Effects of a Diet-Based Weight-Reducing Program with Probiotic Supplementation on Satiety Efficiency, Eating Behaviour Traits, and Psychosocial Behaviours in Obese Individuals
Abstract
:1. Introduction
2. Materials and Methods
2.1. Protocol
2.2. Participants
2.3. Treatments
2.4. Measurements
2.5. Appetite Sensations
2.6. Ad libitum Energy Intake
2.7. Daily Energy Intake and Physical Activity
2.8. Resting Metabolic Rate and Calculation of the Estimated Energy Deficit
2.9. Body Weight and Composition Measurements
2.10. Three-Factor Eating Questionnaire
2.11. State-Trait Food Cravings Questionnaire
2.12. Mood-Related Questionnaires
2.13. Statistics
3. Results
3.1. Body Weight and Composition
3.2. Baseline Data
3.3. Response of Appetite Sensations to Food Intake
3.4. Energy Intake
3.5. Eating Behaviour Traits
3.6. Mood-Related Factors
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chaput, J.P.; Leblanc, C.; Perusse, L.; Despres, J.P.; Bouchard, C.; Tremblay, A. Risk factors for adult overweight and obesity in the Quebec Family Study: Have we been barking up the wrong tree? Obesity 2009, 17, 1964–1970. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S.F.; Murphy, E.F.; Nilaweera, K.; Ross, P.R.; Shanahan, F.; O’Toole, P.W.; Cotter, P.D. The gut microbiota and its relationship to diet and obesity: New insights. Gut Microb. 2012, 3, 186–202. [Google Scholar] [CrossRef] [PubMed]
- Delzenne, N.M.; Cani, P.D. Interaction between obesity and the gut microbiota: Relevance in nutrition. Ann. Rev. Nutr. 2011, 31, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Microbial ecology: Human gut microbes associated with obesity. Nature 2006, 444, 1022–1023. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, S.T.; dos Santos, C.A.; Bressan, J. Intestinal microbiota; relevance to obesity and modulation by prebiotics and probiotics. Nutr. Hosp. 2013, 28, 1039–1048. [Google Scholar] [PubMed]
- Cryan, J.F.; O’Mahony, S.M. The microbiome-gut-brain axis: From bowel to behavior. Neurogastroenterol. Motil. 2011, 23, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Rhee, S.H.; Pothoulakis, C.; Mayer, E.A. Principles and clinical implications of the brain-gut-enteric microbiota axis. Nat. Rev. Gastroent. Hepathol. 2009, 6, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Dinan, T.G.; Cryan, J.F. Melancholic microbes: A link between gut microbiota and depression? Neurogastroenterol. Motil. 2013, 25, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Lyte, M.; Li, W.; Opitz, N.; Gaykema, R.P.; Goehler, L.E. Induction of anxiety-like behavior in mice during the initial stages of infection with the agent of murine colonic hyperplasia Citrobacter rodentium. Physiol. Behav. 2006, 89, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Goehler, L.E.; Park, S.M.; Opitz, N.; Lyte, M.; Gaykema, R.P. Campylobacter jejuni infection increases anxiety-like behavior in the holeboard: Possible anatomical substrates for viscerosensory modulation of exploratory behavior. Brain Behav. Immun. 2008, 22, 354–366. [Google Scholar] [CrossRef] [PubMed]
- Gareau, M.G.; Wine, E.; Rodrigues, D.M.; Cho, J.H.; Whary, M.T.; Philpott, D.J.; Macqueen, G.; Sherman, P.M. Bacterial infection causes stress-induced memory dysfunction in mice. Gut 2011, 60, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Dowd, S.E.; Scurlock, B.; Acosta-Martinez, V.; Lyte, M. Memory and learning behavior in mice is temporally associated with diet-induced alterations in gut bacteria. Physiol. Behav. 2009, 96, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055. [Google Scholar] [CrossRef] [PubMed]
