Dietary Patterns of European Children and Their Parents in Association with Family Food Environment: Results from the I.Family Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Questionnaires and Anthropometric Measurements
2.3. Dietary Information
2.4. Dietary Data Analysis
2.5. Statistical Analysis
3. Results
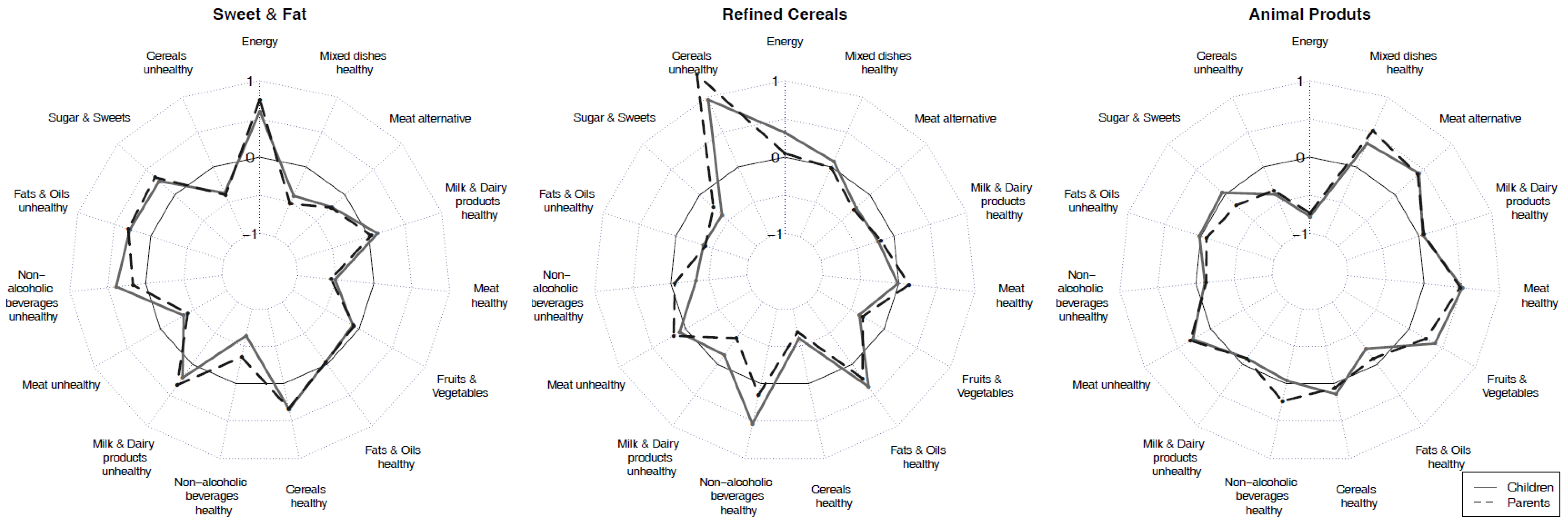
3.1. Dietary Clusters
3.2. Participant Characteristics
3.3. Family Food Environment
4. Discussion
Limitations and Strengths
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
Appendix A.1. Derivation of z-Scores
Appendix A.2. Clustering Procedure
| Parents | Children, Adolescents Sweet & Fat Ref. Cereals Animal Products | ||
|---|---|---|---|
| Sweet & Fat | 0.45 | 2.54 | 2.45 |
| Ref. Cereals | 2.7 | 0.77 | 2.51 |
| Animal Products | 2.48 | 2.25 | 0.49 |
References
- Patrick, H.; Nicklas, T.A. A review of family and social determinants of children’s eating patterns and diet quality. J. Am. Coll. Nutr. 2005, 24, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K.J.; Crawford, D.A.; Ball, K. Family food environment and dietary behaviors likely to promote fatness in 5–6 year-old children. Int. J. Obes. (Lond.) 2006, 30, 1272–1280. [Google Scholar] [CrossRef] [PubMed]
- Draxten, M.; Fulkerson, J.A.; Friend, S.; Flattum, C.F.; Schow, R. Parental role modeling of fruits and vegetables at meals and snacks is associated with children’s adequate consumption. Appetite 2014, 78, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.; Ogden, J. Children’s eating attitudes and behaviour: A study of the modelling and control theories of parental influence. Health Educ. Res. 2004, 19, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.; van Jaarsveld, C.H.; Wardle, J. Individual and family environment correlates differ for consumption of core and non-core foods in children. Br. J. Nutr. 2011, 105, 950–959. [Google Scholar] [CrossRef] [PubMed]
- Freeman, E.; Fletcher, R.; Collins, C.E.; Morgan, P.J.; Burrows, T.; Callister, R. Preventing and treating childhood obesity: Time to target fathers. Int. J. Obes. (Lond.) 2012, 36, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Robinson, L.N.; Rollo, M.E.; Watson, J.; Burrows, T.L.; Collins, C.E. Relationships between dietary intakes of children and their parents: A cross-sectional, secondary analysis of families participating in the family diet quality study. J. Hum. Nutr. Diet. 2014, 28, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Raynor, H.A.; Van Walleghen, E.L.; Osterholt, K.M.; Hart, C.N.; Jelalian, E.; Wing, R.R.; Goldfield, G.S. The relationship between child and parent food hedonics and parent and child food group intake in children with overweight/obesity. J. Am. Diet. Assoc. 2011, 111, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Hall, L.; Collins, C.E.; Morgan, P.J.; Burrows, T.L.; Lubans, D.R.; Callister, R. Children’s intake of fruit and selected energy-dense nutrient-poor foods is associated with fathers’ intake. J. Am. Diet. Assoc. 2011, 111, 1039–1044. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Beydoun, M.A.; Li, J.; Liu, Y.; Moreno, L.A. Do children and their parents eat a similar diet? Resemblance in child and parental dietary intake: Systematic review and meta-analysis. J. Epidemiol. Community Health 2011, 65, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.O.; Mitchell, D.C.; Smiciklas-Wright, H.; Birch, L.L. Parental influences on young girls’ fruit and vegetable, micronutrient, and fat intakes. J. Am. Diet. Assoc. 2002, 102, 58–64. [Google Scholar] [CrossRef]
- Grimm, G.C.; Harnack, L.; Story, M. Factors associated with soft drink consumption in school-aged children. J. Am. Diet. Assoc. 2004, 104, 1244–1249. [Google Scholar] [CrossRef] [PubMed]
- Hanson, N.I.; Neumark-Sztainer, D.; Eisenberg, M.E.; Story, M.; Wall, M. Associations between parental report of the home food environment and adolescent intakes of fruits, vegetables and dairy foods. Public Health Nutr. 2005, 8, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Martens, M.K.; van Assema, P.; Brug, J. Why do adolescents eat what they eat? Personal and social environmental predictors of fruit, snack and breakfast consumption among 12–14-year-old dutch students. Public Health Nutr. 2005, 8, 1258–1265. [Google Scholar] [CrossRef] [PubMed]
- Ray, C.; Roos, E.; Brug, J.; Behrendt, I.; Ehrenblad, B.; Yngve, A.; Te Velde, S.J. Role of free school lunch in the associations between family-environmental factors and children’s fruit and vegetable intake in four european countries. Public Health Nutr. 2013, 16, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Pearson, N.; Biddle, S.J.; Gorely, T. Family correlates of breakfast consumption among children and adolescents. A systematic review. Appetite 2009, 52, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Hannon, P.A.; Bowen, D.J.; Moinpour, C.M.; McLerran, D.F. Correlations in perceived food use between the family food preparer and their spouses and children. Appetite 2003, 40, 77–83. [Google Scholar] [CrossRef]
- Oliveria, S.A.; Ellison, R.C.; Moore, L.L.; Gillman, M.W.; Garrahie, E.J.; Singer, M.R. Parent-child relationships in nutrient intake: The framingham children’s study. Am. J. Clin. Nutr. 1992, 56, 593–598. [Google Scholar] [PubMed]
- Neumark-Sztainer, D.; Story, M.; Resnick, M.D.; Blum, R.W. Correlates of inadequate fruit and vegetable consumption among adolescents. Prev. Med. 1996, 25, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Fiese, B.H.; Hammons, A.; Grigsby-Toussaint, D. Family mealtimes: A contextual approach to understanding childhood obesity. Econ. Hum. Biol. 2012, 10, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Welsh, E.M.; French, S.A.; Wall, M. Examining the relationship between family meal frequency and individual dietary intake: Does family cohesion play a role? J. Nutr. Educ. Behav. 2011, 43, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Expert Group on Health Information. The European Core Health Indicators Shortlist. Available online: http://ec.europa.eu/health/indicators/docs/echi_shortlist_by_policy_area_en.pdf (accessed on 7 February 2017).
