Protein Hydrolysates as Promoters of Non-Haem Iron Absorption
Abstract
:1. Introduction
2. Iron-Chelatable Protein Hydrolysates
2.1. Iron-Chelatable Amino Acids
2.2. Iron-Chelatable Di-Peptides and Tri-Peptides
2.3. Iron-Chelatable Polypeptides and Proteins
2.3.1. Iron-Chelatable Proteins
2.3.2. Iron-Chelatable Protein Fragments
2.3.3. Other Iron-Chelatable Peptides
2.4. Structural Characteristics of Iron-Chelatable Protein Hydrolysates
2.4.1. Structural Characteristics of Iron-Chelatable Amino Acids
2.4.2. Structural Characteristics of Iron-Chelatable Peptides
Special Amino Acids
Size of the Peptides
Sequences of the Peptides
3. Mechanisms of Promoting Non-Haem Iron Absorption
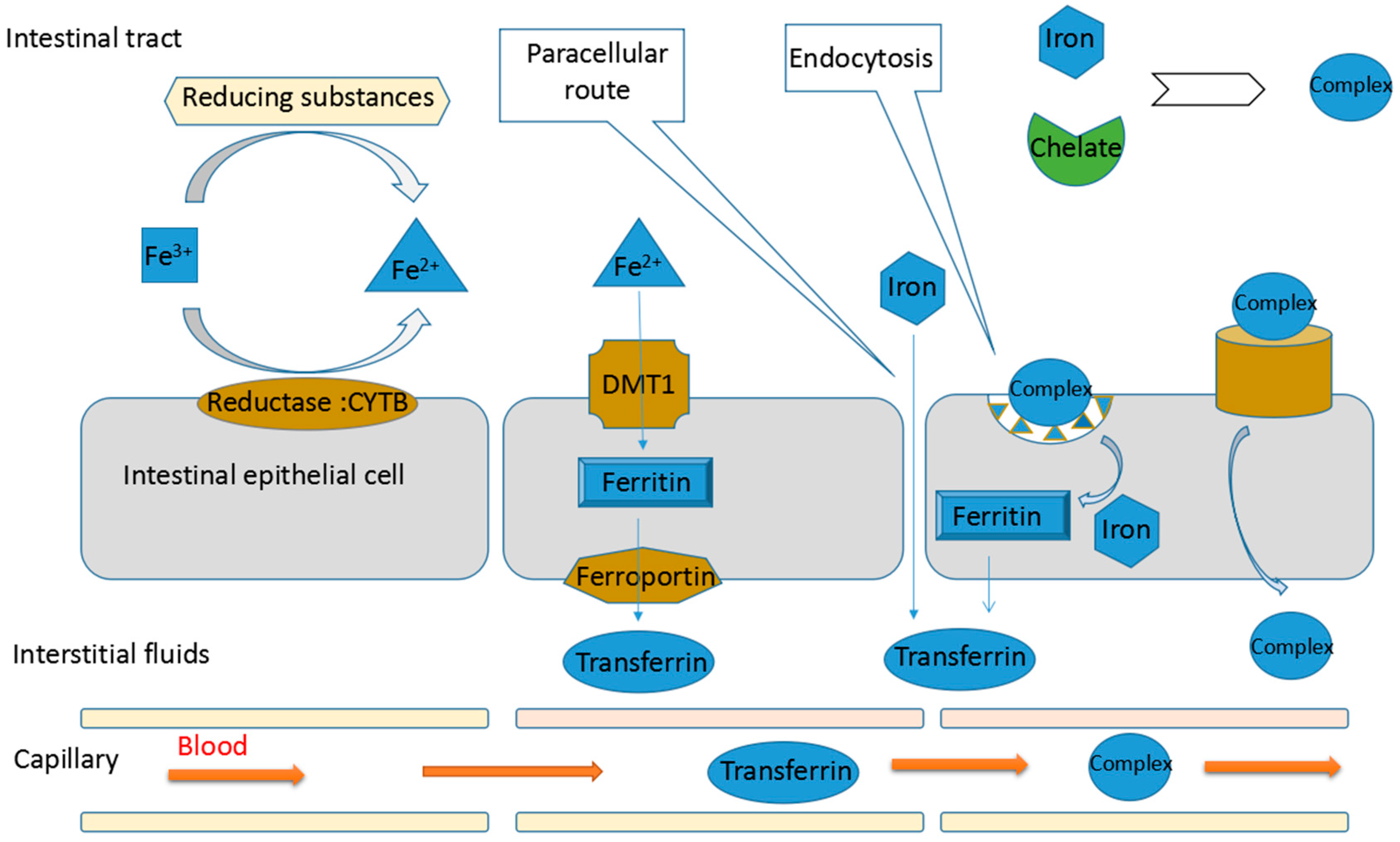
3.1. Iron Absorption
3.2. Mechanisms of Protein Hydrolysates Promoting Non-Haem Iron Absorption
3.2.1. Maintaining the Solubility of Iron
3.2.2. Reducing Ferric Ion to Ferrous Ion
3.2.3. Promoting the Passage of Iron through Intestinal Cell Membranes
4. Usages, Advantages and Challenges of Protein Hydrolysates as Non-Haem Iron Promoters
4.1. Usagesof Protein Hydrolysates as Non-Haem Iron Promoters
4.1.1. Amino Acidsas Non-Haem Iron Promoters
4.1.2. Di-Peptides and Tri-Peptidesas Non-Haem Iron Promoters
4.1.3. Polypeptidesas Non-Haem Iron Promoters
4.2. Advantagesof Protein Hydrolysates as Non-Haem Iron Promoters
4.2.1. Dual-Purpose Nutrients
4.2.2. Reducing the Side Effects of Iron Ions
4.2.3. Bioactivity
4.3. Challenges of Protein Hydrolysates as Non-Haem Iron Promoters
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization (WHO). Global Nutrition Targets 2025: Anaemia Policy Brief; Working Papers; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Baltussen, R.; Knai, C.; Sharan, M. Iron fortification and iron supplementation are cost-effective interventions to reduce iron deficiency in four subregions of the world. J. Nutr. 2016, 134, 2678–2684. [Google Scholar]
- Low, M.; Farrell, A.; Biggs, B.A.; Pasricha, S.R. Effects of daily iron supplementation in primary-school-aged children: Systematic review and meta-analysis of randomized controlled trials. Can. Med. Assoc. J. 2013, 185, 791–802. [Google Scholar] [CrossRef] [PubMed]
- Mfsrh, L.P.; Ffsrh, D.M.; Ffsrh, I.F.D.M. Iron deficiency and iron deficiency anaemia in women. Best Pract. Res. Clin. Obstet. Gynaecol. 2017, 19, 55–67. [Google Scholar] [CrossRef]
- Pineda, O. Iron bis-glycine chelate competes for the non heme-iron absorption pathway. Am. J. Clin. Nutr. 2003, 78, 495–496. [Google Scholar] [PubMed]
- Geisser, P.; Burckhardt, S. The pharmacokinetics and pharmacodynamics of iron preparations. Pharmaceutics 2011, 3, 12–33. [Google Scholar] [CrossRef] [PubMed]
- Pizarro, F.; Olivares, M.; Maciero, E.; Krasnoff, G.; Cócaro, N.; Gaitan, D. Iron Absorption from Two Milk Formulas Fortified with Iron Sulfate Stabilized with Maltodextrin and Citric Acid. Nutrients 2015, 7, 8952–8959. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Harnedy, P.A.; Li, B.; Hou, H.; Zhang, Z.; Zhao, X.; Fitzgerald, R.J. Food protein-derived chelating peptides: Biofunctional ingredients for dietary mineral bioavailability enhancement. Trends Food Sci. Technol. 2014, 37, 92–105. [Google Scholar] [CrossRef]
- Ghasemi, S.; Khoshgoftarmanesh, A.H.; Hadadzadeh, H.; Jafari, M. Synthesis of Iron-amino acid chelates and evaluation of their efficacy as iron source and growth stimulator for tomato in nutrient solution culture. J. Plant Growth Regul. 2012, 31, 498–508. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, Y.; Xue, A.; Li, C. The synthesis of iron chelate with L—Aspartic acid and aspartame. China Food Ind. 2009, 1, 60–61. (In Chinese) [Google Scholar]
- Van Campen, D. Enhancement of iron absorption from ligated segments of rat intestine by histidine, cysteine, and lysine: Effects of removing ionizing groups and of stereoisomerism. J. Nutr. 1973, 103, 139–142. [Google Scholar] [PubMed]
- Bovell-Benjamin, A.C.; Viteri, F.E.; Allen, L.H. Iron absorption from ferrous bisglycinate and ferric trisglycinate in whole maize is regulated by iron status. Am. J. Clin. Nutr. 2000, 71, 1563–1569. [Google Scholar] [PubMed]