- Forsythe, P.; Inman, M.D.; Bienenstock, J. Oral treatment with live Lactobacillus reuteri inhibits the allergic airway response in mice. Am. J. Respir. Crit. Care Med. 2007, 175, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Venault, P.; Chapouthier, G. From the behavioral pharmacology of beta-carbolines to seizures, anxiety, and memory. Sci. World J. 2007, 7, 204–223. [Google Scholar] [CrossRef] [PubMed]
- Doucet, E.; Imbeault, P.; St-Pierre, S.; Almeras, N.; Mauriege, P.; Richard, D.; Tremblay, A. Appetite after weight loss by energy restriction and a low-fat diet-exercise follow-up. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 906–914. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, M.; Kissileff, H.R.; Mayer, L.E.; Hirsch, J.; Leibel, R.L. Energy intake in weight-reduced humans. Brain Res. 2010, 1350, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Chaput, J.P.; Arguin, H.; Gagnon, C.; Tremblay, A. Increase in depression symptoms with weight loss: Association with glucose homeostasis and thyroid function. Appl. Physiol. Nutr. Metab. 2008, 33, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Chaput, J.P.; Drapeau, V.; Hetherington, M.; Lemieux, S.; Provencher, V.; Tremblay, A. Psychobiological effects observed in obese men experiencing body weight loss plateau. Depress. Anxiety 2007, 24, 518–521. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, M.; Darimont, C.; Drapeau, V.; Emady-Azar, S.; Lepage, M.; Rezzonico, E.; Ngom-Bru, C.; Berger, B.; Philippe, L.; Ammon-Zuffrey, C.; et al. Effect of Lactobacillus rhamnosus CGMCC1.3724 supplementation on weight loss and maintenance in obese men and women. Br. J. Nutr. 2014, 111, 1507–1519. [Google Scholar] [CrossRef] [PubMed]
- Drapeau, V.; King, N.; Hetherington, M.; Doucet, E.; Blundell, J.; Tremblay, A. Appetite sensations and satiety quotient: Predictors of energy intake and weight loss. Appetite 2007, 48, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Blundell, J.E.; Hill, A.J. Paradoxical effects of an intense sweetener (aspartame) on appetite. Lancet 1986, 1, 1092–1093. [Google Scholar] [CrossRef]
- Green, S.M.; Delargy, H.J.; Joanes, D.; Blundell, J.E. A satiety quotient: A formulation to assess the satiating effect of food. Appetite 1997, 29, 291–304. [Google Scholar] [CrossRef] [PubMed]
- Drapeau, V.; Blundell, J.; Therrien, F.; Lawton, C.; Richard, D.; Tremblay, A. Appetite sensations as a marker of overall intake. Br. J. Nutr. 2005, 93, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Arvaniti, K.; Richard, D.; Tremblay, A. Reproducibility of energy and macronutrient intake and related substrate oxidation rates in a buffet-type meal. Br. J. Nutr. 2000, 83, 489–495. [Google Scholar] [PubMed]
- Tremblay, A.; Sévigny, J.; Leblanc, C.; Bouchard, C. The reproducibility of a three-day dietary record. Nutr. Res. 1983, 3, 819–830. [Google Scholar] [CrossRef]
- Canada, H. The Canadian Nutrient File; Health Canada: Ottawa, ON, Canada, 2005. [Google Scholar]
- Bouchard, C.; Tremblay, A.; Leblanc, C.; Lortie, G.; Savard, R.; Theriault, G. A method to assess energy expenditure in children and adults. Am. J. Clin. Nutr. 1983, 37, 461–467. [Google Scholar] [PubMed]
- Diabetes Quebec. Meal Planning for People with Diabetes; Government of Quebec: Quebec, QC, Canada, 2003.
- Stunkard, A.J.; Messick, S. The three-factor eating questionnaire to measure dietary restraint, disinhibition and hunger. J. Psychosom. Res. 1985, 29, 71–83. [Google Scholar] [CrossRef]
