Nutrition in Pregnancy Following Bariatric Surgery
Abstract
:1. Introduction
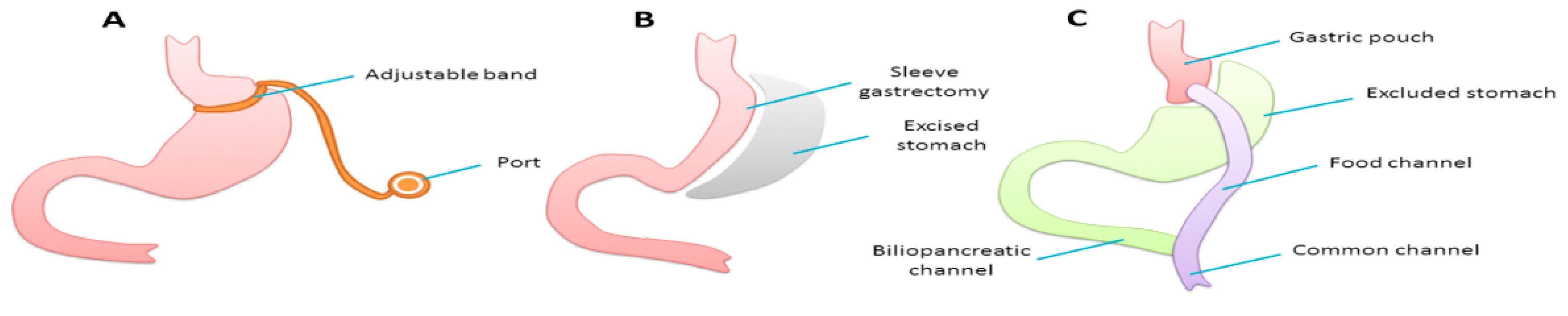
Bariatric Surgery
2. Materials and Methods
3. Results
3.1. Pre-Conception Care
3.2. Antenatal Care
3.2.1. Dietary Assessment
- Taking anti-emetics as prescribed, not waiting for the onset of nausea
- Avoiding foods that induce nausea
- Avoiding cooking smells and other strong odours
- Taking a short walk before meals
- Tracking nausea to establish any patterns and maximising nutritional intake when typically less nauseous
- Experimenting with different foods as taste changes can occur over time
- Following good eating habits by
- Separating consumption of solids and liquids
- Taking small bites
- Chewing well
- Stopping eating when full
3.2.2. Energy Requirements
3.2.3. Protein Intake
3.2.4. Food Hygiene
- Soft, ripe and blue veined cheeses such as brie and stilton.
- Other non-pasteurized dairy products including soft ice creams.
- Shark, marlin and swordfish due to mercury content.
- Liver and liver products such as pate and cod liver oil to avoid consumption of too much retinol-based vitamin A.
- Eggs, unless certified salmonella-safe, should be cooked thoroughly until the yolk and white are solid [45]; unpasteurized foods containing uncertified raw or lightly cooked eggs, such as homemade mayonnaise, salad dressings, custard and eggnog, should be avoided.
3.2.5. Micronutrients
Iron
Calcium and Vitamin D
Folic Acid
Vitamin B12
Vitamin A
Thiamine
3.2.6. Dumping Syndromes
3.2.7. Other Considerations
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- NHS Digital. Health Survey for England 2015; NHS Digital: Leeds, UK, 2016. [Google Scholar]
- Thummler, K.; Britton, A.; Wilhelm, K. Data and Information on Women’s Health in the European Union; European Communities: Brussels, Belgium, 2009. [Google Scholar]
- Vahratian, A. Prevalence of overweight and obesity among women of childbearing age: Results from the 2002 National Survey of Family Growth. Matern. Child Health J. 2009, 13, 268–273. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, H.D.; Gibbons, K.S.; Flenady, V.J.; Callaway, L.K. Overweight and obesity in Australian mothers: Epidemic or endemic? Med. J. Aust. 2012, 196, 184–188. [Google Scholar] [CrossRef] [PubMed]
- World Obesity Federation. About Obesity. Available online: https://www.worldobesity.org/what-we-do/aboutobesity/ (accessed on 1 November 2017).
