Prenatal Vitamin D Intake, Cord Blood 25-Hydroxyvitamin D, and Offspring Body Composition: The Healthy Start Study
Abstract
1. Introduction
2. Methods
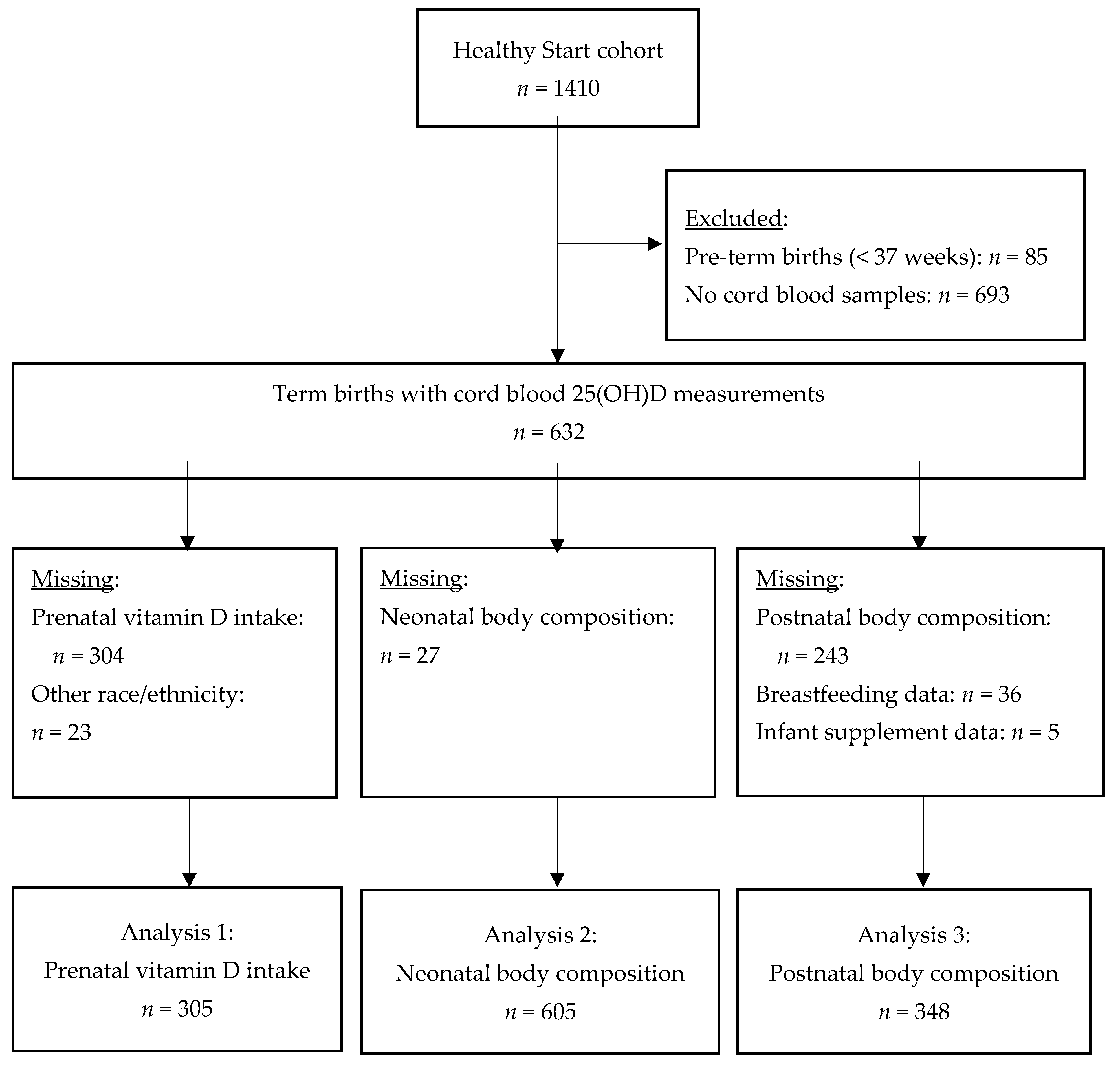
2.1. Participants
2.2. Assessment of Maternal Vitamin D Intake
2.3. Assessment of Offspring Vitamin D Intake
2.4. Assessment of 25(OH)D
2.5. Assessment of Offspring Size and Body Composition
2.6. Covariates
2.7. Statistical analyses
3. Results
3.1. Analysis 1: Vitamin D intake (Table 2)
3.2. Analysis 2: Neonatal Body Size and Composition (Table 3)
3.3. Analysis 3: Postnatal Body Size and Composition
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Christian, P.; Stewart, C.P. Maternal micronutrient deficiency, fetal development, and the risk of chronic disease. J. Nutr. 2010, 140, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Ginde, A.A.; Sullivan, A.F.; Mansbach, J.M.; Camargo, C.A., Jr. Vitamin D insufficiency in pregnant and nonpregnant women of childbearing age in the United States. Am. J. Obstet. Gynecol. 2010, 202, 436. [Google Scholar] [CrossRef] [PubMed]
- Novakovic, B.; Galati, J.C.; Chen, A.; Morley, R.; Craig, J.M.; Saffery, R. Maternal vitamin D predominates over genetic factors in determining neonatal circulating vitamin D concentrations. Am. J. Clin. Nutr. 2012, 96, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Bodnar, L.M.; Platt, R.W.; Simhan, H.N. Early-pregnancy vitamin D deficiency and risk of preterm birth subtypes. Obstet. Gynecol. 2015, 125, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Miliku, K.; Vinkhuyzen, A.; Blanken, L.M.; McGrath, J.J.; Eyles, D.W.; Burne, T.H.; Hofman, A.; Tiemeier, H.; Steegers, E.A.; Gaillard, R.; et al. Maternal vitamin D concentrations during pregnancy, fetal growth patterns, and risks of adverse birth outcomes. Am. J. Clin. Nutr. 2016, 103, 1514–1522. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.H.; Fu, L.; Hao, J.H.; Yu, Z.; Zhu, P.; Wang, H.; Xu, Y.Y.; Zhang, C.; Tao, F.B.; Xu, D.X. Maternal vitamin D deficiency during pregnancy elevates the risks of small for gestational age and low birth weight infants in Chinese population. J. Clin. Endocrinol. Metab. 2015, 100, 1912–1919. [Google Scholar] [CrossRef] [PubMed]
- Gernand, A.D.; Simhan, H.N.; Caritis, S.; Bodnar, L.M. Maternal vitamin D status and small-for-gestational-age offspring in women at high risk for preeclampsia. Obstet. Gynecol. 2014, 123, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Perez-Lopez, F.R.; Pasupuleti, V.; Mezones-Holguin, E.; Benites-Zapata, V.A.; Thota, P.; Deshpande, A.; Hernandez, A.V. Effect of vitamin D supplementation during pregnancy on maternal and neonatal outcomes: A systematic review and meta-analysis of randomized controlled trials. Fertil. Steril. 