Protective Effect of Dietary Calcium Intake on Esophageal Cancer Risk: A Meta-Analysis of Observational Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
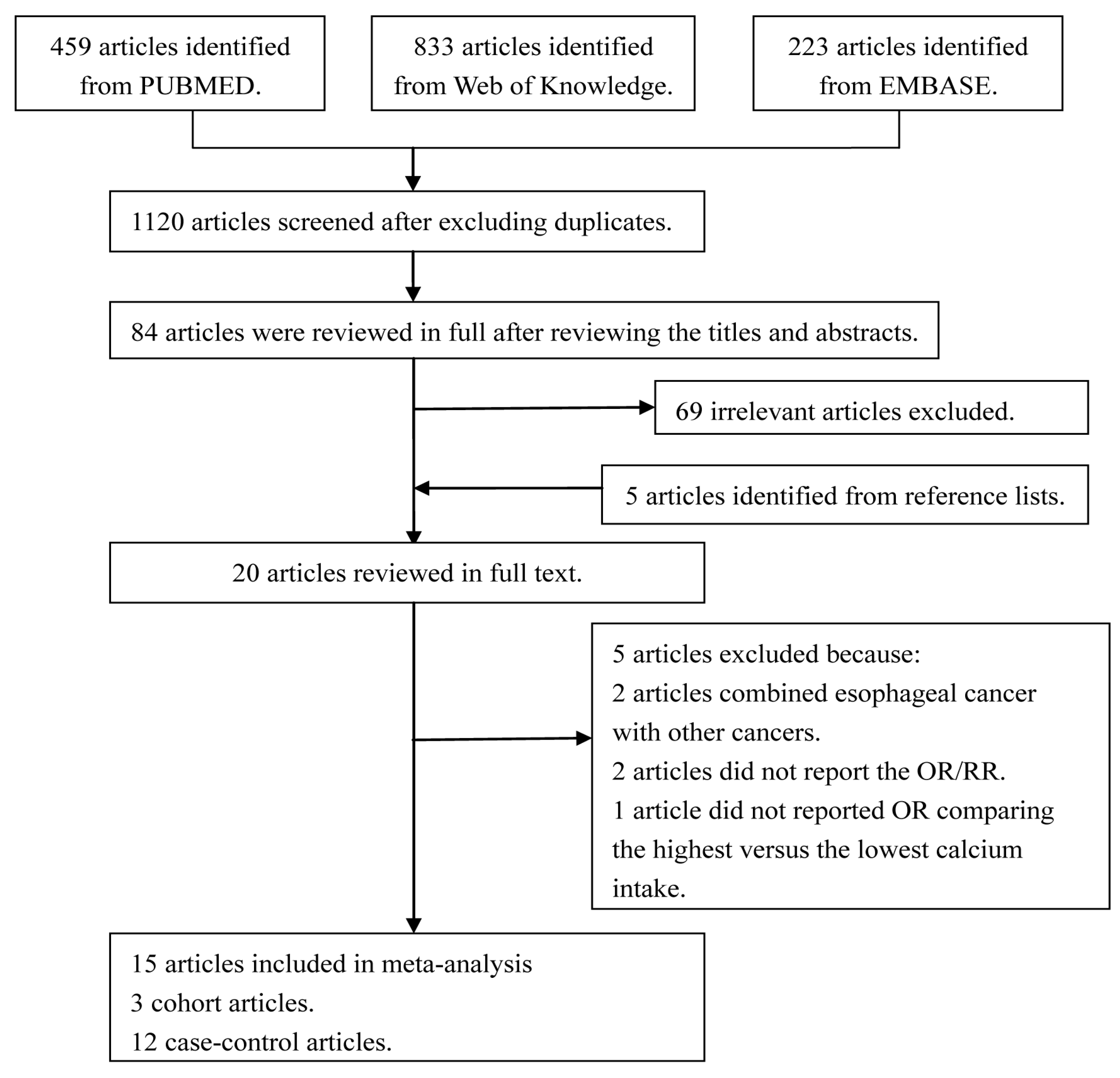
2.2. Study Selection
2.3. Data Extraction
2.4. Statistical Analysis
3. Results
3.1. Characteristics of the Included Studies
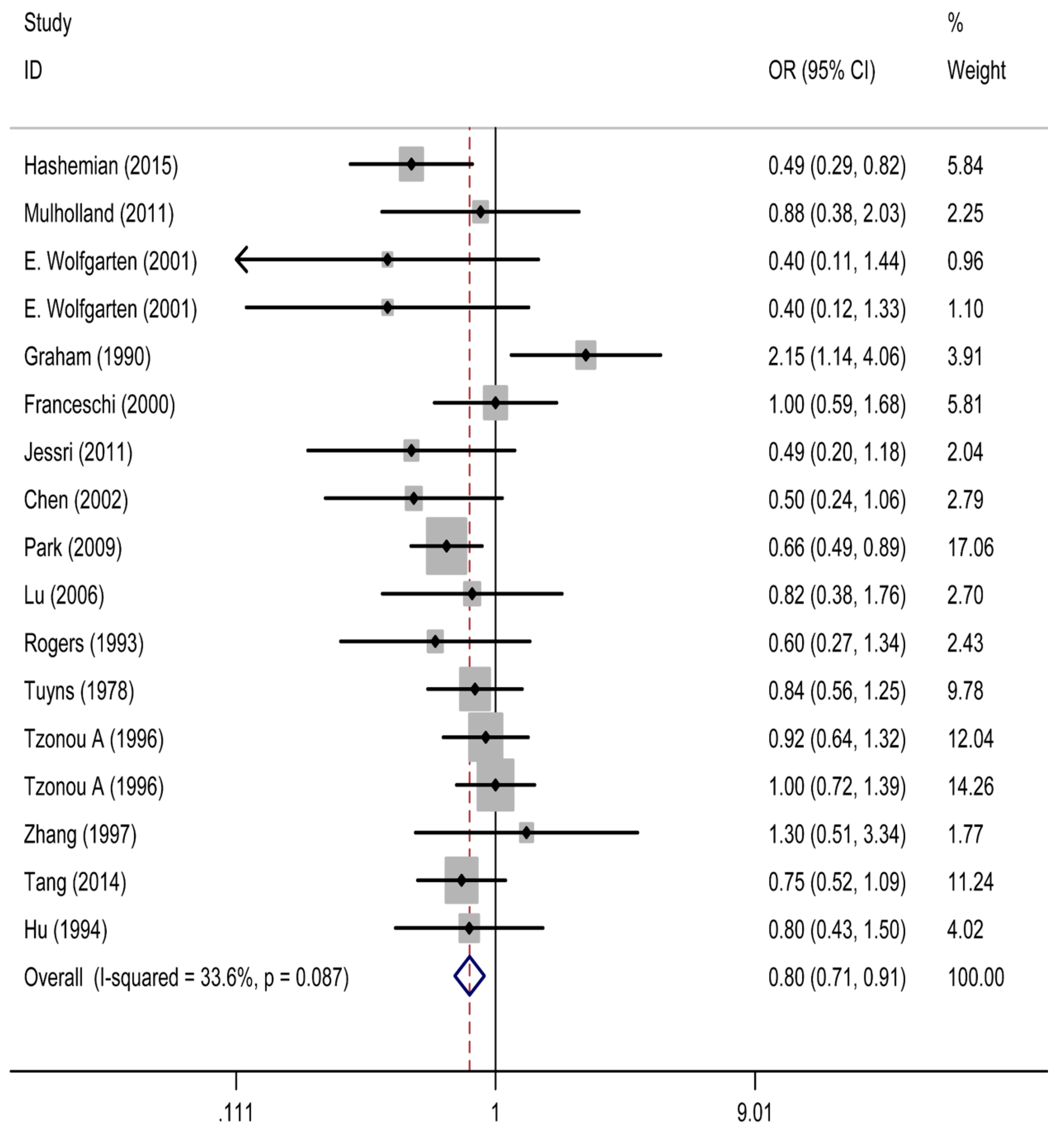
3.2. Meta-Analysis of Calcium Intake and Esophageal Cancer Risk
3.3. Heterogeneity Analysis
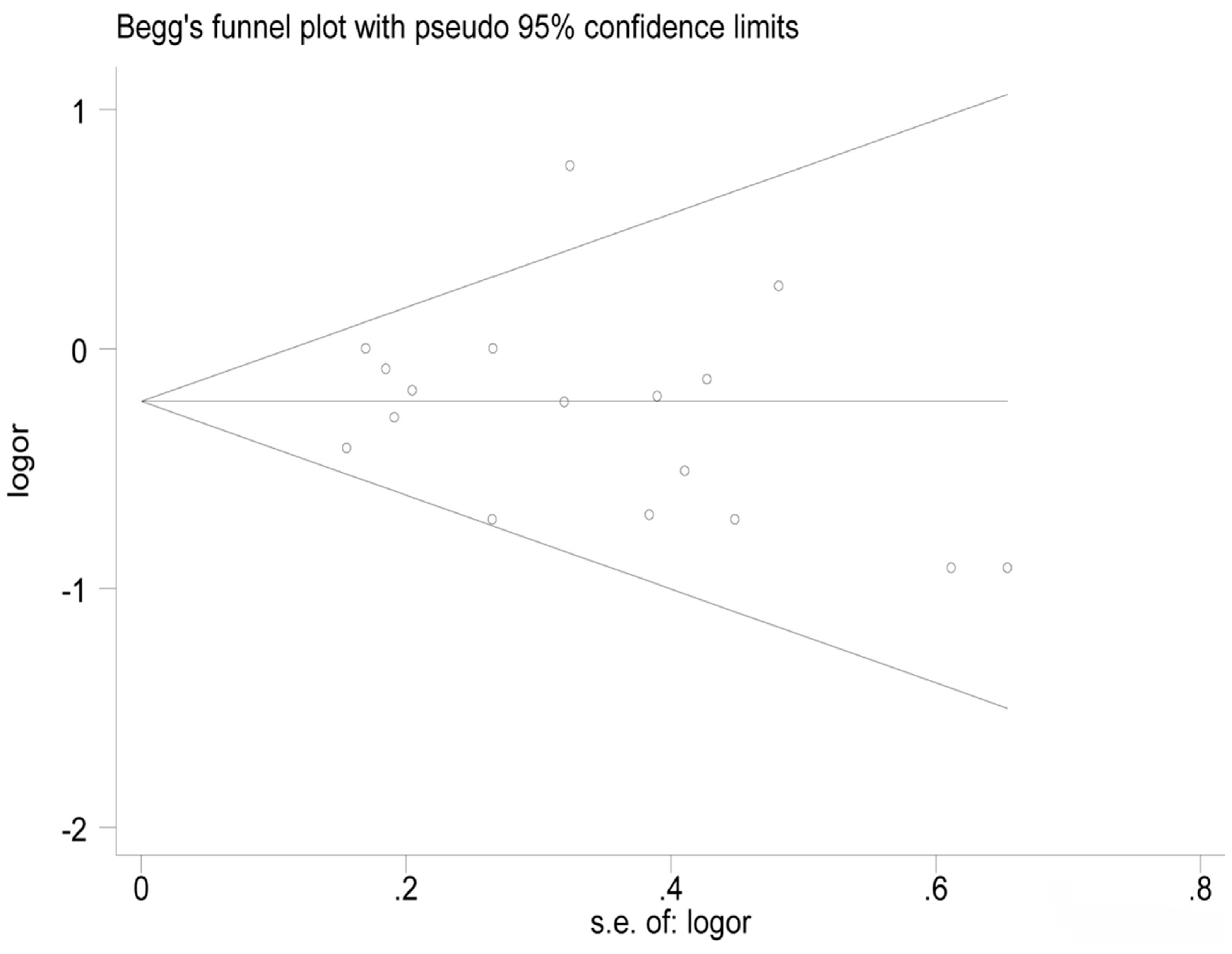
3.4. Sensitivity Analysis and Publication Bias
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflict of Interest
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Palladino-Davis, A.G.; Mendez, B.M.; Fisichella, P.M.; Davis, C.S. Dietary habits and esophageal cancer. Dis. Esophagus 2015, 28, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Islami, F.; Kamangar, F.; Nasrollahzadeh, D.; Moller, H.; Boffetta, P.; Malekzadeh, R. Oesophageal cancer in golestan province, a high-incidence area in northern iran—A review. Eur. J. Cancer 2009, 45, 3156–3165. [Google Scholar] [CrossRef] [PubMed]
- Oze, I.; Matsuo, K.; Wakai, K.; Nagata, C.; Mizoue, T.; Tanaka, K.; Tsuji, I.; Sasazuki, S.; Inoue, M.; Tsugane, S. Alcohol drinking and esophageal cancer risk: An evaluation based on a systematic review of epidemiologic evidence among the japanese population. Jpn. J. Clin. Oncol. 2011, 41, 677–692. [Google Scholar] [CrossRef] [PubMed]
- Mlombe, Y.B.; Rosenberg, N.E.; Wolf, L.L.; Dzamalala, C.P.; Chalulu, K.; Chisi, J.; Shaheen, N.J.; Hosseinipour, M.C.; Shores, C.G. Environmental risk factors for oesophageal cancer in malawi: A case-control study. Malawi Med. J. 2015, 27, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Chen, X.; Tu, S. Etiology and prevention of esophageal cancer. Gastrointest. Tumors 2016, 3, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Ibiebele, T.I.; Hughes, M.C.; Nagle, C.M.; Bain, C.J.; Whiteman, D.C.; Webb, P.M. Dietary antioxidants and risk of barrett’s esophagus and adenocarcinoma of the esophagus in an australian population. Int. J. Cancer 2013, 133, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Hashemian, M.; Poustchi, H.; Abnet, C.C.; Boffetta, P.; Dawsey, S.M.; Brennan, P.J.; Pharoah, P.; Etemadi, A.; Kamangar, F.; Sharafkhah, M.; et al. Dietary intake of minerals and risk of esophageal squamous cell carcinoma: Results from the golestan cohort study. Am. J. Clin. Nutr. 2015, 102, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Peacock, M. Calcium metabolism in health and disease. Clin. J. Am. Soc. Nephrol. 