A Low Glycaemic Index Diet Incorporating Isomaltulose Is Associated with Lower Glycaemic Response and Variability, and Promotes Fat Oxidation in Asians
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Study Protocol
2.3. Treatment Meals
2.4. Glycaemic Measurements
2.5. Energy Expenditure and Substrate Oxidation
fat oxidation (g/min) = 1.67 × O2 (L/min) − 1.67 × CO2 (L/min) − 1.92 × N (g/min)
2.6. Statistics
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dickinson, S.; Colagiuri, S.; Faramus, E.; Petocz, P.; Brand-Miller, J.C. Postprandial hyperglycemia and insulin sensitivity differ among lean young adults of different ethnicities. J. Nutr. 2002, 132, 2574–2579. [Google Scholar] [PubMed]
- Venn, B.S.; Williams, S.M.; Mann, J.I. Comparison of postprandial glycaemia in Asians and Caucasians. Diabetes Med. 2010, 27, 1205–1208. [Google Scholar] [CrossRef]
- Hu, F.B. Globalization of Diabetes The role of diet, lifestyle, and genes. Diabetes Care 2011, 34, 1249–1257. [Google Scholar] [CrossRef] [PubMed]
- Monnier, L.; Mas, E.; Ginet, C.; Michel, F.; Villon, L.; Cristol, J.P.; Colette, C. Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA 2006, 295, 1681–1687. [Google Scholar] [CrossRef] [PubMed]
- Brand-Miller, J.; Hayne, S.; Petocz, P.; Colagiuri, S. Low-glycemic index diets in the management of diabetes: A meta-analysis of randomized controlled trials. Diabetes Care 2003, 26, 2261–2267. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.E.; Elliott, E.J. The use of low-glycaemic index diets in diabetes control. Br. J. Nutr. 2010, 104, 797–802. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, D.S. The glycemic index: Physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA 2002, 287, 2414–2423. [Google Scholar] [CrossRef] [PubMed]
- Foo, L.L.; Vijaya, K.; Sloan, R.A.; Ling, A. Obesity prevention and management: Singapore’s experience. Obes. Rev. 2013, 14, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Roberts, S.B. High-glycemic index foods, hunger, and obesity: Is there a connection? Nutr. Rev. 2000, 58, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Holloway, G.P.; Bonen, A.; Spriet, L.L. Regulation of skeletal muscle mitochondrial fatty acid metabolism in lean and obese individuals. Am. J. Clin. Nutr. 2009, 89, 455S–462S. [Google Scholar] [CrossRef] [PubMed]
- Ravussin, E.; Smith, S.R. Increased fat intake, impaired fat oxidation, and failure of fat cell proliferation result in ectopic fat storage, insulin resistance, and type 2 diabetes mellitus. Ann. N. Y. Acad. Sci. 2002, 967, 363–378. [Google Scholar] [CrossRef] [PubMed]
- Kahlhofer, J.; Karschin, J.; Silberhorn-Buhler, H.; Breusing, N.; Bosy-Westphal, A. Effect of low-glycemic-sugar-sweetened beverages on glucose metabolism and macronutrient oxidation in healthy men. Int. J. Obes. (Lond.) 2016, 40, 990–997. [Google Scholar] [CrossRef] [PubMed]
- Pannacciulli, N.; Salbe, A.D.; Ortega, E.; Venti, C.A.; Bogardus, C.; Krakoff, J. The 24-h carbohydrate oxidation rate in a human respiratory chamber predicts ad libitum food intake. Am. J. Clin. Nutr. 2007, 86, 625–632. [Google Scholar] [PubMed]
- Weyer, C.; Pratley, R.E.; Salbe, A.D.; Bogardus, C.; Ravussin, E.; Tataranni, P.A. Energy expenditure, fat oxidation, and body weight regulation: A study of metabolic adaptation to long-term weight change. J. Clin. Endocrinol. Metab. 2000, 85, 1087–1094. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, E.; Williams, C.; Nute, M. The influence of the glycaemic index of breakfast and lunch on substrate utilisation during the postprandial periods and subsequent exercise. Br. J. Nutr. 2005, 93, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, E.J.; Williams, C.; Mash, L.E.; Phillips, B.; Nute, M.L. Influence of high-carbohydrate mixed meals with different glycemic indexes on substrate utilization during subsequent exercise in women. Am. J. Clin. Nutr. 2006, 84, 354–360. [Google Scholar] [PubMed]
