Oleuropein Decreases Cyclooxygenase-2 and Interleukin-17 Expression and Attenuates Inflammatory Damage in Colonic Samples from Ulcerative Colitis Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients and Sampling
2.2. Ex Vivo Organ Culture
2.3. Extraction of Total Proteins
2.4. Western Blot Analysis
2.5. Measurement of IL-17 in the Culture Supernatants
2.6. Histology
2.7. Statistical Analysis
2.8. Ethical Considerations
3. Results
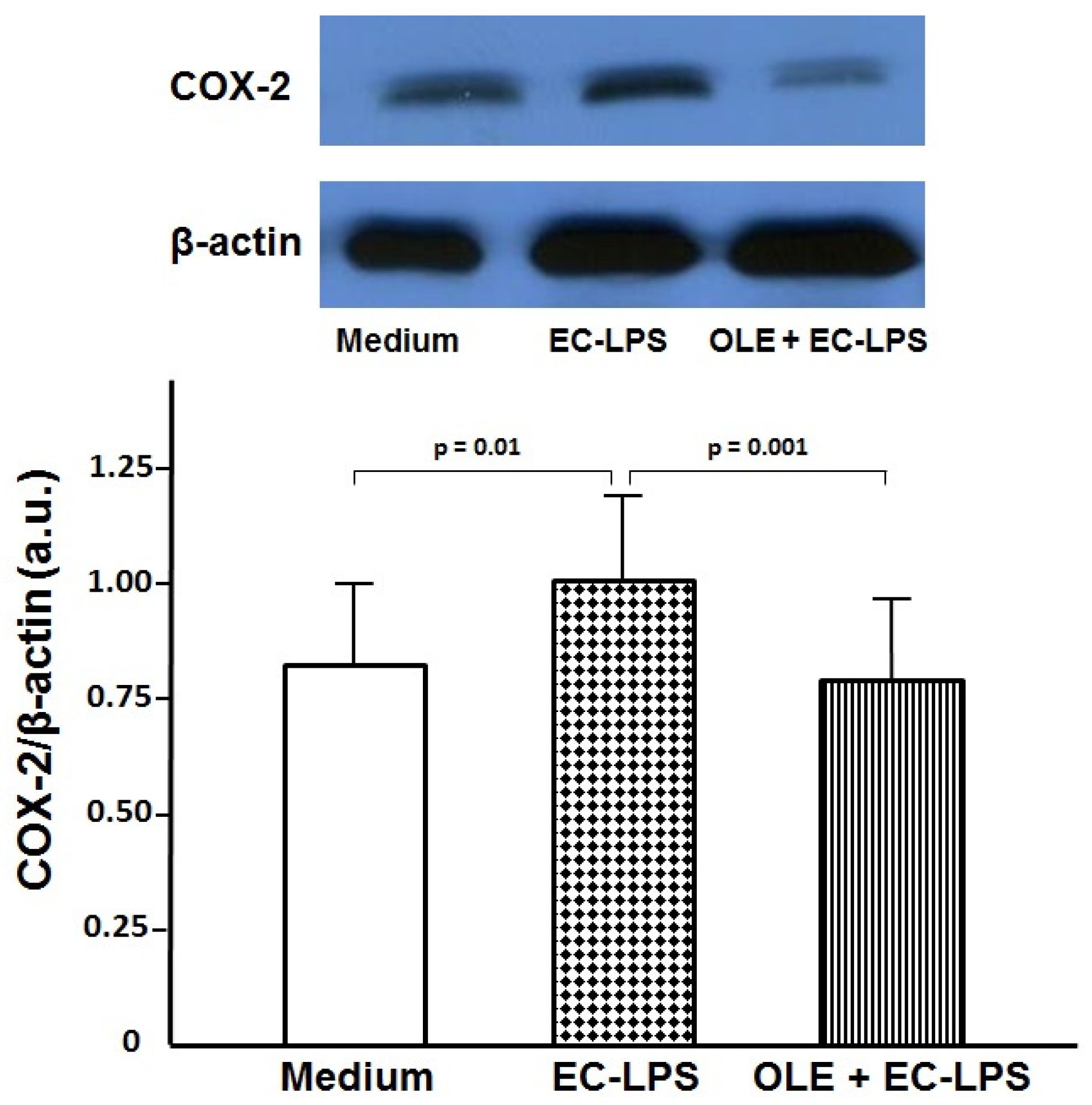
3.1. OLE Decreases COX-2 Expression in Colonic Biopsy Culture
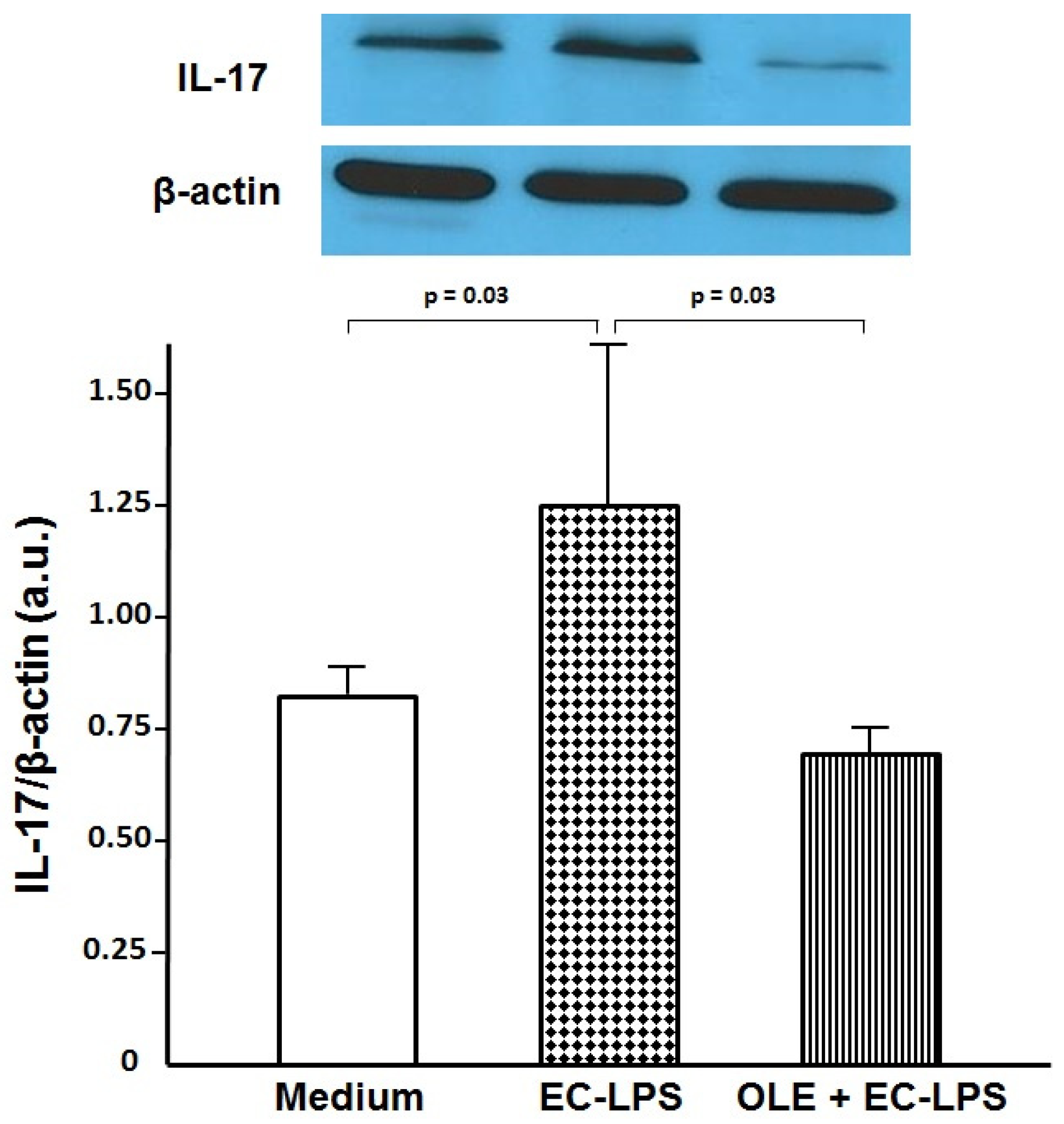
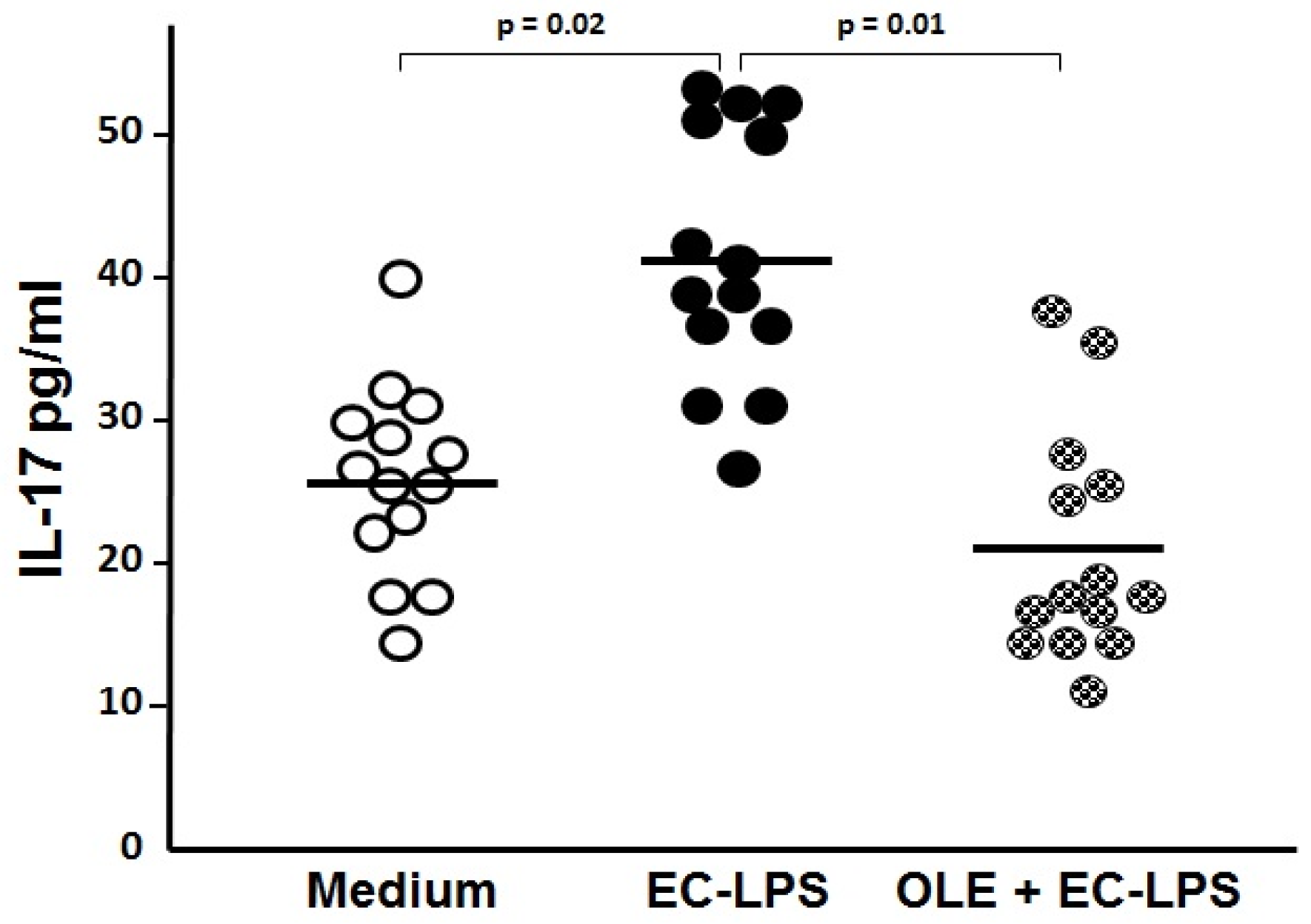
3.2. OLE Suppresses IL-17 in Both Mucosa and Supernatant of Colonic Biopsy Culture
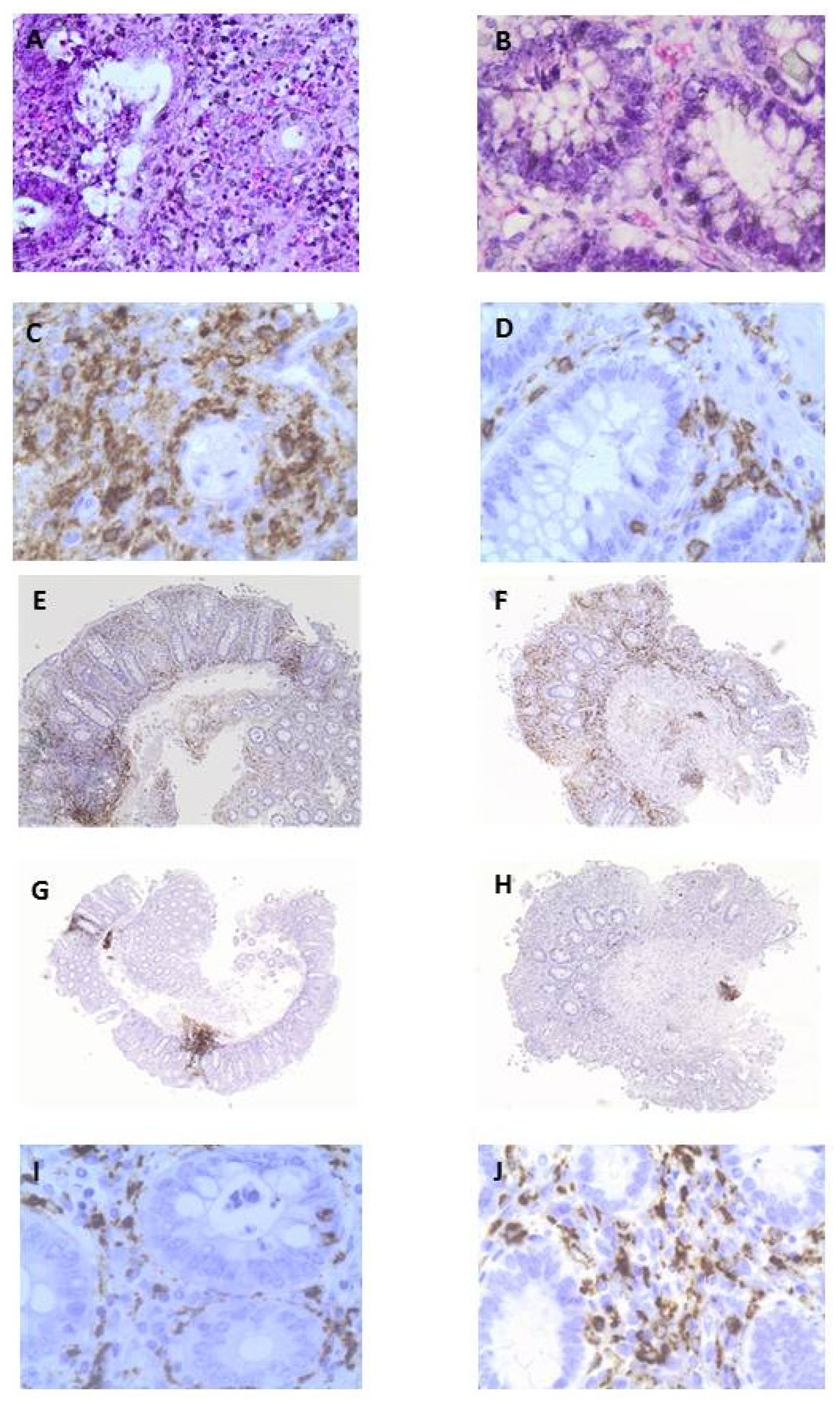
3.3. OLE Attenuates Inflammatory Damage in Colonic Biopsy Samples
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Danese, S.; Fiocchi, C. Ulcerative colitis. N. Engl. J. Med. 2011, 365, 1713–1725. [Google Scholar] [CrossRef] [PubMed]
- Knights, D.; Lassen, K.G.; Xavier, R.J. Advances in inflammatory bowel disease pathogenesis: Linking host genetics and the microbiome. Gut 2013, 62, 1505–1510. [Google Scholar] [CrossRef] [PubMed]
- Levesque, B.G.; Sandborn, W.J.; Ruel, J.; Fegan, B.G.; Sands, B.E.; Colombel, J.F. Converging goals of treatment of inflammatory bowel disease from clinical trials and practice. Gastroenterology 2015, 148, 37–51. [Google Scholar] [CrossRef] [PubMed]
- Park, K.T.; Bass, D. Inflammatory bowel disease-attributable costs and cost-effective strategies in the United States: A review. Inflamm. Bowel Dis. 2011, 17, 1603–1609. [Google Scholar] [CrossRef] [PubMed]