2010, 5, 05910809. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Lamprecht, S.A.; Shinozaki, H.; Fan, K.; Yang, W.; Newmark, H.L.; Kopelovich, L.; Edelmann, W.; Jin, B.; Gravaghi, C.; et al. Dietary calcium and cholecalciferol modulate cyclin d1 expression, apoptosis, and tumorigenesis in intestine of adenomatous polyposis coli1638n/+ mice. J. Nutr. 2008, 138, 1658–1663. [Google Scholar] [PubMed]
- Keum, N.; Lee, D.H.; Greenwood, D.C.; Zhang, X.; Giovannucci, E.L. Calcium intake and colorectal adenoma risk: Dose-response meta-analysis of prospective observational studies. Int. J. Cancer 2015, 136, 1680–1687. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Hu, P.; Xie, D.; Qin, Y.; Wang, F.; Wang, H. Meta-analysis of vitamin D, calcium and the prevention of breast cancer. Breast Cancer Res. Treat. 2010, 121, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; LaValley, M.P.; Tucker, K.L. Prospective studies of dairy product and calcium intakes and prostate cancer risk: A meta-analysis. J. Natl. Cancer Inst. 2005, 97, 1768–1777. [Google Scholar] [CrossRef] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis of observational studies in epidemiology (moose) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Stang, A. Critical evaluation of the newcastle-ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010, 25, 603–605. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G. Controlling the risk of spurious findings from meta-regression. Stat. Med. 2004, 23, 1663–1682. [Google Scholar] [CrossRef] [PubMed]
- Patsopoulos, N.A.; Evangelou, E.; Ioannidis, J.P. Sensitivity of between-study heterogeneity in meta-analysis: Proposed metrics and empirical evaluation. Int. J. Epidemiol. 2008, 37, 1148–1157. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Mulholland, H.G.; Murray, L.J.; Anderson, L.A.; Cantwell, M.M. Vitamin d, calcium and dairy intake, and risk of oesophageal adenocarcinoma and its precursor conditions. Br. J. Nutr. 2011, 106, 732–741. [Google Scholar] [CrossRef] [PubMed]