- Fismen, A.; Smith, O.R.F.; Torsheim, T.; Rasmussen, M.; Pagh, T.P.; Augustine, L.; Ojala, K.; Samdal, O. Trends in food habits and their relation to socioeconomic status among nordic adolescents 2001/2002-2009/2010. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Mathieson, A.; Koller, T. Addressing the Socioeconomic Determinants of Healthy Eating Habits and Physical Activity Levels among Adolescents; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- Pinard, C.A.; Davy, B.M.; Estabrooks, P.A. Beverage intake in low-income parent-child dyads. Eat. Behav. 2011, 12, 313–316. [Google Scholar] [CrossRef] [PubMed]
- Ahrens, W.; Siani, A.; de Henauw, S.; Eiben, G.; Gwozdz, W.; Hebestreit, A.; Hunsberger, M.; Kaprio, J.; Krogh, V.; Lissner, L.; et al. Cohort profile: The transition from childhood to adolescence in european children-how I.Family extends the IDEFICS cohort. Int. J. Epidemiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- UNESCO. International Standard Classification of Education. Available online: http://www.uis.unesco.org/Education/Documents/isced-2011-en.pdf (accessed on 7 February 2017).
- Stomfai, S.; Ahrens, W.; Bammann, K.; Kovacs, E.; Marild, S.; Michels, N.; Moreno, L.A.; Pohlabeln, H.; Siani, A.; Tornaritis, M.; et al. Intra- and inter-observer reliability in anthropometric measurements in children. Int. J. Obes. (Lond.) 2011, 35 (Suppl. S1), S45–S51. [Google Scholar] [CrossRef] [PubMed]
- Suling, M.; Hebestreit, A.; Peplies, J.; Bammann, K.; Nappo, A.; Eiben, G.; Alvira, J.M.; Verbestel, V.; Kovacs, E.; Pitsiladis, Y.P.; et al. Design and results of the pretest of the IDEFICS study. Int. J. Obes. (Lond.) 2011, 35 (Suppl. S1), S30–S44. [Google Scholar] [CrossRef] [PubMed]
- Cole, T.J.; Freeman, J.V.; Preece, M.A. British 1990 growth reference centiles for weight, height, body mass index and head circumference fitted by maximum penalized likelihood. Stat. Med. 1998, 17, 407–429. [Google Scholar] [CrossRef]
- Cole, T.J.; Lobstein, T. Extended international (IOTF) body mass index cut-offs for thinness, overweight and obesity. Pediatr. Obes. 2012, 7, 284–294. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The International Classification of Adult Underweight, Overweight and Obesity according to BMI. Available online: http://apps.who.int/bmi/index.jsp?introPage=intro_3.html (accessed on 7 February 2017).
- Hebestreit, A.; Börnhorst, C.; Barba, G.; Siani, A.; Huybrechts, I.; Tognon, G.; Eiben, G.; Moreno, L.A.; Fernandez Alvira, J.M.; Loit, H.M.; et al. Associations between energy intake, daily food intake and energy density of foods and BMI z-score in 2–9-year-old european children. Eur. J. Nutr. 2014, 53, 673–681. [Google Scholar] [CrossRef] [PubMed]
- Livingstone, M.B.; Robson, P.J. Measurement of dietary intake in children. Proc. Nutr. Soc. 2000, 59, 279–293. [Google Scholar] [CrossRef] [PubMed]
- Hebestreit, A.; Barba, G.; De Henauw, S.; Eiben, G.; Hadjigeorgiou, C.; Kovacs, E.; Krogh, V.; Moreno, L.A.; Pala, V.; Veidebaum, T.; et al. Cross-sectional and longitudinal associations between energy intake and BMI z-score in european children. Int. J. Behav. Nutr. Phys. Act. 2016, 13, 23. [Google Scholar] [CrossRef] [PubMed]
