NAFLD and Atherosclerosis Are Prevented by a Natural Dietary Supplement Containing Curcumin, Silymarin, Guggul, Chlorogenic Acid and Inulin in Mice Fed a High-Fat Diet
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Histological Analysis
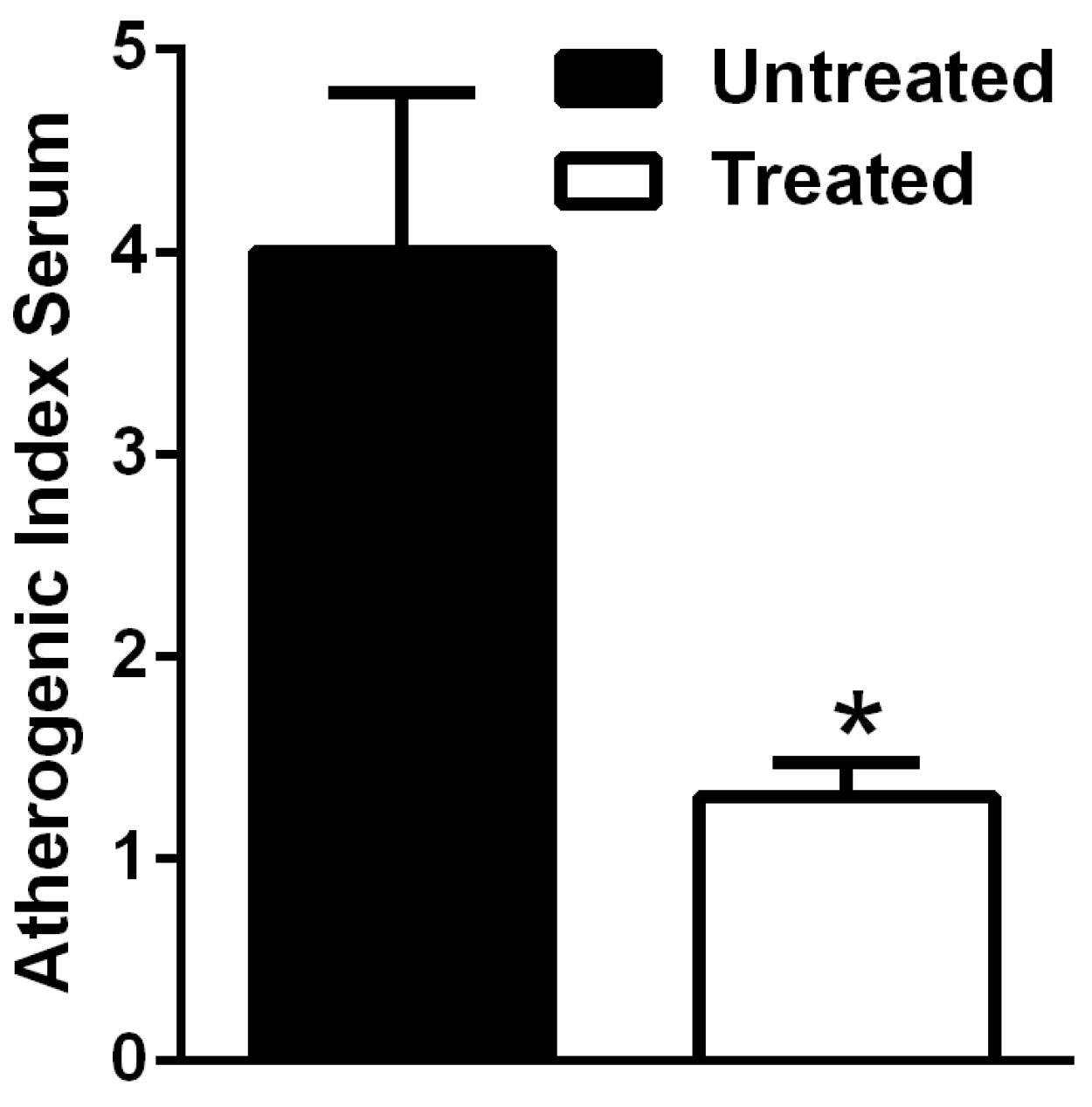
2.3. Atherogenic Index (AIS)
2.4. Biochemical Analysis
2.5. Quantitative Real-Time qPCR
2.6. RT2 Profiler PCR Array
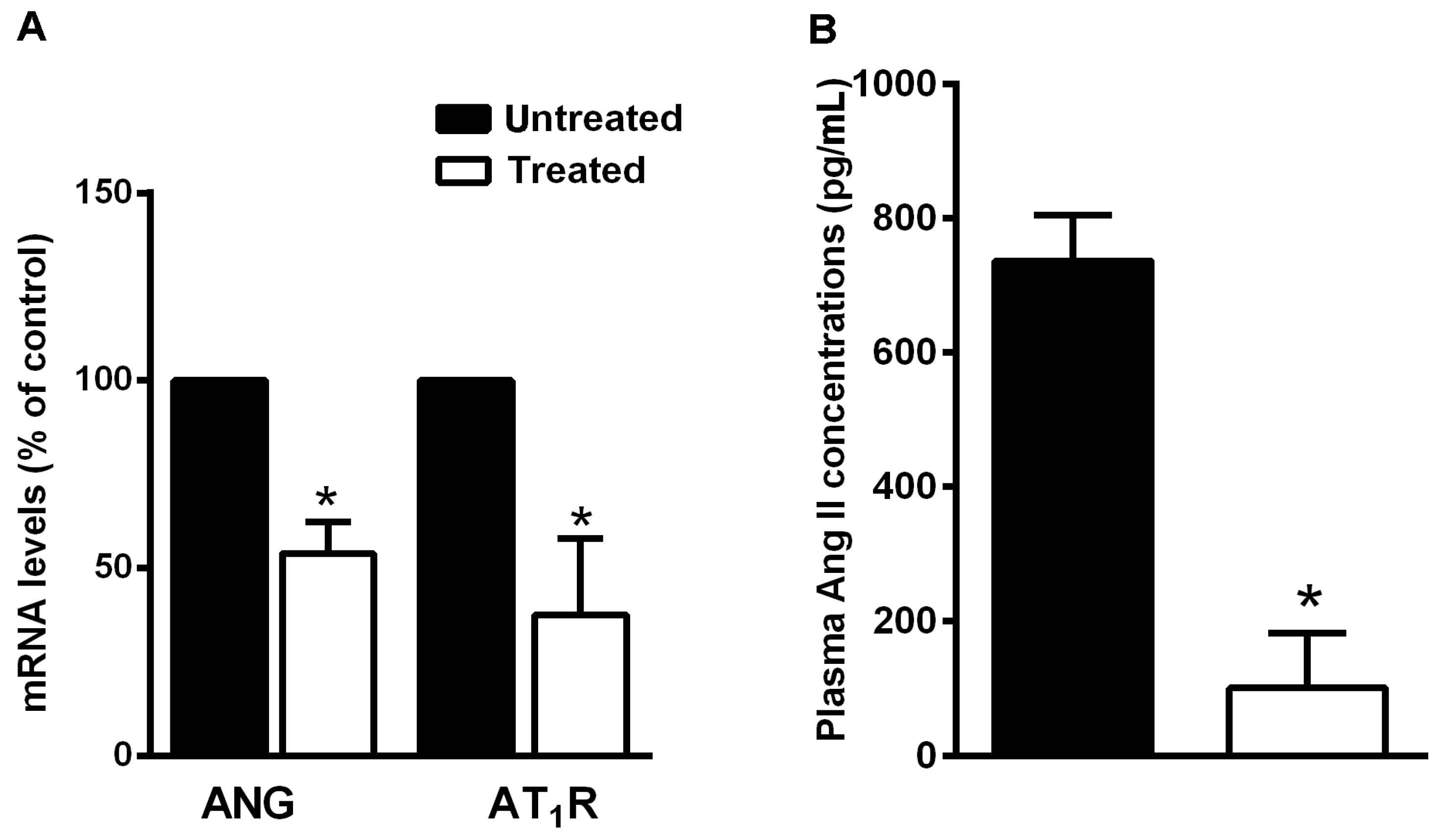
2.7. Measurement of Circulating Levels of Angiotensin II
2.8. Statistical Analyses
3. Results
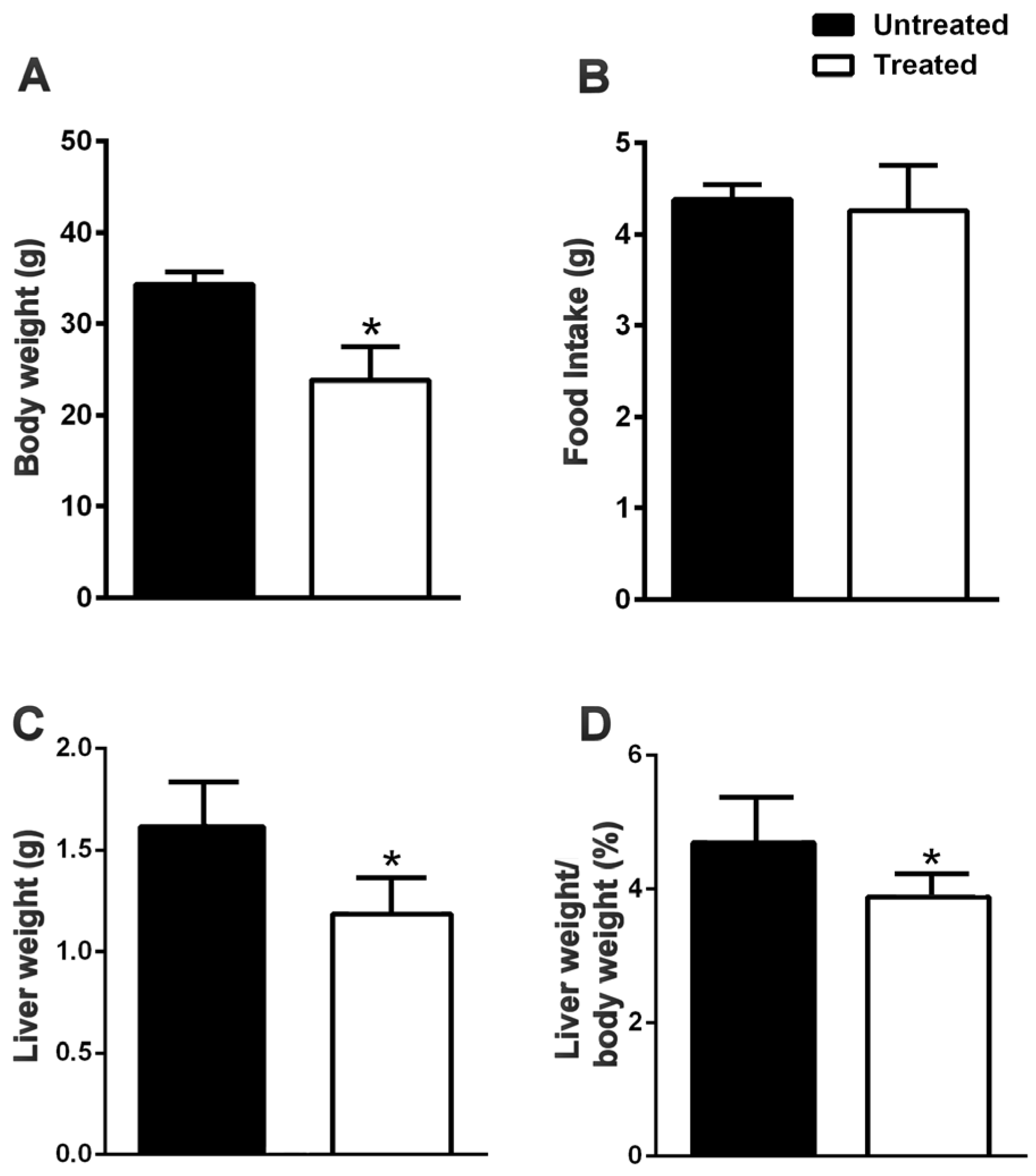
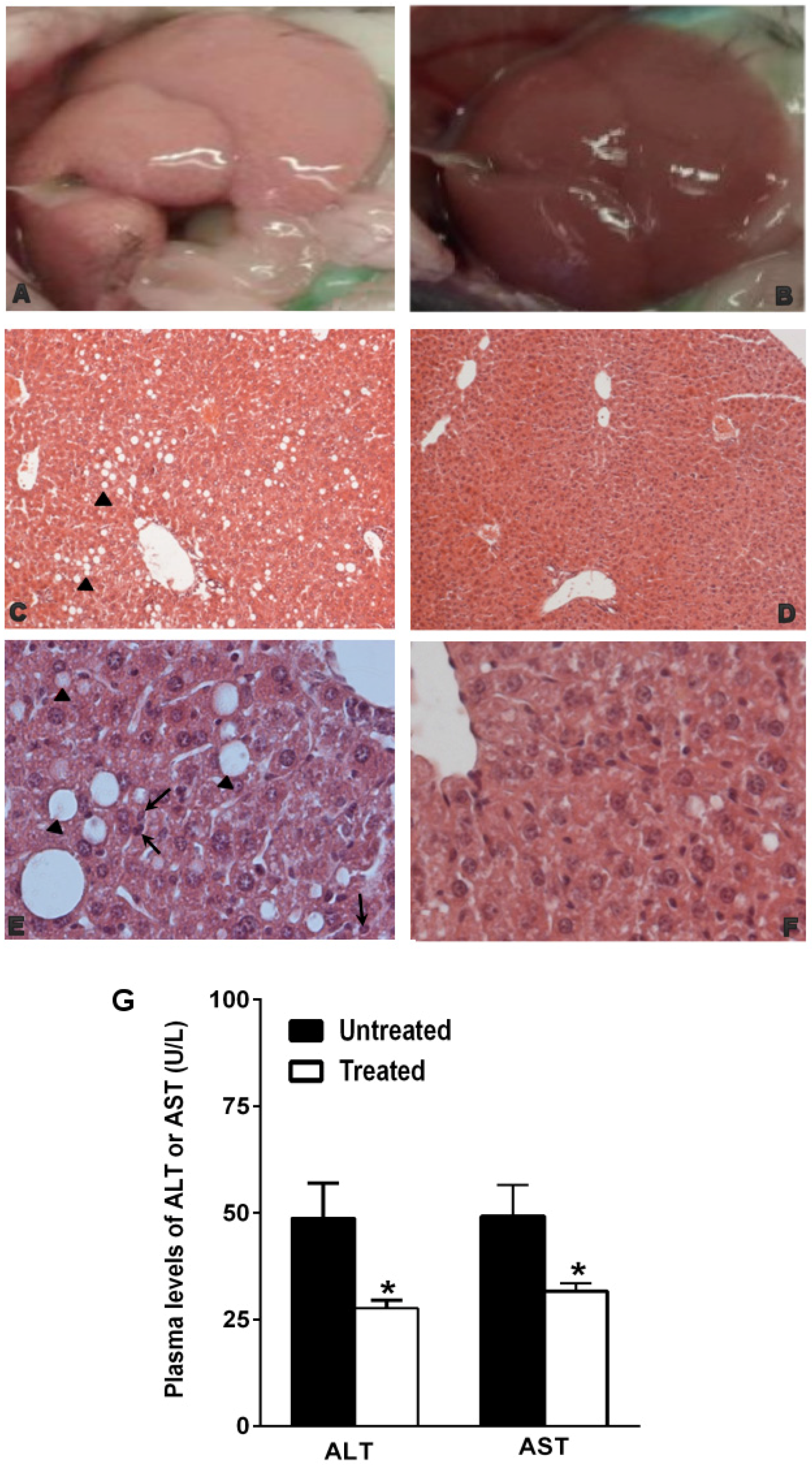
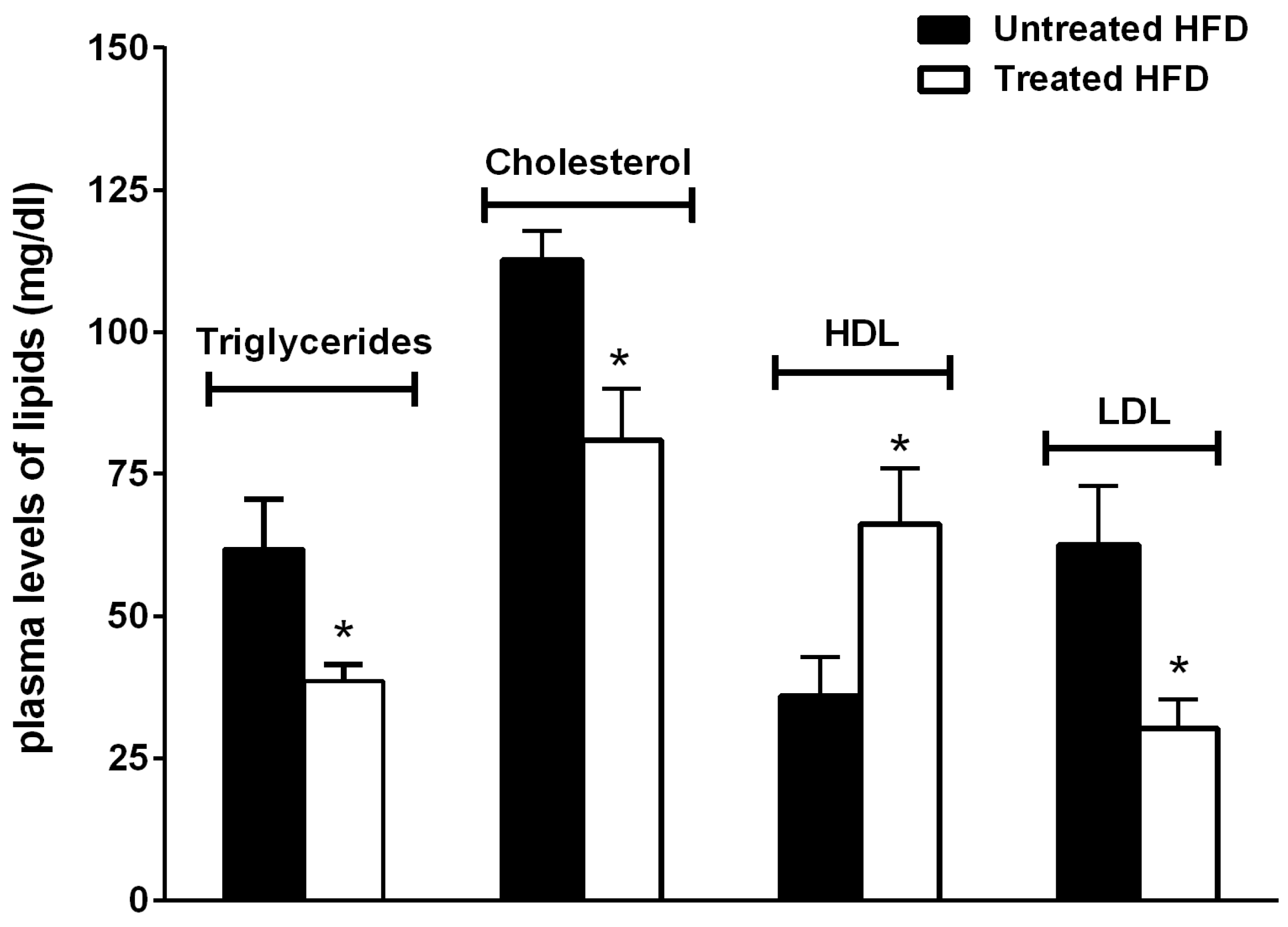
3.1. The Natural Dietary Supplement Prevents the Development of NAFLD
3.2. The Natural Dietary Supplement Modulates the Expression of Genes Involved in NAFLD
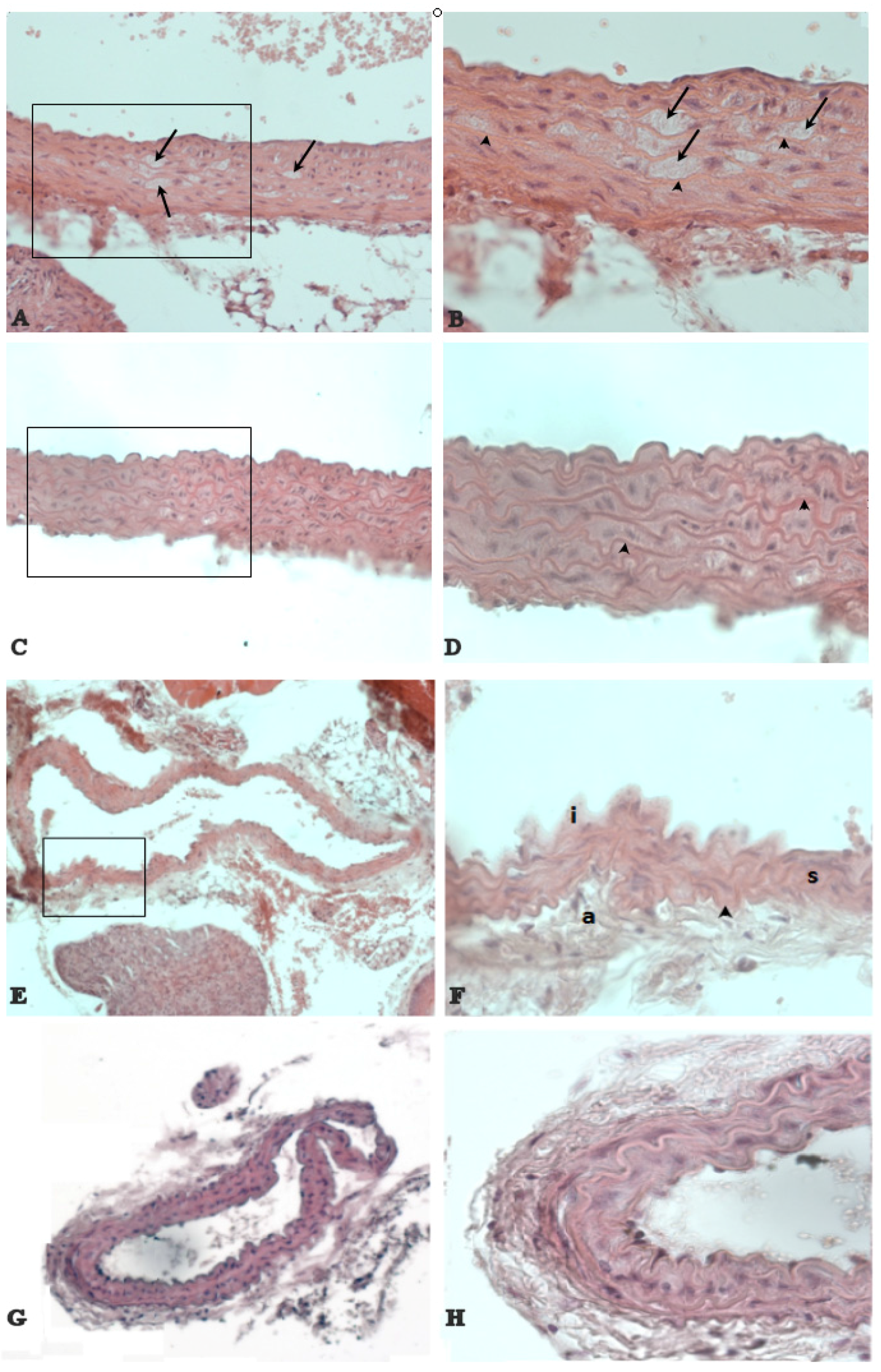
3.3. The Natural Dietary Supplement Prevents Atherosclerosis Development
3.4. The Natural Dietary Supplement Reduces RAS Component Expression
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lim, S.; Oh, T.J.; Koh, K.K. Mechanistic link between nonalcoholic fatty liver disease and cardiometabolic disorders. Int. J. Cardiol. 2015, 201, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.A.; Day, C.P. Benefits of lifestyle modification in NAFLD. Gut 2007, 56, 1760–1769. [Google Scholar] [CrossRef] [PubMed]
- Paschos, P.; Tziomalos, K. Nonalcoholic fatty liver disease and the renin-angiotensin system: Implications for treatment. World J. Hepatol. 2012, 4, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.Z.; Zhang, X.; Wang, L.; Zhang, F.; Qiu, Q.; Liu, M.L.; Zhang, G.R.; Wu, X.L. An increased circulating angiotensin II concentration is associated with hypoadiponectinemia and postprandial hyperglycemia in men with nonalcoholic fatty liver disease. Intern. Med. 2013, 52, 855–861. [Google Scholar] [CrossRef] [PubMed]
