Supplementation with a Polyphenol-Rich Extract, PerfLoad®, Improves Physical Performance during High-Intensity Exercise: A Randomized, Double Blind, Crossover Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Study Population
2.3. Test Supplement
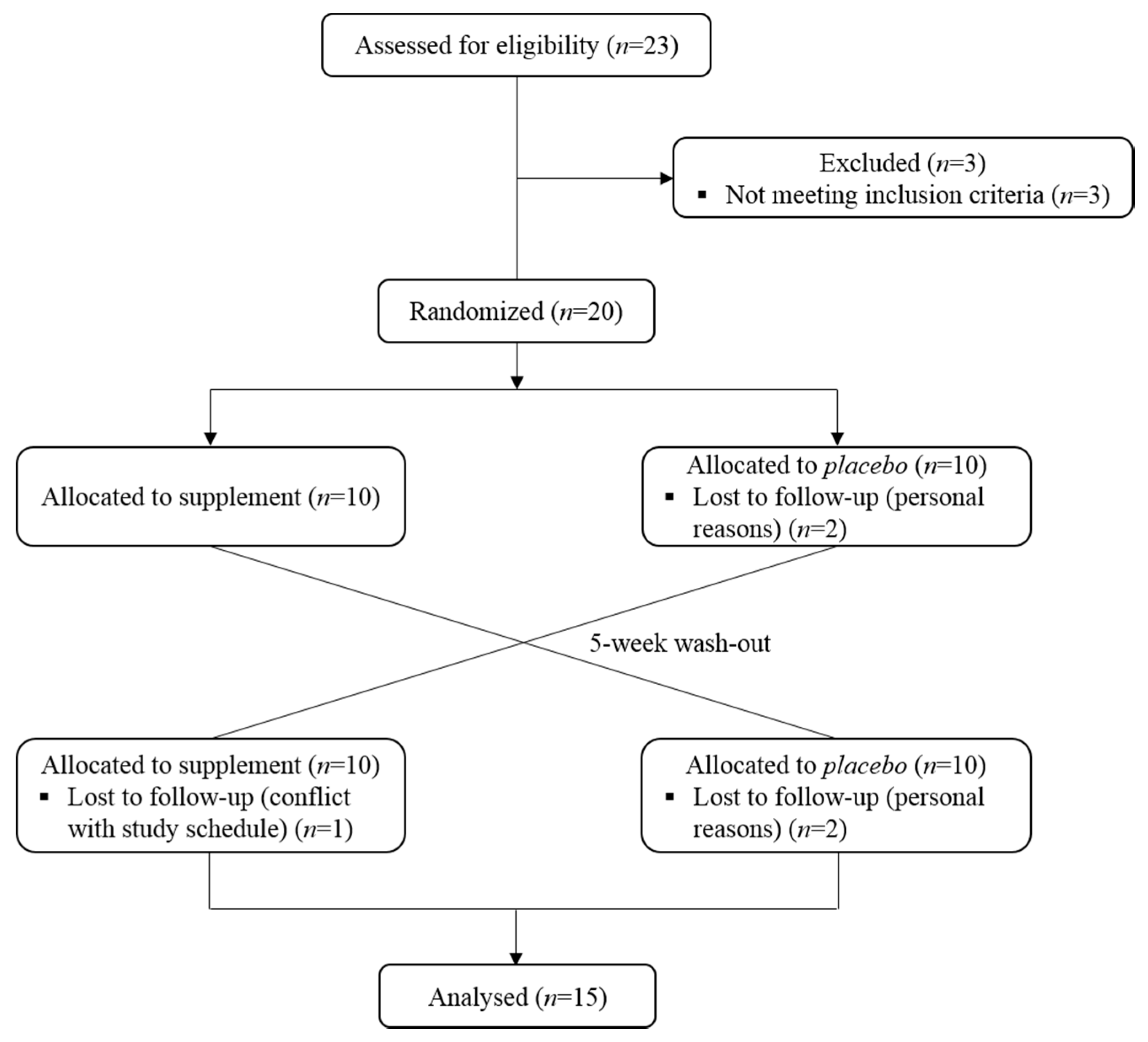
2.4. Study Design
2.5. Exercise Protocol
2.6. Primary Outcome
2.7. Secondary Outcome
2.8. Statistical Analysis
3. Results
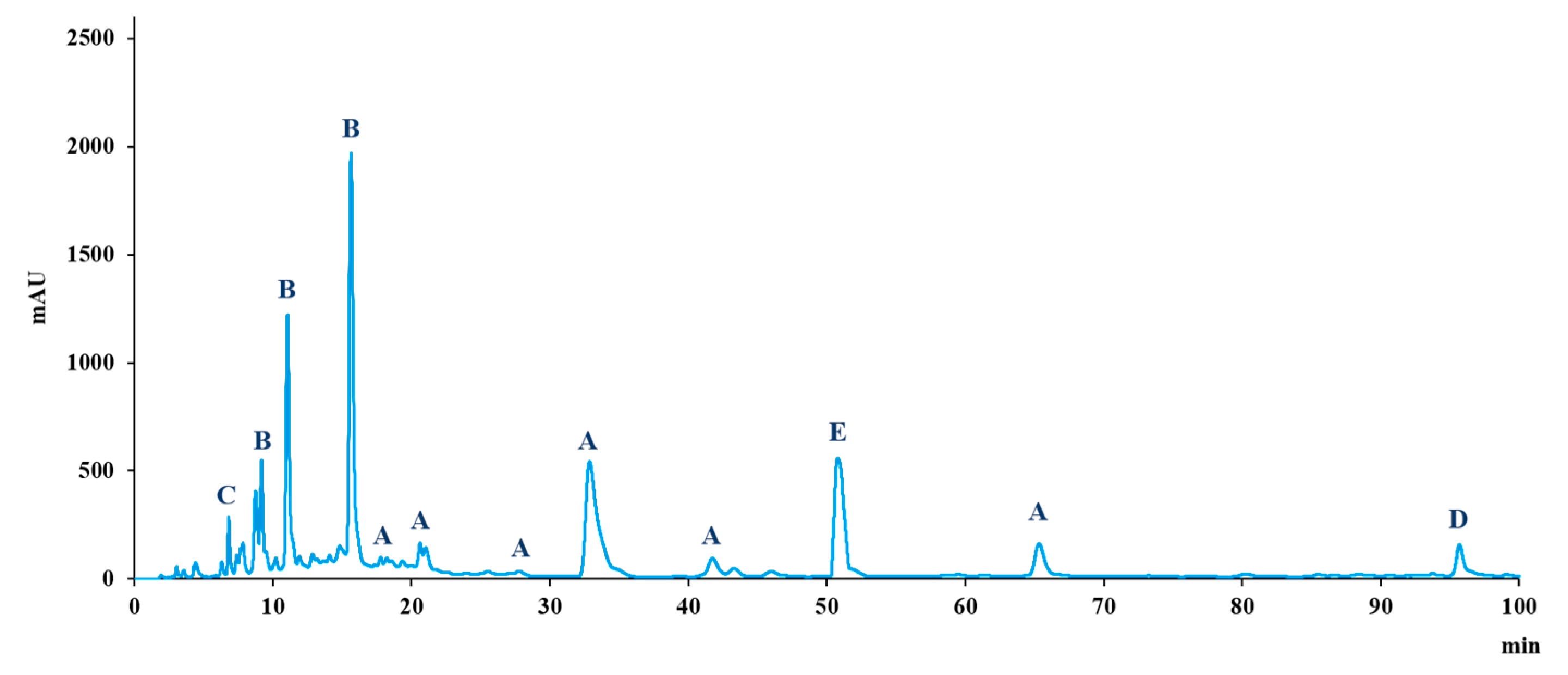
3.1. Characterisation of the Phenolic Profile of the Supplement
3.2. Participant Characteristics
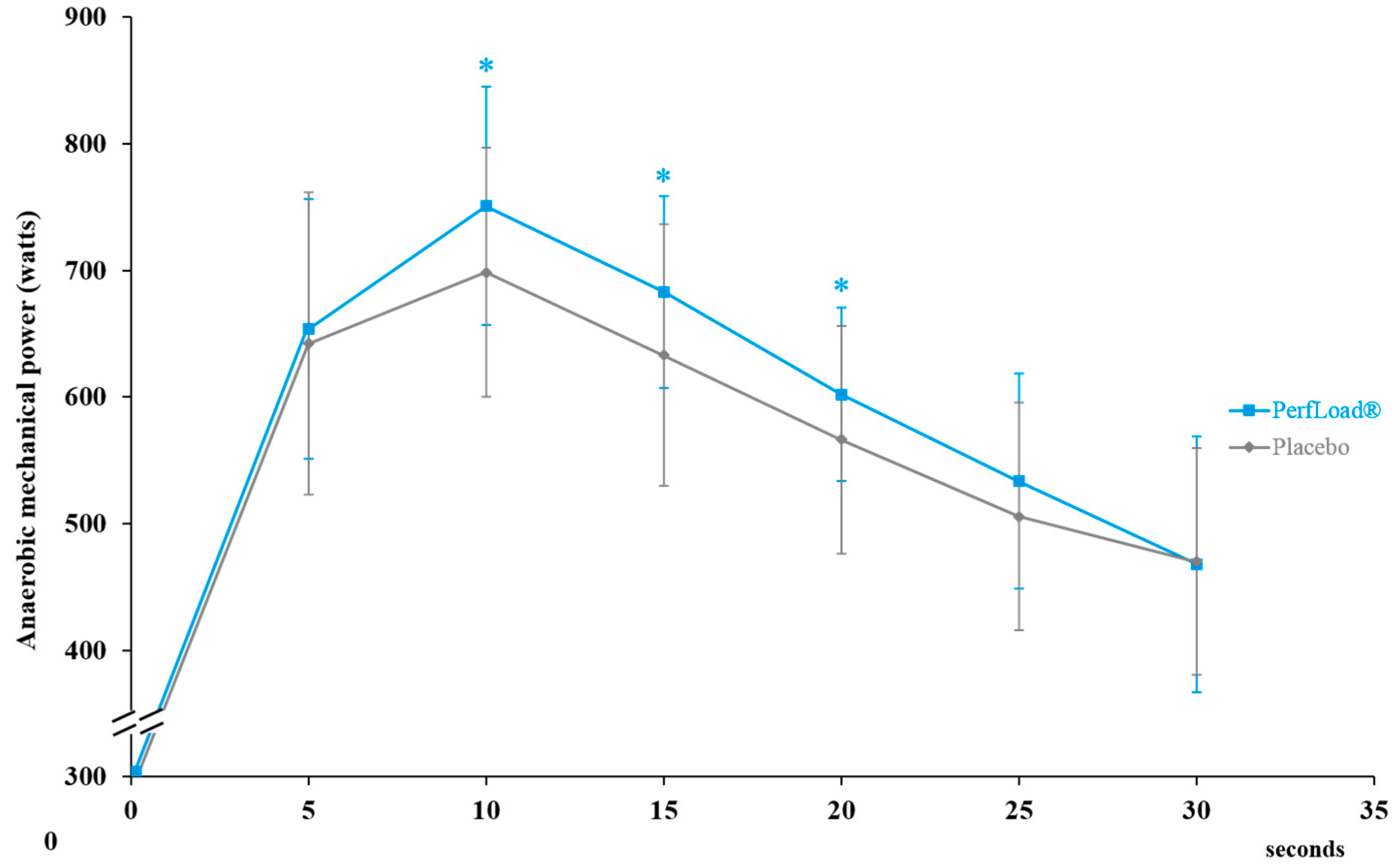
3.3. Power Production
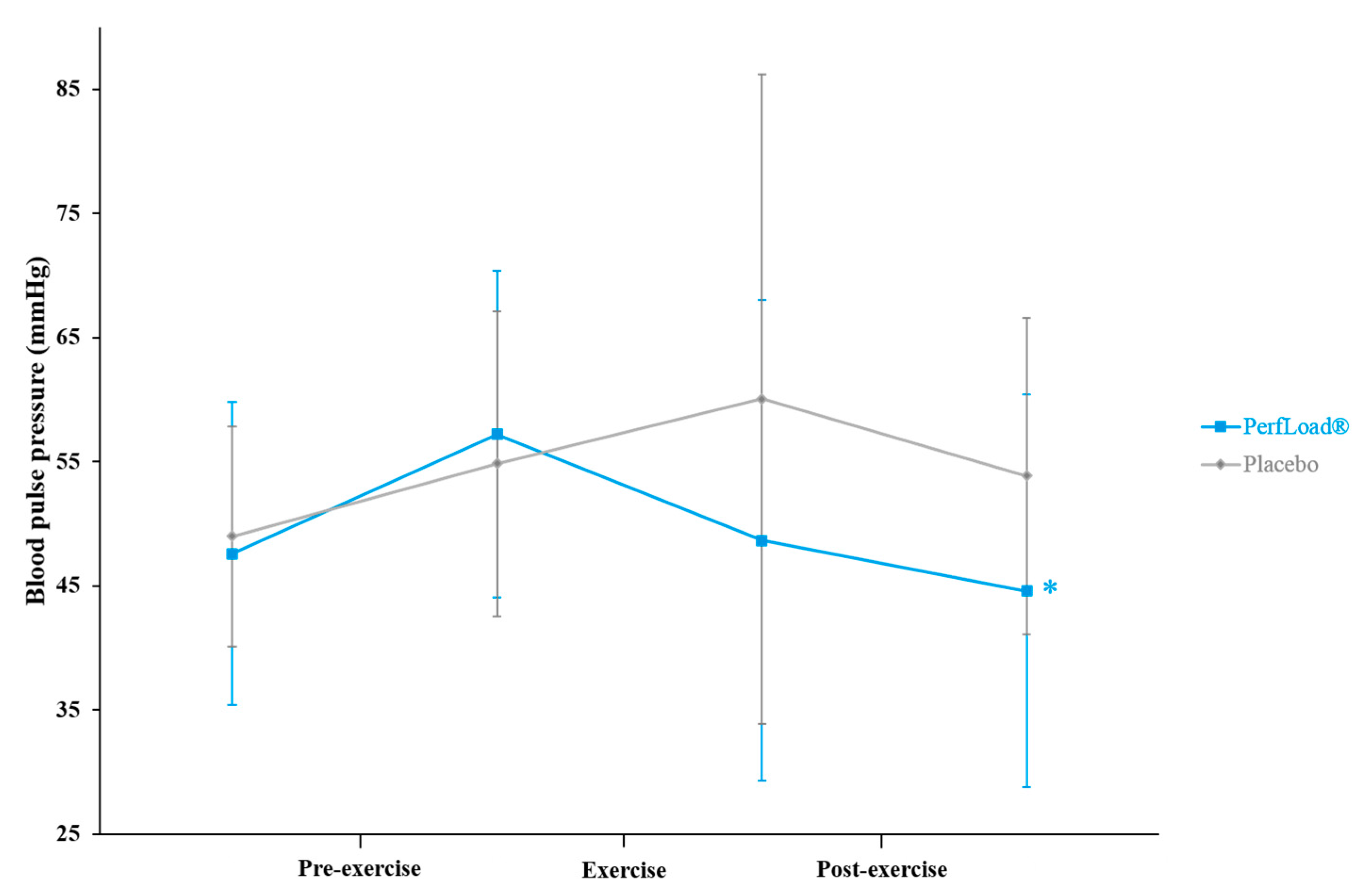
3.4. Blood Pulse Pressure
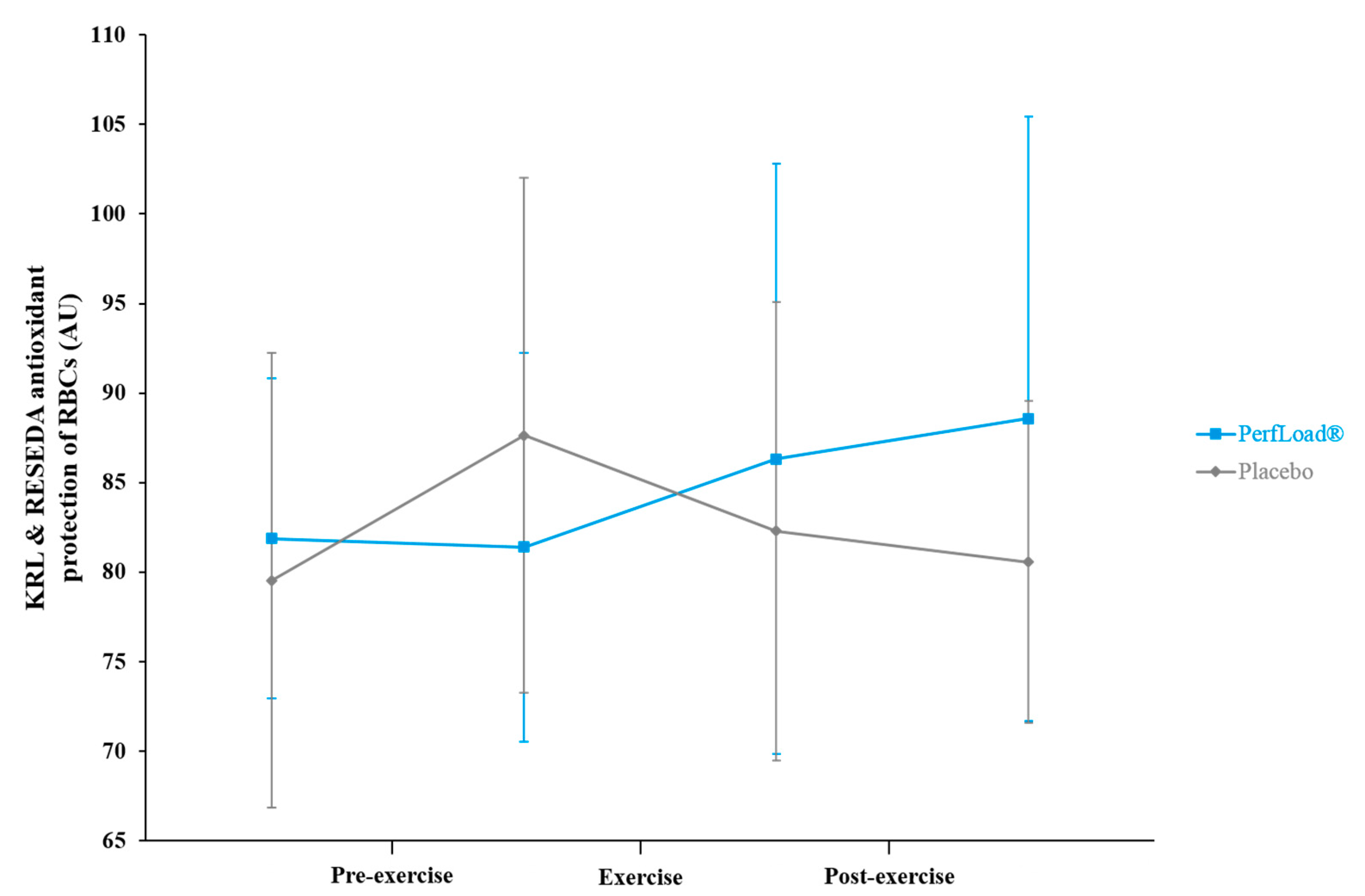
3.5. Protection of Red Blood Cells
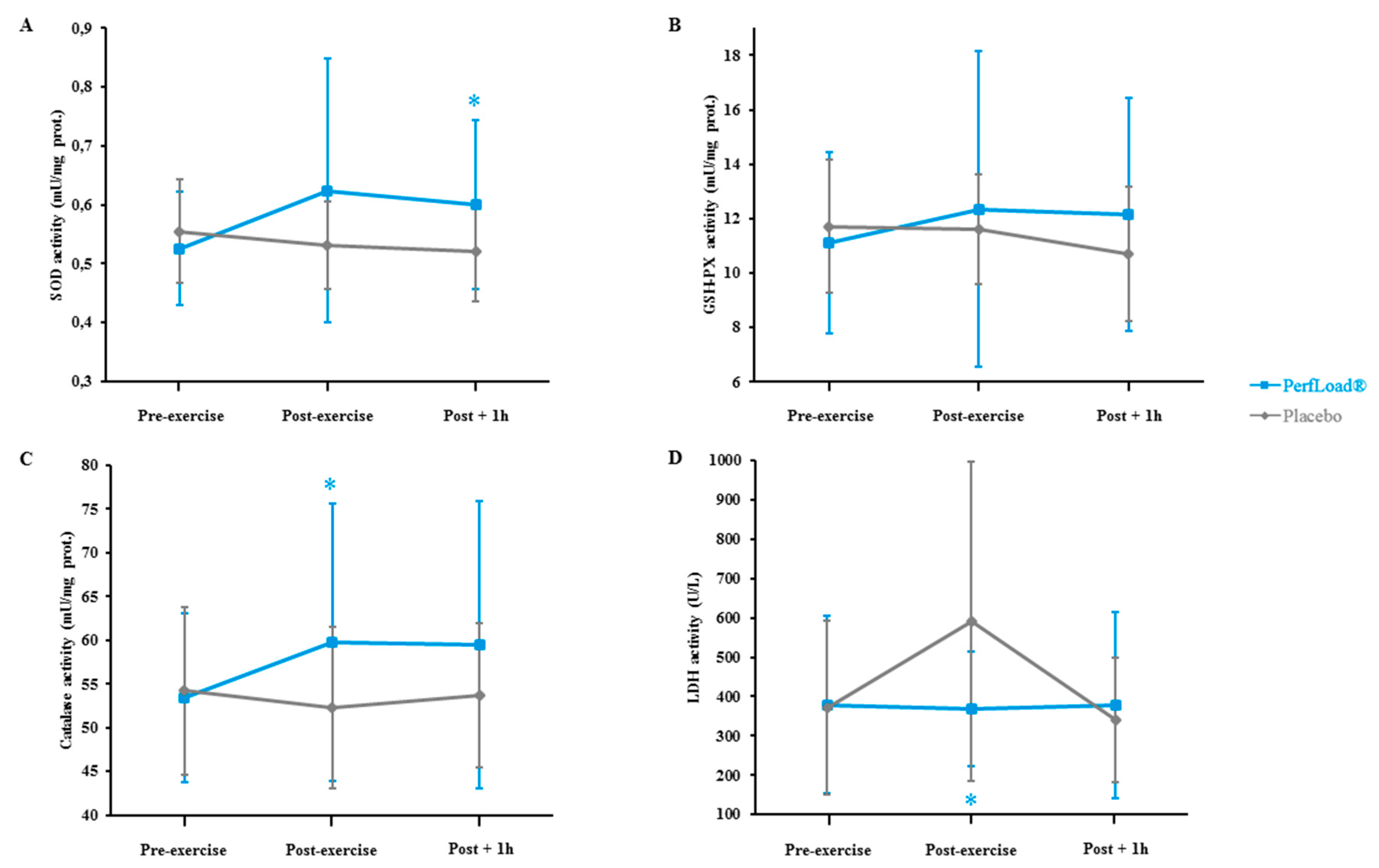
3.6. Antioxidant Enzymes and LDH
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Hall, J.E.; Guyton, A.C. Textbook of Medical Physiology, 12th ed.; Saunders/Elsevier: Philadelphia, PA, USA, 2011. [Google Scholar]
- Gastin, P.B. Energy system interaction and relative contribution during maximal exercise. Sports Med. 2001, 31, 725–741. [Google Scholar] [CrossRef] [PubMed]
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Fuel choice during exercise is determined by intensity and duration of activity. In Biochemistry, 5th ed.; W.H. Freeman Publisher: New York, NY, USA, 2002. [Google Scholar]
- Ferreira de Souza, C.; Fernandes, L.C. Production of reactive oxygen species during the aerobic and anaerobic exercise. Rev. Bras. Cinean. Desemp. Hum. 2006, 8, 102–109. [Google Scholar]
- Baron, B.; Noakes, T.D.; Dekerle, J.; Moullan, F.; Robin, S.; Matran, R.; Pelayo, P. Why does exercise terminate at the maximal lactate steady state intensity? Br. J. Sports Med. 2008, 42, 528–533. [Google Scholar] [CrossRef] [PubMed]
