The Prevalence of Micronutrient Deficiencies and Inadequacies in the Middle East and Approaches to Interventions
Abstract
:1. Introduction
2. Rationale and Approach
Literature Review Approach
- Nutrition, undernutrition, malnutrition, nutrition transition, nutrition status, public health, overweight, obesity, obese, cardiovascular, non-communicable diseases, food consumption
- Prevalence, incidence, burden
- Micronutrient deficiency, vitamins, minerals
- Iron deficiency, folate, folate deficiency, anemia, iron deficiency anemia, iron supplementation
- Vitamin D deficiency, vitamin D insufficiency, vitamin D supplementation
- Vitamin A deficiency, night blindness, vitamin A supplementation
- Dietary supplement, multivitamin mineral supplement, benefit, safety, upper limit, lower limit, clinical, non-clinical
- Fortification, flour, iodine, salt, sodium, vitamins, folic acid, vitamin B12, riboflavin, thiamine, vitamin A, minerals, iron, zinc, calcium
- Healthcare cost, health economics
- Middle East, Egypt, Saudi Arabia, Saudi, KSA, UAE, United Arab Emirates, Kuwait, Bahrain, Qatar, Oman, Lebanon, Jordan, Gulf, GCC, Gulf countries, Arab countries
- Children, preschool, adolescents, adults, men, women, elderly, older adults
3. Prevalence of Micronutrient Deficiencies in the Middle East
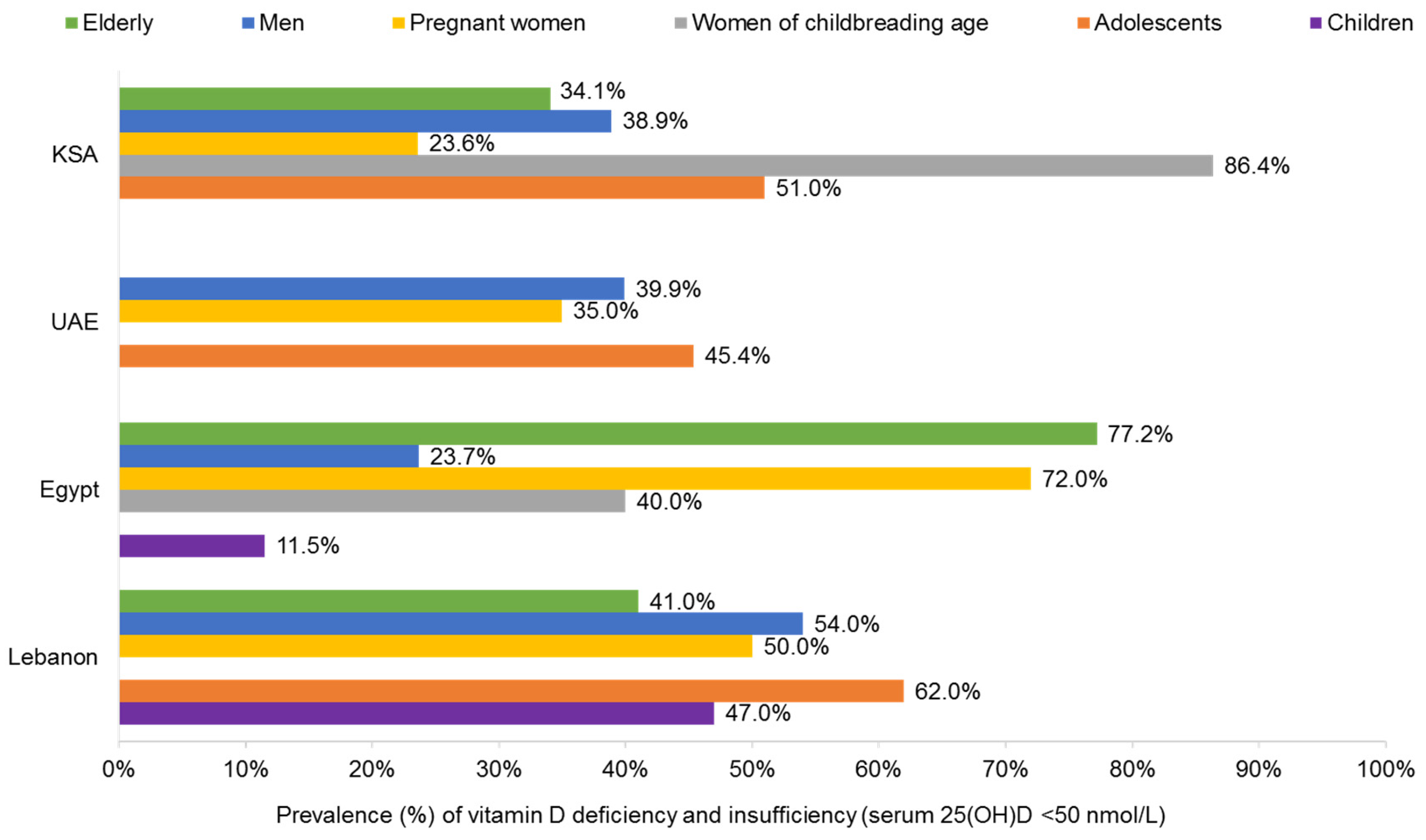
3.1. Vitamin D
3.1.1. Children and Adolescents
3.1.2. Women and Pregnancy
3.1.3. Women of Childbearing Age
3.1.4. Men
3.1.5. Older Adults
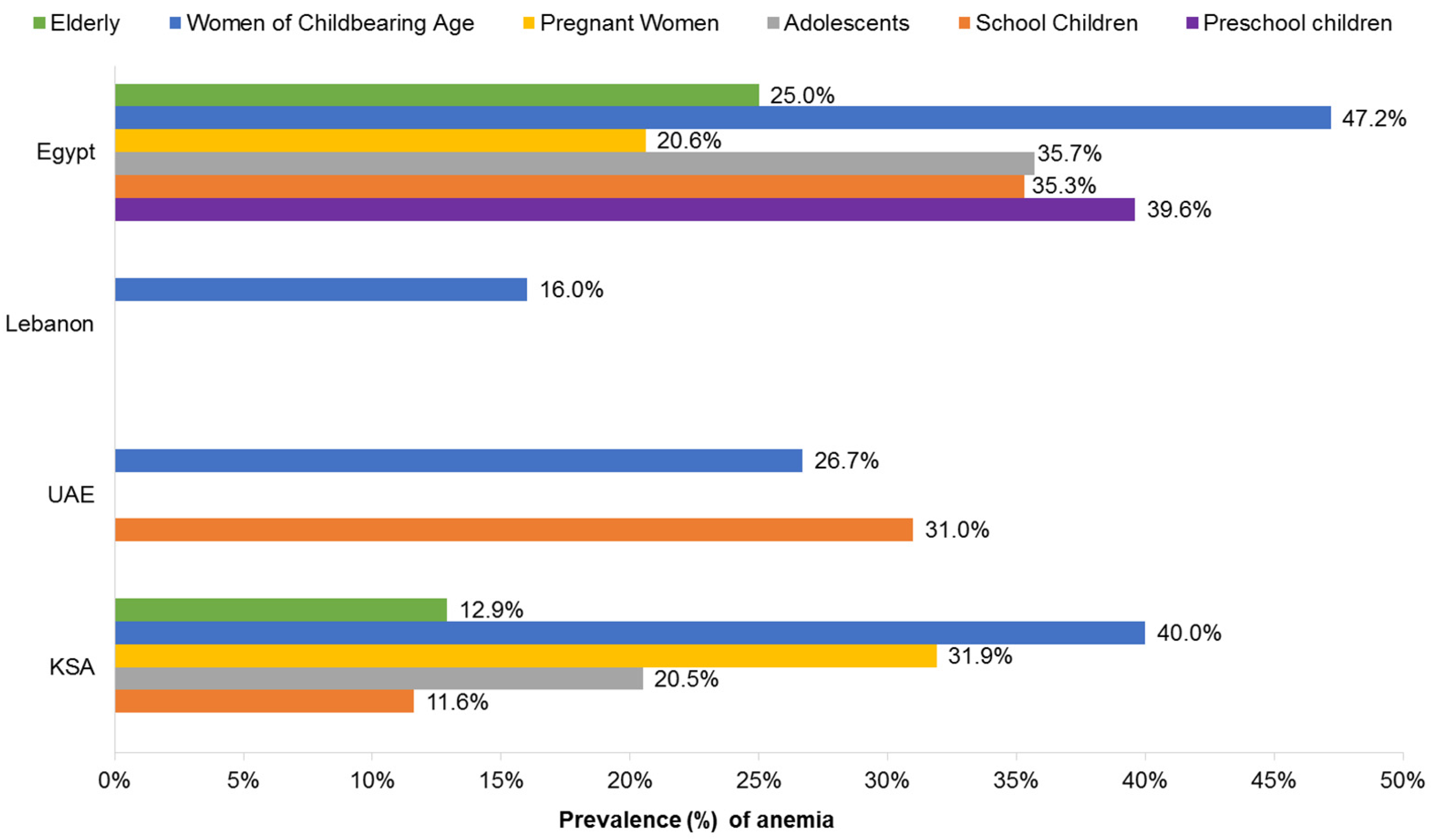
3.2. Iron, Folate, and Anemia
3.2.1. Children and Adolescents
3.2.2. Women and Pregnancy
3.2.3. Women of Childbearing Age
3.2.4. Older Adults
3.2.5. Neural Tube Defects and Folate Deficiency
3.3. Vitamin A
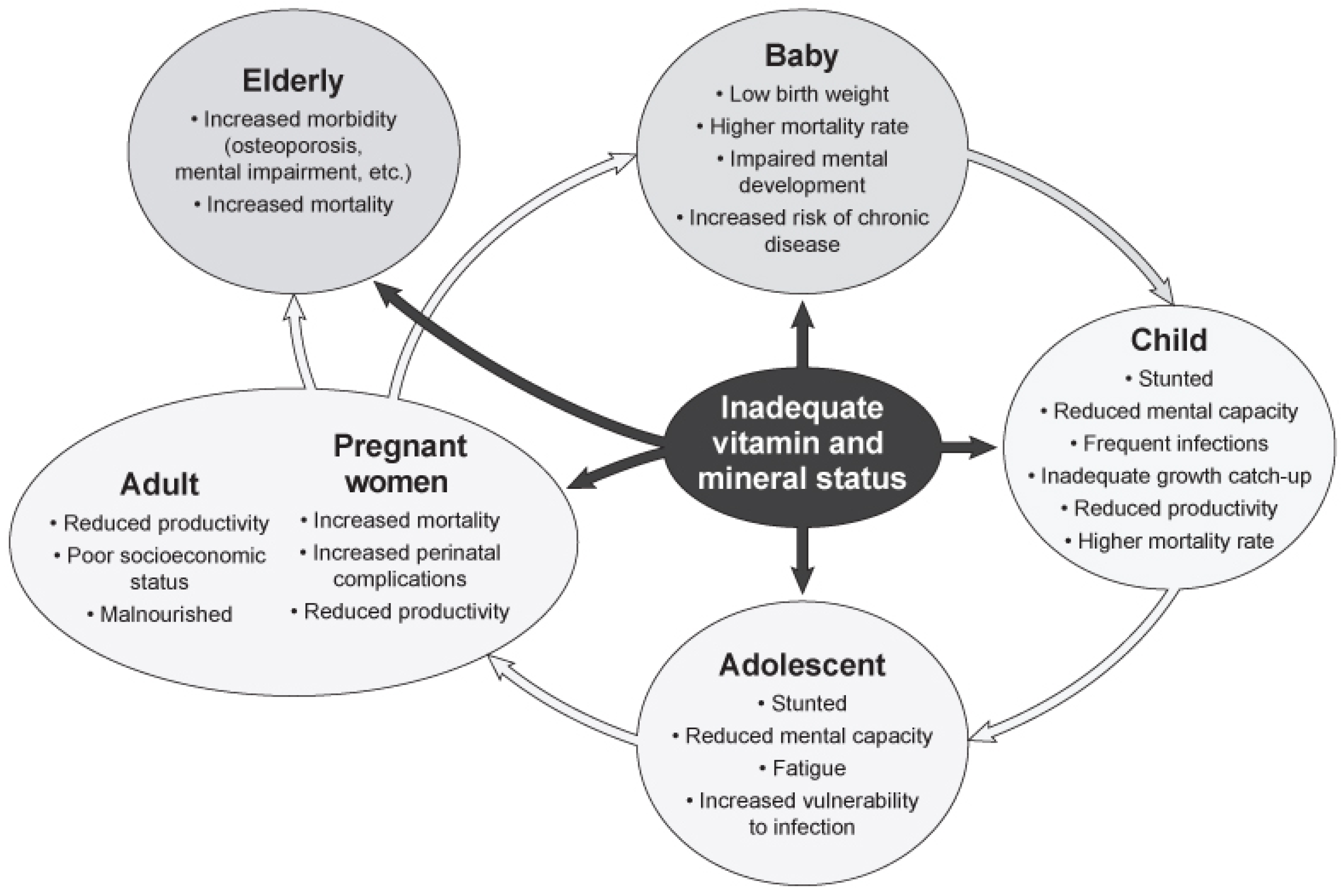
4. Deficient, Inadequate, and Sufficient Micronutrient Intakes
5. Food Fortification and Dietary Supplementation in the Middle East
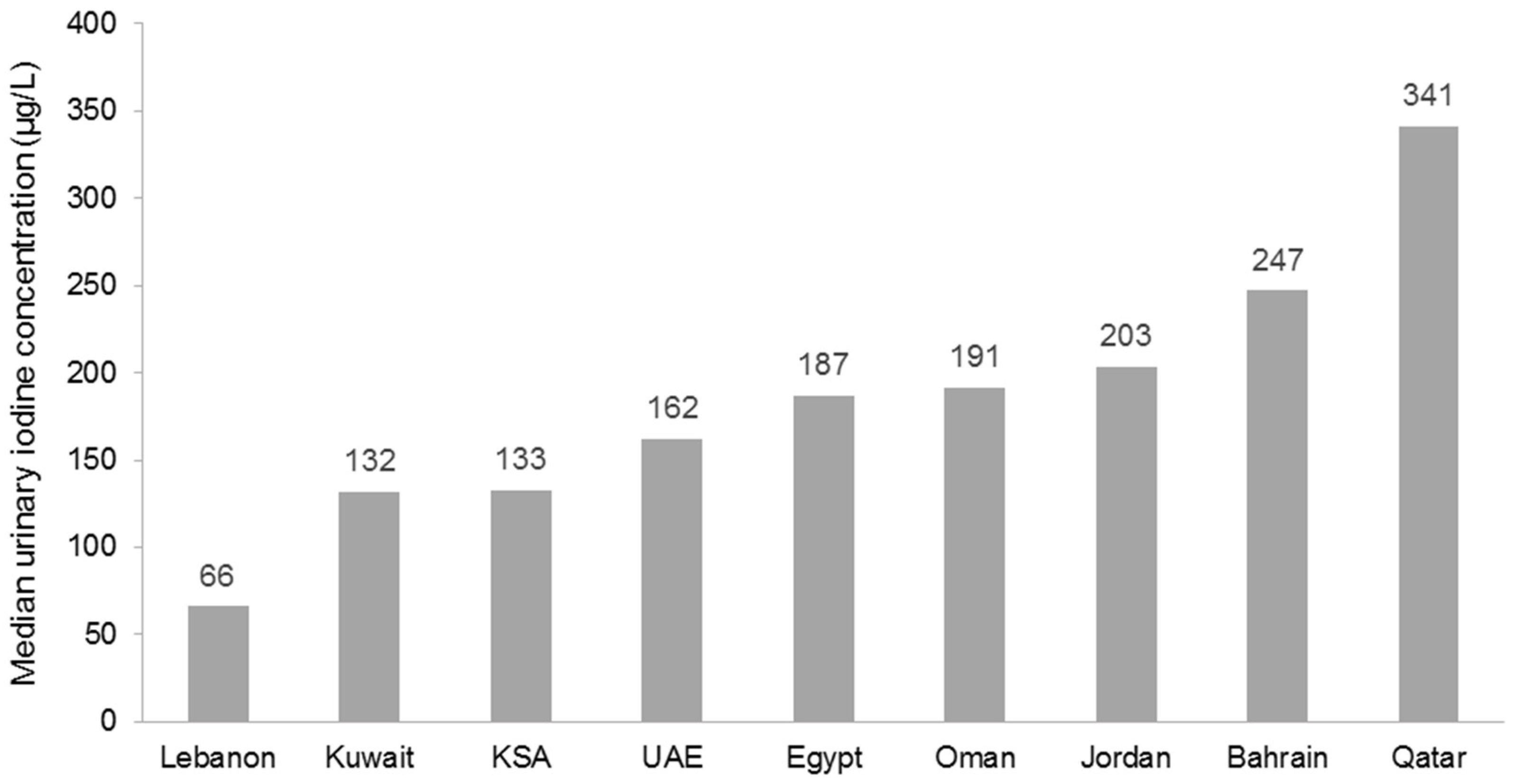
5.1. Fortification Practices in the Middle East
5.2. Dietary Supplementation in the Middle East
6. Benefits and Safety of Dietary Supplementation
6.1. Benefits of Dietary Supplements
6.1.1. Children and Adolescents
6.1.2. Women
6.1.3. Older Adults
6.2. Safety of Dietary Supplements
7. Impact of Malnutrition on Healthcare Costs
8. Proposed Strategies to Address Nutrition and Healthcare Challenges
9. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CMD | cardiometabolic disease |
| CVD | cardiovascular disease |
| EDHS | Egyptian Demographic and Health Survey |
| FAO | Food and Agriculture Organization of the United Nations |
| GDP | Gross Domestic Product |
| Hb | hemoglobin |
| IDA | iron deficiency anemia |
| IU | international units |
| KSA | Kingdom of Saudi Arabia |
| NTD | neural tube defect |
| RCT | randomized controlled trial |
| UAE | United Arab Emirates |
| UNICEF | United Nations Children’s Fund |
| USD | United States dollar |
| WHO | World Health Organization |
References
- World Health Organization. Regional Strategy on Nutrition 2010–2019 and Plan of Action. Available online: http://applications.emro.who.int/dsaf/dsa1230.pdf?ua=1 (accessed on 6 October 2016).
- World Health Organization. Regional Committee for the Eastern Mediterranean. Regional Strategy on Nutrition 2010–2019. EM/RC57/4. Available online: http://applications.emro.who.int/docs/EM_RC57_4_en.pdf (accessed on 6 October 2016).
- Musaiger, A.O.; Hassan, A.S.; Obeid, O. The paradox of nutrition-related diseases in the Arab countries: The need for action. Int. J. Environ. Res. Public Health 2011, 8, 3637–3671. [Google Scholar] [CrossRef] [PubMed]
- Musaiger, A.O.; Arab Centers for Nutrition. The Food Dome: Dietary guidelines for Arab countries. Nutr. Hosp. 2012, 27, 109–115. [Google Scholar] [PubMed]
- General Directorate of Nutrition, Kingdom of Saudi Arabia, Minitry of Health. Dietary Guidelines for Saudis: The Healthy Food Palm. Available online: http://www.moh.gov.sa/ (accessed on 27 October 2016).
