Acute Vitamin D3 Supplementation in Severe Obesity: Evaluation of Multimeric Adiponectin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Study Design
2.3. Body Measurements
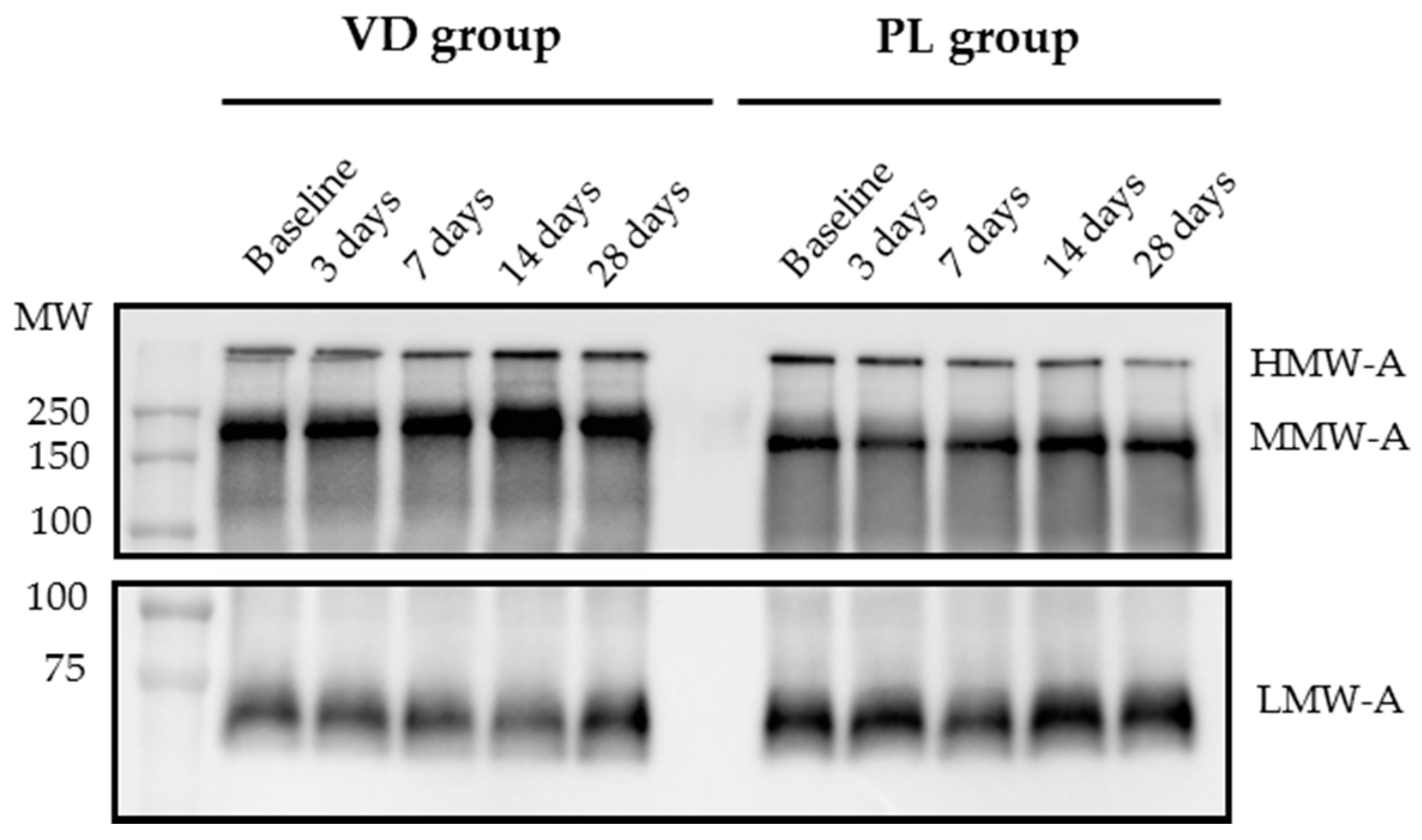
2.4. Western Immunoblot (WIB) of Multimeric Adiponectin
2.5. Adiponectin Immunoassay
2.6. Biochemistry
2.7. Statistics
3. Results
3.1. Metabolic Effects of Cholecalciferol Supplementation
3.2. Analysis of Multimeric Adiponectin
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.; Gao, D.; Wilding, J.; Trayhurn, P.; Bing, C. Vitamin D signalling in adipose tissue. Br. J. Nutr. 2012, 108, 1915–1923. [Google Scholar] [CrossRef] [PubMed]
- Bellan, M.; Guzzaloni, G.; Rinaldi, M.; Merlotti, E.; Ferrari, C.; Tagliaferri, A.; Pirisi, M.; Aimaretti, G.; Scacchi, M.; Marzullo, P. Altered glucose metabolism rather than naive type 2 diabetes mellitus (T2DM) is related to vitamin D status in severe obesity. Cardiovasc. Diabetol. 2014, 13, 57. [Google Scholar] [CrossRef] [PubMed]
- McGill, A.T.; Stewart, J.M.; Lithander, F.E.; Strik, C.M.; Poppitt, S.D. Relationships of low serum vitamin D3 with anthropometry and markers of the metabolic syndrome and diabetes in overweight and obesity. Nutr. J. 2008, 7, 4. [Google Scholar] [CrossRef] [PubMed]
- Palomer, X.; González-Clemente, J.M.; Blanco-Vaca, F.; Mauricio, D. Role of vitamin D in the pathogenesis of type 2 diabetes mellitus. Obes. Metab. 2008, 10, 185–197. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, C.; Gysemans, C.; Giulietti, A.; Bouillon, R. Vitamin D and diabetes. Diabetologia 2005, 48, 1247–1257. [Google Scholar] [CrossRef] [PubMed]
- Baziar, N.; Jafarian, K.; Shadman, Z.; Qorbani, M.; Nikoo, M.K.; Abd Mishani, M. Effect of therapeutic dose of vitamin D on serum adiponectin and glycemia in vitamin D-insufficient or deficient type 2 diabetic patients. Iran. Red Crescent Med. J. 2014, 16, e21458. [Google Scholar] [CrossRef] [PubMed]
- Breslavsky, A.; Frand, J.; Matas, Z.; Boaz, M.; Barnea, Z.; Shargorodsky, M. Effect of high doses of vitamin D on arterial properties, adiponectin, leptin and glucose homeostasis in type 2 diabetic patients. Clin. Nutr. 2013, 32, 970–975. [Google Scholar] [CrossRef] [PubMed]