2015, 103, 1278–1288. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D. Nonclassic actions of vitamin D. J. Clin. Endocrinol. Metab. 2009, 94, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Norman, A.W.; Okamura, W.H.; Sen, A.; Zemel, M.B. 1alpha,25-Dihydroxyvitamin D3 modulates human adipocyte metabolism via nongenomic action. FASEB. J. 2001, 15, 2751–2753. [Google Scholar] [PubMed]
- McCarty, M.F.; Thomas, C.A. PTH excess may promote weight gain by impeding catecholamine-induced lipolysis-implications for the impact of calcium, vitamin D, and alcohol on body weight. Med. Hypotheses 2003, 61, 535–542. [Google Scholar] [CrossRef]
- Kong, J.; Li, Y.C. Molecular mechanism of 1,25-dihydroxyvitamin D3 inhibition of adipogenesis in 3T3-L1 cells. Am. J. Physiol. Endocrinol. Metab. 2006, 290, E916–E924. [Google Scholar] [CrossRef] [PubMed]
- Zemel, M.B. Regulation of adiposity and obesity risk by dietary calcium: Mechanisms and implications. J. Am. Coll. Nutr. 2002, 21, 146S–151S. [Google Scholar] [CrossRef] [PubMed]
- Barebring, L.; Schoenmakers, I.; Glantz, A.; Hulthen, L.; Jagner, A.; Ellis, J.; Barebring, M.; Bullarbo, M.; Augustin, H. Vitamin D Status during Pregnancy in a Multi-Ethnic Population-Representative Swedish Cohort. Nutrients 2016, 8, 655. [Google Scholar] [CrossRef] [PubMed]
- Moon, R.J.; Harvey, N.C.; Cooper, C.; D’Angelo, S.; Crozier, S.R.; Inskip, H.M.; Schoenmakers, I.; Prentice, A.; Arden, N.K.; Bishop, N.J.; et al. Determinants of the Maternal 25-Hydroxyvitamin D Response to Vitamin D Supplementation During Pregnancy. J. Clin. Endocrinol. Metab. 2016, 101, 5012–5020. [Google Scholar] [CrossRef] [PubMed]
- De-Regil, L.M.; Palacios, C.; Lombardo, L.K.; Pena-Rosas, J.P. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef]
- Bjorn Jensen, C.; Thorne-Lyman, A.L.; Hansen, L.V.; Strom, M.; Nielsen, N.O.; Cohen, A.; Olsen, S.F. Development and validation of a vitamin D status prediction model in Danish pregnant women: A study of the Danish National Birth Cohort. PLoS ONE 2013, 8, e53059. [Google Scholar] [CrossRef] [PubMed]
- Morley, R.; Carlin, J.B.; Pasco, J.A.; Wark, J.D. Maternal 25-hydroxyvitamin D and parathyroid hormone concentrations and offspring birth size. J. Clin. Endocrinol. Metab. 2006, 91, 906–912. [Google Scholar] [CrossRef] [PubMed]
- Weiler, H.; Fitzpatrick-Wong, S.; Veitch, R.; Kovacs, H.; Schellenberg, J.; McCloy, U.; Yuen, C.K. Vitamin D deficiency and whole-body and femur bone mass relative to weight in healthy newborns. CMAJ. 2005, 172, 757–761. [Google Scholar] [CrossRef] [PubMed]