- Wolfgarten, E.; Rosendahl, U.; Nowroth, T.; Leers, J.; Metzger, R.; Holscher, A.H.; Bollschweiler, E. Coincidence of nutritional habits and esophageal cancer in germany. Onkologie 2001, 24, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Graham, S.; Marshall, J.; Haughey, B.; Brasure, J.; Freudenheim, J.; Zielezny, M.; Wilkinson, G.; Nolan, J. Nutritional epidemiology of cancer of the esophagus. Am. J. Epidemiol. 1990, 131, 454–467. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, S.; Bidoli, E.; Negri, E.; Zambon, P.; Talamini, R.; Ruol, A.; Parpinel, M.; Levi, F.; Simonato, L.; La Vecchia, C. Role of macronutrients, vitamins and minerals in the aetiology of squamous-cell carcinoma of the oesophagus. Int. J. Cancer 2000, 86, 626–631. [Google Scholar] [CrossRef]
- Jessri, M.; Rashidkhani, B.; Hajizadeh, B.; Jessri, M.; Gotay, C. Macronutrients, vitamins and minerals intake and risk of esophageal squamous cell carcinoma: A case-control study in iran. Nutr. J. 2011, 10, 1475–2891. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Tucker, K.L.; Graubard, B.I.; Heineman, E.F.; Markin, R.S.; Potischman, N.A.; Russell, R.M.; Weisenburger, D.D.; Ward, M.H. Nutrient intakes and adenocarcinoma of the esophagus and distal stomach. Nutr. Cancer 2002, 42, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Leitzmann, M.F.; Subar, A.F.; Hollenbeck, A.; Schatzkin, A. Dairy food, calcium, and risk of cancer in the nih-aarp diet and health study. Arch. Intern. Med. 2009, 169, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Cai, L.; Mu, L.N.; Lu, Q.Y.; Zhao, J.; Cui, Y.; Sul, J.H.; Zhou, X.F.; Ding, B.G.; Elashoff, R.M.; et al. Dietary mineral and trace element intake and squamous cell carcinoma of the esophagus in a chinese population. Nutr. Cancer 2006, 55, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Tuyns, A.J.; Riboli, E.; Doornbos, G.; Pequignot, G. Diet and esophageal cancer in calvados (france). Nutr. Cancer 1987, 9, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.F.; Kurtz, R.C.; Yu, G.P.; Sun, M.; Gargon, N.; Karpeh, M., Jr.; Fein, J.S.; Harlap, S. Adenocarcinomas of the esophagus and gastric cardia: The role of diet. Nutr. Cancer 1997, 27, 298–309. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Lee, A.H.; Xu, F.; Zhang, T.; Lei, J.; Binns, C.W. Fruit and vegetable consumption and risk of esophageal cancer: A case-control study in north-west china. Dis. Esophagus 2014, 27, 777–782. [Google Scholar] [CrossRef] [PubMed]
- Tzonou, A.; Lipworth, L.; Garidou, A.; Signorello, L.B.; Lagiou, P.; Hsieh, C.; Trichopoulos, D. Diet and risk of esophageal cancer by histologic type in a low-risk population. Int. J. Cancer 1996, 68, 300–304. [Google Scholar] [CrossRef]