- Krul-Poel, Y.H.; Ter Wee, M.M.; Lips, P.; Simsek, S. Management of Endocrine Disease: The effect of vitamin D supplementation on glycaemic control in patients with type 2 diabetes mellitus: A systematic review and meta-analysis. Eur. J. Endocrinol. 2017, 176, R1–R14. [Google Scholar] [CrossRef] [PubMed]
- Athyros, V.G.; Tziomalos, K.; Karagiannis, A.; Anagnostis, P.; Mikhailidis, D.P. Should adipokines be considered in the choice of the treatment of obesity-related health problems? Curr. Drug Targets 2010, 11, 122–135. [Google Scholar] [CrossRef] [PubMed]
- Matsuzawa, Y. Adiponectin: Identification, physiology and clinical relevance in metabolic and vascular disease. Atheroscler. Suppl. 2005, 6, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Han, S.H.; Quon, M.J.; Kim, J.A.; Koh, K.K. Adiponectin and cardiovascular disease: Response to therapeutic interventions. J. Am. Coll. Cardiol. 2007, 49, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Shibata, R.; Ouchi, N.; Murohara, T. Adiponectin and cardiovascular disease. Circ. J. 2009, 73, 608–614. [Google Scholar] [CrossRef] [PubMed]
- Bobbert, T.; Rochlitz, H.; Wegewitz, U.; Akpulat, S.; Mai, K.; Weickert, M.O.; Möhlig, M.; Pfeiffer, A.F.; Spranger, J. Changes of adiponectin oligomer composition by moderate weight reduction. Diabetes 2005, 54, 2712–2719. [Google Scholar] [CrossRef] [PubMed]
- Kaser, S.; Tatarczyk, T.; Stadlmayr, A.; Ciardi, C.; Ress, C.; Tschoner, A.; Sandhofer, A.; Paulweber, B.; Ebenbichler, C.F.; Patsch, J.R. Effect of obesity and insulin sensitivity on adiponectin isoform distribution. Eur. J. Clin. Investig. 2008, 38, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Antoniades, C.; Antonopoulos, A.S.; Tousoulis, D.; Stefanadis, C. Adiponectin: From obesity to cardiovascular disease. Obes. Rev. 2009, 10, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Lara-Castro, C.; Luo, N.; Wallace, P.; Klein, R.L.; Garvey, W.T. Adiponectin multimeric complexes and the metabolic syndrome trait cluster. Diabetes 2006, 55, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Tschritter, O.; Fritsche, A.; Thamer, C.; Haap, M.; Shirkavand, F.; Rahe, S.; Staiger, H.; Maerker, E.; Häring, H.; Stumvoll, M. Plasma adiponectin concentrations predict insulin sensitivity of both glucose and lipid metabolism. Diabetes 2003, 52, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Basu, R.; Pajvani, U.B.; Rizza, R.A.; Scherer, P.E. Selective downregulation of the high molecular weight form of adiponectin in hyperinsulinemia and in type 2 diabetes: Differential regulation from nondiabetic subjects. Diabetes 2007, 56, 2174–2177. [Google Scholar] [CrossRef] [PubMed]