- Moreira de Macêdo, S.; Guimarães, T.A.; Feltenberger, J.D.; Sousa Santos, S.H. The role of renin-angiotensin system modulation on treatment and prevention of liver diseases. Peptides 2014, 62, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Kurita, S.; Takamura, T.; Ota, T.; Matsuzawa-Nagata, N.; Kita, Y.; Uno, M.; Nabemoto, S.; Ishikura, K.; Misu, H.; Ando, H.; et al. Olmesartan ameliorates a dietary rat model of non-alcoholic steatohepatitis through its pleiotropic effects. Eur. J. Pharmacol. 2008, 588, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Rector, R.S.; Thyfault, J.P.; Ibdah, J.A. Nonalcoholic fatty liver disease and mitochondrial dysfunction. World J. Gastroenterol. 2008, 14, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Husain, K.; Hernandez, W.; Ansari, R.A.; Ferder, L. Inflammation, oxidative stress and renin angiotensin system in atherosclerosis. World J. Biol. Chem. 2015, 6, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Fargion, S.; Porzio, M.; Fracanzani, A.L. Nonalcoholic fatty liver disease and vascular disease: State-of-the-art. World J. Gastroenterol. 2014, 20, 13306–13324. [Google Scholar] [CrossRef] [PubMed]
- Stepanova, M.; Younossi, Z.M. Independent association between nonalcoholic fatty liver disease and cardiovascular disease in the US population. Clin. Gastroenterol. Hepatol. 2012, 10, 646–650. [Google Scholar] [CrossRef] [PubMed]
- Targher, G.; Day, C.P.; Bonora, E. Risk of cardiovascular disease in patients with Nonalcoholic Fatty Liver Disease. N. Engl. J. Med. 2010, 363, 1341–1350. [Google Scholar] [CrossRef] [PubMed]