- Rogers, M.A.; Thomas, D.B.; Davis, S.; Vaughan, T.L.; Nevissi, A.E. A case-control study of element levels and cancer of the upper aerodigestive tract. Cancer Epidemiol. Biomark. Prev. 1993, 2, 305–312. [Google Scholar]
- Hu, J.; Nyren, O.; Wolk, A.; Bergstrom, R.; Yuen, J.; Adami, H.O.; Guo, L.; Li, H.; Huang, G.; Xu, X.; et al. Risk factors for oesophageal cancer in northeast china. Int. J. Cancer 1994, 57, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J.; Lipp, P.; Bootman, M.D. The versatility and universality of calcium signalling. Nat. Rev. Mol. Cell Biol. 2000, 1, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Lipskaia, L.; Lompre, A.M. Alteration in temporal kinetics of Ca2+ signaling and control of growth and proliferation. Biol. Cell 2004, 96, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Cullen, P.J.; Lockyer, P.J. Integration of calcium and ras signalling. Nat. Rev. Mol. Cell Biol. 2002, 3, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Rock, C.L. Milk and the risk and progression of cancer. Nestle Nutr. Workshop Ser. Pediatr. Program. 2011, 67, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Yuan, Y.; Jiang, Y.; Shrestha, C.; Chen, Y.; Liao, L.; Ji, S.; Deng, X.; Liao, E.; Bikle, D.D. P120-catenin is required for dietary calcium suppression of oral carcinogenesis in mice. J. Cell Physiol. 2017, 232, 1360–1367. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Liao, L.; Shrestha, C.; Li, D.; Li, M.; Mu, Y.; Crumrine, D.; Wang, L.; Xie, Z. Inhibition of 4-nitroquinoline-1-oxide-induced oral carcinogenesis by dietary calcium. Int. J. Clin. Exp. Pathol. 2015, 8, 3529–3542. [Google Scholar] [PubMed]
- Smolinski, K.N.; Abraham, J.M.; Souza, R.F.; Yin, J.; Wang, S.; Xu, Y.; Zou, T.T.; Kong, D.; Fleisher, A.S.; Meltzer, S.J. Activation of the esophagin promoter during esophageal epithelial cell differentiation. Genomics 2002, 79, 875–880. [Google Scholar] [CrossRef] [PubMed]
- Abraham, J.M.; Wang, S.; Suzuki, H.; Jiang, H.Y.; Rosenblum-Vos, L.S.; Yin, J.; Meltzer, S.J. Esophagin cdna cloning and characterization: A tissue-specific member of the small proline-rich protein family that is not expressed in esophageal tumors. Cell Growth Differ. 1996, 7, 855–860. [Google Scholar] [PubMed]
- Cui, L.; Liu, X.; Tian, Y.; Xie, C.; Li, Q.; Cui, H.; Sun, C. Flavonoids, flavonoid subclasses, and esophageal cancer risk: A meta-analysis of epidemiologic studies. Nutrients 2016, 8, 350. [Google Scholar] [CrossRef] [PubMed]