- Kwiecien, M.; Samolinska, W.; Bujanowicz-Haras, B. Effects of iron-glycine chelate on growth, carcass characteristic, liver mineral concentrations and haematological and biochemical blood parameters in broilers. J. Anim. Physiol. Anim. Nutr. 2015, 99, 1184–1196. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Yu, X.; Diao, L.; Huang, X. Optimization of the preparation process of ferrous glutamate by response surface methodology. Food Sci. 2015, 36, 81–85. [Google Scholar] [CrossRef]
- Djurdjevic, P.; Jelic, R. Solution equilibria in l-glutamic acid and l-serine + iron(III) systems. Transit. Met. Chem. 1997, 22, 284–293. [Google Scholar] [CrossRef]
- Van Campen, D.; Gross, E. Effect of histidine and certain other amino acids on the absorption of iron-59 by rats. J. Nutr. 1969, 99, 68–74. [Google Scholar] [PubMed]
- Tao, J.; Hu, X.; Sun, W.; Nie, S.; Xie, M. Study on antioxidant activity of lipids with iron threonine chelate. Sci. Technol. Food Ind. 2012, 33, 137–139. [Google Scholar] [CrossRef]
- Chen, X.; Lei, X.; Wang, Q.; Du, Z.; Lu, J.; Chen, S.; Zhang, M.; Hao, Z.; Ren, F. Effects of a tripeptide iron on iron-deficiency anemia in rats. Biol. Trace Elem. Res. 2015, 169, 211–217. [Google Scholar] [CrossRef]
- Wang, C.; Li, B.; Jing, A. Separation and identification of zinc-chelating peptides from sesame protein hydrolysate using IMAC-Zn2+ and LC-MS/MS. Food Chem. 2012, 134, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sun, X.; Xie, C.; Shu, X.; Oso, A.O.; Ruan, Z.; Deng, Z.Y.; Wu, X.; Yin, Y. Effects of ferrous carbamoyl glycine on iron state and absorption in an iron-deficient rat model. Genes Nutr. 2015, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.M.; Deng, S.G.; Zhang, B.; Pang, J. Separation, Structure Identification and Antimicrobial Activity of Ferrous Chelate of Protein Hydrolysate in Hairtail (Hrichiurus haumela). J. Sin. Mol. Res. 2013, 1, 2–6. [Google Scholar] [CrossRef]
- Kholnazarov, B.M.; Bunyatyan, N.D.; Shakhmatov, A.N.; Bobiev, G.M. Development of an immunotropic drug based on coordination compounds of a synthetic low molecular weight thymus peptide with iron Ions. Pharm. Chem. J. 2014, 48, 632–634. [Google Scholar] [CrossRef]
- Guo, L.; Hou, H.; Li, B.; Zhang, Z.; Wang, S.; Zhao, X. Preparation, isolation and identification of iron-chelating peptides derived from Alaska pollock skin. Process Biochem. 2013, 48, 988–993. [Google Scholar] [CrossRef]
- Guo, L.; Harnedy, P.A.; O’Keeffe, M.B.; Li, Z.; Li, B.; Hu, H.; Fitzgerald, R.J. Fractionation and identification of Alaska pollock skin collagen-derived mineral chelating peptides. Food Chem. 2015, 173, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Eckert, E.; Lu, L.; Unsworth, L.D.; Chen, L.; Xie, J.; Xu, R. Biophysical and in vitro absorption studies of iron chelating peptide from barley proteins. J. Funct. Foods 2016, 25, 291–301. [Google Scholar] [CrossRef]
- Argyri, K.; Miller, D.D.; Glahn, R.P.; Zhu, L.; Kapsokefalou, M. Peptides isolated from in vitro digests of milk enhance iron uptake by Caco-2 cells. J. Agric. Food Chem. 2007, 55, 10221–10225. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Liu, Z.; Zhao, Y.; Zeng, M. Enzymatic preparation and characterization of iron-chelating peptides from anchovy (Engraulis japonicus) muscle protein. Food Res. Int. 2012, 48, 435–441. [Google Scholar] [CrossRef]
- Jaiswal, A.; Bajaj, R.; Mann, B.; Lata, K. Iron (II)-chelating activity of buffalo αS-casein hydrolysed by corolase PP, alcalase and flavourzyme. J. Food Sci. Technol. 2015, 52, 3911–3918. [Google Scholar] [CrossRef] [PubMed]
- Garcíanebot, M.J.; Alegría, A.; Barberá, R.; Gaboriau, F.; Bouhallab, S. Effect of caseinophosphopeptides from αs- and β-Casein on iron bioavailability in HuH7 cells. J. Agric. Food Chem. 2015, 63, 6757–6763. [Google Scholar] [CrossRef] [PubMed]
- Prakash, D.; Lakshmi, A.J. Preparation of caseinophosphopeptides and assessing their efficacy in enhancing the bioaccessibility of iron and zinc. J. Food Sci. Technol. 2015, 52, 7493–7499. [Google Scholar] [CrossRef]