- Frantz, E.D.; Penna-de-Carvalho, A.; Batista Tde, M.; Aguila, M.B.; Mandarim-de-Lacerda, C.A. Comparative effects of the renin-angiotensin system blockers on nonalcoholic fatty liver disease and insulin resistance in C57BL/6 mice. Metab. Syndr. Relat. Disord. 2014, 12, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Fraga-Silva, R.A.; Savergnini, S.Q.; Montecucco, F.; Nencioni, A.; Caffa, I.; Soncini, D.; Costa-Fraga, F.P.; De Sousa, F.B.; Sinisterra, R.D.; Capettini, L.A.; et al. Treatment with Angiotensin-(1-7) reduces inflammation in carotid atherosclerotic plaques. Thromb. Haemost. 2014, 111, 736–747. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, N.; Beaton, M.D. Management of non-alcoholic fatty liver disease in 2015. World J. Hepatol. 2015, 7, 2962–2967. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.B.; Kang, O.H.; Lee, Y.S.; Han, S.H.; Ahn, Y.S.; Cha, S.W.; Seo, Y.S.; Kong, R.; Kwon, D.Y. Hepatoprotective effect and synergism of bisdemethoycurcumin against MCD Diet-Induced Nonalcoholic Fatty Liver Disease in mice. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Patti, A.M.; Al-Rasadi, K.; Katsiki, N.; Banerjee, Y.; Nikolic, D.; Vanella, L.; Giglio, R.V.; Giannone, V.A.; Montalto, G.; Rizzo, M. Effect of a Natural Supplement Containing Curcuma Longa, Guggul, and Chlorogenic Acid in Patients With Metabolic Syndrome. Angiology 2015, 66, 856–861. [Google Scholar] [CrossRef] [PubMed]