- Josefson, J.L.; Feinglass, J.; Rademaker, A.W.; Metzger, B.E.; Zeiss, D.M.; Price, H.E.; Langman, C.B. Maternal obesity and vitamin D sufficiency are associated with cord blood vitamin D insufficiency. J. Clin. Endocrinol. Metab. 2013, 98, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Crozier, S.R.; Harvey, N.C.; Inskip, H.M.; Godfrey, K.M.; Cooper, C.; Robinson, S.M.; Group, S.W.S.S. Maternal vitamin D status in pregnancy is associated with adiposity in the offspring: Findings from the Southampton Women′s Survey. Am. J. Clin. Nutr. 2012, 96, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Godang, K.; Froslie, K.F.; Henriksen, T.; Qvigstad, E.; Bollerslev, J. Seasonal variation in maternal and umbilical cord 25(OH) vitamin D and their associations with neonatal adiposity. Eur. J. Endocrinol. 2014, 170, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Farrant, H.J.; Krishnaveni, G.V.; Hill, J.C.; Boucher, B.J.; Fisher, D.J.; Noonan, K.; Osmond, C.; Veena, S.R.; Fall, C.H. Vitamin D insufficiency is common in Indian mothers but is not associated with gestational diabetes or variation in newborn size. Eur. J. Clin. Nutr. 2009, 63, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Josefson, J.L.; Reisetter, A.; Scholtens, D.M.; Price, H.E.; Metzger, B.E.; Langman, C.B.; Group, H.S.C.R. Maternal BMI Associations with Maternal and Cord Blood Vitamin D Levels in a North American Subset of Hyperglycemia and Adverse Pregnancy Outcome (HAPO) Study Participants. PLoS ONE 2016, 11, e0150221. [Google Scholar] [CrossRef] [PubMed]
- Gale, C.R.; Robinson, S.M.; Harvey, N.C.; Javaid, M.K.; Jiang, B.; Martyn, C.N.; Godfrey, K.M.; Cooper, C.; Princess Anne Hospital Study, G. Maternal vitamin D status during pregnancy and child outcomes. Eur. J. Clin. Nutr. 2008, 62, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Krishnaveni, G.V.; Veena, S.R.; Winder, N.R.; Hill, J.C.; Noonan, K.; Boucher, B.J.; Karat, S.C.; Fall, C.H. Maternal vitamin D status during pregnancy and body composition and cardiovascular risk markers in Indian children: The Mysore Parthenon Study. Am. J. Clin. Nutr. 2011, 93, 628–635. [Google Scholar] [CrossRef] [PubMed]
- Sayers, A.; Tobias, J.H. Estimated maternal ultraviolet B exposure levels in pregnancy influence skeletal development of the child. J. Clin. Endocrinol. Metab. 2009, 94, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Wortsman, J.; Matsuoka, L.Y.; Chen, T.C.; Lu, Z.; Holick, M.F. Decreased bioavailability of vitamin D in obesity. Am. J. Clin. Nutr. 2000, 72, 690–693. [Google Scholar] [PubMed]