- Norman, R.J.; Noakes, M.; Wu, R.; Davies, M.J.; Moran, L.; Wang, J.X. Improving reproductive performance in overweight/obese women with effective weight management. Hum. Reprod. Update 2004, 10, 267–280. [Google Scholar] [CrossRef] [PubMed]
- Bolúmar, F.; Olsen, J.; Rebagliato, M.; Sáez-Lloret, I.; Bisanti, L. Body mass index and delayed conception: A European Multicenter Study on Infertility and Subfecundity. Am. J. Epidemiol. 2000, 151, 1072–1079. [Google Scholar] [CrossRef] [PubMed]
- Sharpe, R.M.; Franks, S. Environment, lifestyle and infertility—An inter-generational issue. Nat. Cell Biol. 2002, 4, S33–S40. [Google Scholar] [CrossRef] [PubMed]
- Dağ, Z.Ö.; Dilbaz, B. Impact of obesity on infertility in women. J. Turk. Ger. Gynecol. Assoc. 2015, 16, 111–117. [Google Scholar] [PubMed]
- Centre for Maternal and Child Enquiries. Maternal Obesity in the UK: Findings from a National Project; Centre for Maternal and Child Enquiries: London, UK, 2010. [Google Scholar]
- Davies, G.A.L.; Maxwell, C.; McLeod, L.; Gagnon, R.; Basso, M.; Bos, H.; Delisle, M.-F.; Farine, D.; Hudon, L.; Menticoglou, S.; et al. Obesity in Pregnancy. J. Obstet. Gynaecol. Can. 2010, 32, 165–173. [Google Scholar] [CrossRef]
- Modder, J.; Fitzsimons, K. CMACE/RCOG Joint Guideline Management of Women with Obesity in Pregnancy Centre for Maternal and Child Enquiries; RCOG: London, UK, 2010. [Google Scholar]
- Shinogle, J.A.; Owings, M.F.; Kozak, L.J. Gastric bypass as treatment for obesity: Trends, characteristics, and complications. Obes. Res. 2005, 13, 2202–2209. [Google Scholar] [CrossRef] [PubMed]
- Welbourn, R.; Small, P.; Finlay, I.; Sareela, A.; Somers, S.; Mahawar, K. National Bariatric Surgery Registry of the British Obesity & Metabolic Surgery Society. Second Registry Report; Dendrite Clinical Systems Ltd.: Henley-on-Thames, UK, 2014. [Google Scholar]
- Booth, H.P.; Khan, O.; Fildes, A.; Prevost, A.T.; Reddy, M.; Charlton, J.; Gulliford, M.C. King’s bariatric surgery study group changing epidemiology of bariatric surgery in the UK: Cohort study using primary care electronic health records. Obes. Surg. 2016, 26, 1900–1905. [Google Scholar] [CrossRef] [PubMed]
- Sjöström, L. Review of the key results from the Swedish Obese Subjects (SOS) trial—A prospective controlled intervention study of bariatric surgery. J. Intern. Med. 2013, 273, 219–234. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.-H.; Stoll, C.R.T.; Song, J.; Varela, J.E.; Eagon, C.J.; Colditz, G.A. The effectiveness and risks of bariatric surgery: An updated systematic review and meta-analysis, 2003–2012. JAMA Surg. 2014, 149, 275–287. [Google Scholar] [CrossRef] [PubMed]
- Buchwald, H.; Avidor, Y.; Braunwald, E.; Jensen, M.D.; Pories, W.; Fahrbach, K.; Schoelles, K. Bariatric Surgery. JAMA 2004, 292, 1724. [Google Scholar] [CrossRef] [PubMed]
- Buchwald, H. Consensus conference panel consensus conference statement. Surg. Obes. Relat. Dis. 2005, 1, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Picot, J.; Jones, J.; Colquitt, J.; Gospodarevskaya, E.; Loveman, E.; Baxter, L.; Clegg, A. The clinical effectiveness and cost-effectiveness of bariatric (weight loss) surgery for obesity: A systematic review and economic evaluation. Health Technol. Assess. 2009, 13, 1–190, 215–357, iii–iv. [Google Scholar] [CrossRef] [PubMed]
- Kennedy-Dalby, A.; Adam, S.; Ammori, B.J.; Syed, A.A. Weight loss and metabolic outcomes of bariatric surgery in men versus women—A matched comparative observational cohort study. Eur. J. Intern. Med. 2014, 25, 922–925. [Google Scholar] [CrossRef] [PubMed]