- Torres-Fuentes, C.; Alaiz, M.; Vioque, J. Iron-chelating activity of chickpea protein hydrolysate peptides. Food Chem. 2012, 134, 1585–1588. [Google Scholar] [CrossRef] [PubMed]
- Cai, B.N.; Chen, X.; Pan, J.Y.; Deng, W.H.; Wan, P.; Chen, D.K.; Sun, H.L. Optimization of preparation process for cod skin collagen peptide-iron (II) Chelate via Response Surface Methodology. Food Sci. 2012, 33, 48–52. [Google Scholar] [CrossRef]
- Suzuki, S.; Fukuda, K.; Irie, M.; Hata, Y. Iron chelated cyclic peptide, ferrichrysin, for oral treatment of iron deficiency: Solution properties and efficacy in anemic rats. Int. J. Vitam. Nutr. Res. 2007, 77, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.M.; Deng, S.G.; Huang, S.B.; Li, Y.J.; Song, R. The effect of ferrous-chelating hairtail peptides on iron deficiency and intestinal flora in rats. J. Sci. Food Agric. 2015, 96, 2839–2844. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Shi, Z.R.; Wang, X.L.; Deng, S.G. The effects of hairtail protein hydrolysate-Fe2+ complexes on growth and non-specific immune response of red swamp crayfish (Procambarus clarkii). Aquac. Int. 2016, 24, 1039–1048. [Google Scholar] [CrossRef]
- Wang, P.F.; Huang, G.R.; Jiang, J.X. Optimization of hydrolysis conditions for the production of iron-binding peptides from mackerel processing byproducts. Adv. J. Food Sci. Technol. 2013, 5, 921–925. [Google Scholar]
- Lin, H.M.; Deng, S.G.; Huang, S.B. Antioxidant activities of ferrous-chelating peptides isolated from five types of low-value fish protein hydrolysates. J. Food Biochem. 2014, 38, 627–633. [Google Scholar] [CrossRef]
- Figueroa-Hernández, C.; Cruz-Guerrero, A.; Rodríguez-Serrano, G.; Gómez-Ruiz, L.; García-Garibay, M.; Jiménez-Guzmán, J. Calcium and iron binding peptides production by Lactococcus lactis sp. Cremoris NCFB 712. Revista Mexicana De Ingeniería Química 2012, 11, 259–267. [Google Scholar]
- Blat, D.; Weiner, L.; Youdim, M.B.; Fridkin, M. A novel iron-chelating derivative of the neuroprotective peptide NAPVSIPQ shows superior antioxidant and antineurodegenerative capabilities. J. Med. Chem. 2008, 51, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Song, K.B. Purification of an iron-binding nona-peptide from hydrolysates of porcine blood plasma protein. Process Biochem. 2009, 44, 378–381. [Google Scholar] [CrossRef]
- Cao, Y.; Chen, Q.; Xiong, H.; Liang, L. Optimal conditions for preparating iron chelate of enzymic hydrolysis peptides from rice protein. Food Ferment. Ind. 2007, 33, 61–64. [Google Scholar] [CrossRef]
- Zhang, W.; Li, Y.; Zhang, J.; Huang, G. Optimization of hydrolysis conditions for the production of iron-binding peptides from Scad (Decapterus maruadsi) processing byproducts. Am. J. Biochem. Biotechnol. 2016. [Google Scholar] [CrossRef]
- Cian, R.E.; Garzón, A.G.; Ancona, D.B.; Guerrero, L.C.; Drago, S.R. Chelating properties of peptides from red seaweed Pyropia columbina and its effect on iron bio-accessibility. Plant Foods Hum. Nutr. 2016, 71, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.J.; Lee, H.-S.; Jung, E.Y.; Park, S.Y.; Lim, W.-T.; Lee, J.-Y.; Yeon, S.-H.; Lee, J.-C.; Suh, H.J. Manufacturing of iron binding peptide using sericin hydrolysate and its bioavailability in iron deficient Rat. J. Korean Soc. Food Sci. Nutr. 2010, 39, 1446–1451. [Google Scholar] [CrossRef]
- Huang, G.; Ren, Z.; Jiang, J.; Chen, W. Purification of a hepta-peptide with iron binding activity from shrimp processing by-products hydrolysates. Adv. J. Food Sci. Technol. 2012, 4, 207–212. [Google Scholar] [CrossRef]
- Huang, G.R.; Ren, Z.Y.; Jiang, J.X. Optimization of hydrolysis conditions for iron binding peptides production from shrimp processing byproducts. Am. J. Food Technol. 2014, 9, 49–55. [Google Scholar] [CrossRef]
- Kim, N.H.; Jung, S.H.; Kim, J.; Kim, S.H.; Ahn, H.J.; Song, K.B. Purification of an iron-chelating peptide from spirulina protein hydrolysates. J. Korean Soc. Appl. Biol. Chem. 2014, 57, 91–95. [Google Scholar] [CrossRef]