- Arunabh, S.; Pollack, S.; Yeh, J.; Aloia, J.F. Body fat content and 25-hydroxyvitamin D levels in healthy women. J. Clin. Endocrinol. Metab. 2003, 88, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Parikh, S.J.; Edelman, M.; Uwaifo, G.I.; Freedman, R.J.; Semega-Janneh, M.; Reynolds, J.; Yanovski, J.A. The relationship between obesity and serum 1,25-dihydroxy vitamin D concentrations in healthy adults. J. Clin. Endocrinol. Metab. 2004, 89, 1196–1199. [Google Scholar] [CrossRef] [PubMed]
- Snijder, M.B.; van Dam, R.M.; Visser, M.; Deeg, D.J.; Dekker, J.M.; Bouter, L.M.; Seidell, J.C.; Lips, P. Adiposity in relation to vitamin D status and parathyroid hormone levels: A population-based study in older men and women. J. Clin. Endocrinol. Metab. 2005, 90, 4119–4123. [Google Scholar] [CrossRef] [PubMed]
- Bodnar, L.M.; Catov, J.M.; Roberts, J.M.; Simhan, H.N. Prepregnancy obesity predicts poor vitamin D status in mothers and their neonates. J. Nutr. 2007, 137, 2437–2442. [Google Scholar] [PubMed]
- Eckhardt, C.L.; Gernand, A.D.; Roth, D.E.; Bodnar, L.M. Maternal vitamin D status and infant anthropometry in a US multi-centre cohort study. Ann. Hum. Biol. 2015, 42, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Leffelaar, E.R.; Vrijkotte, T.G.; van Eijsden, M. Maternal early pregnancy vitamin D status in relation to fetal and neonatal growth: Results of the multi-ethnic Amsterdam born children and their development cohort. Br. J. Nutr. 2010, 104, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Subar, A.F.; Kirkpatrick, S.I.; Mittl, B.; Zimmerman, T.P.; Thompson, F.E.; Bingley, C.; Willis, G.; Islam, N.G.; Baranowski, T.; McNutt, S.; et al. The automated self-administered 24-hour dietary recall (ASA24): A resource for researchers, clinicians, and educators from the National Cancer Institute. J. Acad. Nutr. Diet. 2012, 112, 1134–1137. [Google Scholar] [CrossRef] [PubMed]
- Kipnis, V.; Midthune, D.; Buckman, D.W.; Dodd, K.W.; Guenther, P.M.; Krebs-Smith, S.M.; Subar, A.F.; Tooze, J.A.; Carroll, R.J.; Freedman, L.S. Modeling data with excess zeros and measurement error: Application to evaluating relationships between episodically consumed foods and health outcomes. Biometrics 2009, 65, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- Tooze, J.A.; Kipnis, V.; Buckman, D.W.; Carroll, R.J.; Freedman, L.S.; Guenther, P.M.; Krebs-Smith, S.M.; Subar, A.F.; Dodd, K.W. A mixed-effects model approach for estimating the distribution of usual intake of nutrients: the NCI method. Stat. in Med. 2010, 29, 2857–2868. [Google Scholar] [CrossRef] [PubMed]