- Ward, E. Addressing nutritional gaps with multivitamin and mineral supplements. Nutr. J. 2014, 13, 72. [Google Scholar] [CrossRef] [PubMed]
- Bailey, R.L.; West, K.P., Jr.; Black, R.E. The epidemiology of global micronutrient deficiencies. Ann. Nutr. Metab. 2015, 66, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Fenech, M.F. Dietary reference values of individual micronutrients and nutriomes for genome damage prevention: Current status and a road map to the future. Am. J. Clin. Nutr. 2010, 91, 1438S–1454S. [Google Scholar] [CrossRef] [PubMed]
- Van Ommen, B.; Wopereis, S. Next-generation biomarkers of health. Nestle Nutr. Inst. Workshop Ser. 2016, 84, 25–33. [Google Scholar] [PubMed]
- International Osteroporosis Foundation. Vitamin D Status around the World. Available online: https://www.iofbonehealth.org/facts-and-statistics/vitamin-d-studies-map (accessed on 28 November 2016).
- Al-Daghri, N.M.; Aljohani, N.; Al-Attas, O.S.; Krishnaswamy, S.; Alfawaz, H.; Al-Ajlan, A.; Alokail, M.S. Dairy products consumption and serum 25-hydroxyvitamin D level in Saudi children and adults. Int. J. Clin. Exp. Pathol. 2015, 8, 8480–8486. [Google Scholar] [PubMed]
- Aly, W.W.; Hussein, M.A.; Moahamed Ebeid, S.; Mortagy, A.K. Prevalence of vitamin D insufficiency among community dwelling elderly in Dakahlia as a representative of rural areas in Egypt. Aging Clin. Exp. Res. 2014, 26, 47–51. [Google Scholar] [CrossRef] [PubMed]
- El-Hajj Fuleihan, G.; Nabulsi, M.; Tamim, H.; Maalouf, J.; Salamoun, M.; Khalife, H.; Choucair, M.; Arabi, A.; Vieth, R. Effect of vitamin D replacement on musculoskeletal parameters in school children: A randomized controlled trial. J. Clin. Endocrinol. Metab. 2006, 91, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Al-Faris, N.A. High prevalence of vitamin D deficiency among pregnant Saudi women. Nutrients 2016, 8, 77. [Google Scholar] [CrossRef] [PubMed]
- El-Hajj Fuleihan, G.; Nabulsi, M.; Choucair, M.; Salamoun, M.; Hajj Shahine, C.; Kizirian, A.; Tannous, R. Hypovitaminosis D in healthy schoolchildren. Pediatrics 2001, 107, E53. [Google Scholar] [CrossRef] [PubMed]
- Dawodu, A.; Agarwal, M.; Sankarankutty, M.; Hardy, D.; Kochiyil, J.; Badrinath, P. Higher prevalence of vitamin D deficiency in mothers of rachitic than nonrachitic children. J. Pediatr. 2005, 147, 109–111. [Google Scholar] [CrossRef] [PubMed]
- Al-Jurayyan, N.A.; El-Desouki, M.E.; Al-Herbish, A.S.; Al-Mazyad, A.S.; Al-Qhtani, M.M. Nutritional rickets and osteomalacia in school children and adolescents. Saudi Med. J. 2002, 23, 182–185. [Google Scholar] [PubMed]
- Hoteit, M.; Al-Shaar, L.; Yazbeck, C.; Bou Sleiman, M.; Ghalayini, T.; Fuleihan Gel, H. Hypovitaminosis D in a sunny country: Time trends, predictors, and implications for practice guidelines. Metabolism 2014, 63, 968–978. [Google Scholar] [CrossRef] [PubMed]
- Abu Shady, M.M.; Youssef, M.M.; Salah El-Din, E.M.; Samie, O.M.A.; Megahed, H.S.; Salem, S.M.; Mohsen, M.A.; Aziz, A.A.; El-Toukhy, S. Predictors of serum 25-hydroxyvitamin D concentrations among a sample of Egyptian schoolchildren. Sci. World J. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Muhairi, S.J.; Mehairi, A.E.; Khouri, A.A.; Naqbi, M.M.; Maskari, F.A.; Al Kaabi, J.; Al Dhaheri, A.S.; Nagelkerke, N.; Shah, S.M. Vitamin D deficiency among healthy adolescents in Al Ain, United Arab Emirates. BMC Public Health 2013, 13, 33. [Google Scholar] [CrossRef] [PubMed]
