Correlations between Maternal, Breast Milk, and Infant Vitamin B12 Concentrations among Mother–Infant Dyads in Vancouver, Canada and Prey Veng, Cambodia: An Exploratory Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Populations and Biological Sample Collection
2.1.1. Canadian Sample
2.1.2. Cambodian Sample
2.2. Biological Sample Analysis
2.3. Statistical Analysis
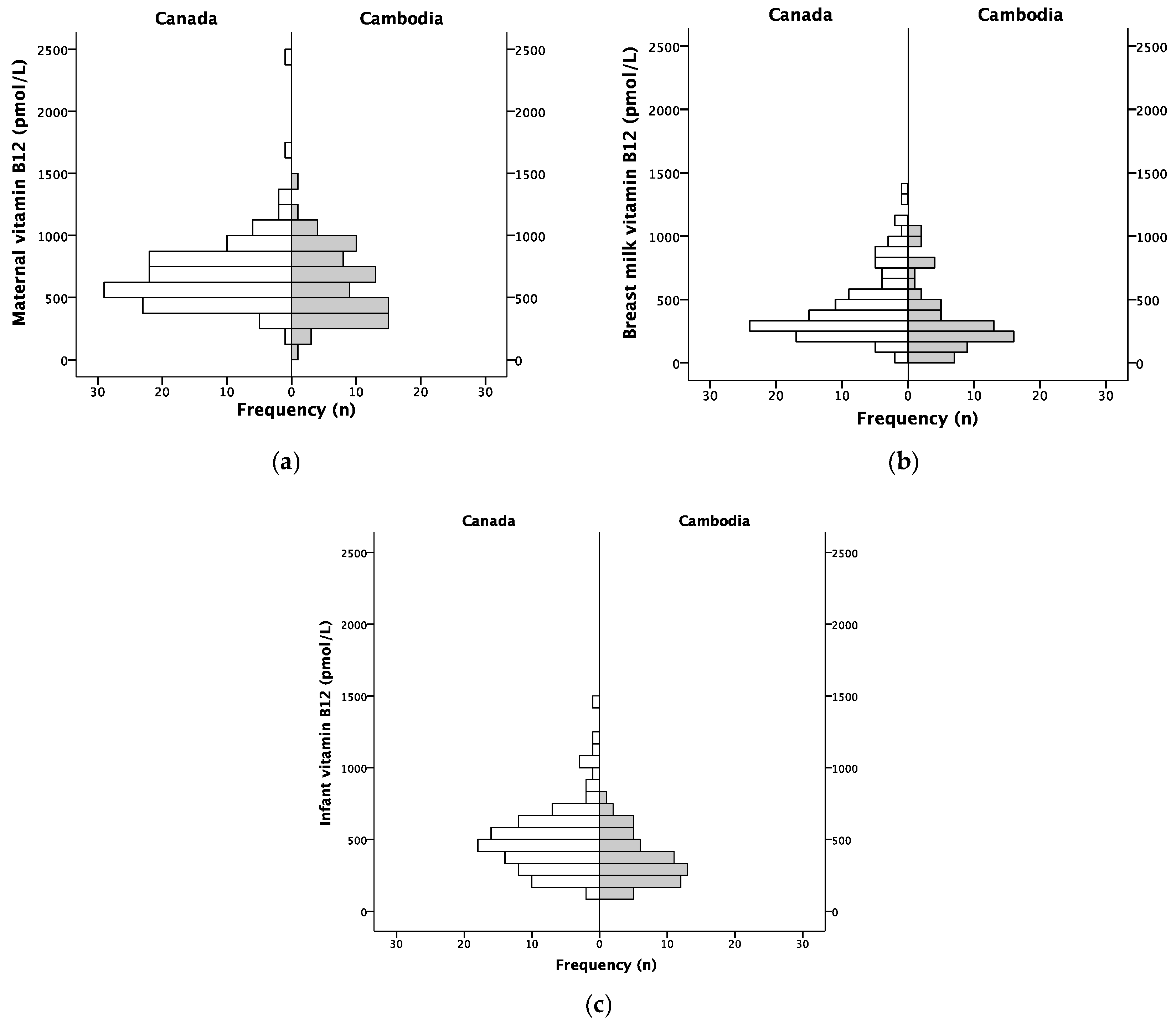
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Allen, L.H. How common is vitamin B-12 deficiency? Am. J. Clin. Nutr. 2009, 89, 693S–696S. [Google Scholar] [CrossRef] [PubMed]
- Dror, D.K.; Allen, L.H. Effect of vitamin B12 deficiency on neurodevelopment in infants: Current knowledge and possible mechanisms. Nutr. Rev. 2008, 66, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.M.; Chantry, C.J.; Young, S.L.; Achando, B.S.; Allen, L.H.; Arnold, B.F.; Colford, J.M.; Dentz, H.N.; Hampel, D.; Kiprotich, M.C.; et al. Vitamin B-12 concentrations in breast milk are low and are not associated with reported household hunger, recent animal-source food, or vitamin B-12 intake in women in rural Kenya. J. Nutr. 2016, 146, 1125–1131. [Google Scholar] [CrossRef] [PubMed]
- Deegan, K.L.; Jones, K.M.; Zuleta, C.; Ramirez-Zea, M.; Lildballe, D.L.; Nexo, E.; Allen, L.H. Breast Milk Vitamin B-12 Concentrations in Guatemalan Women Are Correlated with Maternal but Not Infant Vitamin B-12 Status at 12 Months Postpartum. J. Nutr. 2012, 142, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Allen, L.H. B vitamins in breast milk: Relative importance of maternal status and intake, and effects on infant status and function. Adv. Nutr. 2012, 3, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.S.; Kakuma, R. The Optimal Duration of Exclusive Breastfeeding: A Systematic Review; World Health Organization: Geneva, Switzerland, 2002. [Google Scholar]
- Molloy, A.M.; Kirke, P.N.; Brody, L.C.; Scott, J.M.; Mills, J.L. Effects of folate and vitamin B 12 deficiencies during pregnancy on fetal, infant, and child development. Food Nutr. Bull. 2008, 29, S101–S111. [Google Scholar] [CrossRef] [PubMed]
- Carmel, R. Cobalamin (vitamin B12). In Modern Nutrition in Health and Disease; Ross, A.C., Caballero, B., Cousins, R.J., Tucket, K.L., Ziegler, T.R., Eds.; Lippincott Williams & Wilkins: Baltimore, MD, USA, 2014; pp. 369–389. [Google Scholar]
- Specker, B.L.; Black, A.; Allen, L.H.; Morrow, F. Vitamin B-12: Low milk concentrations are related to low serum concentrations in vegetarian women and to methylmalonic aciduria in their infants. Am. J. Clin. Nutr. 1990, 52, 1073–1076. [Google Scholar] [PubMed]
