The Effect of Dietary Glycemic Properties on Markers of Inflammation, Insulin Resistance, and Body Composition in Postmenopausal American Women: An Ancillary Study from a Multicenter Protein Supplementation Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Dietary Monitoring and Assessment
2.3. Calculation of Dietary Glycemic Index and Load
2.4. Sample Analysis
2.5. Calculation of Insulin Resistance
2.6. Measurement of Body Composition
2.7. Ethics
2.8. Statistical Analysis
3. Results
3.1. Study Population
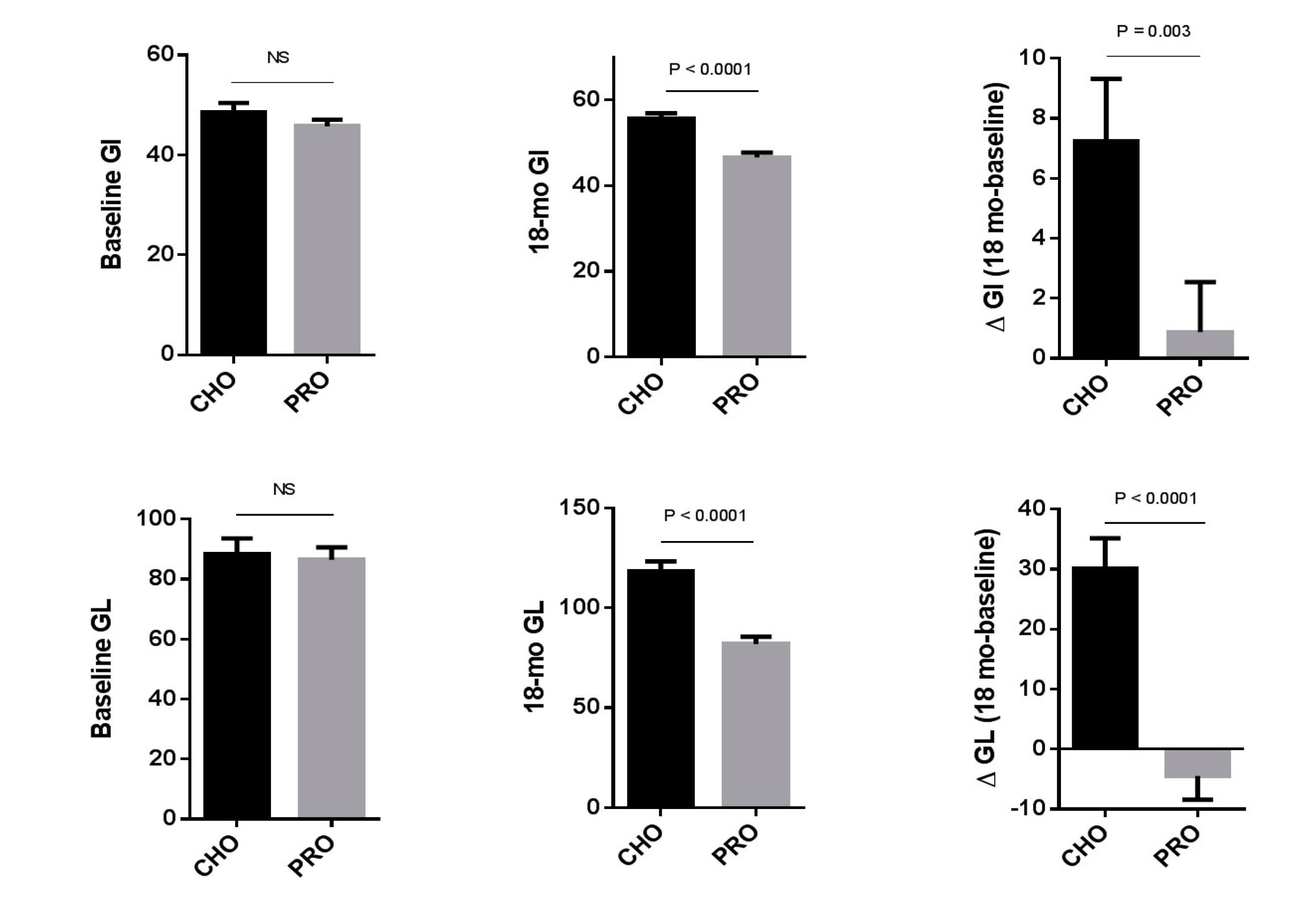
3.2. The Effect of the Study Intervention on Dietary Glycemic Load and Glycemic Index and Nutrient Intake
3.3. Changes in Markers of Inflammation and Estimates of Insulin Resistance
3.4. Changes in BMI and Body Composition
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Libby, P.; Tabas, I.; Fredman, G.; Fisher, E.A. Inflammation and its resolution as determinants of acute coronary syndromes. Circ. Res. 2014, 114, 1867–1879. [Google Scholar] [CrossRef] [PubMed]
- Evangelista, A.F.; Collares, C.V.; Xavier, D.J.; Macedo, C.; Manoel-Caetano, F.S.; Rassi, D.M.; Foss-Freitas, M.C.; Foss, M.C.; Sakamoto-Hojo, E.T.; Nguyen, C.; et al. Integrative analysis of the transcriptome profiles observed in type 1, type 2 and gestational diabetes mellitus reveals the role of inflammation. BMC Med. Genom. 2014. [Google Scholar] [CrossRef] [PubMed]
- Okada, F. Inflammation-related carcinogenesis: Current findings in epidemiological trends, causes and mechanisms. Yonago Acta Med. 2014, 57, 65–72. [Google Scholar] [PubMed]
- Franceschi, C.; Campisi, J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2014, 69, S4–S9. [Google Scholar] [CrossRef] [PubMed]
- Perry, C.D.; Alekel, D.L.; Ritland, L.M.; Bhupathiraju, S.N.; Stewart, J.W.; Hanson, L.N.; Matvienko, O.A.; Kohut, M.L.; Reddy, M.B.; Van Loan, M.D.; et al. Centrally located body fat is related to inflammatory markers in healthy postmenopausal women. Menopause 2008, 15, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Shoelson, S.E.; Lee, J.; Goldfine, A.B. Inflammation and insulin resistance. J. Clin. Investig. 2006, 116, 1793–1801. [Google Scholar] [CrossRef] [PubMed]
- Forbes, L.E.; Storey, K.E.; Fraser, S.N.; Spence, J.C.; Plotnikoff, R.C.; Raine, K.D.; Hanning, R.M.; McCargar, L.J. Dietary patterns associated with glycemic index and glycemic load among Alberta adolescents. Appl. Physiol. Nutr. Metab. 2009, 34, 648–658. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, F.S.; Foster-Powell, K.; Brand-Miller, J.C. International tables of glycemic index and glycemic load values: 2008. Diabetes Care 2008, 31, 2281–2283. [Google Scholar] [CrossRef] [PubMed]