- Banister, E.W.; Cameron, B.J.C. Exercise-induced hyperammonemia: Peripheral and central effects. Int. J. Sports Med. 1990, 2, S129–S142. [Google Scholar] [CrossRef] [PubMed]
- Brouns, F.; Beckers, E.; Wagenmakers, A.J.; Saris, W.H. Ammonia accumulation during highly intensive long-lasting cycling: Individual observations. Int. J. Sports Med. 1990, 2, S78–S84. [Google Scholar] [CrossRef] [PubMed]
- Miramonti, A.A.; Stout, J.R.; Fukuda, D.H.; Robinson, E.H.; Wang, R.; La Monica, M.B.; Hoffman, J.R. Effects of 4 weeks of high-intensity interval training and ß-hydroxy-ß-methylbutyric free acid supplementation on the onset of neuromuscular fatigue. J. Strength Cond. Res. 2016, 30, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Buchheit, M.; Laursen, P.B. High-intensity interval training, solutions to the programming puzzle. Part II: Anaerobic energy, neuromuscular load and practical applications. Sports Med. 2013, 43, 927–954. [Google Scholar] [CrossRef] [PubMed]
- Fisher-Wellman, K.; Bloomer, R.J. Acute exercise and oxidative stress: A 30 year history. Dyn. Med. 2009, 8, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Tarnopolsky, M.A. Caffeine and endurance performance. Sports Med. 1994, 18, 109–125. [Google Scholar] [CrossRef] [PubMed]
- Van der Beek, E.J. Vitamin supplementation and physical exercise performance. J. Sports Sci. 1991, 9, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Chwalbiñska-Moneta, J. Effect of creatine supplementation on aerobic performance and anaerobic capacity in elite rowers in the course of endurance training. Int. J. Sport Nutr. Exerc. Metab. 2003, 13, 173–183. [Google Scholar] [CrossRef] [PubMed]