- Casterline, J.E.; Allen, L.H.; Ruel, M.T. Vitamin B-12 deficiency is very prevalent in lactating Guatemalan women and their infants at three months postpartum. J. Nutr. 1997, 127, 1966–1972. [Google Scholar] [PubMed]
- Greibe, E.; Lildballe, D.L.; Streym, S.; Vestergaard, P.; Rejnmark, L.; Mosekilde, L.; Nexo, E. Cobalamin and haptocorrin in human milk and cobalamin-related variables in mother and child: A 9-mo longitudinal study. Am. J. Clin. Nutr. 2013, 98, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Trugo, N.M.; Donangelo, C.M.; Koury, J.C.; Silva, M.I.; Freitas, L.A. Concentration and distribution pattern of selected micronutrients in preterm and term milk from urban Brazilian mothers during early lactation. Eur. J. Clin. Nutr. 1988, 42, 497–507. [Google Scholar] [PubMed]
- March, K.M.; Chen, N.N.; Karakochuk, C.D.; Shand, A.W.; Innis, S.M.; von Dadelszen, P.; Barr, S.I.; Lyon, M.R.; Whiting, S.J.; Weiler, H.A.; et al. Maternal vitamin D3 supplementation at 50 ug/d protects against low serum 25-hydroxyvitamin D in infants at 8 wk of age: A randomized controlled trial of 3 doses of vitamin D beginning in gestation and continued in lactation. Am. J. Clin. Nutr. 2015, 102, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Whitfield, K.C.; Karakochuk, C.D.; Kroeun, H.; Hampel, D.; Sokhoing, L.; Chan, B.B.; Borath, M.; Sophonneary, P.; McLean, J.; Talukder, A.; et al. Perinatal consumption of thiamine-fortified fish sauce in rural Cambodia: A randomized clinical efficacy trial. JAMA Pediatr. 2016, 170, e162065. [Google Scholar] [CrossRef] [PubMed]
- Hampel, D.; Shahab-Ferdows, S.; Domek, J.M.; Siddiqua, T.; Raqib, R.; Allen, L.H. Competitive chemiluminescent enzyme immunoassay for vitamin B12 analysis in human milk. Food Chem. 2014, 153, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Shahab-Ferdows, S.; Allen, L.H.; Hampel, D.; Bentley, M.E.; Adair, L.S.; Flax, V.L.; Chasela, C.S.; Kacheche, Z.; Tegha, G.; Kourtis, A.P.; et al. Maternal and breastmilk vitamin B12 correlated with infant status but was not influenced by maternal supplementation among HIV-infected Malawian women in the Breastfeeding, Antiretrovirals and Nutrition Study (BAN). FASEB J. 2013, 27, 107.2. [Google Scholar]
- Allen, L.H. Vitamin B12 metabolism and status during pregnancy, lactation and infancy. In Nutrient Regulation during Pregnancy, Lactation, and Infant Growth; Allen, L.H., King, J., Lonnerdal, B., Eds.; Premium Press: New York, NY, USA, 1994; pp. 173–186. [Google Scholar]