- Baldassano, S.; Amato, A.; Cappello, F.; Rappa, F.; Mulè, F. Glucagon-like peptide-2 and mouse intestinal adaptation to a high-fat diet. J. Endocrinol. 2013, 217, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Baldassano, S.; Amato, A.; Rappa, F.; Cappello, F.; Mulè, F. Influence of endogenous glucagon like peptide-2 on lipid disorders in mice fed a high fat diet. Endoc. Res. 2016, 23, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jawień, J.; Nastałek, P.; Korbut, R. Mouse models of experimental atherosclerosis. J. Physiol. Pharmacol. 2004, 55, 503–517. [Google Scholar] [PubMed]
- Nuzzo, D.; Picone, P.; Baldassano, S.; Caruana, L.; Messina, E.; Marino Gammazza, A.; Cappello, F.; Mulè, F.; Di Carlo, M. Insulin Resistance as Common Molecular Denominator Linking Obesity to Alzheimer’s Disease. Curr. Alzheimer Res. 2015, 12, 723–735. [Google Scholar] [CrossRef] [PubMed]
- Amato, A.; Baldassano, S.; Caldara, G.; Mulè, F. Neuronostatin: Peripheral site of action in mouse stomach. Peptides 2015, 64, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Menke, A.L.; Driessen, A.; Koek, G.H.; Lindeman, J.H.; Stoop, R.; Havekes, L.M.; Kleemann, R.; van den Hoek, A.M. Establishment of a general NAFLD scoring system for rodent models and comparison to human liver pathology. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Frediani Brant, N.M.; Mourão Gasparotto, F.; de Oliveira Araújo, V.; Christian Maraschin, J.; Lima Ribeiro Rde, C.; Botelho Lourenço, E.L.; Cardozo Junior, E.L.; Gasparotto Junior, A. Cardiovascular protective effects of Casearia sylvestris Swartz in Swiss and C57BL/6 LDLr-null mice undergoing high fat diet. J. Ethnopharmacol. 