- Salmeron, J.; Manson, J.E.; Stampfer, M.J.; Colditz, G.A.; Wing, A.L.; Willett, W.C. Dietary fiber, glycemic load, and risk of non-insulin-dependent diabetes mellitus in women. JAMA 1997, 277, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Levitan, E.B.; Cook, N.R.; Stampfer, M.J.; Ridker, P.M.; Rexrode, K.M.; Buring, J.E.; Manson, J.E.; Liu, S. Dietary glycemic index, dietary glycemic load, blood lipids, and C-reactive protein. Metab. Clin. Exp. 2008, 57, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Manson, J.E.; Buring, J.E.; Stampfer, M.J.; Willett, W.C.; Ridker, P.M. Relation between a diet with a high glycemic load and plasma concentrations of high-sensitivity C-reactive protein in middle-aged women. Am. J. Clin. Nutr. 2002, 75, 492–498. [Google Scholar] [PubMed]
- Neuhouser, M.L.; Schwarz, Y.; Wang, C.; Breymeyer, K.; Coronado, G.; Wang, C.Y.; Noar, K.; Song, X.; Lampe, J.W. A low-glycemic load diet reduces serum C-reactive protein and modestly increases adiponectin in overweight and obese adults. J. Nutr. 2012, 142, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Gogebakan, O.; Kohl, A.; Osterhoff, M.A.; van Baak, M.A.; Jebb, S.A.; Papadaki, A.; Martinez, J.A.; Handjieva-Darlenska, T.; Hlavaty, P.; Weickert, M.O.; et al. Effects of weight loss and long-term weight maintenance with diets varying in protein and glycemic index on cardiovascular risk factors: The diet, obesity, and genes (DiOGenes) study: A randomized, controlled trial. Circulation 2011, 124, 2829–2838. [Google Scholar] [CrossRef] [PubMed]
- Kelly, K.R.; Haus, J.M.; Solomon, T.P.; Patrick-Melin, A.J.; Cook, M.; Rocco, M.; Barkoukis, H.; Kirwan, J.P. A low-glycemic index diet and exercise intervention reduces TNF(alpha) in isolated mononuclear cells of older, obese adults. J. Nutr. 2011, 141, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Heggen, E.; Klemsdal, T.O.; Haugen, F.; Holme, I.; Tonstad, S. Effect of a low-fat versus a low-gycemic-load diet on inflammatory biomarker and adipokine concentrations. Metab. Syndr. Relat. Disord. 2012, 10, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Juanola-Falgarona, M.; Salas-Salvado, J.; Ibarrola-Jurado, N.; Rabassa-Soler, A.; Diaz-Lopez, A.; Guasch-Ferre, M.; Hernandez-Alonso, P.; Balanza, R.; Bullo, M. Effect of the glycemic index of the diet on weight loss, modulation of satiety, inflammation, and other metabolic risk factors: A randomized controlled trial. Am. J. Clin. Nutr. 2014, 100, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Vrolix, R.; Mensink, R.P. Effects of glycemic load on metabolic risk markers in subjects at increased risk of developing metabolic syndrome. Am. J. Clin. Nutr. 2010, 92, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Shikany, J.M.; Phadke, R.P.; Redden, D.T.; Gower, B.A. Effects of low- and high-glycemic index/glycemic load diets on coronary heart disease risk factors in overweight/obese men. Metab. Clin. Exp. 2009, 58, 1793–1801. [Google Scholar] [CrossRef] [PubMed]