- Wee, S.L.; Williams, C.; Gray, S.; Horabin, J. Influence of high and low glycemic index meals on endurance running capacity. Med. Sci. Sports Exerc. 1999, 31, 393–399. [Google Scholar] [CrossRef] [PubMed]
- DeMarco, H.M.; Sucher, K.P.; Cisar, C.J.; Butterfield, G.E. Pre-exercise carbohydrate meals: Application of glycemic index. Med. Sci. Sports Exerc. 1999, 31, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Livesey, G. Health potential of polyols as sugar replacers, with emphasis on low glycaemic properties. Nutr. Res. Rev. 2003, 16, 163–191. [Google Scholar] [CrossRef] [PubMed]
- Konig, D.; Theis, S.; Kozianowski, G.; Berg, A. Postprandial substrate use in overweight subjects with the metabolic syndrome after isomaltulose (Palatinose) ingestion. Nutrition 2012, 28, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Chandalia, M.; Garg, A.; Lutjohann, D.; von Bergmann, K.; Grundy, S.M.; Brinkley, L.J. Beneficial effects of high dietary fiber intake in patients with type 2 diabetes mellitus. N. Engl. J. Med. 2000, 342, 1392–1398. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Raymond, K. Beta-glucans in the treatment of diabetes and associated cardiovascular risks. Vasc. Health Risk Manag. 2008, 4, 1265–1272. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Keogh, J.B.; Clifton, P.M. Polyphenols and Glycemic Control. Nutrients 2016, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- Lina, B.A.; Jonker, D.; Kozianowski, G. Isomaltulose (Palatinose): A review of biological and toxicological studies. Food Chem. Toxicol. 2002, 40, 1375–1381. [Google Scholar] [CrossRef]
- Holub, I.; Gostner, A.; Theis, S.; Nosek, L.; Kudlich, T.; Melcher, R.; Scheppach, W. Novel findings on the metabolic effects of the low glycaemic carbohydrate isomaltulose (Palatinose). Br. J. Nutr. 2010, 103, 1730–1737. [Google Scholar] [CrossRef] [PubMed]
- Brunner, S.; Holub, I.; Theis, S.; Gostner, A.; Melcher, R.; Wolf, P.; Amann-Gassner, U.; Scheppach, W.; Hauner, H. Metabolic effects of replacing sucrose by isomaltulose in subjects with type 2 diabetes: A randomized double-blind trial. Diabetes Care 2012, 35, 1249–1251. [Google Scholar] [CrossRef] [PubMed]
- Van Can, J.G.; van Loon, L.J.; Brouns, F.; Blaak, E.E. Reduced glycaemic and insulinaemic responses following trehalose and isomaltulose ingestion: Implications for postprandial substrate use in impaired glucose-tolerant subjects. Br. J. Nutr. 2012, 108, 1210–1217. [Google Scholar] [CrossRef] [PubMed]
- Okuno, M.; Kim, M.K.; Mizu, M.; Mori, M.; Mori, H.; Yamori, Y. Palatinose-blended sugar compared with sucrose: Different effects on insulin sensitivity after 12 weeks supplementation in sedentary adults. Int. J. Food Sci. Nutr. 2010, 61, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Baecke, J.A.; Burema, J.; Frijters, J.E. A short questionnaire for the measurement of habitual physical activity in epidemiological studies. Am. J. Clin. Nutr. 1982, 36, 936–942. [Google Scholar] [PubMed]
- Van Strien, T.; Frijters, J.E.R.; Bergers, G.; Defares, P.B. The Dutch Eating Behavior Questionnaire (DEBQ) for assessment of restrained, emotional, and external eating behavior. Int. J. Eat. Disord. 1986, 5, 295–315. [Google Scholar] [CrossRef]
- Randomizer.org. Available online: http://www.randomizer.org/ (accessed on 2 May 2017).