- Bo, Y.; Lu, Y.; Zhao, Y.; Zhao, E.; Yuan, L.; Lu, W.; Cui, L.; Lu, Q. Association between dietary vitamin c intake and risk of esophageal cancer: A dose-response meta-analysis. Int. J. Cancer 2016, 138, 1843–1850. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.; Smith-Warner, S.A.; Spiegelman, D.; Beeson, W.L.; van den Brandt, P.A.; Colditz, G.A.; Folsom, A.R.; Fraser, G.E.; Freudenheim, J.L.; Giovannucci, E.; et al. Dairy foods, calcium, and colorectal cancer: A pooled analysis of 10 cohort studies. J. Natl. Cancer Inst. 2004, 96, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Hidayat, K.; Chen, G.C.; Zhang, R.; Du, X.; Zou, S.Y.; Shi, B.M.; Qin, L.Q. Calcium intake and breast cancer risk: Meta-analysis of prospective cohort studies. Br. J. Nutr. 2016, 116, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Navarro Rosenblatt, D.A.; Chan, D.S.; Vieira, A.R.; Vieira, R.; Greenwood, D.C.; Vatten, L.J.; Norat, T. Dairy products, calcium, and prostate cancer risk: A systematic review and meta-analysis of cohort studies. Am. J. Clin. Nutr. 2015, 101, 87–117. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wang, X.; Yao, Q.; Qin, L.; Xu, C. Dairy product, calcium intake and lung cancer risk: A systematic review with meta-analysis. Sci. Rep. 2016, 6, 20624. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Orsini, N.; Wolk, A. Dietary calcium intake and risk of stroke: A dose-response meta-analysis. Am. J. Clin. Nutr. 2013, 97, 951–957. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Shrubsole, M.J.; Ness, R.M.; Schlundt, D.; Cai, Q.; Smalley, W.E.; Li, M.; Shyr, Y.; Zheng, W. The relation of magnesium and calcium intakes and a genetic polymorphism in the magnesium transporter to colorectal neoplasia risk. Am. J. Clin. Nutr. 2007, 86, 743–751. [Google Scholar] [PubMed]
- Seelig, M.S. The requirement of magnesium by the normal adult. Summary and analysis of published data. Am. J. Clin. Nutr. 1964, 14, 242–290. [Google Scholar] [PubMed]
- Dai, Q.; Cantwell, M.M.; Murray, L.J.; Zheng, W.; Anderson, L.A.; Coleman, H.G. Dietary magnesium, calcium:Magnesium ratio and risk of reflux oesophagitis, barrett’s oesophagus and oesophageal adenocarcinoma: A population-based case-control study. Br. J. Nutr. 2016, 115, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Shu, X.O.; Hebert, J.R.; Jin, F.; Yang, G.; Liu, D.K.; Gao, Y.T.; Zheng, W. Variation in nutrient intakes among women in Shanghai, China. Eur. J. Clin. Nutr. 2004, 58, 1604–1611. [Google Scholar] [CrossRef] [PubMed]