- Cepeda-Benito, A.; Gleaves, D.H.; Fernandez, M.C.; Vila, J.; Williams, T.L.; Reynoso, J. The development and validation of Spanish versions of the State and Trait Food Cravings Questionnaires. Behav. Res. Ther. 2000, 38, 1125–1138. [Google Scholar] [CrossRef]
- White, M.A.; Whisenhunt, B.L.; Williamson, D.A.; Greenway, F.L.; Netemeyer, R.G. Development and validation of the food-craving inventory. Obes. Res. 2002, 10, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Beck, A.T.; Ward, C.H.; Mendelson, M.; Mock, J.; Erbaugh, J. An inventory for measuring depression. Arch. Gen. Psychiatry 1961, 4, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Mendelson, B.K.; Mendelson, M.J.; White, D.R. Body-esteem scale for adolescents and adults. J. Personal. Assess. 2001, 76, 90–106. [Google Scholar] [CrossRef] [PubMed]
- Gormally, J.; Black, S.; Daston, S.; Rardin, D. The assessment of binge eating severity among obese persons. Addict. Behav. 1982, 7, 47–55. [Google Scholar] [CrossRef]
- Cohen, S.; Kamarck, T.; Mermelstein, R. A global measure of perceived stress. J. Health Soc. Behav. 1983, 24, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Spielberger, C.D.; Gorsuch, R.L.; Lushene, R.; Vagg, P.R.; Jacobs, G.A. Manual for the State-Trait Anxiety Inventory; Consulting Psychologists Press: Palo Alto, CA, USA, 1983. [Google Scholar]
- Sanchez, M.; Darimont, C.; Drapeau, V.; Panahi, S.; Laval University, Quebec City, QC, Canada. Unpublished work. 2017.
- Gilbert, J.A.; Drapeau, V.; Astrup, A.; Tremblay, A. Relationship between diet-induced changes in body fat and appetite sensations in women. Appetite 2009, 52, 809–812. [Google Scholar] [CrossRef] [PubMed]
- Jakubowicz, D.; Froy, O.; Wainstein, J.; Boaz, M. Meal timing and composition influence ghrelin levels, appetite scores and weight loss maintenance in overweight and obese adults. Steroids 2012, 77, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Massey, A.; Hill, A.J. Dieting and food craving. A descriptive, quasi-prospective study. Appetite 2012, 58, 781–785. [Google Scholar] [CrossRef] [PubMed]
- Alberts, H.J.; Mulkens, S.; Smeets, M.; Thewissen, R. Coping with food cravings. Investigating the potential of a mindfulness-based intervention. Appetite 2010, 55, 160–163. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Norman, R.J.; Clifton, P.M.; Noakes, M. Psychological effects of prescriptive vs. general lifestyle advice for weight loss in young women. J. Am. Diet. Assoc. 2009, 109, 1917–1921. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.K.; O’Neil, P.M.; Pawlow, L. Changes in food cravings during low-calorie and very-low-calorie diets. Obesity 2006, 14, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Forman, E.M.; Hoffman, K.L.; McGrath, K.B.; Herbert, J.D.; Brandsma, L.L.; Lowe, M.R. A comparison of acceptance- and control-based strategies for coping with food cravings: An analog study. Behav. Res. Ther. 2007, 45, 2372–2386. [Google Scholar] [CrossRef] [PubMed]
- Moreno, S.; Rodriguez, S.; Fernandez, M.C.; Tamez, J.; Cepeda-Benito, A. Clinical validation of the trait and state versions of the Food Craving Questionnaire. Assessment 2008, 15, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Sitton, S.C. Role of craving for carbohydrates upon completion of a protein-sparing fast. Psychol. Rep. 1991, 69, 683–686. [Google Scholar] [CrossRef] [PubMed]
- Riesco, E.; Roussel, M.; Lemoine, S.; Garnier, S.; Sanguignol, F.; Mauriege, P. What is the influence of menopausal status on metabolic profile, eating behaviors, and perceived health of obese women after weight reduction? Appl. Physiol. Nutr. Metab. 2008, 33, 957–965. [Google Scholar] [CrossRef] [PubMed]