- Maggard, M.A.; Yermilov, I.; Li, Z.; Maglione, M.; Newberry, S.; Suttorp, M.; Hilton, L.; Santry, H.P.; Morton, J.M.; Livingston, E.H.; et al. Pregnancy and Fertility Following Bariatric Surgery. JAMA 2008, 300, 2286. [Google Scholar] [CrossRef] [PubMed]
- Alatishe, A.; Ammori, B.J.; New, J.P.; Syed, A.A. Bariatric surgery in women of childbearing age. QJM 2013, 106, 717–720. [Google Scholar] [CrossRef] [PubMed]
- Deitel, M.; Stone, E.; Kassam, H.A.; Wilk, E.J.; Sutherland, D.J. Gynecologic-obstetric changes after loss of massive excess weight following bariatric surgery. J. Am. Coll. Nutr. 1988, 7, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Lapolla, A.; Marangon, M.; Dalfrà, M.G.; Segato, G.; De Luca, M.; Fedele, D.; Favretti, F.; Enzi, G.; Busetto, L. Pregnancy Outcome in Morbidly Obese Women Before and After Laparoscopic Gastric Banding. Obes. Surg. 2010, 20, 1251–1257. [Google Scholar] [CrossRef] [PubMed]
- Scholtz, S.; Balen, A.; Le Roux, C. The Role of Bariatric Surgery in Improving Reproductive Health; Royal College of Obstetricians and Gynaecologists: London, UK, 2015. [Google Scholar]
- Narayanan, R.P.; Syed, A.A. Pregnancy Following Bariatric Surgery—Medical Complications and Management. Obes. Surg. 2016, 26, 2523–2529. [Google Scholar] [CrossRef] [PubMed]
- Milone, M.; De Placido, G.; Musella, M.; Sosa Fernandez, L.M.; Sosa Fernandez, L.V.; Campana, G.; Di Minno, M.N.D.; Milone, F. Incidence of Successful Pregnancy After Weight Loss Interventions in Infertile Women: A Systematic Review and Meta-Analysis of the Literature. Obes. Surg. 2016, 26, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Merhi, Z.O. Challenging Oral Contraception after Weight Loss by Bariatric Surgery. Gynecol. Obstet. Investig. 2007, 64, 100–102. [Google Scholar] [CrossRef] [PubMed]
- Parrott, J.; Frank, L.; Rabena, R.; Craggs-Dino, L.; Isom, K.A.; Greiman, L. American Society for Metabolic and Bariatric Surgery Integrated Health Nutritional Guidelines for the Surgical Weight Loss Patient 2016 Update: Micronutrients. Surg. Obes. Relat. Dis. 2017, 13, 727–741. [Google Scholar] [CrossRef] [PubMed]
- Mechanick, J.I.; Youdim, A.; Jones, D.B.; Garvey, W.T.; Hurley, D.L.; McMahon, M.M.; Heinberg, L.J.; Kushner, R.; Adams, T.D.; Shikora, S.; et al. Clinical practice guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient 2013 update. Cosponsored by American Association of Clinical Endocrinologists, the Obesity Society, and the American Society for Metabolic and Bariatric Surgery. Surg. Obes. Relat. Dis. 2013, 9, 159–191. [Google Scholar] [PubMed]
- O’Kane, M.; Pinkney, J.; Aasheim, E.T.; Barth, J.H.; Batterham, R.L.; Welbourn, R. Bomss Guidelines on Perioperative and Postoperative Biochemical Monitoring and Micronutrient Replacement for Patients Undergoing Bariatric Surgery. Available online: http://www.bomss.org.uk/wp-content/uploads/2014/09/BOMSS-guidelines-Final-version1Oct14.pdf (accessed on 31 October 2017).
- Rogne, T.; Tielemans, M.J.; Chong, M.F.-F.; Yajnik, C.S.; Krishnaveni, G.V.; Poston, L.; Jaddoe, V.W.V.; Steegers, E.A.P.; Joshi, S.; Chong, Y.-S.; et al. Associations of Maternal Vitamin B12 Concentration in Pregnancy With the Risks of Preterm Birth and Low Birth Weight: A Systematic Review and Meta-Analysis of Individual Participant Data. Am. J. Epidemiol. 2017, 185, 212–223. [Google Scholar] [PubMed]
- Queensland Clinical Guidelines. Maternity and Neonatal Clinical Guideline Obesity in Pregnancy; Queensland Health: Brisbane, Australia, 2015.