- Bullo, M.; Casas, R.; Portillo, M.P.; Basora, J.; Estruch, R.; Garcia-Arellano, A.; Lasa, A.; Juanola-Falgarona, M.; Aros, F.; Salas-Salvado, J. Dietary glycemic index/load and peripheral adipokines and inflammatory markers in elderly subjects at high cardiovascular risk. Nutr. Metab. Cardiovascr. Dis. NMCD 2013, 23, 443–450. [Google Scholar] [CrossRef] [PubMed]
- McKeown, N.M.; Meigs, J.B.; Liu, S.; Saltzman, E.; Wilson, P.W.; Jacques, P.F. Carbohydrate nutrition, insulin resistance, and the prevalence of the metabolic syndrome in the Framingham Offspring Cohort. Diabetes Care 2004, 27, 538–546. [Google Scholar] [CrossRef] [PubMed]
- Lau, C.; Faerch, K.; Glumer, C.; Tetens, I.; Pedersen, O.; Carstensen, B.; Jorgensen, T.; Borch-Johnsen, K.; Inter, S. Dietary glycemic index, glycemic load, fiber, simple sugars, and insulin resistance: The Inter99 study. Diabetes Care 2005, 28, 1397–1403. [Google Scholar] [CrossRef] [PubMed]
- Kerstetter, J.E.; Bihuniak, J.D.; Brindisi, J.; Sullivan, R.R.; Mangano, K.M.; Larocque, S.; Kotler, B.M.; Simpson, C.A.; Cusano, A.M.; Gaffney-Stomberg, E.; et al. The Effect of a Whey Protein Supplement on Bone Mass in Older Caucasian Adults. J. Clin. Endocrinol. Metab. 2015, 100, 2214–2222. [Google Scholar] [CrossRef] [PubMed]
- Sydney University Glycemic Index Research Service. The Official Website of the Glycemic Index and GI Database. Available online: www.glycemicindex.com (accessed on 1 October 2014).
- Cray, C.; Rodriguez, M.; Zaias, J.; Altman, N.H. Effects of storage temperature and time on clinical biochemical parameters from rat serum. J. Am. Assoc. Lab. Anim. Sci. JAALAS 2009, 48, 202–204. [Google Scholar] [PubMed]
- Cheng, F.W.; Gao, X.; Mitchell, D.C.; Wood, C.; Still, C.D.; Rolston, D.; Jensen, G.L. Body Mass Index and All-Cause Mortality among Older Adults. Obesity 2016, 24, 2232–2239. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Sasaki, S.; Uenishi, K.; Japan Dietetic Students’ Study for Nutrition and Biomarkers Group. Dietary glycemic index, but not glycemic load, is positively associated with serum homocysteine concentration in free-living young Japanese women. Nutr. Res. 2014, 34, 25–30. [Google Scholar]
- Qi, L.; van Dam, R.M.; Liu, S.; Franz, M.; Mantzoros, C.; Hu, F.B. Whole-grain, bran, and cereal fiber intakes and markers of systemic inflammation in diabetic women. Diabetes Care 2006, 29, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Goletzke, J.; Buyken, A.E.; Joslowski, G.; Bolzenius, K.; Remer, T.; Carstensen, M.; Egert, S.; Nothlings, U.; Rathmann, W.; Roden, M.; et al. Increased Intake of Carbohydrates from Sources with a Higher Glycemic Index and Lower Consumption of Whole Grains during Puberty Are Prospectively Associated with Higher IL-6 Concentrations in Younger Adulthood among Healthy Individuals. J. Nutr. 2014, 144, 1586–1593. [Google Scholar] [CrossRef] [PubMed]