2014, 154, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Balzan, S.; Hernandes, A.; Reichert, C.L.; Donaduzzi, C.; Pires, V.A.; Gasparotto, A., Jr.; Cardozo, E.L., Jr. Lipid-lowering effects of standardized extracts of Ilex paraguariensis in high-fat-diet rats. Fitoterapia 2013, 86, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Gibbons, S. An overview of plant extracts as potential therapeutics. Expert. Opin. Ther. Pat. 2003, 13, 489–497. [Google Scholar] [CrossRef]
- Sahebkar, A. Why it is necessary to translate curcumin into clinical practice for the prevention and treatment of metabolic syndrome? Biofactors 2013, 39, 197–208. [Google Scholar] [CrossRef] [PubMed]
- Salamone, F.; Galvano, F.; Cappello, F.; Mangiameli, A.; Barbagallo, I.; Li Volti, G. Silibinin modulates lipid homeostasis and inhibits nuclear factor kappa B activation in experimental nonalcoholic steatohepatitis. Transl. Res 2012, 159, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Nohr, L.A.; Rasmussen, L.B.; Straand, J. Resin from the mukul myrrh tree, guggul, can it be used for treating hypercholesterolemia? A randomized, controlled study. Complement. Ther. Med. 2009, 17, 16–22. [Google Scholar] [CrossRef]
- Mubarak, A.; Hodgson, J.M.; Considine, M.J.; Croft, K.D.; Matthews, V.B. Correction to Supplementation of a high-fat diet with chlorogenic acid is associated with insulin resistance and hepatic lipid accumulation in mice. J. Agric. Food Chem. 2013, 61, 4371–4378. [Google Scholar] [CrossRef] [PubMed]
- Russo, F.; Riezzo, G.; Chiloiro, M.; De Michele, G.; Chimienti, G.; Marconi, E.; D’Attoma, B.; Linsalata, M.; Clemente, C. Metabolic effects of a diet with inulin-enriched pasta in healthy young volunteers. Curr. Pharm. Des. 2010, 16, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Wilson, L.; Kleiner, D.E.; Cummings, O.W.; Brunt, E.M. Relationship of steatosis grade and zonal location to histological features of steatohepatitis in adult patients with non-alcoholic fatty liver disease. J. Hepatol. 