- Börnhorst, C.; Huybrechts, I.; Ahrens, W.; Eiben, G.; Michels, N.; Pala, V.; Molnar, D.; Russo, P.; Barba, G.; Bel-Serrat, S.; et al. Prevalence and determinants of misreporting among european children in proxy-reported 24 h dietary recalls. Br. J. Nutr. 2012, 109, 1257–1265. [Google Scholar] [CrossRef] [PubMed]
- Tooze, J.A.; Midthune, D.; Dodd, K.W.; Freedman, L.S.; Krebs-Smith, S.M.; Subar, A.F.; Guenther, P.M.; Carroll, R.J.; Kipnis, V. A new statistical method for estimating the usual intake of episodically consumed foods with application to their distribution. J. Am. Diet. Assoc. 2006, 106, 1575–1587. [Google Scholar] [CrossRef] [PubMed]
- Kipnis, V.; Midthune, D.; Buckman, D.W.; Dodd, K.W.; Guenther, P.M.; Krebs-Smith, S.M.; Subar, A.F.; Tooze, J.A.; Carroll, R.J.; Freedman, L.S. Modeling data with excess zeros and measurement error: Application to evaluating relationships between episodically consumed foods and health outcomes. Biometrics 2009, 65, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- Lanfer, A.; Hebestreit, A.; Ahrens, W.; Krogh, V.; Sieri, S.; Lissner, L.; Eiben, G.; Siani, A.; Huybrechts, I.; Loit, H.M.; et al. Reproducibility of food consumption frequencies derived from the children’s eating habits questionnaire used in the IDEFICS study. Int. J. Obes. (Lond.) 2011, 35 (Suppl. S1), S61–S68. [Google Scholar] [CrossRef] [PubMed]
- Bel-Serrat, S.; Mouratidou, T.; Pala, V.; Huybrechts, I.; Bornhorst, C.; Fernandez-Alvira, J.M.; Hadjigeorgiou, C.; Eiben, G.; Hebestreit, A.; Lissner, L.; et al. Relative validity of the children’s eating habits questionnaire-food frequency section among young european children: The IDEFICS study. Public Health Nutr. 2014, 17, 266–276. [Google Scholar] [CrossRef] [PubMed]
- Huybrechts, I.; Bornhorst, C.; Pala, V.; Moreno, L.A.; Barba, G.; Lissner, L.; Fraterman, A.; Veidebaum, T.; Hebestreit, A.; Sieri, S.; et al. Evaluation of the children’s eating habits questionnaire used in the IDEFICS study by relating urinary calcium and potassium to milk consumption frequencies among european children. Int. J. Obes. (Lond.) 2011, 35 (Suppl. S1), S69–S78. [Google Scholar] [CrossRef] [PubMed]
- Drucker, R.R.; Hammer, L.D.; Agras, W.S.; Bryson, S. Can mothers influence their child’s eating behavior? J. Dev. Behav. Pediatr. 1999, 20, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Turrell, G. Compliance with the australian dietary guidelines in the early 1990’s: Have population-based health promotion programs been effective? Nutr. Health 1997, 11, 271–288. [Google Scholar] [CrossRef] [PubMed]
- Orrell-Valente, J.K.; Hill, L.G.; Brechwald, W.A.; Dodge, K.A.; Pettit, G.S.; Bates, J.E. “Just three more bites”: An observational analysis of parents’ socialization of children’s eating at mealtime. Appetite 2007, 48, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Hammons, A.J.; Fiese, B.H. Is frequency of shared family meals related to the nutritional health of children and adolescents? Pediatrics 2011, 127, e1565–e1574. [Google Scholar] [CrossRef] [PubMed]