- Gallant, A.R.; Tremblay, A.; Perusse, L.; Bouchard, C.; Despres, J.P.; Drapeau, V. The Three-Factor Eating Questionnaire and BMI in adolescents: Results from the Quebec family study. Br. J. Nutr. 2010, 104, 1074–1079. [Google Scholar] [CrossRef] [PubMed]
- Drapeau, V.; Despres, J.P.; Bouchard, C.; Allard, L.; Fournier, G.; Leblanc, C.; Tremblay, A. Modifications in food-group consumption are related to long-term body-weight changes. Am. J. Clin. Nutr. 2004, 80, 29–37. [Google Scholar] [PubMed]
- Ewbank, P.P.; Darga, L.L.; Lucas, C.P. Physical activity as a predictor of weight maintenance in previously obese subjects. Obes. Res. 1995, 3, 257–263. [Google Scholar] [CrossRef] [PubMed]
- McGuire, M.T.; Wing, R.R.; Klem, M.L.; Seagle, H.M.; Hill, J.O. Long-term maintenance of weight loss: Do people who lose weight through various weight loss methods use different behaviors to maintain their weight? Int. J. Obes. Relat. Metab. Disord. 1998, 22, 572–577. [Google Scholar] [CrossRef] [PubMed]
- Lejeune, M.P.; Kovacs, E.M.; Westerterp-Plantenga, M.S. Additional protein intake limits weight regain after weight loss in humans. Br. J. Nutr. 2005, 93, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Jackson, S.E.; Steptoe, A.; Beeken, R.J.; Kivimaki, M.; Wardle, J. Psychological changes following weight loss in overweight and obese adults: A prospective cohort study. PLoS ONE 2014, 9, e104552. [Google Scholar] [CrossRef] [PubMed]
- Hemerajata, P.; Versalovic, J. Effects of probiotics on gut microbiota: Mechanisms of intestinal immunomodulation and neuromodulation. Therap. Adv. Gastroenterol. 2013, 6, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Mayer, E.A.; Tillisch, K.; Gupta, A. Gut/brain axis and the microbiota. J. Clin. Invest. 2015, 125, 926–938. [Google Scholar] [CrossRef] [PubMed]
- Steenbergen, L.; Sellaro, R.; van Hemert, S.; Bosch, J.A.; Colzato, L.S. A randomized controlled trial to test the effect of multispecies probiotics on cognitive reactivity to sad mood. Brain Behav. Immun. 2015, 48, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Schrezenmeir, J.; de Vrese, M. Probiotics, prebiotics, and synbiotics—Approaching a definition. Am. J. Clin. Nutr. 2001, 73, 361S–364S. [Google Scholar] [PubMed]
- Mäkeläinen, H.; Forsten, S.; Saarinen, M.; Stowell, J.; Rautonen, N.; Ouwehand, A.C. Xylo-oligosaccharides and lactitol promote the growth of Bifidobacterium lactis and Lactobacillus species in pure cultures. Benef. Microb. 2010, 1, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Kolida, S.; Gibson, G.R. Synbiotics in health and disease. Annu. Rev. Food Sci. Technol. 2011, 2, 373–393. [Google Scholar] [CrossRef] [PubMed]
- Hess, J.R.; Birkett, A.M.; Thomas, W.; Slavin, J.L. Effects of short-chain fructooligosaccharides on satiety responses in healthy men and women. Appetite 2011, 56, 128–134. [Google Scholar] [CrossRef] [PubMed]
| Variables | LPR formulation | Placebo | Men | Women | ||
|---|---|---|---|---|---|---|
| 62 | 63 | LPR Formulation (N = 24) | Placebo (N = 24) | LPR Formulation (N = 38) | Placebo (N = 39) | |
| Subjects characteristics | ||||||
| Age (years) | 35.0 (10.0) | 37.0 (10.0) | 37.0 (10.0) | 38.0 (10.0) | 34.0 (10.0) | 36.0 (10.0) |
| Body weight (kg) | 95.1 (13.9) | 94.0 (14.9) | 104.3 (13.0) | 103.4 (15.0) | 89.3 (11.1) | 88.2 (11.5) |
| BMI (kg/m2) | 33.8 (3.3) | 33.3 (3.2) | 34.0 (2.8) | 33.5 (3.3) | 33.6 (3.6) | 33.2 (3.2) |
| Fat mass (%) | 40.82 (7.68) | 40.08 (8.22) | 34.8 (8.3) | 32.8 (9.1) | 40.8 (8.2) | 40.2 (7.6) |