- CareLink iPro. Available online: http://carelink.minimed.eu (accessed on 2 May 2017).
- Kaur, B.; Quek Yu Chin, R.; Camps, S.; Henry, C.J. The impact of a low glycaemic index (GI) diet on simultaneous measurements of blood glucose and fat oxidation: A whole body calorimetric study. J. Clin. Transl. Endocrinol. 2016, 4, 45–52. [Google Scholar] [CrossRef]
- Schoffelen, P.F.; Westerterp, K.R.; Saris, W.H.; Ten Hoor, F. A dual-respiration chamber system with automated calibration. J. Appl. Physiol. (1985) 1997, 83, 2064–2072. [Google Scholar]
- Janssens, P.L.H.R.; Hursel, R.; Westerterp-Plantenga, M.S. Capsaicin increases sensation of fullness in energy balance, and decreases desire to eat after dinner in negative energy balance. Appetite 2014, 77, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Reddy, N.L.; Peng, C.; Carreira, M.C.; Halder, L.; Hattersley, J.; Piya, M.K.; Tripathi, G.; Randeva, H.S.; Casanueva, F.F.; McTernan, P.G. Enhanced thermic effect of food, postprandial NEFA suppression and raised adiponectin in obese women who eat slowly. Clin. Endocrinol. 2015, 82, 831–837. [Google Scholar] [CrossRef] [PubMed]
- Weir, J.B. New methods for calculating metabolic rate with special reference to protein metabolism. J. Physiol. 1949, 109, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Frayn, K.N. Calculation of substrate oxidation rates in vivo from gaseous exchange. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1983, 55, 628–634. [Google Scholar] [PubMed]
- Allison, D.B.; Paultre, F.; Maggio, C.; Mezzitis, N.; Pi-Sunyer, F.X. The use of areas under curves in diabetes research. Diabetes Care 1995, 18, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Brouns, F.; Bjorck, I.; Frayn, K.N.; Gibbs, A.L.; Lang, V.; Slama, G.; Wolever, T.M.S. Glycaemic index methodology. Nutr. Res. Rev. 2005, 18, 145. [Google Scholar] [CrossRef] [PubMed]
- Molnar, G.D.; Rosevear, J.W.; Ackerman, E.; Gatewood, L.C.; Taylor, W.F. Mean amplitude of glycemic excursions, a measure of diabetic instability. Diabetes 1970, 19, 644–655. [Google Scholar]
- Guerci, B. Asymptomatic glycemic instability: How to measure it and which clinical applications? Diabetes Metab. 2003, 29, 179–188. [Google Scholar] [CrossRef]
- Kovatchev, B.P.; Clarke, W.L.; Breton, M.; Brayman, K.; McCall, A. Quantifying temporal glucose variability in diabetes via continuous glucose monitoring: Mathematical methods and clinical application. Diabetes Technol. Therap. 2005, 7, 849–862. [Google Scholar] [CrossRef] [PubMed]
- EasyGV. Available online: www.easygv.co.uk (accessed on 2 May 2017).