| Author, Year | Country | Study-Design | Pathological Type | Source of Control | Dietary Assessment | Participants (Cases) | Comparison | OR or RR (95% CI) | NOS Score | Adjustment for Covariates |
|---|---|---|---|---|---|---|---|---|---|---|
| Hashemian, 2015 [8] | Iran | Cohort | ESCC | PB | FFQ-116 items, validated | 47,204 (201) | ≥1048.0 vs. <409 (mg/day) | 0.49 (0.29–0.82) | 8 | Age, sex, total energy, place of residence, smoking, wealth score, ethnicity, opiate use, BMI, education, marital status, physical activity score, and fruit and vegetable intakes |
| Mulholland, 2011 [20] | Ireland | Case–control | EAC | PB | FFQ-101 items, validated | 252 (218) | ≥1262.0 vs. <929.3 (mg/day) | 0.88 (0.38–2.03) | 6 | Age, sex, energy intake, smoking status, BMI, education, occupation, alcohol, regular non-steroidal anti-inflammatory drug use, Helicobacter pylori infection, energy-adjusted glycemic index intake, energy-adjusted saturated fat intake, and location |
| Wolfgarten, 2001 [21] | Germany | Case–control | EAC | PB | DHQ, NA | 100 (40) | >1590 vs. <986 (mg/day) | 0.4 (0.1–1.3) | 7 | Age, residence, and nationality |
| Wolfgarten, 2001 [21] | Germany | Case–control | ESCC | PB | DHQ, NA | 100 (45) | >1590 vs. <986 (mg/day) | 0.4 (0.1–1.1) | 7 | Age, residence, and nationality |
| Graham, 1990 [22] | United states | Case–control | Mix type | PB | FFQ, NA | 174 (178) | >1028.5 vs. <543.5 (mg/day) | 2.15 (1.14–4.06) | 4 | Sex, age, education, smoking, and alcohol ingestion |
| Franceschi, 2000 [23] | Italy | Case–control | ESCC | HB | FFQ-78 items, validated | 743 (304) | Q5 vs. Q1 | 1.0 (0.6–1.7) | 8 | Age, gender, area of residence, education, physical activity, BMI, tobacco smoking, alcohol drinking, and non-alcohol energy |
| Jessri, 2011 [24] | Iran | Case–control | ESCC | HB | FFQ-125 items, validated | 96 (47) | T3 vs. T1 | 0.49 (0.15–0.87) | 7 | Age, sex, gastroesophageal reflux disease symptoms, BMI, smoking status, smoking intensity and duration (pack-years), physical activity, and education level |
| Chen, 2002 [25] | United states | Case–control | EAC | PB | DHQ, validated | 449 (124) | Q4 vs. Q1 | 0.5 (0.2–0.9) | 7 | Age, age squared, gender, respondent type, BMI, alcohol use, tobacco use, education level, family history of respective cancers, and vitamin supplement use |
| Park, 2009 [26] | United states | Cohort | Mix type | PB | FFQ-124 items, validated | 29,3439 (468) | >1247 vs. <478 (mg/day) | 0.66 (0.49–0.90) | 9 | Smoking status, time since quitting smoking, smoking dose, antacid use, personal history of diabetes, and hypertension |