- Mai, S.; Walker, G.E.; Brunani, A.; Guzzaloni, G.; Grossi, G.; Oldani, A.; Aimaretti, G.; Scacchi, M.; Marzullo, P. Inherent insulin sensitivity is a major determinant of multimeric adiponectin responsiveness to short-term weight loss in extreme obesity. Sci. Rep. 2014, 4, 5803. [Google Scholar] [CrossRef] [PubMed]
- Polak, J.; Kovacova, Z.; Jacek, M.; Klimcakova, E.; Kovacikova, M.; Vitkova, M.; Kuda, O.; Sebela, M.; Samcova, E.; Stich, V. An increase in plasma adiponectin multimeric complexes follows hypocaloric diet-induced weight loss in obese and overweight pre-menopausal women. Clin. Sci. 2007, 112, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Liu, E.; Meigs, J.B.; Pittas, A.G.; McKeown, N.; Economos, C.D.; Booth, S.L.; Jacques, P.F. Plasma 25-hydroxyvitamin D is associated with markers of the insulin resistant phenotype in nondiabetic adults. J. Nutr. 2009, 139, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, A.; Williams, J.S.; Forman, J.P. The independent association between 25-hydroxyvitamin D and adiponectin and its relation with BMI in two large cohorts: The NHS and the HPFS. Obesity 2012, 20, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Al-Daghri, N.M.; Al-Attas, O.S.; Alokail, M.S.; Alkharfy, K.M.; Al-Othman, A.; Draz, H.M.; Yakout, S.M.; Al-Saleh, Y.; Al-Yousef, M.; Sabico, S.; et al. Hypovitaminosis D associations with adverse metabolic parameters are accentuated in patients with Type 2 diabetes mellitus: A body mass index-independent role of adiponectin? J. Endocrinol. Investig. 2013, 36, 1–6. [Google Scholar]
- Gannagé-Yared, M.H.; Chedid, R.; Khalife, S.; Azzi, E.; Zoghbi, F.; Halaby, G. Vitamin D in relation to metabolic risk factors, insulin sensitivity and adiponectin in a young Middle-Eastern population. Eur. J. Endocrinol. 