- Tooze, J.A.; Midthune, D.; Dodd, K.W.; Freedman, L.S.; Krebs-Smith, S.M.; Subar, A.F.; Guenther, P.M.; Carroll, R.J.; Kipnis, V. A new statistical method for estimating the usual intake of episodically consumed foods with application to their distribution. J. Am. Diet. Assoc. 2006, 106, 1575–1587. [Google Scholar] [CrossRef] [PubMed]
- Crume, T.L.; Ogden, L.; Maligie, M.; Sheffield, S.; Bischoff, K.J.; McDuffie, R.; Daniels, S.; Hamman, R.F.; Norris, J.M.; Dabelea, D. Long-term impact of neonatal breastfeeding on childhood adiposity and fat distribution among children exposed to diabetes in utero. Diabetes Care 2011, 34, 641–645. [Google Scholar] [CrossRef] [PubMed]
- Wagner, C.L.; Greer, F.R. American Academy of Pediatrics Section on B.; American Academy of Pediatrics Committee on N. Prevention of rickets and vitamin D deficiency in infants, children, and adolescents. Pediatrics 2008, 122, 1142–1152. [Google Scholar] [CrossRef] [PubMed]
- Sauder, K.A.; Starling, A.P.; Shapiro, A.L.; Kaar, J.L.; Ringham, B.M.; Glueck, D.H.; Dabelea, D. Exploring the association between maternal prenatal multivitamin use and early infant growth: The Healthy Start Study. Pediatr. Obes. 2016, 11, 434–441. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health Office of Dietary Supplements. Dietary Supplement Label Database. Available online: https://dsld.nlm.nih.gov/dsld/ (accessed on 3 March 2017).
- Centers for Disease Control and Prevention. VDSCP: List of Certified Participants, 2017. Laboratory Quality Assurance and Standardization Programs. Available online: https://www.cdc.gov/labstandards/pdf/hs/CDC_Certified_Vitamin_D_Procedures.pdf (accessed on 5 June 2017).
- U.S. Food and Drug Administration. 510(k) Substantial Equivalence Determination Decision Summary - IDS-iSYS 25-Hydroxy Vitamin DS Assay, (no date). Available online: https://www.accessdata.fda.gov/cdrh_docs/reviews/K140554.pdf (accessed on 5 June 2017).
- Cluse, Z.N.; Fudge, A.N.; Whiting, M.J.; McWhinney, B.; Parkinson, I.; O′Loughlin, P.D. Evaluation of 25-hydroxy vitamin D assay on the immunodiagnostic systems iSYS analyser. Ann. Clin. Biochem. 2012, 49, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Koivula, M.K.; Turpeinen, U.; Laitinen, P.; Risteli, J. Comparison of automated 25-OH vitamin D immunoassays with liquid chromatography isotope dilution tandem mass spectrometry. Clin. Lab. 2012, 58, 1253–1261. [Google Scholar] [PubMed]
- Immunodiagnosticsystems. IDS-iSYS 25-Hydroxy Vitamin Ds: Instructions for Use, 2015. IDS-iSYS 25-Hydroxy Vitamin Ds. Available online: http://www.idsplc.com/products/ids-isys-25-hydroxy-vitamin-d-2/ (accessed on 5 June 2017).
