ± 13.4 | 20.1 ± 6.5 | <0.001 |
| ALT, IU/L | 37.1 ± 30.0 | 16.6 ± 7.8 | <0.001 |
| Fasting glucose, mg/dL | 109.0 ± 31.4 | 94.5 ± 14.4 | <0.001 |
| Triglyceride, mg/dL | 188.1 ± 134.3 | 120.1 ± 101.6 | <0.001 |
| Total cholesterol, mg/dL | 194.2 ± 36.2 | 184.7 ± 32.8 | <0.001 |
| HDL-cholesterol, mg/dL | 47.3 ± 10.4 | 54.3 ± 12.0 | <0.001 |
| 25(OH)D, ng/mL | 16.2 ± 6.2 | 16.2 ± 6.5 | 0.95 |
| Vitamin D insufficiency, n (%) | 317 (77.5) | 1086 (77.4) | 1.00 |
| Variable | Univariable Analysis | Multivariable Analysis | ||
|---|---|---|---|---|
| OR (95% CI) | p-value | OR (95% CI) | p-value | |
| Age, per 1 year | 1.01 (1.00–1.01) | 0.08 | - | - |
| Male gender | 1.63 (1.31–2.04) | <0.001 | 0.80 (0.55–1.17) | 0.24 |
| BMI, per 1 kg/m2 | 2.48 (2.26–2.73) | <0.001 | 2.52 (2.27–2.80) | <0.001 |
| WC, per 1 cm | 1.23 (1.20–1.26) | <0.001 | - | - |
| Diabetes | 2.66 (2.23–3.19) | <0.001 | 7.90 (4.62–13.51) | <0.001 |
| Hypertension | 1.74 (1.54–1.96) | <0.001 | 0.76 (0.52–1.12) | 0.17 |
| Metabolic syndrome | 6.77 (5.32–8.61) | <0.001 | - | - |
| TG, per 50 mg/dL | 1.27 (1.21–1.34) | <0.001 | 1.09 (1.03–1.17) | 0.005 |
| Total cholesterol, per 50 mg/dL | 1.51 (1.28–1.77) | <0.001 | 1.12 (0.84–1.49) | 0.43 |
| HDL-cholesterol, per 10 mg/dL | 0.56 (0.51–0.63) | <0.001 | 0.88 (0.73–1.06) | 0.19 |
| Vitamin D insufficiency † | 0.97 (0.74–1.26) | 0.80 | - | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ha, Y.; Hwang, S.G.; Rim, K.S. The Association between Vitamin D Insufficiency and Nonalcoholic Fatty Liver Disease: A Population-Based Study. Nutrients 2017, 9, 806. https://doi.org/10.3390/nu9080806
Ha Y, Hwang SG, Rim KS. The Association between Vitamin D Insufficiency and Nonalcoholic Fatty Liver Disease: A Population-Based Study. Nutrients. 2017; 9(8):806. https://doi.org/10.3390/nu9080806
Chicago/Turabian StyleHa, Yeonjung, Seong Gyu Hwang, and Kyu Sung Rim. 2017. "The Association between Vitamin D Insufficiency and Nonalcoholic Fatty Liver Disease: A Population-Based Study" Nutrients 9, no. 8: 806. https://doi.org/10.3390/nu9080806
APA StyleHa, Y., Hwang, S. G., & Rim, K. S. (2017). The Association between Vitamin D Insufficiency and Nonalcoholic Fatty Liver Disease: A Population-Based Study. Nutrients, 9(8), 806. https://doi.org/10.3390/nu9080806