- Rodbard, D. Interpretation of continuous glucose monitoring data: Glycemic variability and quality of glycemic control. Diabetes Technol. Therap. 2009, 11, S:55–S:67. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.; Wolever, T.M.; Taylor, R.H.; Griffiths, C.; Krzeminska, K.; Lawrie, J.A.; Bennett, C.M.; Goff, D.V.; Sarson, D.L.; Bloom, S.R. Slow release dietary carbohydrate improves second meal tolerance. Am. J. Clin. Nutr. 1982, 35, 1339–1346. [Google Scholar] [PubMed]
- Liljeberg, H.G.; Akerberg, A.K.; Bjorck, I.M. Effect of the glycemic index and content of indigestible carbohydrates of cereal-based breakfast meals on glucose tolerance at lunch in healthy subjects. Am. J. Clin. Nutr. 1999, 69, 647–655. [Google Scholar] [PubMed]
- Wolever, T.M.; Bolognesi, C. Prediction of glucose and insulin responses of normal subjects after consuming mixed meals varying in energy, protein, fat, carbohydrate and glycemic index. J. Nutr. 1996, 126, 2807–2812. [Google Scholar] [PubMed]
- Lawes, C.M.; Parag, V.; Bennett, D.A.; Suh, I.; Lam, T.H.; Whitlock, G.; Barzi, F.; Woodward, M. Blood glucose and risk of cardiovascular disease in the Asia Pacific region. Diabetes Care 2004, 27, 2836–2842. [Google Scholar] [PubMed]
- Scazzina, F.; Del Rio, D.; Benini, L.; Melegari, C.; Pellegrini, N.; Marcazzan, E.; Brighenti, F. The effect of breakfasts varying in glycemic index and glycemic load on dietary induced thermogenesis and respiratory quotient. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Brand-Miller, J.C.; Stockmann, K.; Atkinson, F.; Petocz, P.; Denyer, G. Glycemic index, postprandial glycemia, and the shape of the curve in healthy subjects: Analysis of a database of more than 1000 foods. Am. J. Clin. Nutr. 2009, 89, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Ritz, P.; Krempf, M.; Cloarec, D.; Champ, M.; Charbonnel, B. Comparative continuous-indirect-calorimetry study of two carbohydrates with different glycemic indices. Am. J. Clin. Nutr. 1991, 54, 855–859. [Google Scholar] [PubMed]
- Blaak, E.E.; Saris, W.H. Postprandial thermogenesis and substrate utilization after ingestion of different dietary carbohydrates. Metabolism 1996, 45, 1235–1242. [Google Scholar] [CrossRef]
- Ellis, A.C.; Hyatt, T.C.; Hunter, G.R.; Gower, B.A. Respiratory quotient predicts fat mass gain in premenopausal women. Obesity (Silver Spring) 2010, 18, 2255–2259. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, D.S. Dietary glycemic index and obesity. J. Nutr. 2000, 130, 280S–283S. [Google Scholar] [PubMed]
- Diaz, E.O.; Galgani, J.E.; Aguirre, C.A.; Atwater, I.J.; Burrows, R. Effect of glycemic index on whole-body substrate oxidation in obese women. Int. J. Obes. (Lond.) 2005, 29, 108–114. [Google Scholar] [CrossRef] [PubMed]
- McDevitt, R.M.; Poppitt, S.D.; Murgatroyd, P.R.; Prentice, A.M. Macronutrient disposal during controlled overfeeding with glucose, fructose, sucrose, or fat in lean and obese women. Am. J. Clin. Nutr. 2000, 72, 369–377. [Google Scholar] [PubMed]
- Bosy-Westphal, A.; Hagele, F.; Nas, A. Impact of dietary glycemic challenge on fuel partitioning. Eur. J. Clin. Nutr. 2016. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Nakamura, K.; Ogata, H.; Miyashita, A.; Nagasaka, S.; Omi, N.; Yamaguchi, S.; Hibi, M.; Umeda, T.; Nakaji, S. Acute effect of late evening meal on diurnal variation of blood glucose and energy metabolism. Obes. Res. Clin. Pract. 2011, 5, e220–e228. [Google Scholar] [CrossRef] [PubMed]