- Al-Mustafa, Z.H.; Al-Madan, M.; Al-Majid, H.J.; Al-Muslem, S.; Al-Ateeq, S.; Al-Ali, A.K. Vitamin D deficiency and rickets in the Eastern Province of Saudi Arabia. Ann. Trop. Paediatr. 2007, 27, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, M.A.; Salhi, H.S.; Bakry, L.A.; Okamoto, E.; Abomelha, A.M.; Stevens, B.; Mousa, F.M. Adolescent rickets in Saudi Arabia: A rich and sunny country. J. Pediatr. Endocrinol. Metab. 2002, 15, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.N.; Alkhenizan, A.H.; El Shaker, M.; Raef, H.; Gabr, A. Increasing trends and significance of hypovitaminosis D: A population-based study in the Kingdom of Saudi Arabia. Arch. Osteoporos. 2014, 9, 190. [Google Scholar] [CrossRef] [PubMed]
- Al-Shaikh, G.K.; Ibrahim, G.H.; Fayed, A.A.; Al-Mandeel, H. Impact of vitamin D deficiency on maternal and birth outcomes in the Saudi population: A cross-sectional study. BMC Pregnancy Childbirth 2016, 16, 119. [Google Scholar] [CrossRef] [PubMed]
- Taha, S.A.; Dost, S.M.; Sedrani, S.H. 25-hydroxyvitamin D and total calcium: Extraordinarily low plasma concentrations in Saudi mothers and their neonates. Pediatr. Res. 1984, 18, 739–741. [Google Scholar] [CrossRef] [PubMed]
- El Rifai, N.M.; Abdel Moety, G.A.; Gaafar, H.M.; Hamed, D.A. Vitamin D deficiency in Egyptian mothers and their neonates and possible related factors. J. Matern. Fetal. Neonatal. Med. 2014, 27, 1064–1068. [Google Scholar] [CrossRef] [PubMed]
- Botros, R.M.; Sabry, I.M.; Abdelbaky, R.S.; Eid, Y.M.; Nasr, M.S.; Hendawy, L.M. Vitamin D deficiency among healthy Egyptian females. Endocrinol. Nutr. 2015, 62, 314–321. [Google Scholar] [CrossRef] [PubMed]
- El Koumi, M.A.; Ali, Y.F.; Abd El Rahman, R.N. Impact of maternal vitamin D status during pregnancy on neonatal vitamin D status. Turk. J. Pediatr. 2013, 55, 371–377. [Google Scholar] [PubMed]
- Dawodu, A.; Absood, G.; Patel, M.; Agarwal, M.; Ezimokhai, M.; Abdulrazzaq, Y.; Khalayli, G. Biosocial factors affecting vitamin D status of women of childbearing age in the United Arab Emirates. J. Biosoc. Sci. 1998, 30, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Haq, A.; Svobodova, J.; Imran, S.; Stanford, C.; Razzaque, M.S. Vitamin D deficiency: A single centre analysis of patients from 136 countries. J. Steroid Biochem. Mol. Biol. 2016, 164, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Al Anouti, F.; Thomas, J.; Abdel-Wareth, L.; Rajah, J.; Grant, W.B.; Haq, A. Vitamin D deficiency and sun avoidance among university students at Abu Dhabi, United Arab Emirates. Dermatoendocrinology 2011, 3, 235–239. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, S.B.; Rao, P.G.; Multani, S.K.; Jain, M. Assessment of prevalence of hypovitaminosis D in multiethnic population of the United Arab Emirates. J. Adv. Pharm. Technol. Res. 2016, 7, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Al Attia, H.M.; Ibrahim, M.A. The high prevalence of vitamin D inadequacy and dress style of women in the sunny UAE. Arch. Osteoporos. 2012, 7, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Gannage-Yared, M.H.; Helou, E.; Zaraket, V.; Abi Akl, S.; Antonios, L.; Moussalli, M.L.; Wakim, S. Serum 25 hydroxyvitamin D in employees of a Middle Eastern university hospital. J. Endocrinol. Investig. 2014, 37, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Abdelkarem, H.M.; El-Sherif, M.A.; Gomaa, S.B. Vitamin D status and insulin resistance among young obese Saudi females. Saudi Med. J. 2016, 37, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Fawzi, M.M.; Swelam, E.; Said, N.S. Plasma levels of 25-hydroxyvitamin D and dress style in a sample of Egyptian female university students. Life Sci. J. 2012, 9, 763–767. [Google Scholar]
- El-Sagheer, G.M.; Soliman, E.; Abdulla, A.M.; Ali, M. Vitamin D deficiency and pseudofractures in child-bearing Egyptian women: Successful medical treatment helps to avoid fractures and surgical interference. Open J. Endocr. Metab. Dis. 2016, 6, 183–191. [Google Scholar] [CrossRef]