2008, 48, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Corey, K.E.; Chalasani, N. Management of dyslipidemia as a cardiovascular risk factor in individuals with nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 2014, 12, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Atshaves, B.P.; Martin, G.G.; Hostetler, H.A.; McIntosh, A.L.; Kier, A.B.; Schroeder, F. Liver fatty acid-binding protein and obesity. J. Nutr. Biochem. 2010, 21, 1015–1032. [Google Scholar] [CrossRef] [PubMed]
- Westerbacka, J.; Kolak, M.; Kiviluoto, T.; Arkkila, P.; Sirén, J.; Hamsten, A.; Fisher, R.M.; Yki-Järvinen, H. Genes involved in fatty acid partitioning and binding, lipolysis, monocyte/macrophage recruitment, and inflammation are overexpressed in the human fatty liver of insulin-resistant subjects. Diabetes 2007, 56, 2759–2765. [Google Scholar] [CrossRef] [PubMed]
- Kotronen, A.; Seppänen-Laakso, T.; Westerbacka, J.; Kiviluoto, T.; Arola, J.; Ruskeepää, A.L.; Oresic, M.; Yki-Järvinen, H. Hepatic stearoyl-CoA desaturase (SCD)-1 activity and diacylglycerol but not ceramide concentrations are increased in the nonalcoholic human fatty liver. Diabetes 2009, 58, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, M.; Dobrzyn, A.; Sampath, H.; Lee, S.H.; Man, W.C.; Chu, K.; Peters, J.M.; Gonzalez, F.J.; Ntambi, J.M. Reduced adiposity and liver steatosis by stearoyl-CoA desaturase deficiency are independent of peroxisome proliferator-activated receptor-alpha. J. Biol. Chem. 2004, 279, 35017–35024. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P.; Miyazaki, M.; Socci, N.D.; Hagge-Greenberg, A.; Liedtke, W.; Soukas, A.A.; Sharma, R.; Hudgins, L.C.; Ntambi, J.M.; Friedman, J.M. Role for stearoyl-CoA desaturase-1 in leptin-mediated weight loss. Science 2002, 297, 240–243. [Google Scholar] [CrossRef] [PubMed]