- De Nigris, F.; Williams-Ignarro, S.; Sica, V.; Lerman, L.O.; D’Armiento, F.P.; Byrns, R.E.; Casamassimi, A.; Carpentiero, D.; Schiano, C.; Sumi, D.; et al. Effects of a pomegranate fruit extract rich in punicalagin on oxidation-sensitive genes and eNOS activity at sites of perturbed shear stress and atherogenesis. Cardiovasc. Res. 2007, 73, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Ladurner, A.; Schachner, D.; Schueller, K.; Pignitter, M.; Heiss, M.H.; Somoza, V.; Dirsch, V.M. Impact of trans-resveratrol-sulfates and -glucuronides on endothelial nitric oxide synthase activity, nitric oxide release and intracellular reactive oxygen species. Molecules 2014, 19, 16724–16736. [Google Scholar] [CrossRef] [PubMed]
- Grau, M.; Bölck, B.; Bizjak, D.A.; Stabenow, C.J.A.; Bloch, W. The red-vine-leaf extract AS195 increases nitric oxide synthase–dependent nitric oxide generation and decreases oxidative stress in endothelial and red blood cells. Pharmacol. Res. Perspect. 2016, 4, e00213. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, M.; Wessler, S.; Follmann, E.; Michaelis, W.; Dusterhoft, T.; Baumann, G.; Stangl, K.; Stangl, V. A constituent of green tea, epigallocatechin-3-gallate, activates endothelial nitric oxide synthase by a phosphatidylinositol-3-OH-kinase-, cAMP-dependent protein kinase-, and Akt-dependent pathway and leads to endothelial-dependent vasorelaxation. J. Biol. Chem. 2004, 279, 6190–6195. [Google Scholar] [CrossRef] [PubMed]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar]
- Vijayananthan, A.; Nawawi, O. The importance of good clinical practice guidelines and its role in clinical trials. Biomed. Imaging Interv. J. 2008, 4, e5. [Google Scholar] [CrossRef] [PubMed]
- Inbar, O.; Bar-Or, O.; Skinners, J.S. The Wingate Anaerobic Test; Human Kinetics: Champaign, IL, USA, 1996. [Google Scholar]
- Gül, I.; Gökbel, H.; Belviranli, M.; Okudan, N.; Büyükbas, S.; Başarali, K. Oxidative stress and antioxidant defense in plasma after repeated bouts of supramaximal exercise: The effect of coenzyme Q10. J. Sports Med. Phys. Fitness 2011, 51, 305–312. [Google Scholar] [PubMed]