- Lv, Y.; Liu, Q.; Bao, X.; Tang, W.; Yang, B.; Guo, S. Identification and characteristics of iron-chelating peptides from soybean protein hydrolysates using IMAC-Fe3+. J. Agric. Food Chem. 2009, 57, 4593–4597. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Bao, X.; Liu, H.; Ren, J.; Guo, S. Purification and characterization of calcium-binding soybean protein hydrolysates by Ca2+/Fe3+ immobilized metal affinity chromatography (IMAC). Food Chem. 2013, 141, 1645–1650. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, T.; Yamamoto, M.; Hirai, Y. Absorption and availability of iron peptide in pregnant sows. Bull. Nippon Vet. Zootech. Coll. 1989, 38, 93–105. [Google Scholar]
- Zhang, M.N.; Huang, G.R.; Jiang, J.X. Iron binding capacity of dephytinised soy protein isolate hydrolysate as influenced by the degree of hydrolysis and enzyme type. J. Food Sci. Technol. 2014, 51, 994–999. [Google Scholar] [CrossRef] [PubMed]
- Caetano-Silva, M.E.; Cilla, A.; Bertoldo-Pacheco, M.T.; Netto, F.M.; Alegría, A. Evaluation of in vitro iron bioavailability in free form and as whey peptide-iron complexes. J. Food Compos. Anal. 2017. [Google Scholar] [CrossRef]
- Małgorzata, K.; Małgorzata, E.; Ewa, I.; Teresa, A.; Hanna, S.; Magdalena, Z.-D.; Dorota, P.-K.; Jadwiga, J.-L. Evaluation of safety of iron-fortified soybean sprouts, a potential component of functional food, in rat. Plant Foods Hum. Nutr. 2016, 71, 1–6. [Google Scholar] [CrossRef]
- And, M.Z.; Hearn, M.T.W. Application of immobilized metal ion chelate complexes as pseudocation exchange adsorbents for protein separation. Biochemistry 1996, 35, 202–211. [Google Scholar] [CrossRef]
- Zhu, C.; Liu, L.; Deng, J.; Ma, X.; Hui, J.; Fan, D. Formation mechanism and biological activity of novel thiolated human-like collagen iron complex. J. Biomater. Appl. 2016, 30, 1205–1218. [Google Scholar] [CrossRef] [PubMed]
- Shilpashree, B.G.; Arora, S.; Sharma, V. Preparation of iron/zinc bound whey protein concentrate complexes and their stability. Lebensm.-Wiss. Technol. 2015, 66, 514–522. [Google Scholar] [CrossRef]
- Freeman, H.J. Clinical relevance of intestinal peptide uptake. World J. Gastrointest. Pharmacol. Ther. 2015, 6, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Glahn, R.P.; Van Campen, D.R. Iron uptake is enhanced in Caco-2 cell monolayers by cysteine and reduced cysteinyl glycine. J. Nutr. 1997, 127, 642–647. [Google Scholar] [PubMed]
- Geissler, S.; Hellwig, M.; Markwardt, F.; Henle, T.; Brandsch, M. Synthesis and intestinal transport of the iron chelator maltosine in free and dipeptide form. Eur. J. Pharm. Biopharm. 2011, 78, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Vij, R.; Reddi, S.; Kapila, S.; Kapila, R. Transepithelial transport of milk derived bioactive peptide VLPVPQK. Food Chem. 2016, 190, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Morishita, M.; Peppas, N.A. Is the oral route possible for peptide and protein drug delivery? Drug Discov. Today 2006, 11, 905–910. [Google Scholar] [CrossRef] [PubMed]
- Kyyaly, M.A.; Powell, C.; Ramadan, E. Preparation of iron-enriched baker’s yeast and its efficiency in recovery of rats from dietary iron deficiency. Nutrition 2015, 31, 1155–1164. [Google Scholar] [CrossRef] [PubMed]
- Palika, R.; Mashurabad, P.C.; Nair, M.K.; Reddy, G.B.; Pullakhandam, R. Characterization of iron-binding phosphopeptide released by gastrointestinal digestion of egg white. Food Res. Int. 2015, 67, 308–314. [Google Scholar] [CrossRef]
- Huang, C.Y.; Wu, C.H.; Yang, J.I.; Li, Y.H.; Kuo, J.M. Evaluation of iron-binding activity of collagen peptides prepared from the scales of four cultivated fishes in Taiwan. J. Food Drug Anal. 2015, 23, 671–678. [Google Scholar] [CrossRef]
- Falanga, A.; Galdiero, M.; Galdiero, S. Membranotropic cell penetrating peptides: The outstanding journey. Int. J. Mol. Sci. 2015, 16, 25323–25337. [Google Scholar] [CrossRef] [PubMed]