- Kandhi, R.; Bobbala, D.; Yeganeh, M.; Mayhue, M.; Menendez, A.; Ilangumaran, S. Negative regulation of the hepatic fibrogenic response by suppressor of cytokine signaling 1. Cytokine 2016, 82, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Klein, T.; Fujii, M.; Sandel, J.; Shibazaki, Y.; Wakamatsu, K.; Mark, M.; Yoneyama, H. Linagliptin alleviates hepatic steatosis and inflammation in a mouse model of non-alcoholic steatohepatitis. Med. Mol. Morphol. 2014, 47, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Whitman, S.C. A practical approach to using mice in atherosclerosis research. Clin. Biochem. Rev. 2004, 25, 81–93. [Google Scholar] [PubMed]
- Villanova, N.; Moscatiello, S.; Ramilli, S.; Bugianesi, E.; Magalotti, D.; Vanni, E.; Zoli, M.; Marchesini, G. Endothelial dysfunction and cardiovascular risk profile in nonalcoholic fatty liver disease. Hepatology 2005, 42, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Wieckowska, A.; Papouchado, B.G.; Li, Z.; Lopez, R.; Zein, N.N.; Feldstein, A.E. Increased hepatic and circulating interleukin-6 levels in human nonalcoholic steatohepatitis. Am. J. Gastroenterol. 2008, 103, 1372–1379. [Google Scholar] [CrossRef] [PubMed]

| Herbal Components | Part Used | Quantity/100 g |
| Cynara scolymus, e.s. tit. 2.5% chlorogenic acid | Leaf | 35 g (extract) 0.87 g (Chlor. acid) |
| Silybum marianum, e.s. tit. 80% silymarin | Seed | 8 g (extract) 6.4 g (silymarin) |