- Ben-Horin, S. Loss of response to anti-tumor necrosis factors: What is the next step? Dig. Dis. 2014, 32, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, L.; Krane, M.K.; Fichera, A. Inflammatory bowel disease surgery in the biologic era. World J. Gastrointest. Surg. 2016, 8, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Uranga, J.A.; López-Miranda, V.; Lombó, F.; Abalo, R. Food, nutrients and nutraceuticals affecting the course of inflammatory bowel disease. Pharmacol. Rep. 2016, 68, 816–826. [Google Scholar] [CrossRef] [PubMed]
- Vezza, T.; Rodríguez-Nogales, A.; Algieri, F.; Utrilla, M.P.; Rodriguez-Cabezas, M.E.; Galvez, J. Flavonoids in Inflammatory Bowel Disease: A Review. Nutrients 2016, 8, 211. [Google Scholar] [CrossRef] [PubMed]
- Martín-Peláez, S.; Castañer, O.; Solà, R.; Motilva, M.J.; Castell, M.; Pérez-Cano, F.J.; Fitó, M. Influence of Phenol-Enriched Olive Oils on Human Intestinal Immune Function. Nutrients 2016, 8, 213. [Google Scholar] [CrossRef] [PubMed]
- El, S.N.; Karakaya, S. Olive tree (Olea europaea) leaves: Potential beneficial effects on human health. Nutr. Rev. 2009, 67, 632–638. [Google Scholar] [CrossRef] [PubMed]
- Corona, G.; Tzounis, X.; Assunta Dessì, M.; Deiana, M.; Debnam, E.S.; Visioli, F.; Spencer, J.P. The fate of olive oil polyphenols in the gastrointestinal tract: Implications of gastric and colonic microflora-dependent biotransformation. Free Radic. Res. 2006, 40, 647–658. [Google Scholar] [CrossRef] [PubMed]