| Lu, 2006 [27] | China | Case–control | ESCC | PB | FFQ-97 items, NA | 415 (218) | ≥344 vs. <157 (mg/day) | 0.82 (0.38–1.75) | 8 | Age, gender, educational level, income, BMI, total energy intake, smoking, and drinking. |
| Rogers, 1993 [32] | United states | Case–control | Mix type | PB | FFQ, NA | 593 (127) | >1419 vs. <571 (mg/day) | 0.6 (0.3–1.5) | 6 | Age, sex, pack-years of cigarette use, drink-years of alcohol, energy intake, β-carotene intake, and ascorbic acid intake |
| Tuyns, 1987 [28] | France | Cohort | mix type | PB | DHQ, validate | 2788 (743) | >1000 vs. <600 (mg/day) | 0.84 (0.56–1.25) | 3 | Age, alcohol consumption, and tobacco smoking |
| Tzonou A 1996 [31] | Greece | Case–control | ESCC | HB | FFQ-115 items, validated | 243 (43) | Q5 vs. Q1 | 0.92 (0.64–1.32) | 7 | Gender, age, birthplace, schooling, height, analgesics, coffee drinking, alcohol intake, tobacco smoking, and energy intake. |
| Tzonou A 1996 [31] | Greece | Case–control | EAC | HB | FFQ-115 items, validated | 256 (56) | Q5 vs. Q1 | 1 (0.72–1.4) | 7 | Gender, age, birthplace, schooling, height, analgesics, coffee drinking, alcohol intake, tobacco smoking, and energy intake, though not mutually |
| Zhang, 1997 [29] | United States | Case–control | EAC | HB | HHHQ, validated | 189 (29) | Q4 vs. Q1 | 1.3 (0.5–3.3) | 5 | Age, sex, race, education, total dietary intake of calories. Smoking, alcohol use, and BMI |
| Tang, 2014 [30] | China | Case–control | mix type | HB | FFQ-137 items, validated | 739 (359) | >470 vs. <260 (mg/day) | 0.75 (0.52–1.1) | 8 | Age, gender, education level, BMI, total energy intake, smoking status, alcohol drinking, and family history of cancer in first-degree relatives. |
| Hu,1994 [33] | China | Case–control | mix type | HB | FFQ-32 items, NA | 588 (196) | Q4 vs. Q1 | 0.8 (0.4–1.4) | 7 | Alcohol intake, smoking. and family income |
| Subgroups | No. of Studies | No. of Cases | Pooled ORs (95% CI) | p | Heterogeneity Test | ||
|---|---|---|---|---|---|---|---|
| Chi-Square | I2 | Phet | |||||
| All studies | 17 | 3396 | 0.80 (0.71, 0.91) | 0.001 | 24.11 | 33.6% | 0.087 |
| Location | |||||||
| Europe | 7 | 1449 | 0.90 (0.75, 1.08) | 0.262 | 3.97 | 0.0% | 0.681 |
| America | 5 | 926 | 0.88 (0.51, 1.49) | 0.625 | 13.83 | 71.1% | 0.008 |
| Asia | 5 | 1021 | 0.67 (0.52, 0.86) | 0.002 | 2.80 | 0.0% | 0.591 |
| Study design | |||||||
| Cohort | 3 | 1412 | 0.67 (0.54, 0.84) | 0.000 | 2.62 | 23.6% | 0.270 |