| Low GI Diet | High GI Diet | |||||||
|---|---|---|---|---|---|---|---|---|
| % of Total | E% CHO | E% Fat | E% Protein | % of Total | E% CHO | E% Fat | E% Protein | |
| Dinner | 35.6 | 68.2 | 11.7 | 20.1 | 35.8 | 70.8 | 11.3 | 17.9 |
| Basmati parboiled rice | Glutinous rice | |||||||
| Chicken stock | Chicken stock | |||||||
| Teriyaki chicken | Teriyaki chicken | |||||||
| Chinese spinach | Carrots | |||||||
| Fruit drink with Palatinose™ | Fruit drink with sucrose | |||||||
| Breakfast | 16.8 | 77.3 | 9.7 | 13.0 | 16.6 | 83.4 | 7.5 | 9.1 |
| All-bran cereal Full fat milk Chamomile tea with Palatinose™ | Koko Krunch cereal Low fat cheese Reduced fat milk Chamomile tea with sucrose | |||||||
| Lunch | 31.0 | 72.7 | 11.6 | 15.7 | 31.0 | 75.5 | 11.1 | 13.3 |
| Basmati parboiled rice | Glutinous rice | |||||||
| Chicken stock | Chicken stock | |||||||
| Chinese spinach | Carrots | |||||||
| Teriyaki chicken | Teriyaki chicken | |||||||
| Extra virgin olive oil | Extra virgin olive oil | |||||||
| Fruit drink with Palatinose™ | Fruit drink with sucrose | |||||||
| Snack | 16.6 | 73.7 | 11.2 | 15.1 | 16.6 | 75.5 | 16.1 | 8.4 |
| Multigrain bread Strawberry jam (low GI) Chamomile tea with Palatinose™ | White bread Strawberry jam Margarine Chamomile tea with sucrose | |||||||
| Total | 100 | 72.0 | 11.3 | 16.7 | 100 | 75.1 | 11.4 | 13.5 |
| Characteristic (n = 20) | Mean ± SD (Range) |
|---|---|
| Age (years) | 23.8 ± 1.8 (21–29) |
| Height (m) | 1.74 ± 0.05 (1.64–1.83) |
| Weight (kg) | 74.2 ± 9.6 (57.1–88.5) |
| BMI (kg/m2) | 24.4 ± 3.1 (18.2–28.4) |
| Fat percentage (%) | 20.1 ± 5.4 (7.8–27.0) |
| Waist circumference (cm) | 80.5 ± 8.2 (64.0–92.7) |
| Hip circumference (cm) | 100.0 ± 6.3 (91.5–112.2) |
| Waist/hip ratio | 0.80 ± 0.05 (0.67–0.89) |