- Gillman, M.W.; Rifas-Shiman, S.L.; Frazier, A.L.; Rockett, H.R.; Camargo, C.A., Jr.; Field, A.E.; Berkey, C.S.; Colditz, G.A. Family dinner and diet quality among older children and adolescents. Arch. Fam. Med. 2000, 9, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Cullen, K.W.; Baranowski, T.; Rittenberry, L.; Olvera, N. Social-environmental influences on children’s diets: Results from focus groups with african-, euro- and mexican-american children and their parents. Health Educ. Res. 2000, 15, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Birch, L.L. Children’s preferences for high-fat foods. Nutr. Rev. 1992, 50, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Boutelle, K.N.; Fulkerson, J.A.; Neumark-Sztainer, D.; Story, M.; French, S.A. Fast food for family meals: Relationships with parent and adolescent food intake, home food availability and weight status. Public Health Nutr. 2007, 10, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Trofholz, A.C.; Tate, A.D.; Draxten, M.L.; Neumark-Sztainer, D.; Berge, J.M. Home food environment factors associated with the presence of fruit and vegetables at dinner: A direct observational study. Appetite 2016, 96, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Hebestreit, A.; Keimer, K.M.; Hassel, H.; Nappo, A.; Eiben, G.; Fernandez, J.M.; Kovacs, E.; Lasn, H.; Shiakou, M.; Ahrens, W. What do children understand? Communicating health behavior in a european multicenter study. J. Public Health 2010, 18, 391–400. [Google Scholar] [CrossRef]
- Subar, A.F.; Freedman, L.S.; Tooze, J.A.; Kirkpatrick, S.I.; Boushey, C.; Neuhouser, M.L.; Thompson, F.E.; Potischman, N.; Guenther, P.M.; Tarasuk, V.; et al. Addressing current criticism regarding the value of self-report dietary data. J. Nutr. 2015, 145, 2639–2645. [Google Scholar] [CrossRef] [PubMed]
- Greenland, P.; Robins, J.M. Confounding and misclassification. Am. J. Epidemiol. 1985, 122, 495–506. [Google Scholar] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Stasinopoulos, D.M.; Rigby, R.A. Generalized additive models for location scale and shape (GAMLSS) in R. J. Stat. Softw. 2007, 23, 1–46. [Google Scholar] [CrossRef]
- Hartigan, J.A.; Wong, M.A. Algorithm AS 136: A k-means clustering algorithm. Appl. Stat. 1979, 28, 100–108. [Google Scholar] [CrossRef]
- Rousseeuw, P.J. Silhouettes: A graphical aid to the interpretation and validation of cluster analysis. J. Compl. Appl. Math. 2015, 20, 53–65. [Google Scholar] [CrossRef]
- Lo, S.G.; Yasui, Y.; Csizmadi, I.; McGregor, S.E.; Robson, P.J. Exploring statistical approaches to diminish subjectivity of cluster analysis to derive dietary patterns: The Tomorrow Project. Am. J. Epidemiol. 2011, 173, 956–967. [Google Scholar]
- Hubert, L.; Arabie, P. Comparing partitions. J. Classif. 1985, 2, 193–218. [Google Scholar] [CrossRef]
| Food Group | Healthy Alternative | Unhealthy Alternative |
|---|---|---|
| Cereals & cereal products | Low sugar content (<15%), low fat content (<20%) and high fiber content (≥5%) | ≥15% sugar, ≥20% fat, and <5% fiber content |
| Sugar & sweets | - | Sugar, sweets, candies, marzipan, chocolate, nut spreads, jam, honey, ice cream, canned/sugared fruit, etc. |
| Fats & oils | From mainly plant origin, and for sauces <40% fat content | Mainly animal and processed origin and ≥40% fat for sauces |
| Non-alcoholic beverages | Non-caloric and non-processed beverages such as table water, plain herbal teas, plain coffee | Sweetened and processed beverages: manufactured juices, sodas, ice tea, energy drinks, coffee/tea with milk/sugar, broth |