2009, 160, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Nimitphong, H.; Chanprasertyothin, S.; Jongjaroenprasert, W.; Ongphiphadhanakul, B. The association between vitamin D status and circulating adiponectin independent of adiposity in subjects with abnormal glucose tolerance. Endocrine 2009, 36, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Dinca, M.; Serban, M.C.; Sahebkar, A.; Mikhailidis, D.P.; Toth, P.P.; Martin, S.S.; Blaha, M.J.; Blüher, M.; Gurban, C.; Penson, P.; et al. Does vitamin D supplementation alter plasma adipokines concentrations? A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2016, 107, 360–371. [Google Scholar] [CrossRef] [PubMed]
- Walker, G.E.; Ricotti, R.; Roccio, M.; Moia, S.; Bellone, S.; Prodam, F.; Bona, G. Pediatric obesity and vitamin D deficiency: A proteomic approach identifies multimeric adiponectin as a key link between these conditions. PLoS ONE 2014, 9, e83685. [Google Scholar] [CrossRef] [PubMed]
- Gueugnon, C.; Mougin, F.; Simon-Rigaud, M.L.; Regnard, J.; Nègre, V.; Dumoulin, G. Effects of an in-patient treatment program based on regular exercise and a balanced diet on high molecular weight adiponectin, resistin levels, and insulin resistance in adolescents with severe obesity. Appl. Physiol. Nutr. Metab. 2012, 37, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Martos-Moren, G.Á.; Barrios, V.; Martínez, G.; Hawkins, F.; Argente, J. Effect of weight loss on high-molecular weight adiponectin in obese children. Obesity 2010, 18, 2288–2294. [Google Scholar] [CrossRef] [PubMed]
- Belenchia, A.M.; Tosh, A.K.; Hillman, L.S.; Peterson, C.A. Correcting vitamin D insufficiency improves insulin sensitivity in obese adolescents: A randomized controlled trial. Am. J. Clin. Nutr. 2013, 97, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Satoh, N.; Naruse, M.; Usui, T.; Tagami, T.; Suganami, T.; Yamada, K.; Kuzuya, H.; Shimatsu, A.; Ogawa, Y. Leptin-to-adiponectin ratio as a potential atherogenic index in obese type 2 diabetic patients. Diabetes Care 2004, 27, 2488–2490. [Google Scholar] [CrossRef] [PubMed]
- Kotani, K.; Sakane, N.; Saiga, K.; Kurozawa, Y. Leptin: Adiponectin ratio as an atherosclerotic index in patients with type 2 diabetes: Relationship of the index to carotid intima-media thickness. Diabetologia 2005, 48, 2684–2686. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Maehata, E.; Yano, M.; Taniyama, M.; Suzuki, S. Correlation between the adiponectin-leptin ratio and parameters of insulin resistance in patients with type 2 diabetes. Metabolism 2005, 54, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Yano, M.; Yamakado, M.; Maehata, E.; Suzuki, S. Relationship between the adiponectin-leptin ratio and parameters of insulin resistance in subjects without hyperglycemia. Metabolism 2006, 55, 1248–1254. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.A. Physiological functions of Vitamin D in adipose tissue. J. Steroid Biochem. Mol. Biol. 2017, 165, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. The vitamin D epidemic and its health consequences. J. Nutr. 2005, 135, 2739S–2748S. [Google Scholar] [PubMed]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: A systematic review. Lancet Diabetes Endocrinol. 2014, 2, 76–89. [Google Scholar] [CrossRef]
- Caan, B.; Neuhouser, M.; Aragaki, A.; Lewis, C.B.; Jackson, R.; LeBoff, M.S.; Margolis, K.L.; Powell, L.; Uwaifo, G.; Whitlock, E.; et al. Calcium plus vitamin D supplementation and the risk of postmenopausal weight gain. Arch. Intern. Med. 2007, 167, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Pittas, A.G.; Harris, S.S.; Stark, P.C.; Dawson-Hughes, B. The effects of calcium and vitamin D supplementation on blood glucose and markers of inflammation in nondiabetic adults. Diabetes Care 2007, 30, 980–986. [Google Scholar] [CrossRef] [PubMed]
- Major, G.C.; Alarie, F.; Doré, J.; Phouttama, S.; Tremblay, A. Supplementation with calcium + vitamin D enhances the beneficial effect of weight loss on plasma lipid and lipoprotein concentrations. Am. J. Clin. Nutr. 2007, 85, 54–59. [Google Scholar] [PubMed]
- Vieth, R.; Bischoff-Ferrari, H.; Boucher, B.J.; Dawson-Hughes, B.; Garland, C.F.; Heaney, R.P.; Holick, M.F.; Hollis, B.W.; Lamberg-Allardt, C.; McGrath, J.J.; et al. The urgent need to recommend an intake of vitamin D that is effective. Am. J. Clin. Nutr. 2007, 85, 649–650. [Google Scholar] [PubMed]
- Aloia, J.F.; Patel, M.; Dimaano, R.; Li-Ng, M.; Talwar, S.A.; Mikhail, M.; Pollack, S.; Yeh, J.K. Vitamin D intake to attain a desired serum 25-hydroxyvitamin D concentration. Am. J. Clin. Nutr. 2008, 87, 1952–1958. [Google Scholar] [PubMed]