- Sadat-Ali, M.; AlElq, A.; Al-Turki, H.; Al-Mulhim, F.; Al-Ali, A. Vitamin D levels in healthy men in eastern Saudi Arabia. Ann. Saudi Med. 2009, 29, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Tuffaha, M.; El Bcheraoui, C.; Daoud, F.; Al Hussaini, H.A.; Alamri, F.; Al Saeedi, M.; Basulaiman, M.; Memish, Z.A.; AlMazroa, M.A.; Al Rabeeah, A.A.; et al. Deficiencies under plenty of sun: Vitamin D status among adults in the Kingdom of Saudi Arabia, 2013. N. Am. J. Med. Sci. 2015, 7, 467–475. [Google Scholar] [PubMed]
- Ardawi, M.S.; Sibiany, A.M.; Bakhsh, T.M.; Qari, M.H.; Maimani, A.A. High prevalence of vitamin D deficiency among healthy Saudi Arabian men: Relationship to bone mineral density, parathyroid hormone, bone turnover markers, and lifestyle factors. Osteoporos. Int. 2012, 23, 675–686. [Google Scholar] [CrossRef] [PubMed]
- Elshafie, D.E.; Al-Khashan, H.I.; Mishriky, A.M. Comparison of vitamin D deficiency in Saudi married couples. Eur. J. Clin. Nutr. 2012, 66, 742–745. [Google Scholar] [CrossRef] [PubMed]
- Sadat-Ali, M.; Al Elq, A.H.; Al-Turki, H.A.; Al-Mulhim, F.A.; Al-Ali, A.K. Influence of vitamin D levels on bone mineral density and osteoporosis. Ann. Saudi Med. 2011, 31, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Gannage-Yared, M.H.; Chemali, R.; Yaacoub, N.; Halaby, G. Hypovitaminosis D in a sunny country: Relation to lifestyle and bone markers. J. Bone Miner. Res. 2000, 15, 1856–1862. [Google Scholar] [CrossRef] [PubMed]
- El Tayeb, I.M.; El-Saghier, E.O.A.; Cheta, S.; Sabbah, N.A.; Raafat, N. Assessment of vitamin D status among senior executive managers in large scale industries in the 10th of Ramadan city, Egypt. IOSR J. Dent. Med. Sci. 2013, 12, 77–83. [Google Scholar]
- Bassil, D.; Rahme, M.; Hoteit, M.; Fuleihan Gel, H. Hypovitaminosis D in the Middle East and North Africa: Prevalence, risk factors and impact on outcomes. Dermatoendocrinology 2013, 5, 274–298. [Google Scholar] [CrossRef] [PubMed]
- Al-Turki, H.A.; Sadat-Ali, M.; Al-Elq, A.H.; Al-Mulhim, F.A.; Al-Ali, A.K. 25-hydoxyvitamin D levels among healthy Saudi Arabian women. Saudi Med. J. 2008, 29, 1765–1768. [Google Scholar] [PubMed]
- Kanan, R.M.; Al Saleh, Y.M.; Fakhoury, H.M.; Adham, M.; Aljaser, S.; Tamimi, W. Year-round vitamin D deficiency among Saudi female out-patients. Public Health Nutr. 2013, 16, 544–548. [Google Scholar] [CrossRef] [PubMed]
- Ardawi, M.S.; Qari, M.H.; Rouzi, A.A.; Maimani, A.A.; Raddadi, R.M. Vitamin D status in relation to obesity, bone mineral density, bone turnover markers and vitamin D receptor genotypes in healthy Saudi pre- and postmenopausal women. Osteoporos. Int. 2011, 22, 463–475. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.H.; Mortagy, A.K. Study of the relationship between vitamin D status and basic functional mobility in ambulatory elderly. Middle East J. Age Ageing 2015, 12, 3–8. [Google Scholar] [CrossRef]
- El Araby, M.; Kamel, H.Y.; Rahman, T.T.A.; Sayed, W.S.; Mortagy, A.K. Vitamin D status in hospitalized male patients in Ain Shams University Hospitals and relation to body composition. Egypt. Rheumatol. Rehabil. 2016, 43, 150–155. [Google Scholar] [CrossRef]
- Gannage-Yared, M.H.; Maalouf, G.; Khalife, S.; Challita, S.; Yaghi, Y.; Ziade, N.; Chalfoun, A.; Norquist, J.; Chandler, J. Prevalence and predictors of vitamin D inadequacy amongst Lebanese osteoporotic women. Br. J. Nutr. 2009, 101, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Alissa, E.M.; Qadi, S.G.; Alhujaili, N.A.; Alshehri, A.M.; Ferns, G.A. Effect of diet and lifestyle factors on bone health in postmenopausal women. J. Bone Miner. Metab. 2011, 29, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Fairfield, K.M.; Fletcher, R.H. Vitamins for chronic disease prevention in adults: Scientific review. JAMA 2002, 287, 3116–3126. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Iron Deficiency Anemia. Assessment, Prevention and Control. A Guide for Programme Managers. Available online: http://apps.who.int/iris/bitstream/10665/66914/1/WHO_NHD_01.3.pdf (accessed on 28 November 2016).