| BMR (kcal/day) | 1540 ± 143 (1307–1809) |
| Fasting blood glucose (mmol/L) | 4.6 ± 0.5(3.6–5.5) |
| Low GI | High GI | p-Value | |
|---|---|---|---|
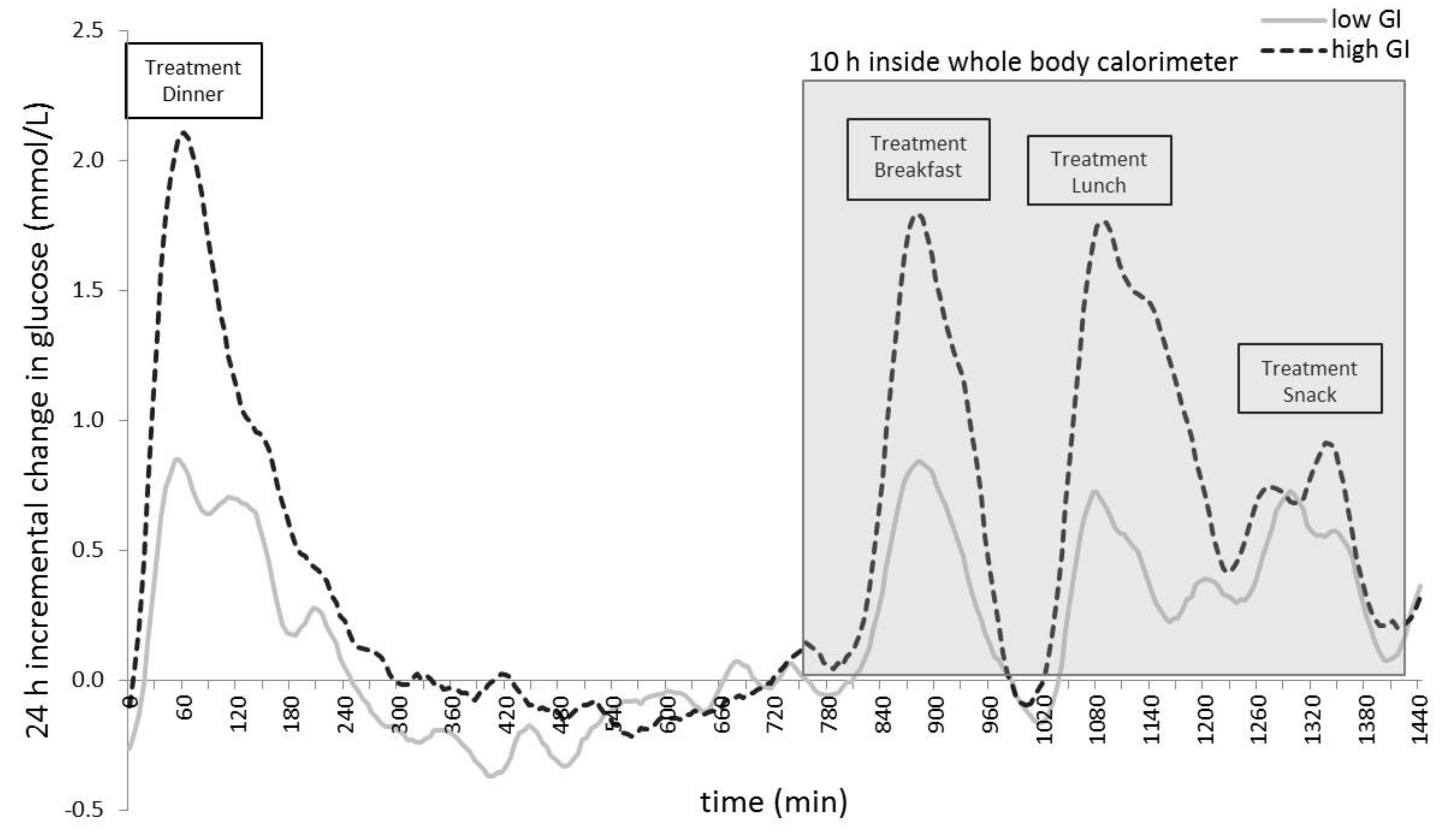
| iAUC overnight after test dinner (mmol/L) | 191.8 ± 168.3 | 318.9 ± 265.1 | 0.027 * |
| iAUC breakfast (mmol/L) | 88.6 ± 53.6 | 177.0 ± 97.9 | <0.001 *** |
| iAUC lunch (mmol/L) | 99.0 ± 51.0 | 233.9 ± 116.1 | <0.001 *** |
| iAUC snack (mmol/L) | 73.9 ± 38.0 | 93.2 ± 62.5 | 0.095 |
| 10 h WBC iAUC (mmol/L) | 288.0 ± 109.8 | 539.9 ± 290.7 | <0.001 *** |
| 24 h iAUC (mmol/L) | 502.5 ± 231.4 | 872.6 ± 493.1 | <0.001 *** |
| iAUC overnight after standard dinner (mmol/L) | 237.0 ± 123.2 | 380.5 ± 297.2 | 0.026 * |
| 42 h iAUC (mmol/L) | 775.7 ± 333.1 | 1379.8 ± 804.7 | 0.002 ** |
| 24 h total AUC (mmol/L) | 43,313 ± 3313 | 43,157 ± 4674 | 0.440 |
| MAGE (24 h) | 1.67 ± 0.53 | 2.68 ± 1.13 | <0.001 *** |
| Low GI | High GI | p-Value | |
|---|---|---|---|
| Energy expenditure | 1.565 ± 0.084 | 1.591 ± 0.097 | 0.19 |
| RQ | 0.907 ± 0.030 | 0.910 ± 0.41 | 0.300 |
| Incremental RQ (10 h) | 0.064 ± 0.030 | 0.070 ± 0.041 | 0.014 * |
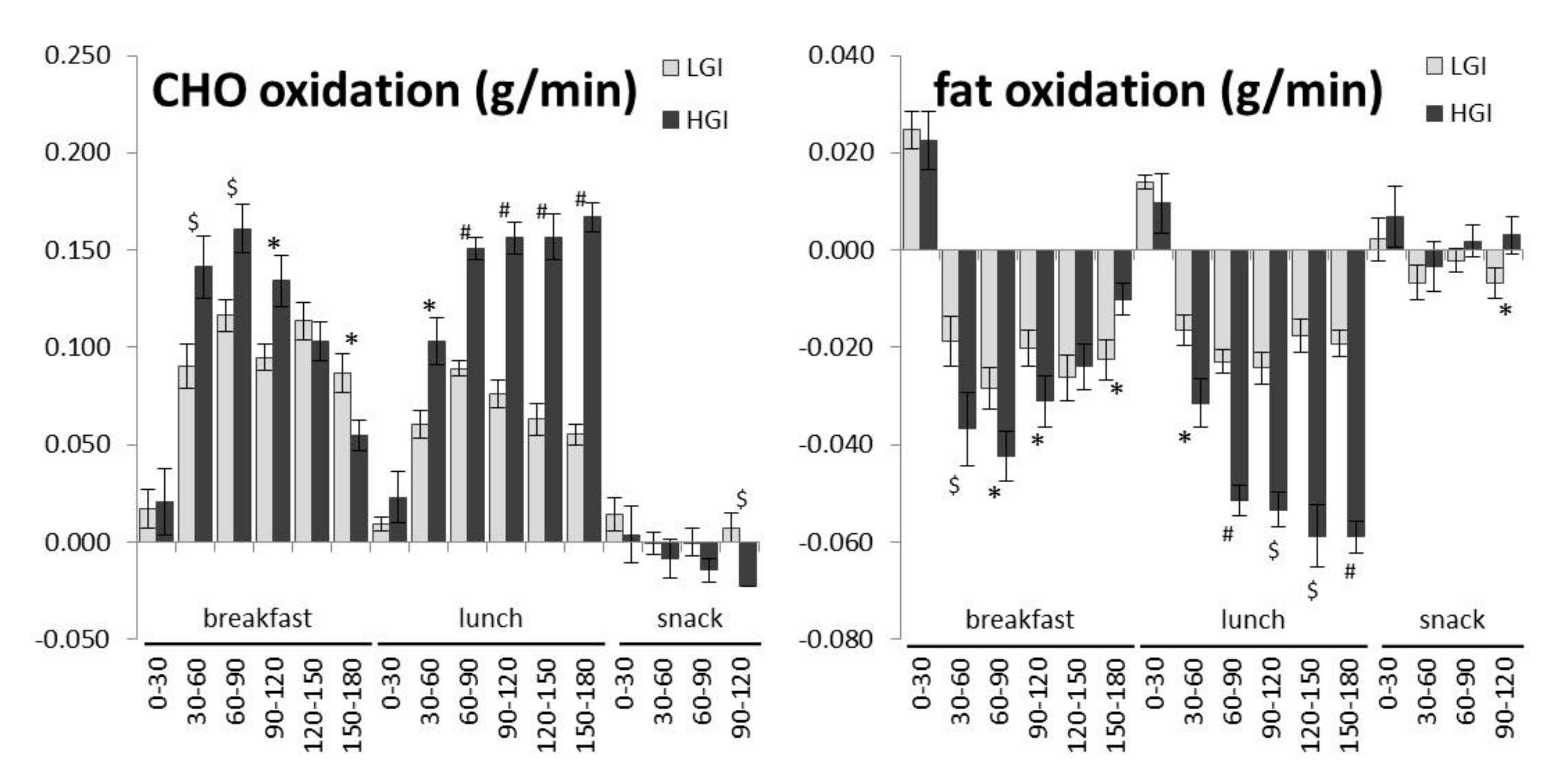
| CHO oxidation (g/min) | 0.268 ± 0.043 | 0.275 ± 0.062 | 0.039 * |
| Incremental CHO oxidation (10 h) (g/min) | 0.121 ± 0.043 | 0.141 ± 0.063 | <0.001 *** |
| Fat oxidation (g/min) | 0.040 ± 0.018 | 0.038 ± 0.024 | 0.237 |
| Incremental fat oxidation (10 h) (g/min) | –0.025 ± 0.018 | –0.028 ± 0.025 | 0.052 |
| Protein oxidation (g/min) | 0.007 ± 0.003 | 0.007 ± 0.002 | 0.53 |
| Low GI | High GI | p-Value | |
|---|---|---|---|
| iRQ breakfast | 0.046 ± 0.034 | 0.054 ± 0.043 | 0.101 |
| iRQ lunch | 0.031 ± 0.024 | 0.074 ± 0.041 | <0.001 *** |