- Katayama, N.; Nozaki, Y.; Okonogi, K.; Harada, S.; Ono, H. Ferrocins, new iron-containing peptide antibiotics produced by bacteria. Taxonomy, fermentation and biological activity. J. Antibiot. 1993, 46, 65–70. [Google Scholar] [CrossRef] [PubMed]
- ReyesMéndez, A.I.; Figueroa-Hernández, C.; Melgar-Lalanne, G.; Hernández-Sánchez, H.; Dávila-Ortiz, G.; Jiménez-Martínez, C. Production of calcium- and iron-binding peptides by probiotic strains of Bacillus subtilis, B. clausii and B. coagulans GBI-30. Revista Mexicana De Ingeniería Química 2015, 14, 239–245. [Google Scholar]
- Barnes, C.L.; Eng-Wilmot, D.L.; Helm, D.V.D. Ferricrocin (C29H44FeN9O13.7H2O), an iron(III)-binding peptide from Aspergillus versicolor. Acta Crystallogr. Sect. C 1984, 40, 922–926. [Google Scholar] [CrossRef]
- Zachariou, M.; Hearn, M.T.W. Protein selectivity in immobilized metal affinity chromatography based on the surface accessibility of aspartic and glutamic acid residues. J. Protein Chem. 1995, 14, 419–430. [Google Scholar] [CrossRef] [PubMed]
- Gurd, F.R.N.; Wilcox, P.E. Complex formation between metallic cations and proteins, peptides, and amino acids. Adv. Protein Chem. 1956, 11, 311–427. [Google Scholar] [CrossRef] [PubMed]
- Amadou, I.; Le, G.W.; Shi, Y.H.; Jin, S. Reducing, Reducing, radical scavenging, and chelation properties of fermented soy protein meal hydrolysate by lactobacillus plantarum lp6. Int. J. Food Prop. 2011, 14, 654–665. [Google Scholar] [CrossRef]
- Taylor, P.G.; Martínez-Torres, C.; Romano, E.L.; Layrisse, M. The effect of cysteine-containing peptides released during meat digestion on iron absorption in humans. Am. J. Clin. Nutr. 1986, 43, 68–71. [Google Scholar] [PubMed]
- García-Nebot, M.J.; Barberá, R.; Alegría, A. Iron and zinc bioavailability in Caco-2 cells: Influence of caseinophosphopeptides. Food Chem. 2013, 138, 1298–1303. [Google Scholar] [CrossRef] [PubMed]
- Na, S.; Cui, P.; Jin, Z.; Wu, H.; Wang, Y.; Lin, S. Contributions of molecular size, charge distribution, and specific amino acids to the iron-binding capacity of sea cucumber (Stichopus japonicus) ovum hydrolysates. Food Chem. 2017, 230, 627–636. [Google Scholar] [CrossRef]
- Fillebeen, C.; Gkouvatsos, K.; Fragoso, G.; Calve, A.; Garcia-Santos, D.; Buffler, M.; Becker, C.; Schumann, K.; Ponka, P.; Santos, M.M.; et al. Mice are poor heme absorbers and do not require intestinal Hmox1 for dietary heme iron assimilation. Haematologica 2015, 100, 334–337. [Google Scholar] [CrossRef] [PubMed]
- Taghavi, S.; Amiri, A.; Amin, A.; Ehsani, A.; Maleki, M.; Naderi, N. Oral iron therapy with polysaccharide-iron complex may be useful in increasing the ferritin level for a short time in patients with dilated cardiomyopathy. Res. Cardiovasc. Med. 2016. [Google Scholar] [CrossRef]
- Cao, C.; Thomas, C.E.; Insogna, K.L.; O’Brien, K.O. Duodenal absorption and tissue utilization of dietary heme and nonheme iron differ in rats. J. Nutr. 2014, 144, 1710–1717. [Google Scholar] [CrossRef] [PubMed]
- Santos, T.; Connolly, C.; Murphy, R. Trace element inhibition of phytase activity. Biol. Trace Elem. Res. 2015, 163, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Cilla, A.; Perales, S.; Lagarda, M.J.; Barbera, R.; Farre, R. Iron bioavailability in fortified fruit beverages using ferritin synthesis by Caco-2 cells. J. Agric. Food Chem. 2008, 56, 8699–8703. [Google Scholar] [CrossRef] [PubMed]
- Kibangou, I.B.; Bouhallab, S.; Henry, G.; Bureau, F.; Allouche, S.; Blais, A.; Guérin, P.; Arhan, P.; Bouglé, G.L. Milk proteins and iron absorption: Contrasting effects of different caseinophosphopeptides. Pediatric Res. 2005, 58, 731–734. [Google Scholar] [CrossRef] [PubMed]
- Pérès, J.M.; Bouhallab, S.; Bureau, F.; Neuville, D.; Maubois, J.L.; Devroede, G.; Arhan, P.; Bouglé, D. Mechanisms of absorption of caseinophosphopeptide bound iron. J. Nutr. Biochem. 1999, 10, 215–222. [Google Scholar] [CrossRef]
- Ma, W.-Q.; Wu, J.; Zhuo, Z.; Sun, H.; Yue, M.; Feng, J. Comparison of Absorption Characteristics of Iron Glycine Chelate and Ferrous Sulfate in Caco-2 Cells. Int. J. Agric. Biol. 2013, 15, 372–376. [Google Scholar]