- World Health Organization. Serum Ferritin Concentrations for the Assessment of Iron Status and Iron Deficiency in Populations. Available online: http://www.who.int/vmnis/indicators/serum_ferritin.pdf (accessed on 28 November 2016).
- Elalfy, M.S.; Hamdy, A.M.; Maksoud, S.S.; Megeed, R.I. Pattern of milk feeding and family size as risk factors for iron deficiency anemia among poor Egyptian infants 6 to 24 months old. Nutr. Res. 2012, 32, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Mhanna, R.G.; Rahal, M.; Iskandarani, M.; Hammoudi, D. Incidence and risk factors associated with iron deficiency anaemia among hospitalised Lebanese infants. Int. J. Pharm. Pract. 2016, 24, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Flour Fortification Initiative. Country Profile: Lebanon. Available online: http://www.ffinetwork.org/country_profiles/country.php?record=121 (accessed on 6 October 2016).
- Austin, A.M.; Fawzi, W.; Hill, A.G. Anaemia among Egyptian Children between 2000 and 2005: Trends and predictors. Matern. Child. Nutr. 2012, 8, 522–532. [Google Scholar] [CrossRef] [PubMed]
- Abou-Zeid, A.H.; Abdel-Fattah, M.M.; Al-Shehri, A.S.; Hifnawy, T.M.; Al-Hassan, S.A. Anemia and nutritional status of schoolchildren living at Saudi high altitude area. Saudi Med. J. 2006, 27, 862–869. [Google Scholar] [PubMed]
- Tawfik, A.S.; Hanna, E.T.; Abdel-Maksoud, A.M. Anemia and iron deficiency anemia in Egypt. IOSR J. Pharm. 2015, 5, 30–34. [Google Scholar]
- WHO. The Global Prevalence of Anaemia in 2011. Available online: http://apps.who.int/iris/bitstream/10665/177094/1/9789241564960_eng.pdf (accessed on 6 October 2016).
- Musaiger, A.O.; Abdulghafoor, A.; Radwan, H. Anaemia among 6 year old children in the United Arab Emirates. Eur. J. Clin. Nutr. 1996, 50, 636–637. [Google Scholar] [PubMed]
- Miller, C.J.; Dunn, E.V.; Berg, B.; Abdouni, S.F. A hematological survey of preschool children of the United Arab Emirates. Saudi Med. J. 2003, 24, 609–613. [Google Scholar] [PubMed]
- Babiker, M.A.; Bahakim, H.M.; Al-Omair, A.O.; Al-Jishi, N.; Al-Habib, S.A. Prevalence of iron deficiency in Saudi children from birth to 15 months of age. Ann. Trop. Paediatr. 1989, 9, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Al Hawsawi, Z.M.; Al-Rehali, S.A.; Mahros, A.M.; Al-Sisi, A.M.; Al-Harbi, K.D.; Yousef, A.M. High prevalence of iron deficiency anemia in infants attending a well-baby clinic in northwestern Saudi Arabia. Saudi Med. J. 2015, 36, 1067–1070. [Google Scholar] [CrossRef] [PubMed]
- Abalkhail, B.; Shawky, S. Prevalence of daily breakfast intake, iron deficiency anaemia and awareness of being anaemic among Saudi school students. Int. J. Food Sci. Nutr. 2002, 53, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Alquaiz, A.J.; Khoja, T.A.; Alsharif, A.; Kazi, A.; Mohamed, A.G.; Al Mane, H.; Aldiris, A.; Shaikh, S.A. Prevalence and correlates of anaemia in adolescents in Riyadh city, Kingdom of Saudi Arabia. Public Health Nutr. 2015, 18, 3192–3200. [Google Scholar] [CrossRef] [PubMed]
- Al Ghwass, M.M.; Halawa, E.F.; Sabry, S.M.; Ahmed, D. Iron deficiency anemia in an Egyptian pediatric population: A cross-sectional study. Ann. Afr. Med. 2015, 14, 25–31. [Google Scholar] [PubMed]
- Barakat, A.A.; Nada, K.H.; Ezzat, D.A. Prevalence and determining factors of anemia and malnutrition among Egyptian children. Ind. J. Med. Sci. 2013, 67, 168–177. [Google Scholar]
- Mousa, S.M.O.; Saleh, S.M.; Higazi, A.M.M.; Ali, H.A.A. Iron deficiency and iron deficiency anemia in adolescent girls in rural upper Egypt. IBRR 2016, 5, 1–6. [Google Scholar] [CrossRef]
- Ministry of Health and Population; El-Zanaty and Associates; DHS Program ICF International. Egypt Demographic and Health Survey 2014: Main Findings. Available online: http://dhsprogram.com/pubs/pdf/PR54/PR54.pdf (accessed on 2 November 2016).