| Meat | Containing <10% fat, and meat products with <20% fat from poultry, rabbit or game | Meat from all other origins than poultry, including offal, with ≥10% fat and meat products containing ≥20% fat |
| Meat alternative | Soy products, meat and dairy substitutes from soy, vegetarian burgers, tempeh, tofu, seitan and lean prepared fish and eggs | - |
| Milk & dairy products | Low fat and unsweetened | Full fat and sweetened, flavored |
| Fruit & vegetables | Fresh fruit and vegetables, their fresh juices/smoothies, or lean preparation, without added sugars | - |
| Mixed dishes | Based on cereals, legumes, vegetables/potatoes with small amounts of fish, egg or dairy, soups, veloutés, mixed salad | Based on meat; fried foods (also fried vegetables), fast food, snack foods (not included in the final cluster analysis) |
| Children | Sweet & Fat (N = 697; 35%) | Ref. Cereals (N = 563; 28%) | Animal Products (N = 716; 36%) | |||
| Mean | SD | Mean | SD | Mean | SD | |
| Energy | 0.60 a | 0.82 | 0.32 | 0.83 | −0.78 b | 0.79 |
| Non-alcoholic beverages—healthy | −0.64 b | 0.89 | 0.54 a | 0.81 | −0.04 | 1.01 |
| Non-alcoholic beverages—unhealthy | 0.39 a | 1.24 | −0.33 b | 0.67 | −0.12 | 0.82 |
| Cereals—unhealthy | −0.37 | 0.7 | 0.96 a | 1.06 | −0.40 b | 0.61 |
| Cereals—healthy | 0.34 a | 1.05 | −0.61 b | 0.64 | 0.14 | 0.97 |
| Sugar & Sweets | 0.27 a | 0.94 | −0.39 b | 0.94 | 0.05 | 1.00 |
| Fats & Oils—unhealthy | 0.29 a | 1.21 | −0.38 b | 0.61 | 0.02 | 0.93 |
| Fats & Oils—healthy | −0.03 | 1.03 | 0.36 a | 1.18 | −0.25 b | 0.69 |
| Fruit & Vegetables | −0.09 | 0.96 | −0.37 b | 0.87 | 0.39 a | 1.00 |
| Meat—unhealthy | −0.35 b | 0.88 | 0.09 | 1.02 | 0.27 a | 1.00 |
| Meat—healthy | −0.51 b | 0.73 | −0.01 | 0.96 | 0.51 a | 1.01 |
| Milk & Dairy products—unhealthy | 0.22 a | 1.13 | −0.15 b | 0.91 | −0.1 | 0.89 |
| Milk & Dairy products—healthy | 0.12 a | 1.09 | −0.22 b | 0.82 | 0.05 | 1.02 |
| Meat alternative | −0.24 | 0.78 | −0.25 b | 0.71 | 0.42 a | 1.22 |
| Mixed dishes—healthy | −0.41 b | 0.89 | 0.08 | 0.97 | 0.34 a | 0.98 |
| Parents | Sweet & Fat (N = 728; 39%) | Ref. Cereals (N = 410; 22%) | Animal Products (N = 747; 40%) | |||
| Mean | SD | Mean | SD | Mean | SD | |
| Energy | 0.75 a | 0.76 | 0.05 | 0.83 | −0.73 b | 0.75 |
| Non-alcoholic beverages—healthy | −0.36 b | 1.01 | 0.15 | 0.87 | 0.23 a | 0.96 |
| Non-alcoholic beverages—unhealthy | 0.17 a | 1.12 | −0.05 | 1.03 | −0.14 b | 0.82 |
| Cereals—unhealthy | −0.40 b | 0.59 | 1.32 | 1.09 | −0.34 | 0.55 |
| Cereals—healthy | 0.33 a | 1.02 | −0.69 b | 0.58 | 0.06 | 0.97 |
| Sugar & Sweets | 0.34 a | 0.96 | −0.24 b | 0.93 | −0.20 | 0.99 |
| Fats & Oils—unhealthy | 0.31 a | 1.17 | −0.41 b | 0.66 | −0.08 | 0.87 |
| Fats & Oils—healthy | −0.03 | 1.04 | 0.23 a | 1.11 | −0.09 b | 0.87 |
| Fruit & Vegetables | −0.08 | 0.94 | −0.32 b | 0.95 | 0.25 a | 1.02 |
| Meat—unhealthy | −0.41 b | 0.87 | 0.18 | 0.91 | 0.30 a | 1.03 |
| Meat—healthy | −0.56 b | 0.74 | 0.13 | 0.84 | 0.48 a | 1.03 |
| Milk & Dairy products—unhealthy | 0.34 a | 1.14 | −0.42 b | 0.72 | −0.09 | 0.87 |
| Milk & Dairy products—healthy | 0.03 | 1.02 | −0.18 b | 0.93 | 0.07 a | 1.00 |