- Zhuo, Z.; Fang, S.; Yue, M.; Zhang, Y.; Feng, J. Kinetics absorption characteristics of ferrous glycinate in SD rats and its impact on the relevant transport protein. Biol. Trace Elem. Res. 2014, 158, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Bouhallab, S.; Bouglé, D. Biopeptides of milk: Caseinophosphopeptides and mineral bioavailability. Reprod. Nutr. Dev. 2004, 44, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Hoz, L.D.L.; Silva, V.S.N.D.; Morgano, M.A.; Pacheco, M.T.B. Small peptides from enzymatic whey hydrolyzates increase dialyzable iron. Int. Dairy J. 2014, 38, 145–147. [Google Scholar] [CrossRef]
- Fouad, G.T.; Evans, M.; Sharma, P.; Baisley, J.; Crowley, D.; Guthrie, N. A randomized, double-blind clinical study on the safety and tolerability of an iron multi-amino acid chelate preparation in premenopausal women. J. Diet. Suppl. 2013, 10, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Bisbe, E.; Molto, L.; Arroyo, R.; Muniesa, J.M.; Tejero, M. Randomized trial comparing ferric Carboxymaltose vs oral ferrous glycine sulphate for postoperative anaemia after total knee arthroplasty. Br. J. Anaesth. 2014, 113, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Wu, X.; Shu, X. A kind of dipeptide-iron used for feed supplement and its preparation method. CN201010540740.6, 2 November 2011. [Google Scholar]
- Galaffu, N.; Habeych Narvaez, E.A.; Ho Dac, T.; Sabatier, M. Food Composition Comprising Desferrichrysin. WO 2014/111532 A1, 24 July 2014. [Google Scholar]
- Li, W.L.; Han, H.F.; Zhang, L.; Zhang, Y.; Qu, H.B. Manufacturer identification and storage time determination of “Dong’e Ejiao” using near infrared spectroscopy and chemometrics. J. Zhejiang Univ.-Sci. B Biomed. Biotechnol. 2016, 17, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Kongkachuichai, R.; Yasumoto, K. Effects of amino acids and dipeptides on the uptake and transport of iron by Caco-2 cells cultured in serum-free medium. Food Sci. Technol. Int. 1997, 3, 279–284. [Google Scholar] [CrossRef]
- Abdel Moety, G.A.; Ali, A.M.; Fouad, R.; Ramadan, W.; Belal, D.S.; Haggag, H.M. Amino acid chelated iron versus an iron salt in the treatment of iron deficiency anemia with pregnancy: A randomized controlled study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 210, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Rojas, M.L.; Sanchez, J.; Villada, O.; Montoya, L.; Diaz, A.; Vargas, C.; Chica, J.; Herrera, A.M. Effectiveness of iron amino acid chelate versus ferrous sulfate as part of a food complement in preschool children with iron deficiency, Medellin, 2011. Biomedica 2013, 33, 350–360. [Google Scholar] [CrossRef] [PubMed]
- Shi, R.; Liu, D.; Sun, J.; Jia, Y.; Zhang, P. Effect of replacing dietary FeSO4 with equal Fe-levelled iron glycine chelate on broiler chickens. Czech J. Anim. Sci. 2015, 60, 233–239. [Google Scholar] [CrossRef]
- Layrisse, M.; Garcia-Casal, M.N.; Solano, L.; Baron, M.A.; Arguello, F.; Llovera, D.; Ramirez, J.; Leets, I.; Tropper, E. Iron bioavailability in humans from breakfasts enriched with iron bis-glycine chelate, phytates and polyphenols. J. Nutr. 2000, 130, 2195–2199. [Google Scholar] [PubMed]
- Osman, A.K.; Al-Othaimeen, A. Experience with ferrous bis-glycine chelate as an iron fortificant in milk. Int. J. Vitam. Nutr. Res. 2002, 72, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Layrisse, M.; Martínez-Torres, C.; Leets, I.; Taylor, P.; Ramírez, J. Effect of histidine, cysteine, glutathione or beef on iron absorption in humans. J. Nutr. 1984, 114, 217–223. [Google Scholar] [PubMed]
- Kobayashi, Y.; Wakasugi, E.; Yasui, R.; Kuwahata, M.; Kido, Y. Egg Yolk Protein Delays Recovery while Ovalbumin Is Useful in Recovery from Iron Deficiency Anemia. Nutrients 2015, 7, 4792–4803. [Google Scholar] [CrossRef] [PubMed]
- Smialowska, A.; Matiamerino, L.; Carr, A.J. Assessing the iron chelation capacity of goat casein digest isolates. J. Dairy Sci. 2017, 100, 2553–2563. [Google Scholar] [CrossRef] [PubMed]