| Energy intake reported (3-day dietary record) | ||||||
| Mean daily kcal | 2510 (676) | 2362 (611) | 2898 (550) | 2752 (479) | 2247 (637) | 2122 (562) |
| Proteins (%) | 17.1 (2.6) | 16.9 (2.4) | 17.2 (2.3) | 17.1 (2.6) | 16.9 (2.7) | 16.7 (2.4) |
| Carbohydrates (%) | 46.4 (6.9) | 47.2 (5.6) | 46.3 (5.5) | 46.6 (5.9) | 46.5 (7.6) | 47.6 (5.4) |
| Lipids (%) | 33.7 (6.2) | 34.1 (4.9) | 33.5 (4.4) | 34.5 (5.2) | 33.9 (7.1) | 33.8 (4.5) |
| Alcohol (%) | 2.8 (2.9) | 1.8 (2.7) | 3.0 (2.7) | 1.8 (2.6) | 2.7 (3.1) | 1.9 (2.8) |
| Energy intake measured (buffet type meal) | ||||||
| Kcal | 908.6 (437.3) | 906 (424.5) | 1017 (407) | 1096 (503) | 799 (419) | 750 (279) |
| Proteins (%) | 17.3 (3.4) | 17.3 (5.3) | 17.5 (3.0) | 16.8 (5.8) | 16.4 (3.7) | 16.8 (5.3) |
| Carbohydrates (%) | 42.8 (8.2) | 44.7 (10.1) | 45.2 (8.2) | 46.9 (11.9) | 41.7 (8.4) | 43.8 (9.2) |
| Lipids (%) | 39.9 (8.5) | 38.0 (10.2) | 37.3 (8.3) | 36.3 (11.2) | 41.9 (8.7) | 39.4 (10.0) |
| Appetite sensations in fasting state (mm) | ||||||
| Desire to eat | 96.6 (31.7) | 96.5 (33.0) | 98.1 (28.9) | 96.0 (34.7) | 95.7 (33.7) | 96.8 (32.4) |
| Fullness | 24.0 (25.4) | 24.0 (27.7) | 27.4 (30.0) | 33.9 (33.7) | 21.8 (22.1) | 17.9 (21.6) |
| SQ at breakfast (mm/ kcal) | ||||||
| Desire to eat | 10.2 (4.9) | 10.4 (5.8) | 8.6 (4.1) | 6.8 (4.3) | 11.2 (5.2) | 12.7 (5.5) |
| Fullness | 11.1 (5.6) | 10.8 (7.4) | 8.4 (5.0) | 6.0 (4.6) | 12.8 (5.4) | 13.8 (7.3) |
| SQ at buffet (mm/ kcal) | ||||||
| Desire to eat | 10.3 (6.2) | 10.2 (5.9) | 7.1 (3.8) | 7.7 (3.9) | 12.5 (6.5) | 11.6 (6.3) |
| Fullness | 10.3 (5.6) | 9.9 (5.9) | 8.2 (3.7) | 8.8 (4.0) | 11.7 (6.3) | 10.4 (6.7) |
| Eating behaviour traits (TFEQ) | ||||||
| Cognitive restraint | 8.3 (4.0) | 8.3 (3.7) | 8.0 (3.2) | 7.3 (3.3) | 8.4 (4.5) | 8.9 (3.9) |
| Disinhibition | 7.8 (3.0) | 8.0 (3.1) | 6.7 (3.2) | 8.2 (3.2) | 8.4 (2.8) | 7.9 (3.0) |
| Hunger | 6.3 (3.3) | 6.2 (3.5) | 5.6 (3.7) | 8.2 (2.6) | 6.7 (3.0) * | 5.0 (3.5) |
| State-Trait Food Cravings Questionnaire-Trait (FCQ-T) | ||||||
| Total score | 108.8 (25.0) | 110.3 (28.3) | 100.8 (24.6) | 111.3 (24.2) | 114.4 (24.0) | 109.8 (30.6) |
| BECK Depression Inventory | ||||||
| Total score | 4.4 (4.1) | 4.7 (4.2) | 3.6 (3.5) | 3.6 (2.9) | 4.96 (4.4) | 5.2 (4.7) |
| Body Esteem Scale (BE) | ||||||
| Total score | 38.7 (10.9) | 35.8 (10.8) | 45.6 (11.3) | 37.8 (11.4) | 34.6 (8.4) | 34.6 (10.4) |
| Binge Eating Scale (BES) | ||||||
| Total score | 12.0 (6.6) | 12.2 (6.6) | 10.0 (6.6) | 12.8 (6.9) | 13.4 (6.4) | 11.8 (6.5) |
| Perceived Stress Scale (EPS) | ||||||
| Total score | 14.1 (6.0) | 14.4 (6.0) | 12.1 (5.8) | 12.9 (5.7) | 15.4 (5.9) | 15.3 (6.1) |
| Spielberger State-Trait Anxiety Inventory (State Anxiety) | ||||||
| Total score | 28.9 (6.5) | 28.2 (5.9) | 29.8 (7.8) | 27.4 (5.9) | 28.3 (5.6) | 28.7(5.9) |
| Spielberger State-Trait Anxiety Inventory (Trait Anxiety) | ||||||
| Total score | 37.3 (7.8) | 37.0 (6.7) | 35.8 (7.9) | 37.9 (7.6) | 38.3 (7.7) | 36.5 (6.2) |
| Variables Changes | All participants | Men | Women | |||
|---|---|---|---|---|---|---|
| LPR Formulation | Placebo | LPR Formulation | Placebo | LPR formulation | Placebo | |
| Number of participants | ||||||
| ∆ Phase 1-baseline | 52 | 53 | 23 | 22 | 29 | 31 |
| ∆ Phase 2-Phase 1 | 45 | 48 | 19 | 20 | 26 | 28 |
| ∆ Phase 2-baseline | 45 | 48 | 19 | 20 | 26 | 28 |
| Body weight (kg) ** | ||||||
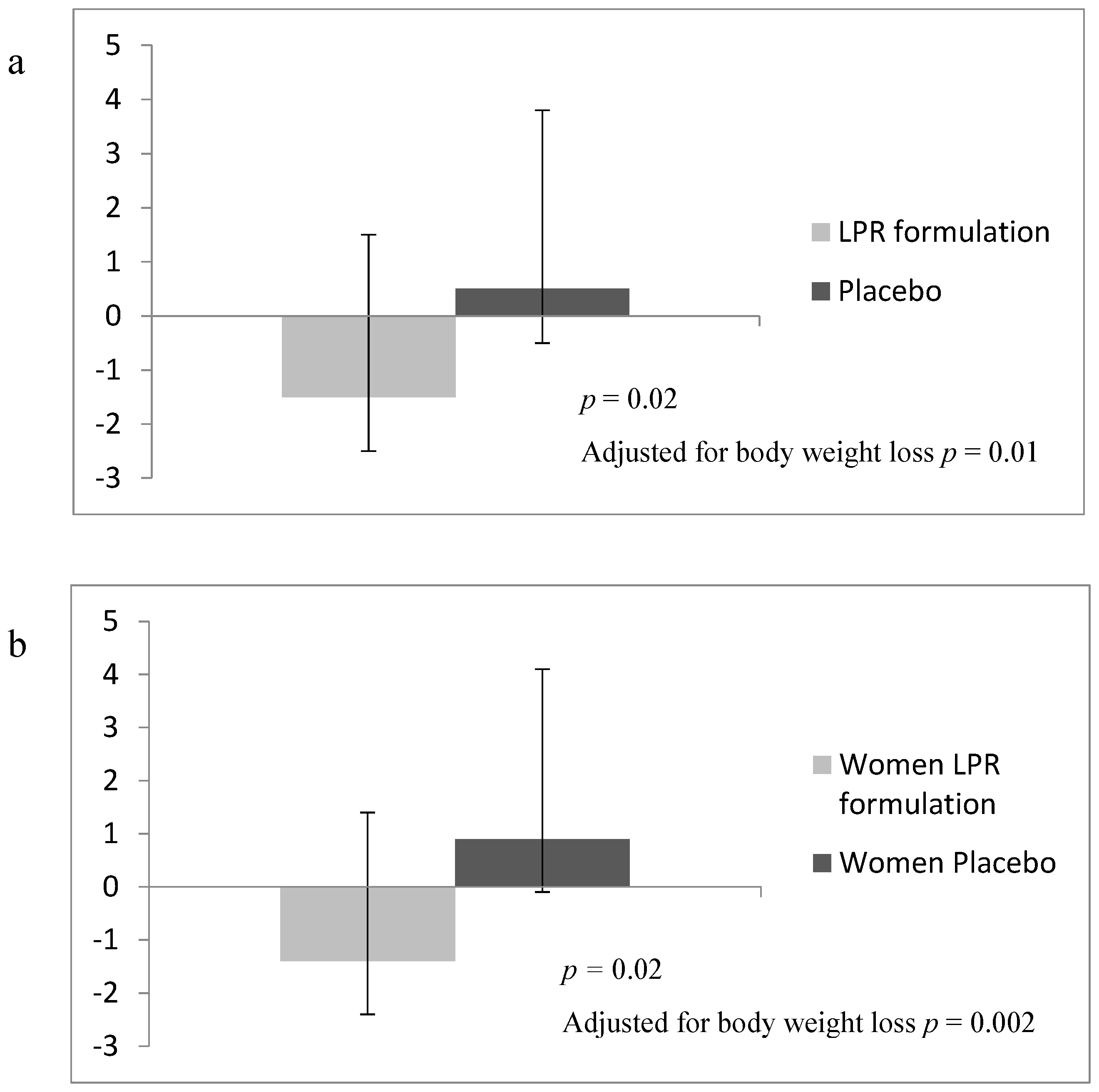
| ∆ Phase 1-baseline | −4.2 (3.2) | −3.4 (2.9) | −4.0 (3.4) | −4.6 (3.2) | −4.4 (3.0) * | −2.6 (2.3) |