- Ministry of Health and Population; El-Zanaty and Associates; The DHS Program ICF International. Egypt Demographic and Health Survey 2014. Available online: https://dhsprogram.com/pubs/pdf/FR302/FR302.pdf (accessed on 2 November 2016).
- Bhutta, Z.A.; Das, J.K.; Rizvi, A.; Gaffey, M.F.; Walker, N.; Horton, S.; Webb, P.; Lartey, A.; Black, R.E.; Lancet Nutrition Interventions Review Group; et al. Evidence-based interventions for improvement of maternal and child nutrition: What can be done and at what cost? Lancet 2013, 382, 452–477. [Google Scholar] [CrossRef]
- Rezk, M.; Marawan, H.; Dawood, R.; Masood, A.; Abo-Elnasr, M. Prevalence and risk factors of iron-deficiency anaemia among pregnant women in rural districts of Menoufia governorate, Egypt. J. Obstet. Gynaecol. 2015, 35, 663–666. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, Z.M.; El-Hamid, A.A.; Mikhail, H.; Khattab, M.S. Assessment of adherence to iron and folic acid supplementation and prevalence of anemia in pregnant women. Med. J. Cairo Univ. 2011, 79, 115–121. [Google Scholar]
- El-Zanaty, F.; Way, A.A. Egypt Demographic and Health Survey 2000. Available online: http://dhsprogram.com/pubs/pdf/FR117/00FrontMatter.pdf (accessed on 2 November 2016).
- El-Zoghby, Z.M.; Way, A.A. Egypt Demographic and Health Survey 2005. Available online: http://www.dhsprogram.com/pubs/pdf/FR176/FR176.pdf (accessed on 2 November 2016).
- Flour Fortification Initiative. Country Profile: Oman. Available online: http://www.ffinetwork.org/country_profiles/country.php?record=221 (accessed on 6 October 2016).
- Flour Fortification Initiative. Country Profile: United Arab Emirates. Available online: http://www.ffinetwork.org/country_profiles/country.php?record=225 (accessed on 6 October 2016).
- Flour Fortification Initiative. Country Profile: Saudi Arabia. Available online: http://www.ffinetwork.org/country_profiles/country.php?record=194 (accessed on 6 October 2016).
- Sultan, A.H. Anemia among female college students attending the University of Sharjah, UAE: Prevalence and classification. J. Egypt. Public Health Assoc. 2007, 82, 261–271. [Google Scholar] [PubMed]
- Alquaiz, A.M.; Gad Mohamed, A.; Khoja, T.A.; Alsharif, A.; Shaikh, S.A.; Al Mane, H.; Aldiris, A.; Kazi, A.; Hammad, D. Prevalence of anemia and associated factors in child bearing age women in Riyadh, Saudi Arabia. J. Nutr. Metab. 2013, 2013, 636585. [Google Scholar] [CrossRef] [PubMed]
- Al-Quaiz, J.M. Iron deficiency anemia. A study of risk factors. Saudi Med. J. 2001, 22, 490–496. [Google Scholar] [PubMed]
- Al Khatib, L.; Obeid, O.; Sibai, A.M.; Batal, M.; Adra, N.; Hwalla, N. Folate deficiency is associated with nutritional anaemia in Lebanese women of childbearing age. Public Health Nutr. 2006, 9, 921–927. [Google Scholar] [CrossRef] [PubMed]
- Alsaeed, A.H. An analysis of hematological parameters to assess the prevalence of anemia in elderly subjects from Saudi Arabia. Genet. Test. Mol. Biomark. 2011, 15, 697–700. [Google Scholar] [CrossRef] [PubMed]
- FAO. Food and Nutrition Paper 84. In The Double Burden of Malnutrition: Case Studies from Six Developing Countries; FAO: Rome, Italy, 2006; Available online: ftp://ftp.fao.org/docrep/fao/009/a0442e/a0442e00.pdf (accessed on 4 November 2016).
- Mahfouz, A.A.; el-Said, M.M.; Alakija, W.; Badawi, I.A.; al-Erian, R.A.; Moneim, M.A. Anemia among pregnant women in the Asir region, Saudi Arabia: An epidemiologic study. Southeast Asian J. Trop. Med. Public Health 1994, 25, 84–87. [Google Scholar] [PubMed]
- Tawfik, A.A.; Hanna, E.T.; Freig, S.A. Folate status in Egypt. IOSR J. Nurs. Health Sci. 2014, 3, 32–36. [Google Scholar] [CrossRef]
- Seidahmed, M.Z.; Abdelbasit, O.B.; Shaheed, M.M.; Alhussein, K.A.; Miqdad, A.M.; Khalil, M.I.; Al-Enazy, N.M.; Salih, M.A. Epidemiology of neural tube defects. Saudi Med. J. 2014, 35, S29–S35. [Google Scholar] [PubMed]
- Safdar, O.Y.; Al-Dabbagh, A.A.; Abuelieneen, W.A.; Kari, J.A. Decline in the incidence of neural tube defects after the national fortification of flour (1997–2005). Saudi Med. J. 2007, 28, 1227–1229. [Google Scholar] [PubMed]
- Al Rakaf, M.S.; Kurdi, A.M.; Ammari, A.N.; Al Hashem, A.M.; Shoukri, M.M.; Garne, E.; Majeed-Saidan, M.A. Patterns of folic acid use in pregnant Saudi women and prevalence of neural tube defects—Results from a nested case-control study. Prev. Med. Rep. 2015, 2, 572–576. [Google Scholar] [CrossRef] [PubMed]
- Flour Fortification Initiative. Country Profile: Qatar. Available online: http://www.ffinetwork.org/country_profiles/country.php?record=179 (accessed on 6 October 2016).
- Flour Fortification Initiative. Country Profile: Kuwait. Available online: http://www.ffinetwork.org/country_profiles/country.php?record=118 (accessed on 6 October 2016).
- Flour Fortification Initiative. Country Profile: Jordan. Available online: http://www.ffinetwork.org/country_profiles/country.php?record=112 (accessed on 6 October 2016).
- Flour Fortification Initiative. Country Profile: Egypt. Available online: http://www.ffinetwork.org/country_profiles/country.php?record=59 (accessed on 6 October 2016).
- Flour Fortification Initiative. Country Profile: Bahrain. Available online: http://www.ffinetwork.org/country_profiles/country.php?record=13 (accessed on 6 October 2016).
- Stevens, G.A.; Bennett, J.E.; Hennocq, Q.; Lu, Y.; de-Regil, L.M.; Rogers, L.; Danaei, G.; Li, G.; White, R.A.; Flaxman, S.R.; et al. Trends and mortality effects of vitamin A deficiency in children in 138 low-income and middle-income countries between 1991 and 2013: A pooled analysis of population-based surveys. Lancet Glob. Health 2015, 3, e528–e536. [Google Scholar] [CrossRef]
- Al-Saleh, I.; Billedo, G.; El-Doush, I.; El-Din Mohamed, G.; Yosef, G. Selenium and vitamins status in Saudi children. Clin. Chim. Acta 2006, 368, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Tolba, A.M.; Hewedy, F.M.; al-Senaidy, A.M.; al-Othman, A.A. Neonates’ vitamin A status in relation to birth weight, gestational age, and sex. J. Trop. Pediatr. 1998, 44, 174–177. [Google Scholar] [CrossRef] [PubMed]
- Al Senaidy, A.M. Serum concentration of retinol, beta-carotene, cholesterol, and triglycerides in Saudi school children. J. Trop. Pediatr. 2000, 46, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Tawfik, A.A.; Gohar, A.S.; Abdel Halim, S.; Abdel Rahman, M.K. Current vitamin A status in Egypt. In Bulletin of the National Nutrition Institute of the Arab Republic of Egypt; National Nutrition Institute: Cairo, Egypt, 2010; pp. 4–12. [Google Scholar]
- Qazaq, H.S.; Aladeeb, N.Z.; Al-Masri, J. Is vitamin A deficiency a problem among pregnant Emirati women in Al-Ain City? East. Mediterr. Health J. 2005, 11, 648–656. [Google Scholar] [PubMed]
- WHO. Global Prevalence of Vitamin A Deficiency in Populations at Risk 1995–2005: WHO Global Database on Vitamin A Deficiency. Available online: http://apps.who.int/iris/bitstream/10665/44110/1/9789241598019_eng.pdf (accessed on 24 February 2016).