- Ait-Oukhatar, N.; Peres, J.M.; Bouhallab, S.; Neuville, D.; Bureau, F.; Bouvard, G.; Arhan, P.; Bougle, D. Bioavailability of caseinophosphopeptide-bound iron. J. Lab. Clin. Med. 2002, 140, 290–294. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Dong, Y.; Qi, B.; Liu, L.; Zhou, G.; Bai, X.; Yang, C.; Zhao, D.; Zhao, Y. Preventive effects of collagen Peptide from deer sinew on bone loss in ovariectomized rats. Evid. Based Complement. Altern. Med. 2014, 2014, 627285. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.-B.; Lin, H.-C.; Chang, Y.-W. Analysis of proteins and potential bioactive peptides from tilapia (Oreochromis spp.) processing co-products using proteomic techniques coupled with BIOPEP database. J. Funct. Foods 2015, 19, 629–640. [Google Scholar] [CrossRef]
- Chen, Q.; Guo, L.; Du, F.; Chen, T.; Hou, H.; Li, B. The chelating peptide (GPAGPHGPPG) derived from Alaska pollock skin enhances calcium, zinc and iron transport in Caco-2 cells. Int. J. Food Sci. Technol. 2017, 52, 1283–1290. [Google Scholar] [CrossRef]
- Torresfuentes, C.; Alaiz, M.; Vioque, J. Affinity purification and characterisation of chelating peptides from chickpea protein hydrolysates. J. Neurochem. 2011, 98, 1746–1762. [Google Scholar] [CrossRef]
- Delgado, M.C.O.; Galleano, M.; Añón, M.C.; Tironi, V.A. Amaranth peptides from simulated gastrointestinal digestion: Antioxidant activity against reactive species. Plant Foods Hum. Nutr. 2015, 70, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Pan, Y.; Yuan, Z.H.; Zhang, H.; Peng, B.Y.; Wang, F.F.; Qian, W. Two-component signaling system VgrRS directly senses extracytoplasmic and intracellular iron to control bacterial adaptation under iron depleted stress. Pathogens 2016. [Google Scholar] [CrossRef] [PubMed]
- Lysionek, A.E.; Zubillaga, M.B.; Salgueiro, M.J.; Caro, R.A.; Leonardi, N.M.; Ettlin, E.; Boccio, J.R. Stabilized ferrous gluconate as iron source for food fortification: Bioavailability and toxicity. Biol. Trace Elem. Res. 2003, 94, 73–77. [Google Scholar] [CrossRef]
- Salama, S.A.; Al-Harbi, M.S.; Abdel-Bakky, M.S.; Omar, H.A. Glutamyl cysteine dipeptide suppresses ferritin expression and alleviates liver injury in iron-overload rat model. Biochimie 2015, 115, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Pas, M.; Piskur, B.; Sustaric, M.; Raspor, P. Iron enriched yeast biomass—A promising mineral feed supplement. Bioresour. Technol. 2007, 98, 1622–1628. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.Q.; Sun, H.; Zhou, Y.; Wu, J.; Feng, J. Effects of iron glycine chelate on growth, tissue mineral concentrations, fecal mineral excretion, and liver antioxidant enzyme activities in broilers. Biol. Trace Elem. Res. 2012, 149, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Chen, D.; Xu, C.; Han, T.; Sheng, B.; Gao, X. Effect of iron supplementation on the expression of hypoxia-Inducible factor and antioxidant status in rats exposed to high-altitude hypoxia environment. Biol. Trace Elem. Res. 2014, 162, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, G.; Ponmurugan, P.; Begum, M.S. Effect of S-allylcysteine, a sulphur containing amino acid on iron metabolism in streptozotocin induced diabetic rats. J. Trace Elem. Med. Biol. 2013, 27, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H. Salsolinol, a catechol neurotoxin, induces modification of ferritin: Protection by histidine dipeptide. Environ. Toxicol. Pharmacol. 2010, 29, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Yılmaz, Z.; Kalaz, E.B.; Aydın, A.F.; Soluktekkeşin, M.; Doğruabbasoğlu, S.; Uysal, M.; Koçaktoker, N. The effect of carnosine on methylglyoxal-induced oxidative stress in rats. Arch. Physiol. Biochem. 2017, 123, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Jarosz, L.; Kwiecien, M.; Marek, A.; Gradzki, Z.; Winiarska-Mieczan, A.; Kalinowski, M.; Laskowska, E. Effects of feed supplementation with glycine chelate and iron sulfate on selected parameters of cell-mediated immune response in broiler chickens. Res. Vet. Sci. 2016, 107, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.Y.; Jan, H.J.; Fu, C.H.; Chen, T.T.; Chen, M.K.; Lee, H.M. Use of Composition Containing Iron(II) Amino Acid Chelate in Preparation of Drug for Ameliorating Diabetes. U.S. Patent 2017/0007568 A1, 12 January 2017. [Google Scholar]