| ∆ Phase 2-baseline | −5.3 (4.3) | −3.9 (4.2) | −5.4 (4.8) | −5.7 (4.5) | −5.2 (4.0) * | −2.5 (3.5) |
| Fasting state Desire to eat (mm) | ||||||
| ∆ Phase 1-baseline | 9.3 (39.8) | 3.6 (37.0) | 0.2 (46.5) | −4.2 (33.3) | 16.2 (33.0) | 9.1 (39.0) |
| ∆ Phase 2-Phase 1 | 6.9 (30.6) | 9.3 (40.5) | 7.9 (27.6) | 23.9 (34.2) | 6.3 (33.0) | -1.1 (42.0) |
| ∆ Phase 2-baseline | 16.3 (32.8) | 13.5 (37.5) | 8.0 (35.6) | 22.9 (34.3) | 22.2 (29.9) * | 6.8 (38.9) |
| Fullness (mm) | ||||||
| ∆ Phase 1-baseline | −3.2 (27.8) | 1.9 (25.0) | −3.3 (33.3) | 3.1 (31.6) | −3.1 (23.4) | 1.1 (19.6) |
| ∆ Phase 2-Phase 1 | 1.9 (17.1) | −3.4 (19.1) | 5.8 (17.0) * | −10.0 (18.1) | −0.9 (16.9) | 1.3 (18.7) |
| ∆ Phase 2-baseline | −1.1 (30.0) | −0.2 (22.4) | 2.6 (39.2) | −3.1 (25.0) | −3.7 (21.9) | 1.9 (20.6) |
| SQ at breakfast Desire to eat (mm/kcal) | ||||||
| ∆ Phase 1-baseline | 1.2 (6.6) | 0.7 (6.2) | −1.1 (5.9) | 0.2 (4.6) | 2.8 (6.8) | 1.1 (7.2) |
| ∆ Phase 2-Phase 1 | 1.1 (5.8) | 0.7 (6.5) | 0.8 (4.7) | 2.5 (4.1) | 1.3 (6.4) | −0.6 (7.6) |
| ∆ Phase 2-baseline | 2.3 (5.3) | 1.4 (6.5) | 0.6 (4.6) | 3.0 (5.0) | 3.5 (5.5) | 0.2 (7.2) |
| Fullness (mm/kcal) | ||||||
| ∆ Phase 1-baseline | 1.4 (5.1) | 0.3 (5.0) | 0.2 (5.0) | 0.2 (4.1) | 2.3 (5.1) | 0.4 (5.7) |
| ∆ Phase 2-Phase 1 | −0.3 (4.0) | 0.7 (4.4) | −0.5 (3.8) | 1.1 (3.1) | −0.3 (4.2) | 0.4 (5.2) |
| ∆ Phase 2-baseline | 1.5 (5.3) | 1.1 (5.5) | 0.6 (6.1) | 1.2 (5.6) | 2.1 (4.7) | 1.1 (5.6) |
| SQ at buffet Desire to eat (mm/kcal) | ||||||
| ∆ Phase 1-baseline | 1.6 (7.1) * | −0.5 (4.8) | 1.5 (5.0) | −0.2 (4.2) | 1.6 (8.2) * | -0.7 (5.3) |
| ∆ Phase 2-Phase 1 | −0.5 (6.2) | 0.9 (5.9) | −0.6 (3.0) | 0.3 (3.7) | −0.4 (7.6) | 1.4 (7.4) |
| ∆ Phase 2-baseline | 1.0 (7.5) | −0.1 (6.5) | 1.8 (5.0) | −1.1 (3.8) | 0.6 (8.6) | 0.6 (7.9) |
| Fullness (mm/kcal) | ||||||
| ∆ Phase 1-baseline | −22.0 (12.9) | −17.9 (9.0) | −16.4 (6.6) | −15.3 (6.4) | −25.4 (14.5) | -20.2 (10.4) |
| ∆ Phase 2-Phase 1 | 0.9 (6.8) | −1.8 (5.1) | 1.5 (3.0) | −0.5 (3.6) | 0.6 (8.3) | −3.0 (6.0) |
| ∆ Phase 2-baseline | −21.0 (11.6) | −18.8 (7.6) | −14.3 (6.0) | −15.0 (5.7) | −24.4 (12.3) | -21.9 (7.6) |
| Energy intake reported (3-day dietary record) Mean daily kcal | ||||||
| ∆ Phase 1-baseline | −426.3 (65.2) | 405.6 (65.7) | −405 (454) | −435 (454) | -488 (505) | -375 (423) |
| ∆ Phase 2-Phase 1 | 100.0 (67.0) | 78.8 (67.5) | 44 (619) | 46 (431) | 156 (275) | 112 (438) |
| ∆ Phase 2-baseline | −343.0 (68.0) | −323.2 (68.1) | −341 (432) | −375 (457) | −345 (455) | −272 (456) |
| Energy intake reported (3-day dietary record) Proteins (%) | ||||||
| ∆ Phase 1-baseline | 2.2 (0.4) | 2.5 (0.4) | 0.6 (3.4) | 1.7 (3.5) | 3.7 (3.9) | 3.4 (3.5) |
| ∆ Phase 2-Phase 1 | −0.1 (0.4) | 0.1 (0.4) | 0.7 (2.7) | 0.5 (4.0) | −0.1 (3.7) | −0.5 (3.2) |
| ∆ Phase 2-baseline | 2.5 (0.5) | 2.8 (0.5) | 1.0 (3.8) | 2.3 (4.1) | 3.9 (3.6) | 3.0 (2.5) |
| Energy intake reported (3-day dietary record) Carbohydrates (%) | ||||||
| ∆ Phase 1-baseline | 3.2 (0.8) | 2.1 (0.8) | 4.5 (8.1) | 1.8 (7.5) | 2.6 (6.8) | 1.6 (6.3) |
| ∆ Phase 2-Phase 1 | −1.6 (0.8) | −0.1 (0.8) | −3.9 (5.6) | 1.6 (6.6) * | −0.5 (6.3) | −0.9(7.4) |
| ∆ Phase 2-baseline | 1.3 (0.8) | 2.3 (0.8) | 1.0 (8.0) | 2.6 (7.7) | 2.5 (8.3) | 0.7 (5.5) |
| Energy intake reported (3-day dietary record) Lipids (%) | ||||||
| ∆ Phase 1-baseline | −4.6 (0.8) | −4.2 (0.8) | −3.8 (5.7) | −3.8 (6.4) | −5.4 (8.0) | −4.4 (6.6) |
| ∆ Phase 2-Phase 1 | 1.8 (0.8) | 0.2 (0.8) | 2.7 (4.8) | −0.9 (6.4) * | 1.2 (6.3) | 0.8 (7.0) |
| ∆ Phase 2-baseline | −2.9 (0.8) | −4.2 (0.8) | −1.3 (6.5) | −4.2 (8.1) | −4.7 (7.8) | −3.5 (4.9) |