- Zouhal, H.; Rannou, F.; Gratas-Delamarche, A.; Monnier, M.; Bentue-Ferrer, D.; Delamarche, P. Adrenal medulla responsiveness to the sympathetic nervous activity in sprinters and untrained subjects during a supramaximal exercise. Int. J. Sports Med. 1998, 19, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Blache, D.; Prost, M. Free radical attack: Biological test for human resistance capability. In Proceedings of the IX College Park Colloquium on Chemical Evolution: A Lunar-Based Chemical Analysis Laboratory (LBCAL 1989); Ponnamperuma, C., Gehrke, C.W., Eds.; NASA: Washington, DC, USA, 1992; pp. 82–98. [Google Scholar]
- Lesgards, J.F.; Durand, P.; Lassare, M.; Stocker, P.; Lesgards, G.; Lanteaume, A.; Prost, M.; Lehucher-Michel, M.P. Assessment of lifestyle effects on the overall antioxidant capacity of healthy subjects. Environ. Health Perspect. 2002, 110, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, J.; Kawaura, T.; Miyazaki, T.; Prost, M.; Prost, E.; Watanabe, M.; Quentin-Leclercq, J. Test for antioxidant ability by scavenging long-lived mutagenic radicals in mammalian cells and by blood test with intentional radicals: An application of gallic acid. Radiat. Phys. Chem. 2003, 66, 17–25. [Google Scholar] [CrossRef]
- Stocker, P.; Lesgards, J.F.; Vidal, N.; Chalier, F.; Prost, M. ESR study of a biological assay on whole blood. Antioxidant efficiency of various vitamins. Biochim. Biophys. Acta 2003, 1621, 1–8. [Google Scholar] [CrossRef]
- Caspar-Bauguil, S.; Maestre, N.; Segafredo, C.; Galinier, A.; Garcia, J.; Prost, M.; Périquet, B.; Pénicaud, L.; Salvayre, R.; Casteilla, L. Evaluation of whole antioxidant defenses of human mononuclear cells by a new in vitro biological test: Lack of correlation between erythrocyte and mononuclear cell resistance to oxidative stress. Clin. Biochem. 2009, 42, 510–514. [Google Scholar] [CrossRef] [PubMed]