- Institute of Medicine Vitamin B12. In Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academy Press: Washington, DC, USA, 1998; pp. 306–356. [Google Scholar]
- United States Department of Agriculture (USDA). Ready-to-Serve Fish Sauce (06179). Available online: https://ndb.nal.usda.gov/ndb/foods/show/1311?fgcd=&manu=&lfacet=&format=&count=&max=50&offset=&sort=default&order=asc&qlookup=fish+sauce&ds= (accessed on 3 October 2016).
- Codex Alimentarius International Food Standards. Standard for Fish Sauce (CODEX STAN 302-2011); Amendment 2012; Codex Alimentarius International Food Standards: Geneva, Switzerland, 2011. [Google Scholar]
- National Institute of Statistics; Directorate General for Health; ICF International. Cambodia Demographic and Health Survey 2014; National Institute of Statistics: Phnom Penh, Cambodia; Rockville, MD, USA, 2015. [Google Scholar]
- Hampel, D.; Shahab-Ferdows, S.; Islam, M.M.; Peerson, J.M.; Allen, L.H. Vitamin concentrations in human milk vary with time within feed, circadian rhythym, and single-dose supplementation. J. Nutr. 2017. [Google Scholar] [CrossRef] [PubMed]
- Fedosov, S.N.; Brito, A.; Miller, J.W.; Green, R.; Allen, L.H. Combined indicator of vitamin B12 status: modification for missing biomarkers and folate status and recommendations for revised cut-points. Clin. Chem. Lab. Med. 2015, 53, 1215–1225. [Google Scholar] [CrossRef] [PubMed]
| Vitamin B12 Marker | n | Vancouver, Canada | n | Prey Veng, Cambodia | p Value |
|---|---|---|---|---|---|
| Maternal vitamin B12 | 124 | 698 (648, 747) | 69 | 620 (552, 687) | 0.009 |
| Deficient (<148) | - | 1 (~1%) | 0.37 | ||
| Marginal (≥148 to 221) | 1 (1%) | 1 (~1%) | |||
| Sufficient (≥221) | 123 (99%) | 67 (97%) | |||
| Infant vitamin B12 | 102 | 506 (459, 552) | 50 | 357 (312, 402) | <0.001 |
| Deficient (<148) | 2 (2%) | 4 (8%) | 0.10 | ||
| Marginal (≥148 to 221) | 4 (4%) | 4 (8%) | |||
| Sufficient (≥221) | 96 (94%) | 42 (84%) | |||
| Breast milk vitamin B12 3 | 109 | 452 (400, 504) | 59 | 317 (256, 378) | <0.001 |
| Vitamin B12 marker | n | Correlation 1 | p Value |
|---|---|---|---|
| Maternal vitamin B12 and breast milk vitamin B12 | |||
| Vancouver, Canada | 109 | 0.498 | <0.001 |
| Prey Veng, Cambodia | 59 | 0.105 | 0.43 |
| Maternal vitamin B12 and infant vitamin B12 | |||
| Vancouver, Canada | 102 | 0.208 | 0.04 |
| Prey Veng, Cambodia | 49 | 0.562 | <0.001 |
| Breast milk vitamin B12 and infant vitamin B12 | |||
| Vancouver, Canada | 88 | 0.370 | <0.001 |
| Prey Veng, Cambodia | 45 | 0.073 | 0.64 |
| Vitamin B12 Marker | Adjusted R2 | Unstandardized | Standardized β | p Value | |
|---|---|---|---|---|---|
| β | 95% CI | ||||