- Ellis, K.J.; Yao, M.; Shypailo, R.J.; Urlando, A.; Wong, W.W.; Heird, W.C. Body-composition assessment in infancy: Air-displacement plethysmography compared with a reference 4-compartment model. Am. J. Clin. Nutr. 2007, 85, 90–95. [Google Scholar] [PubMed]
- Ma, G.; Yao, M.; Liu, Y.; Lin, A.; Zou, H.; Urlando, A.; Wong, W.W.; Nommsen-Rivers, L.; Dewey, K.G. Validation of a new pediatric air-displacement plethysmograph for assessing body composition in infants. Am. J. Clin. Nutr. 2004, 79, 653–660. [Google Scholar] [PubMed]
- Demerath, E.W.; Fields, D.A. Body composition assessment in the infant. Am. J. Hum. Biol. 2014, 26, 291–304. [Google Scholar] [CrossRef] [PubMed]
- Starling, A.P.; Brinton, J.T.; Glueck, D.H.; Shapiro, A.L.; Harrod, C.S.; Lynch, A.M.; Siega-Riz, A.M.; Dabelea, D. Associations of maternal BMI and gestational weight gain with neonatal adiposity in the Healthy Start study. Am. J. Clin. Nutr. 2015, 101, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.M.; Crozier, S.R.; Harvey, N.C.; Barton, B.D.; Law, C.M.; Godfrey, K.M.; Cooper, C.; Inskip, H.M. Modifiable early-life risk factors for childhood adiposity and overweight: An analysis of their combined impact and potential for prevention. Am. J. Clin. Nutr. 2015, 101, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Harrod, C.S.; Chasan-Taber, L.; Reynolds, R.M.; Fingerlin, T.E.; Glueck, D.H.; Brinton, J.T.; Dabelea, D. Physical activity in pregnancy and neonatal body composition: The Healthy Start study. Obstet. Gynecol. 2014, 124, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Harrod, C.S.; Fingerlin, T.E.; Chasan-Taber, L.; Reynolds, R.M.; Glueck, D.H.; Dabelea, D. Exposure to prenatal smoking and early-life body composition: The Healthy Start Study. Obes. 2015, 23, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Catalano, P.M. The impact of gestational diabetes and maternal obesity on the mother and her offspring. J. Dev. Orig. Health. Dis. 2010, 1, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Seto, T.L.; Tabangin, M.E.; Langdon, G.; Mangeot, C.; Dawodu, A.; Steinhoff, M.; Narendran, V. Racial disparities in cord blood vitamin D levels and its association with small-for-gestational-age infants. J. Perinatol. 2016, 36, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Vinkhuyzen, A.A.; Eyles, D.W.; Burne, T.H.; Blanken, L.M.; Kruithof, C.J.; Verhulst, F.; Jaddoe, V.W.; Tiemeier, H.; McGrath, J.J. Prevalence and predictors of vitamin D deficiency based on maternal mid-gestation and neonatal cord bloods: The Generation R Study. J. Steroid. Biochem. Mol. Biol. 2016, 164, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Streym, S.V.; Moller, U.K.; Rejnmark, L.; Heickendorff, L.; Mosekilde, L.; Vestergaard, P. Maternal and infant vitamin D status during the first 9 months of infant life—a cohort study. Eur. J. Clin. Nutr. 2013, 67, 1022–1028. [Google Scholar] [CrossRef] [PubMed]