| PBCC | 7 | 950 | 0.84 (0.61, 1.14) | 0.261 | 13.68 | 56.2% | 0.033 |
| HBCC | 7 | 1034 | 0.89 (0.74, 1.06) | 0.182 | 4.00 | 0.0% | 0.677 |
| Pathological type | |||||||
| ESCC | 6 | 858 | 0.76 (0.60, 0.96) | 0.019 | 6.97 | 28.3% | 0.223 |
| EAC | 5 | 467 | 0.89 (0.68, 1.16) | 0.381 | 4.84 | 17.4% | 0.304 |
| Mixed type | 6 | 2071 | 0.84 (0.63, 1.13) | 0.252 | 11.50 | 56.5% | 0.042 |
| Dietary assessment | |||||||
| Validated method | 11 | 2592 | 0.79 (0.69, 0.90) | 0.001 | 11.84 | 15.5% | 0.296 |
| Not Validated method | 6 | 804 | 0.81 (0.48, 1.35) | 0.413 | 11.68 | 57.2% | 0.039 |
| NOS score | |||||||
| Low quality | 1 | 743 | 0.84 (0.56, 1.25) | 0.395 | 0.00 | N/A | N/A |
| Moderate quality | 4 | 552 | 1.14 (0.63, 2.05) | 0.671 | 6.64 | 54.8% | 0.084 |
| High quality | 12 | 2101 | 0.76 (0.66, 0.87) | 0.000 | 12.60 | 12.7% | 0.320 |
| Adjustment for energy intake | |||||||
| Yes | 8 | 1251 | 0.83 (0.70, 0.98) | 0.031 | 7.26 | 3.6% | 0.402 |
| No | 9 | 2145 | 0.78 (0.58, 1.04) | 0.093 | 16.61 | 51.8% | 0.034 |
| Adjustment forBMI | |||||||
| Yes | 8 | 1500 | 0.72 (0.58, 0.90) | 0.003 | 7.16 | 2.2% | 0.413 |
| No | 9 | 1896 | 0.85 (0.73, 0.99) | 0.037 | 15.57 | 48.6% | 0.049 |
| Publication year | |||||||
| Before/in 2000 | 8 | 1676 | 0.97 (0.82, 1.16) | 0.767 | 8.75 | 20.0% | 0.271 |
| After 2000 | 9 | 1720 | 0.64 (0.53, 0.77) | 0.000 | 4.58 | 0.0% | 0.802 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Q.; Cui, L.; Tian, Y.; Cui, H.; Li, L.; Dou, W.; Li, H.; Wang, L. Protective Effect of Dietary Calcium Intake on Esophageal Cancer Risk: A Meta-Analysis of Observational Studies. Nutrients 2017, 9, 510. https://doi.org/10.3390/nu9050510
Li Q, Cui L, Tian Y, Cui H, Li L, Dou W, Li H, Wang L. Protective Effect of Dietary Calcium Intake on Esophageal Cancer Risk: A Meta-Analysis of Observational Studies. Nutrients. 2017; 9(5):510. https://doi.org/10.3390/nu9050510
Chicago/Turabian StyleLi, Qianwen, Lingling Cui, Yalan Tian, Han Cui, Li Li, Weifeng Dou, Haixia Li, and Ling Wang. 2017. "Protective Effect of Dietary Calcium Intake on Esophageal Cancer Risk: A Meta-Analysis of Observational Studies" Nutrients 9, no. 5: 510. https://doi.org/10.3390/nu9050510
APA StyleLi, Q., Cui, L., Tian, Y., Cui, H., Li, L., Dou, W., Li, H., & Wang, L. (2017). Protective Effect of Dietary Calcium Intake on Esophageal Cancer Risk: A Meta-Analysis of Observational Studies. Nutrients, 9(5), 510. https://doi.org/10.3390/nu9050510