| Maternal plasma vitamin B12 | 0.038 | −0.015 | −0.031, 0.001 | −0.229 | 0.06 |
| Milk vitamin B12 | 0.049 | −0.028 | −0.056, 0.000 | −0.256 | 0.05 |
| Infant plasma vitamin B12 | 0.021 | −0.013 | −0.032, 0.005 | −0.202 | 0.16 |
| Vitamin B12 Marker | n | Infant ≤ 8 weeks | n | Infant > 8 weeks | p Value |
|---|---|---|---|---|---|
| Maternal plasma vitamin B12 | 18 | 634 (464, 803) | 51 | 615 (541, 688) | 0.85 |
| Deficient (<148) | - | - | 0.73 | ||
| Marginal (≥148 to 221) | - | 1 (2%) | |||
| Sufficient (≥221) | 18 (100%) | 50 (98%) | |||
| Infant plasma vitamin B12 | 12 | 343 (253, 433) | 38 | 361 (307, 415) | 0.93 |
| Deficient (<148) | - | 4 (10%) | 0.50 | ||
| Marginal (≥148 to 221) | 1 (7%) | 3 (8%) | |||
| Sufficient (≥221) | 11 (93%) | 31 (82%) | |||
| Breast milk vitamin B12 2 | 13 | 427 (266, 588) | 46 | 286 (222, 351) | 0.051 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chebaya, P.; Karakochuk, C.D.; March, K.M.; Chen, N.N.; Stamm, R.A.; Kroeun, H.; Sophonneary, P.; Borath, M.; Shahab-Ferdows, S.; Hampel, D.; et al. Correlations between Maternal, Breast Milk, and Infant Vitamin B12 Concentrations among Mother–Infant Dyads in Vancouver, Canada and Prey Veng, Cambodia: An Exploratory Analysis. Nutrients 2017, 9, 270. https://doi.org/10.3390/nu9030270
Chebaya P, Karakochuk CD, March KM, Chen NN, Stamm RA, Kroeun H, Sophonneary P, Borath M, Shahab-Ferdows S, Hampel D, et al. Correlations between Maternal, Breast Milk, and Infant Vitamin B12 Concentrations among Mother–Infant Dyads in Vancouver, Canada and Prey Veng, Cambodia: An Exploratory Analysis. Nutrients. 2017; 9(3):270. https://doi.org/10.3390/nu9030270
Chicago/Turabian StyleChebaya, Philip, Crystal D. Karakochuk, Kaitlin M. March, Nancy N. Chen, Rosemary A. Stamm, Hou Kroeun, Prak Sophonneary, Mam Borath, Setareh Shahab-Ferdows, Daniela Hampel, and et al. 2017. "Correlations between Maternal, Breast Milk, and Infant Vitamin B12 Concentrations among Mother–Infant Dyads in Vancouver, Canada and Prey Veng, Cambodia: An Exploratory Analysis" Nutrients 9, no. 3: 270. https://doi.org/10.3390/nu9030270
APA StyleChebaya, P., Karakochuk, C. D., March, K. M., Chen, N. N., Stamm, R. A., Kroeun, H., Sophonneary, P., Borath, M., Shahab-Ferdows, S., Hampel, D., Barr, S. I., Lamers, Y., Houghton, L. A., Allen, L. H., Green, T. J., & Whitfield, K. C. (2017). Correlations between Maternal, Breast Milk, and Infant Vitamin B12 Concentrations among Mother–Infant Dyads in Vancouver, Canada and Prey Veng, Cambodia: An Exploratory Analysis. Nutrients, 9(3), 270. https://doi.org/10.3390/nu9030270