- Liu, K. Statistical issues related to semiquantitative food-frequency questionnaires. Am. J. Clin. Nutr. 1994, 59, 262S–265S. [Google Scholar] [PubMed]
- Schaefer, E.J.; Augustin, J.L.; Schaefer, M.M.; Rasmussen, H.; Ordovas, J.M.; Dallal, G.E.; Dwyer, J.T. Lack of efficacy of a food-frequency questionnaire in assessing dietary macronutrient intakes in subjects consuming diets of known composition. Am. J. Clin. Nutr. 2000, 71, 746–751. [Google Scholar] [PubMed]
- Hare-Bruun, H.; Flint, A.; Heitmann, B.L. Glycemic index and glycemic load in relation to changes in body weight, body fat distribution, and body composition in adult Danes. Am. J. Clin. Nutr. 2006, 84, 871–879. [Google Scholar] [PubMed]
- Maki, K.C.; Rains, T.M.; Kaden, V.N.; Raneri, K.R.; Davidson, M.H. Effects of a reduced-glycemic-load diet on body weight, body composition, and cardiovascular disease risk markers in overweight and obese adults. Am. J. Clin. Nutr. 2007, 85, 724–734. [Google Scholar] [PubMed]
- Pfutzner, A.; Kunt, T.; Hohberg, C.; Mondok, A.; Pahler, S.; Konrad, T.; Lübben, G.; Forst, T. Fasting intact proinsulin is a highly specific predictor of insulin resistance in type 2 diabetes. Diabetes Care 2004, 27, 682–687. [Google Scholar] [CrossRef] [PubMed]
- Basiotis, P.P.; Welsh, S.O.; Cronin, F.J.; Kelsay, J.L.; Mertz, W. Number of Days of Food Intake Records Required to Estimate Individual and Group Nutrient Intakes with Defined Confidence. J. Nutr. 1987, 117, 1638–1641. [Google Scholar] [PubMed]
| CHO | PRO | |||||
|---|---|---|---|---|---|---|
| Baseline | 18 Months | Δ (18 Months-Baseline) | Baseline | 18-Months | Δ (18 Months-Baseline) | |
| N | 46 | 38 | ||||
| Age | 69.3 ± 0.9 | 68.9 ± 0.9 | ||||
| Weight (kg) | 66.5 ± 1.5 | 66.8 ± 1.6 | 0.3 ± 0.6 | 68.1 ± 1.7 | 68.0 ± 1.6 | −0.2 ± 0.4 |
| BMI (kg/m2) | 25.8 ± 0.6 | 26.0 ± 0.6 | 0.2 ± 0.2 | 26.0 ± 0.6 | 26.0 ± 0.6 | 0.009 ± 0.1 |
| Body lean mass (kg) | 39.0 ± 0.6 | 38.4 ± 0.6 a | −0.6 ± 0.3 a | 40.1 ± 0.7 | 40.3 ± 0.7 a | 0.2 ± 0.2 a |
| Trunk lean mass (kg) | 19.6 ± −0.3 | 19.1 ± −0.3 a,b | −0.5 ±0.2 a | 20.0 ± 0.3 | 20.1 ± 0.4 a | 0.1 ± 0.1 a |
| Body fat mass (kg) | 25.5 ± 1.1 | 26.7 ± 1.2 | 1.2 ± 0.5 | 25.9 ± 1.3 | 25.9 ± 1.2 | −0.009 ± 0.4 |
| Lean/Fat | 1.7 ± 0.1 | 1.6 ± 0.1 | −0.1 ± 0.03 a | 1.7 ± 0.1 | 1.7 ± 0.1 | −0.001 ± 0.03 a |
| Dietary Calories (kcal) | 1661 ± 51.6 | 1626 ± 55.5 | −34.5 ± 54.9 | 1627 ± 45.6 | 1551 ± 57.1 | −70.4 ± 53.5 |
| Total Calories (kcal) | 1661 ± 51.6 | 1729 ± 55.5 | 68.2 ± 54.5 | 1627 ± 45.6 | 1678 ± 58.0 | 56.5 ± 54.2 |
| Dietary Carbohydrate (g) | 201.2 ± 6.9 | 202.8 ± 8.7 | 1.6 ± 7.5 | 207.2 ± 9.0 | 198.9 ± 8.9 | −8.6 ± 8.4 |
| Fiber (g) | 19.4 ± 1.0 | 18.9 ± 1.0 | −1.1 ± 1.1 | 21.0 ± 1.2 | 18.3 ± 1.0 | −2.8 ± 1.4 |