- De Bock, M.; Thorstensen, E.B.; Derraik, J.G.; Henderson, H.V.; Hofman, P.L.; Cutfield, W.S. Human absorption and metabolism of oleuropein and hydroxytyrosol ingested as olive (Olea europaea L.) leaf extract. Mol. Nutr. Food Res. 2013, 57, 2079–2085. [Google Scholar] [CrossRef] [PubMed]
- D’Angelo, S.; Manna, C.; Migliardi, V.; Mazzoni, O.; Morrica, P.; Capasso, G.; Pontoni, G.; Galletti, P.; Zappia, V. Pharmacokinetics and metabolism of hydroxytyrosol, a natural antioxidant from olive oil. Drug Metab. Dispos. 2001, 29, 1492–1498. [Google Scholar] [PubMed]
- Biel, S.; Mesa, M.D.; de la Torre, R.; Espejo, J.A.; Fernández-Navarro, J.R.; Fitó, M.; Sánchez-Rodriguez, E.; Rosa, C.; Marchal, R.; Alche, J.D.; et al. The NUTRAOLEOUM Study, a randomized controlled trial, for achieving nutritional added value for olive oils. BMC Complement. Altern. Med. 2016, 16, 404. [Google Scholar] [CrossRef] [PubMed]
- Giner, E.; Recio, M.-C.; Ríos, J.-L.; Giner, R.-M. Oleuropein protects against dextran sodium sulfate-induced chronic colitis in mice. J. Nat. Prod. 2013, 76, 1113–1120. [Google Scholar] [CrossRef] [PubMed]
- Giner, E.; Recio, M.C.; Ríos, J.L.; Cerda-Nicolas, J.M.; Giner, R.M. Chemopreventive effect of oleuropein in colitis-associated colorectal cancer in c57bl/6 mice. Mol. Nutr. Food Res. 2016, 60, 242–255. [Google Scholar] [CrossRef] [PubMed]
- Lennard-Jones, J.E. Classification of inflammatory bowel disease. Scand. J. Gastroenterol. 1989, 170, 16–19. [Google Scholar] [CrossRef]
- Schroeder, K.W.; Tremaine, W.J.; Ilstrup, D.M. Coated oral 5-aminosalicylic acid therapy for mildly to moderately active ulcerative colitis. A randomized study. N. Engl. J. Med. 1987, 317, 1625–1629. [Google Scholar] [CrossRef] [PubMed]