| Taraxacum officinale e.s. tit. 2% inulin | Root | 10 g (extract) 0.2 g (inulin) |
| Curcuma Longa e.s. tit. 95% curcumin | Rhizome | 10 g (powder) 9.5 g (curcumin) |
| Commiphora mukul Guggul e.s. tit. 10% guggulipids | Resin | 15 g (extract) 1.5 g (guggulipids) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amato, A.; Caldara, G.-F.; Nuzzo, D.; Baldassano, S.; Picone, P.; Rizzo, M.; Mulè, F.; Di Carlo, M. NAFLD and Atherosclerosis Are Prevented by a Natural Dietary Supplement Containing Curcumin, Silymarin, Guggul, Chlorogenic Acid and Inulin in Mice Fed a High-Fat Diet. Nutrients 2017, 9, 492. https://doi.org/10.3390/nu9050492
Amato A, Caldara G-F, Nuzzo D, Baldassano S, Picone P, Rizzo M, Mulè F, Di Carlo M. NAFLD and Atherosclerosis Are Prevented by a Natural Dietary Supplement Containing Curcumin, Silymarin, Guggul, Chlorogenic Acid and Inulin in Mice Fed a High-Fat Diet. Nutrients. 2017; 9(5):492. https://doi.org/10.3390/nu9050492
Chicago/Turabian StyleAmato, Antonella, Gaetano-Felice Caldara, Domenico Nuzzo, Sara Baldassano, Pasquale Picone, Manfredi Rizzo, Flavia Mulè, and Marta Di Carlo. 2017. "NAFLD and Atherosclerosis Are Prevented by a Natural Dietary Supplement Containing Curcumin, Silymarin, Guggul, Chlorogenic Acid and Inulin in Mice Fed a High-Fat Diet" Nutrients 9, no. 5: 492. https://doi.org/10.3390/nu9050492
APA StyleAmato, A., Caldara, G.-F., Nuzzo, D., Baldassano, S., Picone, P., Rizzo, M., Mulè, F., & Di Carlo, M. (2017). NAFLD and Atherosclerosis Are Prevented by a Natural Dietary Supplement Containing Curcumin, Silymarin, Guggul, Chlorogenic Acid and Inulin in Mice Fed a High-Fat Diet. Nutrients, 9(5), 492. https://doi.org/10.3390/nu9050492