- Rossi, R.; Pastorelli, G.; Corino, C. Application of KRL test to assess total antioxidant activity in pigs: Sensitivity to dietary antioxidants. Res. Vet. Sci. 2013, 94, 372–377. [Google Scholar] [CrossRef] [PubMed]
- Prost, M. Utilisation de Générateur de Radicaux Libres dans le Domaine des Dosages Biologiques. French Patent 2,642,526, 27 January 1989. [Google Scholar]
- Prost, M. Process for the Determination by Means of Free Radicals of the Antioxidant Properties of a Living Organism or Potentially Aggressive Agents. U.S. Patent 5,135,850 A, 27 January 1989. [Google Scholar]
- Prost, M. Method for Determining the Antiradical Defense Potential and Use Thereof, in Particular in Veterinary and Human Preventive Therapeutics. U.S. Patent 20060234329 A1, 22 October 2003. [Google Scholar]
- Driss, T.; Vandewalle, H. The measurement of maximal (anaerobic) power output on a cycle ergometer: A critical review. BioMed Res. Int. 2013, 2013, 589361. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.A.; Sharpe, G.R.; Brown, P.I. Inspiratory muscle training improves cycling time-trial performance and anaerobic work capacity but not critical power. Eur. J. Appl. Physiol. 2007, 101, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Cooper, R.; Naclerio, F.; Allgrove, J.; Jimenez, A. Creatine supplementation with specific view to exercise/sports performance: An update. J. Int. Soc. Sports Nutr. 2012, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Collomp, K.; Ahmaidi, S.; Audran, M.; Chanal, J.L.; Préfaut, C. Effects of caffeine ingestion on performance and anaerobic metabolism during the Wingate Test. Int. J. Sports Med. 1991, 12, 439–443. [Google Scholar] [CrossRef] [PubMed]