| Supplement Carbohydrate (g) | 0 | 29.5 ± 0.9 | 0 | |||
| Total carbohydrate (g) | 201.2 ± 6.9 | 232.3 ± 8.7 a,b | 31.1 ± 7.4 a | 207.2 ± 9.0 | 198.9 ± 8.9 a | −8.6 ± 8.4 a |
| Dietary Protein (g) | 71.5 ± 2.2 | 69.8 ± 2.5 | −1.7 ± 2.3 | 73.5 ± 2.7 | 68.3 ± 2.5 | −5.1 ± 2.9 |
| Supplement Protein (g) | 0 | 0 | 30.4 ± 0.9 | |||
| Total protein (g) | 71.5 ± 2.2 | 69.8 ± 2.5 a | −1.7 ± 2.3 a | 73.5 ± 2.7 | 98.5 ± 2.8 a,b | 25.1 ± 3.1 a |
| Dietary Fat (g) | 62.5 ± 3.9 | 57.1 ± 2.8 | −5.4 ± 3.7 | 56.1 ± 2.7 | 51.6 ± 2.5 | −4.4 ± 3.2 |
| CHO | PRO | ||||||
|---|---|---|---|---|---|---|---|
| Baseline | 18-Months | Δ | Baseline | 18-Months | Δ | ||
| IL-6 (pg/mL) | 1.7 ± 0.2 | 1.9 ± 0.2 | 0.2 ± 0.2 | 1.5 ± 0.1 | 1.8 ± 0.1 | 0.3 ± 0.1 | |
| CRP (mg/L) | 1.5 ± 0.2 | 2.0 ± 0.2 | 0.5 ± 0.2 | 1.9 ± 0.2 | 2.5 ± 0.4 | 0.6 ± 0.3 | |
| HOMA | % B 2 | 132.8 ± 7.1 | 117.1 ± 6.2 | −15.7 ± 8.4 | 137.0 ± 8.6 | 133.3 ± 6.1 | −3.7 ± 9.3 |
| % S 2 | 75.0 ± 4.5 | 75.7 ± 4.5 | 0.66 ± 4.7 | 66.9 ± 3.6 | 64.1 ± 3.3 | −2.9 ± 2.6 | |
| % IR 2 | 1.6 ± 0.1 | 1.5 ± 0.1 | −0.02 ± 0.08 | 1.7 ± 0.1 | 1.7 ± 0.1 | 0.02 ± 0.07 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stojkovic, V.; Simpson, C.A.; Sullivan, R.R.; Cusano, A.M.; Kerstetter, J.E.; Kenny, A.M.; Insogna, K.L.; Bihuniak, J.D. The Effect of Dietary Glycemic Properties on Markers of Inflammation, Insulin Resistance, and Body Composition in Postmenopausal American Women: An Ancillary Study from a Multicenter Protein Supplementation Trial. Nutrients 2017, 9, 484. https://doi.org/10.3390/nu9050484
Stojkovic V, Simpson CA, Sullivan RR, Cusano AM, Kerstetter JE, Kenny AM, Insogna KL, Bihuniak JD. The Effect of Dietary Glycemic Properties on Markers of Inflammation, Insulin Resistance, and Body Composition in Postmenopausal American Women: An Ancillary Study from a Multicenter Protein Supplementation Trial. Nutrients. 2017; 9(5):484. https://doi.org/10.3390/nu9050484
Chicago/Turabian StyleStojkovic, Violeta, Christine A. Simpson, Rebecca R. Sullivan, Anna Maria Cusano, Jane E. Kerstetter, Anne M. Kenny, Karl L. Insogna, and Jessica D. Bihuniak. 2017. "The Effect of Dietary Glycemic Properties on Markers of Inflammation, Insulin Resistance, and Body Composition in Postmenopausal American Women: An Ancillary Study from a Multicenter Protein Supplementation Trial" Nutrients 9, no. 5: 484. https://doi.org/10.3390/nu9050484
APA StyleStojkovic, V., Simpson, C. A., Sullivan, R. R., Cusano, A. M., Kerstetter, J. E., Kenny, A. M., Insogna, K. L., & Bihuniak, J. D. (2017). The Effect of Dietary Glycemic Properties on Markers of Inflammation, Insulin Resistance, and Body Composition in Postmenopausal American Women: An Ancillary Study from a Multicenter Protein Supplementation Trial. Nutrients, 9(5), 484. https://doi.org/10.3390/nu9050484