- Procopio, A.; Alcaro, S.; Nardi, M.; Oliverio, M.; Ortuso, F.; Sacchetta, P.; Pieragostino, D.; Sindona, G. Synthesis, biological evaluation, and molecular modeling of oleuropein and its semisynthetic derivatives as cyclooxygenase inhibitors. J. Agric. Food Chem. 2009, 57, 11161–11167. [Google Scholar] [CrossRef] [PubMed]
- Fabiani, R. Anti-cancer properties of olive oil secoiridoid phenols: A systematic review of in vivo studies. Food Funct. 2016, 7, 4145–4159. [Google Scholar] [CrossRef] [PubMed]
- Giner, E.; Andújar, I.; Recio, M.C.; Rios, J.L.; Cerdá-Nicolás, J.M.; Giner, R.M. Oleuropein ameliorates acute colitis in mice. J. Agric. Food Chem. 2011, 59, 12882–12892. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Fidalgo, S.; Cárdeno, A.; Sánchez-Hidalgo, M.; Aparicio-Soto, M.; de la Lastra, C.A. Dietary extra virgin olive oil polyphenols supplementation modulates DSS-induced chronic colitis in mice. J. Nutr. Biochem. 2013, 24, 1401–1413. [Google Scholar] [CrossRef] [PubMed]
- Zamuner, S.R.; Warrier, N.; Buret, A.G.; MacNaughton, W.K.; Wallace, J.L. Cyclooxygenase 2 mediates post-inflammatory colonic secretory and barrier dysfunction. Gut 2003, 52, 1714–1720. [Google Scholar] [CrossRef] [PubMed]
- Singer, I.I.; Kawka, D.W.; Schloemann, S.; Tessner, T.; Riehl, T.; Stenson, W.F. Cyclooxygenase 2 is induced in colonic epithelial cells in inflammatory bowel disease. Gastroenterology 1998, 115, 297–306. [Google Scholar] [CrossRef]
- Wiercinska-Drapało, A.; Flisiak, R.; Prokopowicz, D. Effects of ulcerative colitis activity on plasma and mucosal prostaglandin E2 concentration. Prostaglandins Other Lipid Mediat. 1999, 58, 159–165. [Google Scholar] [CrossRef]