- Tallis, J.; Duncan, M.J.; Leddington Wright, S.; Eyre, E.L.J.; Bryant, E.; Langdon, D.; James, R.S. Assessment of the ergogenic effect of caffeine supplementation on mood, anticipation timing, and muscular strength in older adults. Physiol. Rep. 2013, 1, e00072. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, A.B.; Randell, R.K.; Jeukendrup, A.E. The metabolic and performance effects of caffeine compared to coffee during endurance exercise. PLoS ONE 2013, 8, e59561. [Google Scholar] [CrossRef] [PubMed]
- Strait, J.B.; Lakatta, E.G. Aging-associated cardiovascular changes and their relationship to heart failure. Heart Fail. Clin. 2012, 8, 143–164. [Google Scholar] [CrossRef] [PubMed]
- Kitzman, D.W.; Groban, L. Exercise intolerance. Heart Fail. Clin. 2008, 4, 99–115. [Google Scholar] [CrossRef] [PubMed]
- Keshavarz-Motamed, Z.; Garcia, J.; Gaillard, E.; Capoulade, R.; Le Ven, F.; Cloutier, G.; Kadem, L.; Pibarot, P. Non-invasive determination of left ventricular workload in patients with aortic stenosis using magnetic resonance imaging and doppler echocardiography. PLoS ONE 2014, 9, e86793. [Google Scholar]
- Vita, J.A. Polyphenols and cardiovascular disease: Effects on endothelial and platelet function. Am. J. Clin. Nutr. 2005, 81, 292S–297S. [Google Scholar] [PubMed]
- Ras, R.T.; Zock, P.L.; Draijer, R. Tea consumption enhances endothelial-dependent vasodilation; a meta-analysis. PLoS ONE 2011, 6, e16974. [Google Scholar] [CrossRef] [PubMed]
- Li, S.H.; Tian, H.B.; Zhao, H.J.; Chen, L.H.; Cui, L.Q. The acute effects of grape polyphenols supplementation on endothelial function in adults: Meta-analyses of controlled trials. PLoS ONE 2013, 8, e69818. [Google Scholar] [CrossRef] [PubMed]
- Kelishadi, R.; Gidding, S.S.; Hashemi, M.; Hashemipour, M.; Zakerameli, A.; Poursafa, P. Acute and long term effects of grape and pomegranate juice consumption on endothelial dysfunction in pediatric metabolic syndrome. J. Res. Med. Sci. 2011, 16, 245–253. [Google Scholar] [PubMed]