- Zittermann, A.; Frisch, S.; Berthold, H.K.; Götting, C.; Kuhn, J.; Kleesiek, K.; Stehle, P.; Koertke, H.; Koerfer, R. Vitamin D supplementation enhances the beneficial effects of weight loss on cardiovascular disease risk markers. Am. J. Clin. Nutr. 2009, 89, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Cipriani, C.; Romagnoli, E.; Scillitani, A.; Chiodini, I.; Clerico, R.; Carnevale, V.; Mascia, M.L.; Battista, C.; Viti, R.; Pileri, M.; et al. Effect of a single oral dose of 600,000 IU of cholecalciferol on serum calciotropic hormones in young subjects with vitamin D deficiency: A prospective intervention study. J. Clin. Endocrinol. Metab. 2010, 95, 4771–4777. [Google Scholar] [CrossRef] [PubMed]
- Camozzi, V.; Frigo, A.C.; Zaninotto, M.; Sanguin, F.; Plebani, M.; Boscaro, M.; Schiavon, L.; Luisetto, G. 25-Hydroxycholecalciferol response to single oral cholecalciferol loading in the normal weight, overweight, and obese. Osteoporos. Int. 2016, 27, 2593–2602. [Google Scholar] [CrossRef] [PubMed]
- Swarbrick, M.M.; Austrheim-Smith, I.T.; Stanhope, K.L.; Van Loan, M.D.; Ali, M.R.; Wolfe, B.M.; Havel, P.J. Circulating concentrations of high-molecular-weight adiponectin are increased following Roux-en-Y gastric bypass surgery. Diabetologia 2006, 49, 2552–2558. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, J.M.; Tzameli, I.; Astapova, I.; Lam, F.S.; Flier, J.S.; Hollenberg, A.N. Complex role of the vitamin D receptor and its ligand in adipogenesis in 3T3-L1 cells. J. Biol. Chem. 2006, 281, 11205–11213. [Google Scholar] [CrossRef] [PubMed]
- Nimitphong, H.; Holick, M.F.; Fried, S.K.; Lee, M.J. 25-hydroxyvitamin D3 and 1,25-dihydroxyvitamin D3 promote the differentiation of human subcutaneous preadipocytes. PLoS ONE 2012, 7, e52171. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Zemel, M.B. Calcium and 1,25-dihydroxyvitamin D3 regulation of adipokine expression. Obesity 2007, 15, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Mallard, S.R.; Howe, A.S.; Houghton, L.A. Vitamin D status and weight loss: A systematic review and meta-analysis of randomized and nonrandomized controlled weight-loss trials. Am. J. Clin. Nutr. 2016, 104, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Piccolo, B.D.; Dolnikowski, G.; Seyoum, E.; Thomas, A.P.; Gertz, E.R.; Souza, E.C.; Woodhouse, L.R.; Newman, J.W.; Keim, N.L.; Adams, S.H.; et al. Association between subcutaneous white adipose tissue and serum 25-hydroxyvitamin D in overweight and obese adults. Nutrients 2013, 5, 3352–3366. [Google Scholar] [CrossRef] [PubMed]