| Meat alternative | −0.25 | 0.77 | −0.29 b | 0.65 | 0.40 a | 1.21 |
| Mixed dishes—healthy | −0.53 b | 0.91 | −0.01 | 0.84 | 0.52 a | 0.89 |
| Covariates | Plausible Reporters | Misreporters | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Children, Adolescents | Parents | Children, Adolescents | Parents | |||||||||||||
| Sweet & Fat (N = 697; 35%) | Ref. Cereals (N = 563; 28%) | Animal Products (N = 716; 36%) | Sweet & Fat (N = 728; 39%) | Ref. Cereals (N = 410; 22%) | Animal Products (N = 747; 40%) | (N = 484) | (N = 471) | |||||||||
| Age mean (SD) | 10.9 (2.1) | 11.4 (2.1) | 11.2 (2.1) | 41.8 (5.4) | 44.2 (5.8) | 41.8 (5.4) | 12.2 (1.9) | 42.3 (5.7) | ||||||||
| Age range (min; max) | 6.0; 15.8 | 6.0; 16.0 | 6.0; 16.0 | 28.1; 58.5 | 30.3; 65.4 | 27.0; 63.0 | 6.5; 15.7 | 27.8; 58.7 | ||||||||
| Sex (N, %) | ||||||||||||||||
| Male | 322 | 32 | 311 | 30 | 387 | 38 | 149 | 26 | 173 | 30 | 257 | 44 | 238 | 49 | 139 | 30 |
| Female | 375 | 39 | 252 | 26 | 329 | 34 | 579 | 44 | 237 | 18 | 490 | 38 | 246 | 51 | 332 | 71 |
| Weight group (N, %N) a | ||||||||||||||||
| Normal weight | 588 | 37 | 403 | 25 | 613 | 38 | 428 | 43 | 175 | 18 | 388 | 39 | 310 | 64 | 159 | 34 |
| Overweight | 93 | 32 | 115 | 39 | 84 | 32 | 209 | 34 | 157 | 25 | 255 | 41 | 123 | 25 | 156 | 33 |
| Obese | 16 | 20 | 45 | 56 | 19 | 24 | 91 | 33 | 78 | 29 | 104 | 38 | 51 | 11 | 156 | 33 |
| ISCED-Level of parents b (N, %) | ||||||||||||||||
| Low Education | 5 | 12 | 31 | 76 | 5 | 12 | 1 | 3 | 24 | 71 | 9 | 26 | 26 | 5 | 31 | 7 |
| Medium Education | 200 | 31 | 245 | 38 | 207 | 32 | 185 | 30 | 190 | 31 | 234 | 38 | 227 | 47 | 235 | 50 |
| High Education | 482 | 39 | 271 | 22 | 491 | 39 | 529 | 44 | 182 | 15 | 488 | 41 | 226 | 47 | 199 | 42 |
| Missing ISCED c | 10 | 26 | 16 | 41 | 13 | 33 | 13 | 30 | 14 | 33 | 16 | 37 | 5 | 1 | 6 | 1 |
| County (N, %) | ||||||||||||||||
| Italy | 8 | 3 | 232 | 85 | 32 | 12 | 14 | 6 | 179 | 77 | 38 | 16 | 59 | 12 | 77 | 16 |
| Estonia | 275 | 50 | 29 | 5 | 246 | 45 | 341 | 56 | 28 | 5 | 244 | 40 | 148 | 31 | 82 | 17 |
| Cyprus | 28 | 17 | 66 | 39 | 75 | 44 | 33 | 21 | 40 | 26 | 81 | 53 | 27 | 6 | 41 | 9 |
| Belgium | 67 | 44 | 14 | 9 | 73 | 47 | 66 | 55 | 14 | 12 | 39 | 33 | 24 | 5 | 18 | 4 |
| Sweden | 133 | 39 | 120 | 35 | 89 | 26 | 113 | 40 | 58 | 20 | 113 | 40 | 55 | 11 | 66 | 14 |
| Germany | 149 | 49 | 40 | 13 | 113 | 37 | 133 | 47 | 39 | 14 | 114 | 40 | 143 | 30 | 147 | 31 |
| Hungary | 29 | 32 | 38 | 42 | 23 | 26 | 11 | 12 | 41 | 45 | 39 | 43 | 17 | 4 | 25 | 5 |
| Spain | 8 | 8 | 24 | 25 | 65 | 67 | 17 | 16 | 11 | 10 | 79 | 74 | 11 | 2 | 15 | 3 |
| Parental Dietary Pattern | Children’s Dietary Pattern | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sweet & Fat | Ref. Cereals | Animal Products | |||||||
| N | OR | 95%CI | N | OR | 95%CI | N | OR | 95%CI | |
| Maternal dietary pattern (N = 1662) | |||||||||
| Different & <1 shared meal (reference) | 132 | 1.00 | 265 | 1.00 | 183 | 1.00 | |||
| Different & ≥1 shared meals | 771 | 0.97 | 0.59; 1.58 | 1096 | 1.20 | 0.70; 2.07 | 877 | 1.06 | 0.70; 1.60 |
| Identical & <1 shared meal | 158 | 2.12 | 1.18; 3.81 | 25 | 5.70 | 1.51; 21.48 | 107 | 2.18 | 1.21; 3.92 |