- West, K.P., Jr. Extent of vitamin A deficiency among preschool children and women of reproductive age. J. Nutr. 2002, 132, 2857S–2866S. [Google Scholar] [PubMed]
- Huskisson, E.; Maggini, S.; Ruf, M. The role of vitamins and minerals in energy metabolism and well-being. J. Int. Med. Res. 2007, 35, 277–289. [Google Scholar] [CrossRef] [PubMed]
- WHO. Principles and Methods for the Assessment of Risk from Essential Trace Elements. Available online: http://www.inchem.org/documents/ehc/ehc/ehc228.htm (accessed on 27 October 2016).
- Alpers, D.H. Subclinical micronutrient deficiency: A problem in recognition. Curr. Opin. Gastroenterol. 2012, 28, 135–138. [Google Scholar] [CrossRef] [PubMed]
- ACC/SCN in Collaboration with IFPRI. Fourth Report on the World Nutrition Situation: Nutrition throughout the Life Cycle. Available online: http://www.unscn.org/layout/modules/resources/files/rwns4.pdf (accessed on 27 October 2016).
- Fletcher, R.H.; Fairfield, K.M. Vitamins for chronic disease prevention in adults: Clinical applications. JAMA 2002, 287, 3127–3129. [Google Scholar] [CrossRef] [PubMed]
- Assmann, K.E.; Andreeva, V.A.; Jeandel, C.; Hercberg, S.; Galan, P.; Kesse-Guyot, E. Healthy aging 5 years after a period of daily supplementation with antioxidant nutrients: A post hoc analysis of the French randomized trial SU.VI.MAX. Am. J. Epidemiol. 2015, 182, 694–704. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.C.; McBurney, M.; Fulgoni, V.L. Multivitamin/mineral supplement contribution to micronutrient intakes in the United States, 2007–2010. J. Am. Coll. Nutr. 2014, 33, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Fulgoni, V.L., 3rd; Keast, D.R.; Bailey, R.L.; Dwyer, J. Foods, fortificants, and supplements: Where do Americans get their nutrients? J. Nutr. 2011, 141, 1847–1854. [Google Scholar] [CrossRef] [PubMed]
- Ames, B.N. Low micronutrient intake may accelerate the degenerative diseases of aging through allocation of scarce micronutrients by triage. Proc. Natl. Acad. Sci. USA 2006, 103, 17589–17594. [Google Scholar] [CrossRef] [PubMed]
- Supplementation, Food Fortification and Dietary Diversification. A Three-Pronged Approach to Reducing Hidden Hunger. Available online: https://www.bmz.de/en/zentrales_downloadarchiv/themen_und_schwerpunkte/ernaehrung/food_fortification.pdf (accessed on 6 October 2016).
- WHO. Joint WHO/Flour Fortification Initiative Harmonization Workshop for Wheat and Maize Flour Fortification; Document WHO-EM/NUT/259/E; WHO: Amman, Jordan, 20–22 February 2012; Available online: http://applications.emro.who.int/docs/IC_Meet_Rep_2012_EN_14767.pdf (accessed on 6 October 2016).
- Global Iodine Nutrition Scorecard 2015. Available online: http://ign.org/cm_data/Scorecard_2015_August_26.pdf (accessed on 6 September 2016).
- Ahmed, A.; Khalid, N.; David, A.; Sandhu, M.A.; Randhawa, M.A.; Suleria, H.A. A question mark on iron deficiency in 185 million people of Pakistan: Its outcomes and prevention. Crit. Rev. Food Sci. Nutr. 2014, 54, 1617–1635. [Google Scholar] [CrossRef] [PubMed]
- Sultan, S.; Anjum, F.M.; Butt, M.S.; Huma, N.; Suleria, H.A. Concept of double salt fortification; a tool to curtail micronutrient deficiencies and improve human health status. J. Sci. Food Agric. 2014, 94, 2830–2838. [Google Scholar] [CrossRef] [PubMed]
- Al-Akhfash, A.A.; Abdulla, A.M.; Osman, A.M.; Abdulgafar, J.I.; Almesned, A.A. Maternal knowledge and use of folic acid among Saudi females. Saudi Med. J. 2013, 34, 1173–1178. [Google Scholar] [PubMed]
- Kari, J.A.; Bardisi, E.S.; Baitalmal, R.M.; Ageely, G.A. Folic acid awareness among female college students: Neural tube defects prevention. Saudi Med. J. 2008, 29, 1749–1751. [Google Scholar] [PubMed]
- Abdulrazzaq, Y.M.; Al-Gazali, L.I.; Bener, A.; Hossein, M.; Verghese, M.; Dawodu, A.; Padmanabhan, R. Folic acid awareness and intake survey in the United Arab Emirates. Reprod. Toxicol. 2003, 17, 171–176. [Google Scholar] [CrossRef]
- Al-Darzi, W.; Al-Mudares, F.; Farah, A.; Ali, A.; Marzouk, D. Knowledge of periconceptional folic acid use among pregnant women at Ain Shams University Hospital, Cairo, Egypt. East. Mediterr. Health J. 2014, 20, 561–568. [Google Scholar] [PubMed]
- Nasr Hage, C.; Jalloul, M.; Sabbah, M.; Adib, S.M. Awareness and intake of folic acid for the prevention of neural tube defects among Lebanese women of childbearing age. Matern. Child. Health J. 2012, 16, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Tamim, H.; Harrison, G.; Atoui, M.; Mumtaz, G.; El-Kak, F.; Seoud, M.; Yunis, K.; National Collaborative Perinatal Neonatal Network. Preconceptional folic acid supplement use in Lebanon. Public Health Nutr. 2009, 12, 687–692. [Google Scholar] [CrossRef] [PubMed]
- Abdulrazzaq, Y.M.; Nagelkerke, N.; Abdulla, S.; Belhaj, G. Nutrient intake of infants and toddlers in the United Arab Emirates: The Feeding Infants and Toddlers Study. East. Mediterr. Health J. 2016, 22, 293–300. [Google Scholar] [PubMed]
- Egypt Nutritional Landscape Analysis Report 2012. Available online: http://www.unicef.org/egypt/Landscape_Anaylsis_Report_January_2013.pdf (accessed on 6 October 2016).