- Grassi, D.; Desideri, G.; Di Giosia, P.; De Feo, M.; Fellini, E.; Cheli, P.; Ferri, L.; Ferri, C. Tea, flavonoids, and cardiovascular health: Endothelial protection. Am. J. Clin. Nutr. 2013, 98, 1660S–1666S. [Google Scholar] [CrossRef] [PubMed]
- Morillas-Ruiz, J.; Zafrilla, P.; Almar, M.; Cuevas, M.J.; López, F.J.; Abellán, P.; Villegas, J.A.; González-Gallego, J. The effects of an antioxidant-supplemented beverage on exercise-induced oxidative stress: Results from a placebo-controlled double-blind study in cyclists. Eur. J. Appl. Physiol. 2005, 95, 543–549. [Google Scholar] [CrossRef] [PubMed]
- Powers, S.K.; De Ruisseau, K.C.; Quindry, J.; Hamilton, K.L. Dietary antioxidants and exercise. J. Sports Sci. 2004, 22, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Tauler, P.; Aguiló, A.; Fuentespina, G.E. Response of blood cell antioxidant enzyme defenses to antioxidant diet supplementation and to intense exercise. Eur. J. Nutr. 2006, 45, 187–195. [Google Scholar] [CrossRef] [PubMed]
- MacRae, H.S.H.; Mefferd, K.M. Dietary antioxidant supplementation combined with quercetin improves cycling time trial performance. Int. J. Sport Nutr. Exerc. Met. 2006, 16, 405–419. [Google Scholar] [CrossRef]
- Bloomer, R.J.; Goldfarb, A.H.; McKenzie, M.J. Oxidative stress response to aerobic exercise: Comparison of antioxidant supplements. Med. Sci. Sports Exerc. 2006, 38, 1098–1105. [Google Scholar] [CrossRef] [PubMed]
| Bioactive | Dose |
|---|---|
| Total polyphenols | 290 mg |
| Bioflavonoids | 170 mg |
| Punicalagins | 100 mg |
| Trans-resveratrol | 20 mg |
| Caffeine | 120 mg |
| Characteristics | All Volunteers |
|---|---|
| N | 15 |
| Age (years, mean ± SD) | 22.2 ± 2.2 |
| Height (cm, mean ± SD) | 178.1 ± 4.3 |
| Weight (kg, mean ± SD) | 75.1 ± 6.6 |
| BMI (kg/m2, mean ± SD) | 22.3 ± 1.9 |
| Variables | Placebo Group | PerfLoad® Group | p Value |