- Lauritsen, K.; Laursen, L.S.; Kjeldsen, J.; Bukhave, K.; Hansen, T.K.; Rask-Madsen, J. Effects of mesalazine on the formation of lipoxygenase and cyclooxygenase products. Adv. Exp. Med. Biol. 1995, 371B, 1301–1306. [Google Scholar] [PubMed]
- Raso, G.M.; Meli, R.; Di Carlo, G.; Pacilio, M.; Di Carlo, R. Inhibition of inducible nitric oxide synthase and cyclooxygenase-2 expression by flavonoids in macrophage J774A.1. Life Sci. 2001, 68, 921–931. [Google Scholar] [CrossRef]
- Tsatsanis, C.; Androulidaki, A.; Venihaki, M.; Margioris, A.N. Signalling networks regulating cyclooxygenase-2. Int. J. Biochem. Cell Biol. 2006, 38, 1654–1661. [Google Scholar]
- Tonai, T.; Taketani, Y.; Ueda, N.; Nishisho, T.; Ohmoto, Y.; Sakata, Y.; Muraguchi, M.; Wada, K.; Yamamoto, S. Possible involvement of interleukin-1 in cyclooxygenase-2 induction after spinal cord injury in rats. J. Neurochem. 1999, 72, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Khalatbary, A.R.; Zarrinjoei, G.R. Anti-inflammatory effect of oleuropein in experimental rat spinal cord trauma. Iran Red Crescent Med. J. 2012, 14, 229–234. [Google Scholar] [PubMed]
- Boss, A.; Bishop, K.S.; Marlow, G.; Barnett, M.P.; Ferguson, L.R. Evidence to Support the Anti-Cancer Effect of Olive Leaf Extract and Future Directions. Nutrients 2016, 8, 513. [Google Scholar] [CrossRef] [PubMed]
- Neurath, M.F. Cytokines in inflammatory bowel disease. Nat. Rev. Immunol. 2014, 14, 329–342. [Google Scholar] [CrossRef] [PubMed]