- Allam, A.R.; Taha, I.M.; Al-Nozha, O.M.; Sultan, I.E. Nutritional and health status of medical students at a university in Northwestern Saudi Arabia. Saudi Med. J. 2012, 33, 1296–1303. [Google Scholar] [PubMed]
- Wessells, K.R.; Brown, K.H. Estimating the global prevalence of zinc deficiency: Results based on zinc availability in national food supplies and the prevalence of stunting. PLoS ONE 2012, 7, e50568. [Google Scholar] [CrossRef] [PubMed]
- De-Regil, L.M.; Palacios, C.; Lombardo, L.K.; Pena-Rosas, J.P. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2016, 1. [Google Scholar] [CrossRef]
- Roberfroid, D.; Huybregts, L.; Lanou, H.; Ouedraogo, L.; Henry, M.C.; Meda, N.; Kolsteren, P.; MISAME Study Group. Impact of prenatal multiple micronutrients on survival and growth during infancy: A randomized controlled trial. Am. J. Clin. Nutr. 2012, 95, 916–924. [Google Scholar] [CrossRef] [PubMed]
- Ter Borg, S.; Verlaan, S.; Hemsworth, J.; Mijnarends, D.M.; Schols, J.M.; Luiking, Y.C.; de Groot, L.C. Micronutrient intakes and potential inadequacies of community-dwelling older adults: A systematic review. Br. J. Nutr. 2015, 113, 1195–1206. [Google Scholar] [CrossRef] [PubMed]
- McNulty, H.; Pentieva, K.; Hoey, L.; Ward, M. Homocysteine, B-vitamins and CVD. Proc. Nutr. Soc. 2008, 67, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Mason, P. One is okay, more is better? Pharmacological aspects and safe limits of nutritional supplements. Proc. Nutr. Soc. 2007, 66, 493–507. [Google Scholar] [CrossRef] [PubMed]
- Mulholland, C.A.; Benford, D.J. What is known about the safety of multivitamin-multimineral supplements for the generally healthy population? Theoretical basis for harm. Am. J. Clin. Nutr. 2007, 85, 318s–322s. [Google Scholar] [PubMed]
- Datta, M.; Vitolins, M.Z. Food fortification and supplement use - are there health implications? Crit. Rev. Food Sci. Nutr. 2016, 56, 2149–2159. [Google Scholar] [CrossRef] [PubMed]
- Bjelakovic, G.; Nikolova, D.; Gluud, C. Antioxidant supplements to prevent mortality. JAMA 2013, 310, 1178–1179. [Google Scholar] [CrossRef] [PubMed]
- Mursu, J.; Robien, K.; Harnack, L.J.; Park, K.; Jacobs, D.R., Jr. Dietary supplements and mortality rate in older women: The Iowa Women’s Health Study. Arch. Intern. Med. 2011, 171, 1625–1633. [Google Scholar] [CrossRef] [PubMed]
- Macpherson, H.; Pipingas, A.; Pase, M.P. Multivitamin-multimineral supplementation and mortality: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2013, 97, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.N. Drug-nutrient interaction in clinical nutrition. Curr. Opin. Clin. Nutr. Metab. Care 2002, 5, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Horton, S.; Steckel, R.H. Malnutrition. Global Economic Losses Attributable to Malnutrition 1900–2000 and Projections to 2050. Assessment Paper Copenhagen Consensus on Human Challenges 2011. Available online: http://www.copenhagenconsensus.com/sites/default/files/malnutrition.pdf (accessed on 10 April 2016).
- Vitamin and Mineral Deficiency: A Global Progress Report. Available online: http://www.unicef.org/media/files/vmd.pdf (accessed on 7 April 2016).
- Investing in the Future. A United Call to Action on Vitamin and Mineral Deficiencies. Global Report 2009. Available online: http://www.unitedcalltoaction.org/documents/Investing_in_the_future.pdf (accessed on 24 March 2016).
- Berti, C.; Faber, M.; Smuts, C.M. Prevention and control of micronutrient deficiencies in developing countries: Current perspectives. Nutr. Diet. Suppl. 2014, 6, 41–57. [Google Scholar]
| Country | Preschool Children | Pregnant Women | ||
|---|---|---|---|---|
| Prevalence of Night Blindness | Prevalence of Vitamin A Deficiency (<0.7 µmol/L) | Prevalence of Night Blindness | Prevalence of Vitamin A Deficiency (<0.7 µmol/L) | |
| Lebanon | 0.6% | 2% | 3.7% | 3% |
| Jordan | 0.6% | 4% | 4.4% | 7% |
| Egypt | 0.1% | 9% | 9.4% b | 10.2% b |
| KSA | 0.4% | 12% | 5.1% | 32% |
| Oman | 0.4 % | 1 % | % | 3 % |
| Country | Iron | Zinc | Folic Acid | B12 | Niacin | Riboflavin | Thiamine | Vitamin A | Percent Flour Fortified |
|---|---|---|---|---|---|---|---|---|---|
| Lebanon | No fortification | ||||||||
| Jordan | 32.25 (ferrous sulfate) | 20 | 1 | 0.007 | 35 | 3.6 | 3.575 | 1.5 | 100% (mandatory) |
| Egypt | 30 (ferrous sulfate) | - | 1.5 | - | - | - | - | - | Unavailable |
| KSA | 36.3 (type unknown) | - | 1.5 | - | 52.9 | 3.96 | 6.38 | - | 100% (mandatory) |
| UAE | 60 (electrolyte) | - | 1.5 | - | - | - | - | - | 90% (mandatory) |
| Oman | 60 (electrolyte) | - | 1.5–2.0 | - | - | - | - | - | 89% (mandatory) |
| Bahrain | 60 (electrolyte) | - | 1.5 | - | - | - | - | - | 90% (mandatory) |
| Qatar | 60 (electrolyte) | - | 1.5 | - | - | - | - | - | 90% (voluntary) |
| Kuwait | 60 (electrolyte) | - | 1.5 | - | 52.91 | 3.96 | 6.38 | 1.5 | 100% (mandatory) |
| Country | Neural Tube Defects per 10,000 births | Percent Anemia in Non-Pregnant Women of Reproductive Age | Percent Anemia in Pre-School Children |
|---|---|---|---|
| Lebanon | 18.0 | 28 | 24 |
| Jordan | 33.0 | 29 | 31 |
| Egypt | 10 | 35 | 45 |
| KSA | 12.0 | 40 | 39 |
| UAE | 12.0 | 26 | 29 |
| Oman | 12 | 35 | 41 |
| Bahrain | 11.8 | 38 | 32 |
| Qatar | 12 | 28 | 26 |
| Kuwait | 12 | 22 | 26 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hwalla, N.; Al Dhaheri, A.S.; Radwan, H.; Alfawaz, H.A.; Fouda, M.A.; Al‐Daghri, N.M.; Zaghloul, S.; Blumberg, J.B. The Prevalence of Micronutrient Deficiencies and Inadequacies in the Middle East and Approaches to Interventions. Nutrients 2017, 9, 229. https://doi.org/10.3390/nu9030229
Hwalla N, Al Dhaheri AS, Radwan H, Alfawaz HA, Fouda MA, Al‐Daghri NM, Zaghloul S, Blumberg JB. The Prevalence of Micronutrient Deficiencies and Inadequacies in the Middle East and Approaches to Interventions. Nutrients. 2017; 9(3):229. https://doi.org/10.3390/nu9030229
Chicago/Turabian StyleHwalla, Nahla, Ayesha Salem Al Dhaheri, Hadia Radwan, Hanan Abdullah Alfawaz, Mona A. Fouda, Nasser Mohammed Al‐Daghri, Sahar Zaghloul, and Jeffrey B. Blumberg. 2017. "The Prevalence of Micronutrient Deficiencies and Inadequacies in the Middle East and Approaches to Interventions" Nutrients 9, no. 3: 229. https://doi.org/10.3390/nu9030229
APA StyleHwalla, N., Al Dhaheri, A. S., Radwan, H., Alfawaz, H. A., Fouda, M. A., Al‐Daghri, N. M., Zaghloul, S., & Blumberg, J. B. (2017). The Prevalence of Micronutrient Deficiencies and Inadequacies in the Middle East and Approaches to Interventions. Nutrients, 9(3), 229. https://doi.org/10.3390/nu9030229