| iRQ snack | 0.006 ± 0.013 | –0.011 ± 0.017 | <0.001*** |
| iCHO oxidation breakfast (g/min) | 0.086 ± 0.039 | 0.102 ± 0.058 | 0.013 * |
| iCHO oxidation lunch (g/min) | 0.059 ± 0.029 | 0.126 ± 0.056 | <0.001 *** |
| iCHO oxidation snack (g/min) | 0.005 ± 0.018 | −0.013 ± 0.027 | 0.003 ** |
| iFat oxidation breakfast (g/min) | −0.015 ± 0.021 | −0.021 ± 0.025 | 0.026 * |
| iFat oxidation lunch (g/min) | −0.014 ± 0.015 | −0.041 ± 0.027 | <0.001 *** |
| iFat oxidation snack (g/min) | −0.003 ± 0.009 | 0.002 ± 0.011 | 0.013 * |
| iRQ breakfast | 0.046 ± 0.034 | 0.054 ± 0.043 | 0.101 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Henry, C.J.; Kaur, B.; Quek, R.Y.C.; Camps, S.G. A Low Glycaemic Index Diet Incorporating Isomaltulose Is Associated with Lower Glycaemic Response and Variability, and Promotes Fat Oxidation in Asians. Nutrients 2017, 9, 473. https://doi.org/10.3390/nu9050473
Henry CJ, Kaur B, Quek RYC, Camps SG. A Low Glycaemic Index Diet Incorporating Isomaltulose Is Associated with Lower Glycaemic Response and Variability, and Promotes Fat Oxidation in Asians. Nutrients. 2017; 9(5):473. https://doi.org/10.3390/nu9050473
Chicago/Turabian StyleHenry, Christiani Jeyakumar, Bhupinder Kaur, Rina Yu Chin Quek, and Stefan Gerardus Camps. 2017. "A Low Glycaemic Index Diet Incorporating Isomaltulose Is Associated with Lower Glycaemic Response and Variability, and Promotes Fat Oxidation in Asians" Nutrients 9, no. 5: 473. https://doi.org/10.3390/nu9050473
APA StyleHenry, C. J., Kaur, B., Quek, R. Y. C., & Camps, S. G. (2017). A Low Glycaemic Index Diet Incorporating Isomaltulose Is Associated with Lower Glycaemic Response and Variability, and Promotes Fat Oxidation in Asians. Nutrients, 9(5), 473. https://doi.org/10.3390/nu9050473