- Ohman, L.; Dahlén, R.; Isaksson, S.; Sjoling, A.; Wick, M.J.; Sjövall, H.; van Oudenhove, L.; Simrén, M.; Strid, H. Serum IL-17A in newly diagnosed treatment-naive patients with ulcerative colitis reflects clinical disease severity and predicts the course of disease. Inflamm. Bowel Dis. 2013, 19, 2433–2439. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Su, J.; Zhang, X.; Cheng, X.; Zhou, J.; Shi, R.; Zhang, H. Elevated levels of Th17 cells and Th17-related cytokines are associated with disease activity in patients with inflammatory bowel disease. Inflamm. Res. 2014, 63, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Qian, Y. The activation and regulation of IL-17 receptor mediated signaling. Cytokine. 2013, 62, 175–182. [Google Scholar] [CrossRef] [PubMed]
- DeRoche, T.C.; Xiao, S.Y.; Liu, X. Histological evaluation in ulcerative colitis. Gastroenterol. Rep. (Oxf.) 2014, 2, 178–192. [Google Scholar] [CrossRef] [PubMed]
- Harpaz, N.; Polydorides, A.D. Colorectal dysplasia in chronic inflammatory bowel disease: Pathology, clinical implications, and pathogenesis. Arch. Pathol. Lab. Med. 2010, 13, 876–895. [Google Scholar]
- Vossenkämper, A.; Hundsrucker, C.; Page, K.; van Maurik, A.; Sanders, T.J.; Stagg, A.J.; Das, L.; MacDonald, T.T. A CD3-specific antibody reduces cytokine production and alters phosphoprotein profiles in intestinal tissues from patients with inflammatory bowel disease. Gastroenterology 2014, 147, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Gerner, R.R.; Moschen, A.R.; Tilg, H. Targeting T and B lymphocytes in inflammatory bowel diseases: Lessons from clinical trials. Dig. Dis. 2013, 31, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Leiper, K.; Martin, K.; Ellis, A.; Subramanian, S.; Watson, A.J.; Christmas, S.E.; Howarth, D.; Campbell, F.; Rhodes, J.M. Randomised placebo-controlled trial of rituximab (anti-CD20) in active ulcerative colitis. Gut 2011, 60, 1520–1526. [Google Scholar] [CrossRef] [PubMed]
- Caprioli, F.; Bose, F.; Rossi, R.L.; Petti, L.; Viganò, C.; Ciafardini, C.; Raeli, L.; Basilisco, G.; Ferrero, S.; Pagani, M.; et al. Reduction of CD68 C macrophages and decreased IL-17 expression in intestinal mucosa of patients with inflammatory bowel disease strongly correlate with endoscopic response and mucosal healing following infliximab therapy. Inflamm. Bowel Dis. 2013, 19, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Cosín-Roger, J.; Ortiz-Masiá, D.; Calatayud, S.; Hernandez, C.; Alvarez, A.; Hinojosa, J.; Esplugues, J.V.; Barrachina, M.D. M2 macrophages activate WNT signaling pathway in epithelial cells: Relevance in ulcerative colitis. PLoS ONE 2013, 8, e78128. [Google Scholar] [CrossRef] [PubMed]
- Omar, S.H. Oleuropein in olive and its pharmacological effects. Sci. Pharm. 2010, 78, 133–154. [Google Scholar] [CrossRef] [PubMed]
- Commission Regulation (EU) No 432/2012 Establishing a List of Permitted Health Claims Made on Foods, Other than Those Referring to the Reduction of Disease Risk and to Children’s Development and Health. Available online: https://www.fsai.ie/uploadedFiles/Reg432_2012.pdf (accessed on 13 April 2017).
| Characteristic | Values |
|---|---|
| Sex | 8 males; 6 females |
| Age (years) | 59 (range 39–80) |
| Mayo Score | 6 (range 4–6) |
| Pancolitis | 8 |
| Left-sided colitis | 6 |
| Oral/Topic Mesalamine | 14 |