|---|---|---|---|
| Peak power (W, mean ± SD) | 734 ± 104.7 | 761 ± 92.1 * | 0.048 |
| Average power (W, mean ± SD) | 586 ± 78.9 | 615 ± 75.4 * | 0.025 |
| Fatigue index (%, mean ± SD) | 38 ± 12.9 | 39 ± 10.4 | 0.395 |
| Heart rate (bpm, mean ± SD) | 129 ± 15.1 | 132 ± 25.9 | 0.344 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cases, J.; Romain, C.; Marín-Pagán, C.; Chung, L.H.; Rubio-Pérez, J.M.; Laurent, C.; Gaillet, S.; Prost-Camus, E.; Prost, M.; Alcaraz, P.E. Supplementation with a Polyphenol-Rich Extract, PerfLoad®, Improves Physical Performance during High-Intensity Exercise: A Randomized, Double Blind, Crossover Trial. Nutrients 2017, 9, 421. https://doi.org/10.3390/nu9040421
Cases J, Romain C, Marín-Pagán C, Chung LH, Rubio-Pérez JM, Laurent C, Gaillet S, Prost-Camus E, Prost M, Alcaraz PE. Supplementation with a Polyphenol-Rich Extract, PerfLoad®, Improves Physical Performance during High-Intensity Exercise: A Randomized, Double Blind, Crossover Trial. Nutrients. 2017; 9(4):421. https://doi.org/10.3390/nu9040421
Chicago/Turabian StyleCases, Julien, Cindy Romain, Cristian Marín-Pagán, Linda H. Chung, José Miguel Rubio-Pérez, Caroline Laurent, Sylvie Gaillet, Emmanuelle Prost-Camus, Michel Prost, and Pedro E. Alcaraz. 2017. "Supplementation with a Polyphenol-Rich Extract, PerfLoad®, Improves Physical Performance during High-Intensity Exercise: A Randomized, Double Blind, Crossover Trial" Nutrients 9, no. 4: 421. https://doi.org/10.3390/nu9040421
APA StyleCases, J., Romain, C., Marín-Pagán, C., Chung, L. H., Rubio-Pérez, J. M., Laurent, C., Gaillet, S., Prost-Camus, E., Prost, M., & Alcaraz, P. E. (2017). Supplementation with a Polyphenol-Rich Extract, PerfLoad®, Improves Physical Performance during High-Intensity Exercise: A Randomized, Double Blind, Crossover Trial. Nutrients, 9(4), 421. https://doi.org/10.3390/nu9040421