- Min, K.A.; Cho, J.H.; Song, Y.K.; Kim, C.K. Iron casein succinylate-chitosan coacervate for the liquid oral delivery of iron with bioavailability and stability enhancement. Arch. Pharm. Res. 2016, 39, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Pizarro, F.; Olivares, M.; Hertrampf, E.; Mazariegos, D.I.; Arredondo, M.; Letelier, A.; Gidi, V. Iron bis-glycine chelate competes for the nonheme-iron absorption pathway. Am. J. Clin. Nutr. 2002, 76, 577–581. [Google Scholar] [PubMed]
| Class | Iron-Chelatable Substance | Sequence | Iron Valence | Reference |
|---|---|---|---|---|
| Amino acids | Arginine | R | II | [9] |
| Aspartic acid | D | II | [10] | |
| Cysteine | C | III | [11] | |
| Glycine | G | III/II | [12,13] | |
| Glutamic acid | E | II/III | [14,15] | |
| Glutamine | Q | III | [16] | |
| Histidine | H | II/III | [9,11] | |
| Lysine | K | III | [11] | |
| Methionine | M | III | [16] | |
| Serine | S | III | [15] | |
| Threonine | T | II | [17] | |
| Small peptides | Aspartame | II | [10] | |
| Arg-Glu-Glu | REE | II | [18] | |
| Asn-Cys-Ser | NCS | II | [19] | |
| Carbamyl glycine | II | [20] | ||
| His-Tyr-Asp | HYD | II | [21] | |
| Isoleucyl-tryptophan | IW | II | [22] | |
| Leu-Ala-Asn | LAN | II | [19] | |
| Reduced glutathione | GSH | II | [19] | |
| Ser-Met | SM | II | [19] | |
| Ser-Cys-His | SCH | II | [23] | |
| Ser-Ala-Cys | SAC | II | [24] | |
| Val-Pro-Leu | VPL | II | [25] | |
| Poly-peptides | α-lactalbumin and β-lactoglobulin hydrolysate | II | [25] | |
| β-casein peptide | PGPIPN | III | [26] | |
| Anchovy peptide | S(G)7LGS(G)2SIR | II | [27] | |
| Barley protein hydrolysate | SVNVPLY | II | [25] | |
| Buffalo αS-casein | II | [28] | ||
| Caseinophosphopeptide | (SpSpSpEE)n | II | [29,30] | |
| Chickpea protein hydrolysate | II/III | [31] | ||
| Cod skin peptides | II | [32] | ||
| Ferrichrysin/ferrocins | III | [33] | ||
| Hairtail protein hydrolysate | II | [21,34,35] | ||
| Hydrolysate of Alaskan pollock skin | GPAGPHGPPG/SGSTGH | II | [23,24] | |
| Mackerel hydrolysate | NPVRGN/NPDRGN | II | [36,37] | |
| Lactein | II | [38] | ||
| Peptide-hydroxamate | NAPVSIPQ | II/III | [39] | |
| Plasma hydrolysate | DLGEQYFKG | II | [40] | |
| Rice protein hydrolysate | II | [41] | ||
| Scad protein hydrolysate | III | [42] | ||
| Seaweed protein hydrolysate | II | [43] | ||
| Sericin hydrolysate | II | [44] | ||
| Shrimp protein hydrolysates | LPTGPKS | II | [45,46] | |
| Spirulina protein hydrolysate | TDPI(L)AACI(L) | II | [47] | |
| Soybean protein hydrolysate | DEGEQPRPFPFP | III/II | [48,49,50,51] | |
| Whey peptide | II | [52] | ||
| Protein | Ferritin | III | [53] | |
| Hen egg white lysozyme | III | [54] | ||
| Thiolated human-like collagen | II | [55] | ||
| Whey proteins | II | [56] |
| Class | Substance | Product Branch | Nation | References |
|---|---|---|---|---|
| Amino acids | The full spectrum of amino acids | FerrActiv® | America | [86] |
| Glycine | Ferbisol® | Spain | [87] | |
| Small peptides | Carbamyl glycine | China | [88] | |
| Polypeptides | Deferrichrysin | Worldwide | [89] | |
| Donkey-hide gelatine | Dong E® | China | [90] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Jiang, H.; Huang, G. Protein Hydrolysates as Promoters of Non-Haem Iron Absorption. Nutrients 2017, 9, 609. https://doi.org/10.3390/nu9060609
Li Y, Jiang H, Huang G. Protein Hydrolysates as Promoters of Non-Haem Iron Absorption. Nutrients. 2017; 9(6):609. https://doi.org/10.3390/nu9060609
Chicago/Turabian StyleLi, Yanan, Han Jiang, and Guangrong Huang. 2017. "Protein Hydrolysates as Promoters of Non-Haem Iron Absorption" Nutrients 9, no. 6: 609. https://doi.org/10.3390/nu9060609
APA StyleLi, Y., Jiang, H., & Huang, G. (2017). Protein Hydrolysates as Promoters of Non-Haem Iron Absorption. Nutrients, 9(6), 609. https://doi.org/10.3390/nu9060609