| Energy intake reported (3-day dietary record) Alcohol (%) | ||||||
| ∆ Phase 1-baseline | −0.9 (0.3) | −0.3 0(0.3) | −1.3 (2.6) | 0.3 (3.2) * | −1.0 .(2.1) | −0.6 (1.7) |
| ∆ Phase 2-Phase 1 | −0.1 (0.3) | −0.2 (0.3) | 0.5 (2.4) | −1.2 (3.1) | −0.6 (3.0) | 0.6 (3.1) |
| ∆ Phase 2-baseline | −0.9 (0.3) | −0.9 (0.3) | −0.7 (3.3) | −0.8 (2.9) | −1.8 (3.4) | −0,3 (3.7) |
| Energy intake measured (buffet type meal) Mean daily kcal | ||||||
| ∆ Phase 1-baseline | −18.6 (39.2) | −35.2 (38.4) | −30 (464) | −62 (340) | −22 (179) | −39 (336) |
| ∆ Phase 2-Phase 1 | 0.5 (68.1) | 102.9 (68.1) | 12 (235) | 115 (194) | −16 (141) | 62 (377) |
| ∆ Phase 2-baseline | −38.5 (50.4) | 35.0 (49.4) | −46 (583) | 47(396) | −45 (197) | 20 (413) |
| Energy intake measured (buffet type meal) Proteins (%) | ||||||
| ∆ Phase 1-baseline | 0.6 (0.5) | 0.5 (0.5) | −0.8 (3.0) | 0.4 (3.1) | 2.0 (4.0) | 0.5 (5.5) |
| ∆ Phase 2-Phase 1 | 0.7 (0.7) | −0.1 (0.7) | 2.3 (4.9) | −0.3 (3.3) | −0.7 (5.3) | 0.1 (5.2) |
| ∆ Phase 2-baseline | 1.4 (0.7) | 0.5 (0.7) | 1.6 (6.2) | 0.2 (5.0) | 1.5 (4.1) | 0.4 (5.7) |
| Energy intake measured (buffet type meal) Carbohydrates (%) | ||||||
| ∆ Phase 1-baseline | 0.1 (1.1) | 2.9 (1.1) | −0.1 (7.7) | −0.1 (11.5) | 1.5 (9.7) | 4.1 (10.8) |
| ∆ Phase 2-Phase 1 | −3.7 (1.2) | −1.4 (1.1) | −7.0 (9.7) * | −1.2 (6.6) | 0.5 (8.0) | −2.6 (8.9) |
| ∆ Phase 2-baseline | −2.5 (1.3) | 0.6 (1.3) | −5.8 (10.2) | −2.1 (12.6) | 1.2 (9.9) | 2.2 (9.4) |
| Energy intake measured (buffet type meal) Lipids (%) | ||||||
| ∆ Phase 1-baseline | −0.7 (1.2) | −3.3 (1.2) | 0.8 (8.8) | −0.3 (10.4) | −3.5 (10.5) | −4.6 (10.5) |
| ∆ Phase 2-Phase 1 | 2.9 (1.1) | 1.6 (1.0) | 4.7 (8.0) | 1.5 (5.5) | 0.2 (8.2) | 2.5 (7.6) |
| ∆ Phase 2-baseline | −1.1 (1.3) | −1.1 (1.2) | 4.2 (9.0) | 1.8 (11.5) | −2.7 (9.1) | −2.7(10.2) |
| Variables Changes | All Participants | Men | Women | |||
|---|---|---|---|---|---|---|
| LPR Formulation | Placebo | LPR Formulation | Placebo | LPR Formulation | Placebo | |
| Number of Participants | ||||||
| ∆ Phase 1-baseline | 52 | 53 | 23 | 22 | 29 | 31 |
| ∆ Phase 2-Phase 1 | 45 | 48 | 19 | 20 | 26 | 28 |
| ∆ Phase 2-baseline | 45 | 48 | 19 | 20 | 26 | 28 |
| Three-Factor Eating Questionnaire Cognitive restraint | ||||||
| ∆ Phase 1-baseline | 3.5 (3.5) * | 4.8 (3.7) | 3.0 (3.6) * | 5.9 (3.8) | 3.9 (3.5) | 4.0 (3.5) |
| ∆ Phase 2-Phase 1 | 0.1 (2.6) | 0.0 (3.0) | −0.3 (1.9) | 0.6 (2.8) | 0.5 (3.1) | −0.5 (3.2) |
| ∆ Phase 2-baseline | 3.7 (4.4) * | 5.3 (4.3) | 2.6 (4.3) * | 6.7 (4.5) | 4.4 (4.4) | 4.2 (4.0) |
| Disinhibition | ||||||
| ∆ Phase 1-baseline | −2.0 (2.0) | −1.3 (2.3) | −1.6 (1.8) | −1.5 (2.3) | −2.2 (2.1) * | −1.1 (2.3) |
| ∆ Phase 2-Phase 1 | 0.3 (1.4) | −0.4 (1.9) | 0.5 (1.4) | −0.4 (1.6) | 0.2 (1.4) | −0.5 (2.1) |
| ∆ Phase 2-baseline | −1.7 (2.3) | −1.8 (2.1) | −1.2 (2.5) | −1.7 (2.1) | −2.1 (2.2) | −1.8 (2.2) |
| Hunger | ||||||
| ∆ Phase 1-baseline | −2.6 (3.0) | −1.8 (2.7) | −1.8 (3.5) | −2.9 (3.0) | −3.2 (2.6) * | −1.1 (2.2) |
| ∆ Phase 2-Phase 1 | 0.3 (2.0) | −0.2 (2.7) | 0.3 (1.7) | −0.3 (2.9) | 0.4 (2.2) | −0.0(2.5) |
| ∆ Phase 2-baseline | −2.4 (2.4) | −2.0 (2.7) | −1.8 (3.1) | −3.3 (2.5) | −2.8 (1.8) * | −1.0 (2.5) |
| State-Trait Food Cravings Questionnaire-Trait (FCQ-T) | ||||||
| ∆ Phase 1-baseline | −16.1 (15.6) | −8.1 (18.0) | −11.5 (9.7) | −6.8 (21.7) | −19.5 (18.2) * | −8.9 (15.2) |
| ∆ Phase 2-Phase 1 | −3.1 (10.4) | −4.1 (11.9) | −3.6 (9.7) | −5.1 (9.8) | −2.8 (11.1) | −3.4 (13.5) |
| ∆ Phase 2-baseline | −19.2 (18.8) | −13.2 (18.2) | −13.8 (14.6) | −14.5 (22.7) | −23.2 (20.8) * | −12.4 (14.8) |