- Ong, Y.L.; Quah, P.L.; Tint, M.T.; Aris, I.M.; Chen, L.W.; van Dam, R.M.; Heppe, D.; Saw, S.M.; Godfrey, K.M.; Gluckman, P.D.; et al. The association of maternal vitamin D status with infant birth outcomes, postnatal growth and adiposity in the first 2 years of life in a multi-ethnic Asian population: the Growing Up in Singapore Towards healthy Outcomes (GUSTO) cohort study. Br. J. Nutr. 2016, 116, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Merewood, A.; Mehta, S.D.; Grossman, X.; Chen, T.C.; Mathieu, J.S.; Holick, M.F.; Bauchner, H. Widespread vitamin D deficiency in urban Massachusetts newborns and their mothers. Pediatrics 2010, 125, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.W.; Johnson, D.; Hulsey, T.C.; Ebeling, M.; Wagner, C.L. Vitamin D supplementation during pregnancy: Double-blind, randomized clinical trial of safety and effectiveness. J. Bone Miner. Res. 2011, 26, 2341–2357. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P.; Armas, L.A. Quantifying the vitamin D economy. Nutr. Rev. 2015, 73, 51–67. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D; The National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
| Mean (SD) or N (%) | ||
|---|---|---|
| Maternal characteristics | ||
| Age (years) | 27.6 | (6.3) |
| Race | ||
| Hispanic | 156 | (26%) |
| Non-Hispanic white | 328 | (54%) |
| Black | 87 | (14%) |
| Other | 34 | (6%) |
| Education | ||
| <High school degree | 94 | (16%) |
| High school degree | 114 | (19%) |
| A college or 2-year degree | 139 | (23%) |
| 4-year degree | 126 | (21%) |
| Graduate degree | 132 | (22%) |
| Household income | ||
| <$40,000 | 185 | (31%) |
| $40,000–$70,000 | 112 | (19%) |
| >$70,000 | 192 | (32%) |
| Missing/do not know | 116 | (19%) |
| Pre-pregnancy BMI (kg/m2) | 26.1 | (6.5) |
| Gestational weight gain (kg) | 14.0 | (6.6) |
| Prenatal oral vitamin D intake (IU/day) (n = 305) | 415 | (453) |
| Neonatal characteristics | ||
| Female (n) | 289 | (48%) |
| Gestational age at birth (weeks) | 39.5 | (1.1) |
| Season of birth | ||
| Summer (June, July, August) | 189 | (31%) |
| Fall (September, October, November) | 140 | (23%) |
| Winter (December, January, February) | 128 | (21%) |
| Spring (March, April, May) | 148 | (24%) |
| Total 25(OH)D in cord blood (nmol/L) | 56.0 | (21.2) |
| Body size and composition | ||
| Total mass (g) | 3158 | (412) |
| Fat-free mass (g) | 2858 | (332) |
| Fat mass (g) | 300 | (147) |
| Adiposity (%) | 9.3 | (3.8) |
| Postnatal characteristics (n = 348) | ||
| Age at postnatal visit (months) | 5.1 | (1.3) |
| Exclusive breastfeeding (%) | 151 | (43%) |
| Use of infant vitamin D-containing supplements (%) | 156.0 | (30%) |
| Body size and composition | ||
| Total mass (g) | 6860 | (881) |
| Fat-free mass (g) | 5185 | (626) |
| Fat mass (g) | 1675 | (507) |
| Adiposity (%) | 24.1 | (5.5) |
| Beta | (SE) | p-Value | |
|---|---|---|---|
| Maternal pre-pregnancy BMI (per 5 kg/m2) | –3.2 | (0.8) | <0.001 |
| Race/ethnicity | |||
| Non-Hispanic white | 1.0 | (ref) | |
| Non-Hispanic black | –15.6 | (3.4) | <0.001 |