| Previous azathioprine/anti-TNF alpha | - |
| Current oral/topic Steroids | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Larussa, T.; Oliverio, M.; Suraci, E.; Greco, M.; Placida, R.; Gervasi, S.; Marasco, R.; Imeneo, M.; Paolino, D.; Tucci, L.; et al. Oleuropein Decreases Cyclooxygenase-2 and Interleukin-17 Expression and Attenuates Inflammatory Damage in Colonic Samples from Ulcerative Colitis Patients. Nutrients 2017, 9, 391. https://doi.org/10.3390/nu9040391
Larussa T, Oliverio M, Suraci E, Greco M, Placida R, Gervasi S, Marasco R, Imeneo M, Paolino D, Tucci L, et al. Oleuropein Decreases Cyclooxygenase-2 and Interleukin-17 Expression and Attenuates Inflammatory Damage in Colonic Samples from Ulcerative Colitis Patients. Nutrients. 2017; 9(4):391. https://doi.org/10.3390/nu9040391
Chicago/Turabian StyleLarussa, Tiziana, Manuela Oliverio, Evelina Suraci, Marta Greco, Roberta Placida, Serena Gervasi, Raffaella Marasco, Maria Imeneo, Donatella Paolino, Luigi Tucci, and et al. 2017. "Oleuropein Decreases Cyclooxygenase-2 and Interleukin-17 Expression and Attenuates Inflammatory Damage in Colonic Samples from Ulcerative Colitis Patients" Nutrients 9, no. 4: 391. https://doi.org/10.3390/nu9040391
APA StyleLarussa, T., Oliverio, M., Suraci, E., Greco, M., Placida, R., Gervasi, S., Marasco, R., Imeneo, M., Paolino, D., Tucci, L., Gulletta, E., Fresta, M., Procopio, A., & Luzza, F. (2017). Oleuropein Decreases Cyclooxygenase-2 and Interleukin-17 Expression and Attenuates Inflammatory Damage in Colonic Samples from Ulcerative Colitis Patients. Nutrients, 9(4), 391. https://doi.org/10.3390/nu9040391