| Identical & ≥1 shared meals | 601 | 1.91 | 1.17; 3.13 | 276 | 2.70 | 1.34; 5.45 | 495 | 2.19 | 1.41; 3.40 |
| Paternal dietary pattern (N = 789) | |||||||||
| Different & <1 shared meal (reference) | 149 | 1.00 | 153 | 1.00 | 112 | 1.00 | |||
| Different & ≥1 shared meals | 430 | 1.31 | 0.82; 2.09 | 396 | 0.83 | 0.45;1.54 | 338 | 0.55 | 0.32; 0.92 |
| Identical & <1 shared meal | 58 | 2.30 | 1.15; 4.57 | 54 | 1.66 | 0.68;4.06 | 95 | 1.45 | 0.78; 2.71 |
| Identical & ≥1 shared meals | 152 | 3.18 | 1.84; 5.47 | 186 | 1.99 | 0.98;4.08 | 244 | 1.54 | 0.91; 2.59 |
| Parental Dietary Pattern | Children’s Dietary Pattern | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sweet & Fat | Ref. Cereals | Animal Products | |||||||
| N | OR | 95%CI | N | OR | 95%CI | N | OR | 95%CI | |
| Maternal dietary pattern (N = 1607) | |||||||||
| Different & soft drink not available (reference) | 742 | 1.00 | 1017 | 1.00 | 767 | 1.00 | |||
| Different & soft drink is available | 138 | 1.97 | 1.20; 3.25 | 294 | 0.46 | 0.25; 0.84 | 256 | 0.95 | 0.65; 1.38 |
| Identical & soft drink not available | 521 | 2.04 | 1.49; 2.80 | 246 | 2.48 | 1.43; 4.27 | 496 | 2.16 | 1.59; 2.92 |
| Identical & soft drink is available | 206 | 2.78 | 1.80; 4.28 | 50 | 1.67 | 0.66; 4.22 | 88 | 1.42 | 0.82; 2.47 |
| Paternal dietary pattern (N = 763) | |||||||||
| Different & soft drink not available (reference) | 465 | 1.00 | 407 | 1.00 | 360 | 1.00 | |||
| Different & soft drink is available | 92 | 1.55 | 0.90; 2.68 | 122 | 0.43 | 0.18; 1.04 | 80 | 0.83 | 0.45; 1.52 |
| Identical & soft drink not available | 151 | 2.48 | 1.58; 3.87 | 209 | 2.05 | 1.22; 3.45 | 256 | 2.48 | 1.62; 3.79 |
| Identical & soft drink is available | 55 | 4.26 | 2.16; 8.41 | 25 | 1.97 | 0.61; 6.39 | 67 | 1.80 | 0.96; 3.36 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hebestreit, A.; Intemann, T.; Siani, A.; De Henauw, S.; Eiben, G.; Kourides, Y.A.; Kovacs, E.; Moreno, L.A.; Veidebaum, T.; Krogh, V.; et al. Dietary Patterns of European Children and Their Parents in Association with Family Food Environment: Results from the I.Family Study. Nutrients 2017, 9, 126. https://doi.org/10.3390/nu9020126
Hebestreit A, Intemann T, Siani A, De Henauw S, Eiben G, Kourides YA, Kovacs E, Moreno LA, Veidebaum T, Krogh V, et al. Dietary Patterns of European Children and Their Parents in Association with Family Food Environment: Results from the I.Family Study. Nutrients. 2017; 9(2):126. https://doi.org/10.3390/nu9020126
Chicago/Turabian StyleHebestreit, Antje, Timm Intemann, Alfonso Siani, Stefaan De Henauw, Gabriele Eiben, Yiannis A. Kourides, Eva Kovacs, Luis A. Moreno, Toomas Veidebaum, Vittorio Krogh, and et al. 2017. "Dietary Patterns of European Children and Their Parents in Association with Family Food Environment: Results from the I.Family Study" Nutrients 9, no. 2: 126. https://doi.org/10.3390/nu9020126
APA StyleHebestreit, A., Intemann, T., Siani, A., De Henauw, S., Eiben, G., Kourides, Y. A., Kovacs, E., Moreno, L. A., Veidebaum, T., Krogh, V., Pala, V., Bogl, L. H., Hunsberger, M., Börnhorst, C., & Pigeot, I. (2017). Dietary Patterns of European Children and Their Parents in Association with Family Food Environment: Results from the I.Family Study. Nutrients, 9(2), 126. https://doi.org/10.3390/nu9020126