| Hispanic | –8.2 | (3.0) | 0.006 |
| Daily prenatal oral vitamin D intake (per 100 IU) | 0.6 | (0.2) | 0.01 |
| Season of birth | |||
| Summer | 1.0 | (ref) | |
| Fall | –6.8 | (2.9) | 0.02 |
| Winter | –12.1 | (3.1) | <0.001 |
| Spring | –10.4 | (2.9) | <0.001 |
| Simple Model | Simple Model + BMI | Simple Model + GWG | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Beta | (SE) | p-Value | Beta | (SE) | p-Value | Beta | (SE) | p-Value | |
| Total mass (g) | |||||||||
| Total 25(OH)D (nmol/L) | –1.44 | (0.76) | 0.06 | –6.22 | (2.67) | 0.02 | –5.13 | (1.68) | 0.002 |
| Pre-pregnancy BMI (5 kg/m2) | 2.08 | (27.11) | 0.94 | ||||||
| Total 25(OH)D × BMI | 1.05 | (0.50) | 0.04 | ||||||
| Gestational weight gain (kg) | –4.19 | (6.13) | 0.49 | ||||||
| Total 25(OH)D × GWG | 0.26 | (0.11) | 0.02 | ||||||
| Fat-free mass (g) | |||||||||
| Total 25(OH)D (nmol/L) | –0.91 | (0.59) | 0.12 | –0.58 | (0.60) | 0.33 | –3.74 | (1.31) | 0.005 |
| Pre-pregnancy BMI (5 kg/m2) | 28.64 | (9.46) | 0.003 | ||||||
| Gestational weight gain (kg) | –3.86 | (4.79) | 0.42 | ||||||
| Total 25(OH)D x GWG | 0.20 | (0.09) | 0.02 | ||||||
| Fat mass (g) | |||||||||
| Total 25(OH)D (nmol/L) | –0.48 | (0.30) | 0.11 | –0.19 | (0.30) | 0.51 | –0.52 | (0.30) | 0.08 |
| Pre-pregnancy BMI (5 kg/m2) | 24.75 | (4.71) | 0.0001 | ||||||
| Gestational weight gain (kg) | 2.72 | (0.90) | 0.003 | ||||||
| Adiposity (%) | |||||||||
| Total 25(OH)D (nmol/L) | –0.02 | (0.01) | 0.05 | –0.01 | (0.01) | 0.25 | –0.02 | (0.01) | 0.04 |
| Pre-pregnancy BMI (5 kg/m2) | 0.57 | (0.12) | 0.0001 | ||||||
| Gestational weight gain (kg) | 0.05 | (0.02) | 0.02 | ||||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sauder, K.A.; Koeppen, H.J.; Shapiro, A.L.B.; Kalata, K.E.; Stamatoiu, A.V.; Ringham, B.M.; Glueck, D.H.; Norris, J.M.; Dabelea, D. Prenatal Vitamin D Intake, Cord Blood 25-Hydroxyvitamin D, and Offspring Body Composition: The Healthy Start Study. Nutrients 2017, 9, 790. https://doi.org/10.3390/nu9070790
Sauder KA, Koeppen HJ, Shapiro ALB, Kalata KE, Stamatoiu AV, Ringham BM, Glueck DH, Norris JM, Dabelea D. Prenatal Vitamin D Intake, Cord Blood 25-Hydroxyvitamin D, and Offspring Body Composition: The Healthy Start Study. Nutrients. 2017; 9(7):790. https://doi.org/10.3390/nu9070790
Chicago/Turabian StyleSauder, Katherine A., Hallie J. Koeppen, Allison L.B. Shapiro, Kathryn E. Kalata, Alexandra V. Stamatoiu, Brandy M. Ringham, Deborah H. Glueck, Jill M. Norris, and Dana Dabelea. 2017. "Prenatal Vitamin D Intake, Cord Blood 25-Hydroxyvitamin D, and Offspring Body Composition: The Healthy Start Study" Nutrients 9, no. 7: 790. https://doi.org/10.3390/nu9070790
APA StyleSauder, K. A., Koeppen, H. J., Shapiro, A. L. B., Kalata, K. E., Stamatoiu, A. V., Ringham, B. M., Glueck, D. H., Norris, J. M., & Dabelea, D. (2017). Prenatal Vitamin D Intake, Cord Blood 25-Hydroxyvitamin D, and Offspring Body Composition: The Healthy Start Study. Nutrients, 9(7), 790. https://doi.org/10.3390/nu9070790