- Duggan, C.; de Dieu Tapsoba, J.; Mason, C.; Imayama, I.; Korde, L.; Wang, C.Y.; McTiernan, A. Effect of Vitamin D3 Supplementation in Combination with Weight Loss on Inflammatory Biomarkers in Postmenopausal Women: A Randomized Controlled Trial. Cancer Prev. Res. 2015, 8, 628–635. [Google Scholar] [CrossRef] [PubMed]
- Lilja, M.; Rolandsson, O.; Norberg, M.; Söderberg, S. The impact of leptin and adiponectin on incident type 2 diabetes is modified by sex and insulin resistance. Metab. Syndr. Relat. Disord. 2012, 10, 143–151. [Google Scholar] [CrossRef] [PubMed]
| Vitamin D Group (n = 12) | Placebo Group (n = 12) | p Value | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Baseline | At the end of the study | ∆ (%) | Baseline | At the end of the study | ∆ (%) | Time | Treatment | Time × Treatment | |
| Male/Female | 6/6 | - | - | 7/5 | - | - | - | - | - |
| Age (years) | 38 ± 2.4 | - | - | 37 ± 3.0 | - | - | - | - | - |
| BMI (kg/m2) | 42.7 ± 1.3 | 40.1 ± 1.2 ** | −6.0 ± 0.7 | 39.8 ± 0.9 | 37.6 ± 0.7 ** | −5.5 ± 0.5 | <0.0001 | 0.07 | 0.41 |
| Body weight (kg) | 115.9 ± 4.4 | 109.2 ± 4.9 ** | −5.8 ± 0.4 | 112.7 ± 4.7 | 106.4 ± 4.3 ** | −5.5 ± 0.5 | <0.0001 | 0.64 | 0.65 |
| Fat mass (%) | 44.8 ± 1.6 | - | - | 40.4 ± 1.6 | - | - | - | - | - |
| Free fat mass (kg) | 63.4 ± 2.9 | - | - | 66.7 ± 3.3 | - | - | - | - | - |
| Waist (cm) | 126.2 ± 3.5 | - | - | 119.0 ± 4.0 | - | - | - | - | - |
| Glucose (mg/dL) | 97.0 ± 7.1 | 90.2 ± 3.1 | −7.1 ± 4.1 | 88.5 ± 3.2 | 83.0 ± 1.6 * | −5.5 ± 2.1 | 0.07 | 0.45 | 0.10 |
| Insulin (mU/L) | 13.9 ± 2.5 | 14.5 ± 1.7 | 5.9 ± 12.7 | 17.5 ± 2.2 | 14.1 ± 1.3 | −18.5 ± 8.1 | 0.06 | 0.17 | 0.30 |
| HbA1c (%) | 5.5 ± 0.1 | 5.7 ± 0.3 | 0.7 ± 3.2 | 5.6 ± 0.6 | 5.3 ± 0.2 * | −4.7 ± 1.3 | 0.21 | 0.53 | 0.07 |
| HOMA−IR | 3.9 ± 0.6 | 3.3 ± 0.4 | −3.2 ± 11.3 | 3.7 ± 0.5 | 3.0 ± 0.2 | −9.7 ± 8.3 | 0.11 | 0.67 | 0.99 |
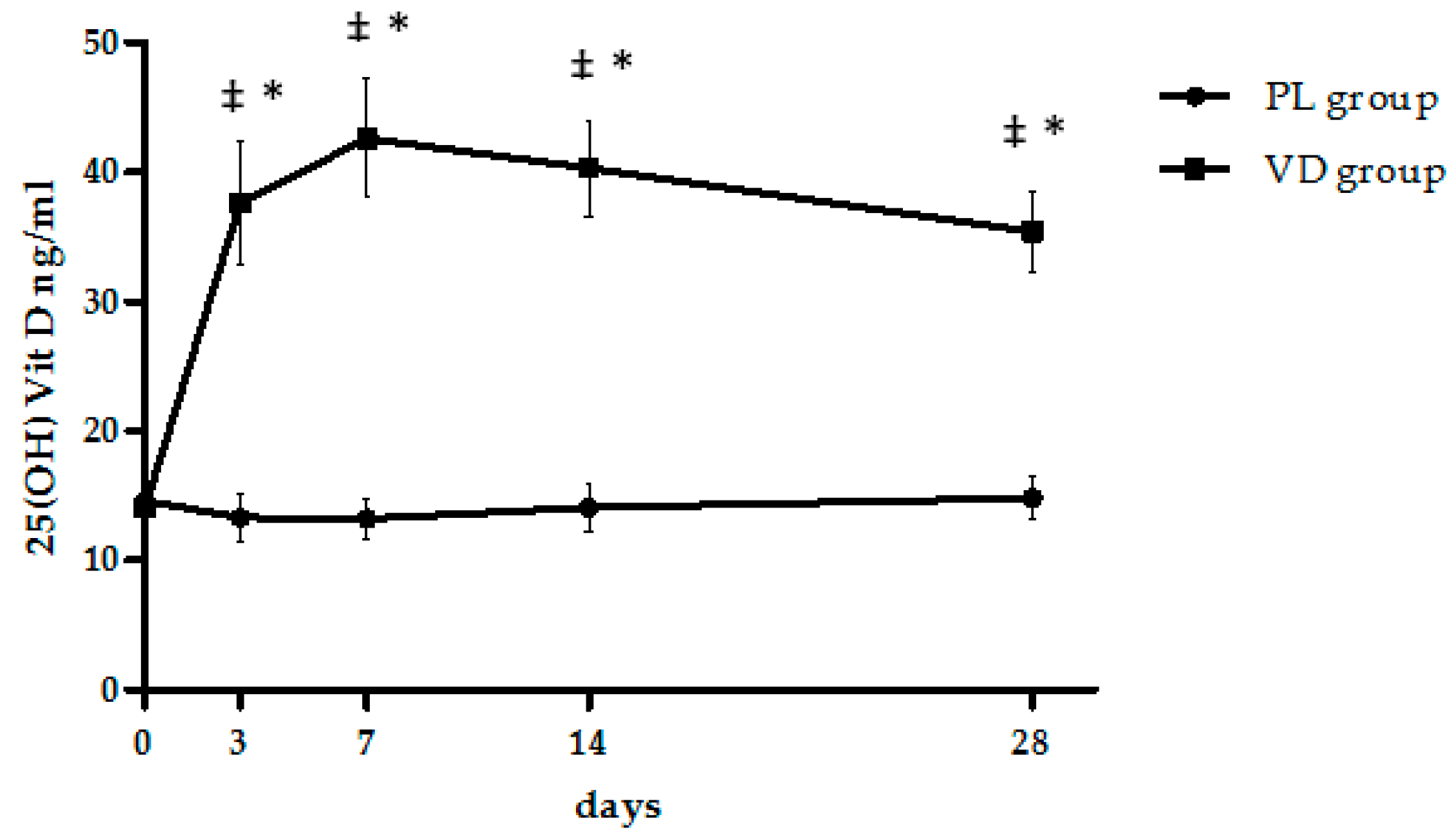
| 25(OH)Vit D (ng/mL) | 14.2 ± 1.9 | 35.0 ± 3.2 #,** | 191.0 ± 39.6 | 14.5 ± 1.9 | 14.8 ± 1.6 | 7.2 ± 6.3 | <0.0001 | <0.0001 | <0.0001 |
| Total adiponectin (µg/mL) | 3.6 ± 0.5 | 3.5 ± 0.4 | 0.2 ± 5.9 | 4.0 ± 1.1 | 3.9 ± 1.0 | 1.9 ± 9.1 | 0.39 | 0.75 | 0.89 |