| BECK Depression Inventory | ||||||
| ∆ Phase 1-baseline | 0.1 (2.9) | −0.6 (3.1) | 0.5 (2.7) | −0.1 (2.5) | −0.2 (3.1) | −1.0 (3.5) |
| ∆ Phase 2-Phase 1 | −1.3 (2.7) * | 0.6 (3.2) | −2.0 (3.1) | −0.2 (3.2) | −0.8 (2.3) * | 1.4 (3.1) |
| ∆ Phase 2-baseline | −1.5 (3.0) * | 0.5 (3.3) | −1.5 (3.3) | −0.1 (3.6) | −1.4 (2.8) * | 0.9 (3.2) |
| Body Esteem Scale (BE) | ||||||
| ∆ Phase 1-baseline | 6.2 (6.7) | 6.6 (6.9) | 4.2 (5.7) | 6.5 (7.1) | 7.4 (7.1) | 6.7 (6.9) |
| ∆ Phase 2-Phase 1 | 4.2 (7.4) | 3.0 (6.4) | 3.1 (7.7) | 6.2 (4.9) | 4.8 (7.3) * | 0.7 (6.4) |
| ∆ Phase 2-baseline | 9.8 (7.7) | 10.2 (6.3) | 7.9 (8.3) | 14.3 (5.0) | 11.0 (7.2) † | 7.3 (6.4) |
| Binge Eating Scale (BES) | ||||||
| ∆ Phase 1-baseline | −4.0 (4.5) | −3.6 (4.7) | −2.5 (3.9) | −2.6 (5.0) | −5.2 (4.7) | −4.3 (4.6) |
| ∆ Phase 2-Phase 1 | −0.5 (2.6) | −0.7 (3.3) | −0.6 (2.8) | −0.4 (3.1) | −0.5 (2.5) | −1.0 (3.4) |
| ∆ Phase 2-baseline | −3.8 (3.3) | −4.3 (4.7) | −2.8 (3.6) | −3.6 (5.5) | −4.5 (30.) | −4.7 (4.2) |
| Perceived Stress Scale (PSS) | ||||||
| ∆ Phase 1-baseline | −1.3 (5.2) | −1.2 (3.8) | −0.4 (4.4) | −0.5 (3.9) | −1.9 (5.7) | −1.6 (3.7) |
| ∆ Phase 2-Phase 1 | −0.0 (4.5) | 0.2 (5.0) | −0.4 (4.0) | −0.4 (5.1) | 0.2 (4.9) | 0.7 (5.0) |
| ∆ Phase 2-baseline | −1.6 (6.0) | −1.0 (4.6) | −1.5 (4.7) | −1.3 (4.8) | −1.7 (6.9) | −0.8 (4.5) |
| State-Trait Anxiety Inventory (State Anxiety) | ||||||
| ∆ Phase 1-baseline | 0.2 (7.6) | −0.0 (7.5) | −0.4 (7.5) | 1.9 (7.3) | 0.6 (7.8) | −1.4 (7.6) |
| ∆ Phase 2-Phase 1 | 0.4 (7.8) | 1.7 (6.7) | −1.0 (6.2) | 0.3 (6.6) | 1.3 (8.7) | 2.7 (6.8) |
| ∆ Phase 2-baseline | −0.6 (8.1) | 1.5 (8.9) | −1.7 (5.7) | 2.7 (10.7) | 0.1 (9.4) | 0.7 (7.4) |
| State-Trait Anxiety Inventory (Trait Anxiety) | ||||||
| ∆ Phase 1-baseline | −0.9 (5.4) | −1.8 (4.0) | −0.5 (3.6) | −2.4 (4.4) | −1.2 (6.4) | −1.5 (3.8) |
| ∆ Phase 2-Phase 1 | −1.1 (4.2) | 0.5 (5.2) | −1.4 (4.1) | −0.8 (4.6) | −1.0 (4.3) † | 1.6 (5.4) |
| ∆ Phase 2-baseline | −2.7 (5.4) | −1.4 (5.8) | −2.6 (3.8) | −2.9 (5.0) | −2.8 (6.3) | −0.5 (6.2) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanchez, M.; Darimont, C.; Panahi, S.; Drapeau, V.; Marette, A.; Taylor, V.H.; Doré, J.; Tremblay, A. Effects of a Diet-Based Weight-Reducing Program with Probiotic Supplementation on Satiety Efficiency, Eating Behaviour Traits, and Psychosocial Behaviours in Obese Individuals. Nutrients 2017, 9, 284. https://doi.org/10.3390/nu9030284
Sanchez M, Darimont C, Panahi S, Drapeau V, Marette A, Taylor VH, Doré J, Tremblay A. Effects of a Diet-Based Weight-Reducing Program with Probiotic Supplementation on Satiety Efficiency, Eating Behaviour Traits, and Psychosocial Behaviours in Obese Individuals. Nutrients. 2017; 9(3):284. https://doi.org/10.3390/nu9030284
Chicago/Turabian StyleSanchez, Marina, Christian Darimont, Shirin Panahi, Vicky Drapeau, André Marette, Valerie H. Taylor, Jean Doré, and Angelo Tremblay. 2017. "Effects of a Diet-Based Weight-Reducing Program with Probiotic Supplementation on Satiety Efficiency, Eating Behaviour Traits, and Psychosocial Behaviours in Obese Individuals" Nutrients 9, no. 3: 284. https://doi.org/10.3390/nu9030284
APA StyleSanchez, M., Darimont, C., Panahi, S., Drapeau, V., Marette, A., Taylor, V. H., Doré, J., & Tremblay, A. (2017). Effects of a Diet-Based Weight-Reducing Program with Probiotic Supplementation on Satiety Efficiency, Eating Behaviour Traits, and Psychosocial Behaviours in Obese Individuals. Nutrients, 9(3), 284. https://doi.org/10.3390/nu9030284