| Leptin (ng/mL) | 44.1 ± 7.2 | 31.6 ± 5.9 * | −27.3 ± 5.4 | 41.1 ± 4.6 | 9.8 ± 4.1 ** | −29.9 ± 4.0 | <0.0001 | 0.75 | 0.72 |
| Leptin/adiponectin | 15.9 ± 4.3 | 10.5 ± 2.7 * | −23.6 ± 5.4 | 14.8 ± 2.5 | 10.3 ± 1.6 | −21.2 ± 12.9 | <0.01 | 0.86 | 0.73 |
| Leptin/HMW−A | 49.1 ± 12.0 | 24.7 ± 4.2 * | −26.8 ± 10.6 | 37.4 ± 7.4 | 35.5 ± 9.9 | −6.9 ± 12.8 | <0.05 | 0.96 | <0.05 |
| Vitamin D Group (n = 12) | Placebo Group (n = 12) | p Value | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Baseline | 14 Days | 28 Days | Baseline | 14 Days | 28 Days | Time | Treatment | Time × Treatment | |
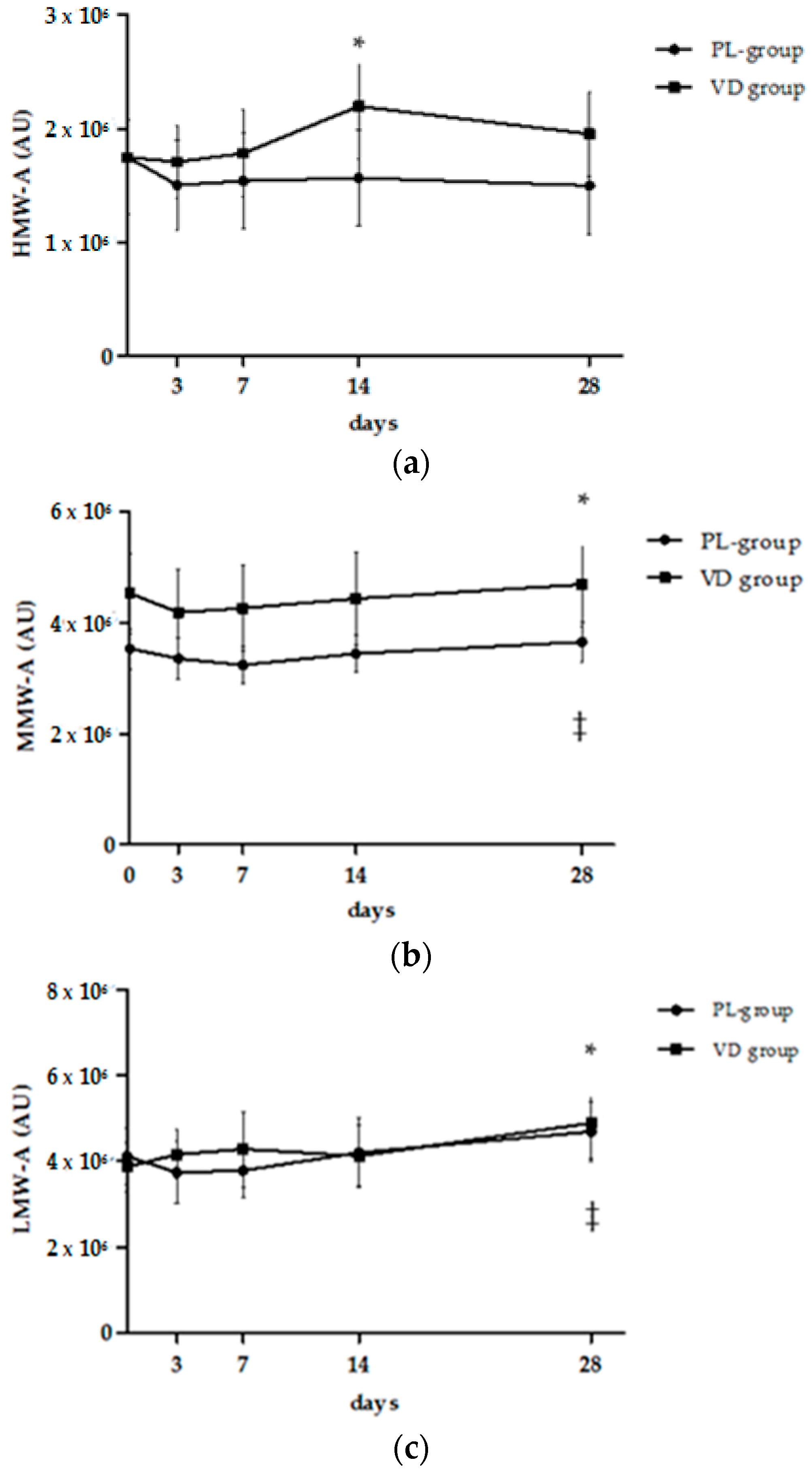
| Total Adiponectin (µg/mL) | 3.6 ± 0.5 | 3.7 ± 0.4 | 3.5 ± 0.4 | 4.0 ± 1.1 | 3.9 ± 0.9 | 3.8 ± 1.0 | 0.94 | 0.51 | 0.99 |
| HMW-A (µg/mL) | 1.6 ± 0.4 | 1.6 ± 0.2 | 1.5 ± 0.2 | 2.1 ± 0.8 | 1.9 ± 0.5 | 1.8 ± 0.7 | 0.92 | 0.38 | 0.97 |
| MMW-A (µg/mL) | 0.7 ± 0.1 | 0.6 ± 0.1 | 0.7 ± 0.1 | 0.5 ± 0.2 | 0.4 ± 0.2 | 0.8 ± 0.2 | 0.63 | 0.93 | 0.87 |
| LMW-A (µg/mL) | 1.5 ± 0.2 | 1.5 ± 0.2 | 1.4 ± 0.2 | 1.5 ± 0.3 | 1.8 ± 0.4 | 1.5 ± 0.4 | 0.64 | 0.51 | 0.74 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mai, S.; Walker, G.E.; Vietti, R.; Cattaldo, S.; Mele, C.; Priano, L.; Mauro, A.; Bona, G.; Aimaretti, G.; Scacchi, M.; et al. Acute Vitamin D3 Supplementation in Severe Obesity: Evaluation of Multimeric Adiponectin. Nutrients 2017, 9, 459. https://doi.org/10.3390/nu9050459
Mai S, Walker GE, Vietti R, Cattaldo S, Mele C, Priano L, Mauro A, Bona G, Aimaretti G, Scacchi M, et al. Acute Vitamin D3 Supplementation in Severe Obesity: Evaluation of Multimeric Adiponectin. Nutrients. 2017; 9(5):459. https://doi.org/10.3390/nu9050459
Chicago/Turabian StyleMai, Stefania, Gillian E. Walker, Roberta Vietti, Stefania Cattaldo, Chiara Mele, Lorenzo Priano, Alessandro Mauro, Gianni Bona, Gianluca Aimaretti, Massimo Scacchi, and et al. 2017. "Acute Vitamin D3 Supplementation in Severe Obesity: Evaluation of Multimeric Adiponectin" Nutrients 9, no. 5: 459. https://doi.org/10.3390/nu9050459
APA StyleMai, S., Walker, G. E., Vietti, R., Cattaldo, S., Mele, C., Priano, L., Mauro, A., Bona, G., Aimaretti, G., Scacchi, M., & Marzullo, P. (2017). Acute Vitamin D3 Supplementation in Severe Obesity: Evaluation of Multimeric Adiponectin. Nutrients, 9(5), 459. https://doi.org/10.3390/nu9050459