- Rand, C.S.; Macgregor, A.M. Adolescents having obesity surgery: A 6-year follow-up. South. Med. J. 1994, 87, 1208–1213. [Google Scholar] [CrossRef] [PubMed]
- Dixon, J.B.; Dixon, M.E.; O’Brien, P.E. Elevated homocysteine levels with weight loss after Lap-Band® surgery: Higher folate and vitamin B12 levels required to maintain homocysteine level. Int. J. Obes. 2001, 25, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Royal College of Obstetricians and Gynaecologists. The Management of Nausea and Vomiting of Pregnancy and Hyperemesis Gravidarum; Royal College of Obstetricians and Gynaecologists: London, UK, 2016. [Google Scholar]
- British Nutrition Foundation. Nutrition Requirements; British Nutrition Foundation: London, UK, 2016. [Google Scholar]
- Thomas, B.; Bishop, J. British Dietetic Association. In Manual of Dietetic Practice, 4th ed.; Blackwell Publishing: Chichester, UK, 2007; ISBN 9781405135252. [Google Scholar]
- Xanthakos, S.A. Nutritional deficiencies in obesity and after bariatric surgery. Pediatr. Clin. N. Am. 2009, 56, 1105–1121. [Google Scholar] [CrossRef] [PubMed]
- Blumfield, M.; Nowson, C.; Hure, A.; Smith, R.; Simpson, S.; Raubenheimer, D.; MacDonald-Wicks, L.; Collins, C. Lower Protein-to-carbohydrate ratio in maternal diet is associated with higher childhood systolic blood pressure up to age four years. Nutrients 2015, 7, 3078–3093. [Google Scholar] [CrossRef] [PubMed]
- Zohdi, V.; Lim, K.; Pearson, J.; Black, M. Developmental programming of cardiovascular disease following intrauterine growth restriction: Findings utilising a rat model of maternal protein restriction. Nutrients 2014, 7, 119–152. [Google Scholar] [CrossRef] [PubMed]
- American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 105: Bariatric Surgery and Pregnancy. Obstet. Gynecol. 2009, 113, 1405–1413. [Google Scholar]
- Landsberger, E.J.; Gurewitsch, E.D. Reproductive implications of bariatric surgery: Pre- and postoperative considerations for extremely obese women of childbearing age. Curr. Diabetes Rep. 2007, 7, 281–288. [Google Scholar] [CrossRef]
- Food Standards Agency Review of Advice to Consumers (including Vulnerable Groups) on Eating Raw or lightly Cooked Shell Eggs and Their Products in the UK/Food Standards Agency. Available online: https://www.food.gov.uk/news-updates/help-shape-our-policies/review-of-advice-to-consumers-on-eggs (accessed on 30 October 2017).
- Gautam, C.S.; Saha, L.; Sekhri, K.; Saha, P.K. Iron deficiency in pregnancy and the rationality of iron supplements prescribed during pregnancy. Medscape J. Med. 2008, 10, 283. [Google Scholar] [PubMed]
- Allen, L.H. Anemia and iron deficiency: Effects on pregnancy outcome. Am. J. Clin. Nutr. 2000, 71, 1280S–1284S. [Google Scholar] [PubMed]
- Confidential Enquiry into Maternal and Child Health. Saving Mothers’ Lives: Reviewing Maternal Deaths to Make Motherhood Safer—2003–2005. In The Seventh Report of the Confidential Enquiries into Maternal Deaths in the United Kingdom; CEMACH: London, UK, 2007. [Google Scholar]
- Mahadevan, S.; Kumaravel, V.; Bharath, R. Calcium and bone disorders in pregnancy. Indian J. Endocrinol. Metab. 2012, 16, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Brzozowska, M.M.; Sainsbury, A.; Eisman, J.A.; Baldock, P.A.; Center, J.R. Bariatric surgery, bone loss, obesity and possible mechanisms. Obes. Rev. 2013, 14, 52–67. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, C.S.; Kronenberg, H.M. Maternal-fetal calcium and bone metabolism during pregnancy, puerperium, and lactation. Endocr. Rev. 1997, 18, 832–872. [Google Scholar] [CrossRef] [PubMed]
- Kominiarek, M.A. Preparing for and Managing a Pregnancy after Bariatric Surgery. Semin. Perinatol. 2011, 35, 356–361. [Google Scholar] [CrossRef] [PubMed]
- John, S.; Hoegerl, C. Nutritional Deficiencies after Gastric Bypass Surgery. J. Am. Osteopat. Assoc. 2009, 109, 601–604. [Google Scholar]
- Moliterno, J.A.; DiLuna, M.L.; Sood, S.; Roberts, K.E.; Duncan, C.C. Gastric bypass: A risk factor for neural tube defects? J. Neurosurg. Pediatr. 2008, 1, 406–409. [Google Scholar] [CrossRef] [PubMed]
- Scientific Advisory Committee on Nutrition. Update on Folic Acid; Scientific Advisory Committee on Nutrition: London, UK, 2017.
- Bebber, F.E.; Rizzolli, J.; Casagrande, D.S.; Rodrigues, M.T.; Padoin, A.V.; Mottin, C.C.; Repetto, G. Pregnancy after Bariatric Surgery: 39 Pregnancies Follow-up in a Multidisciplinary Team. Obes. Surg. 2011, 21, 1546–1551. [Google Scholar] [CrossRef] [PubMed]
- Majumder, S.; Soriano, J.; Louie Cruz, A.; Dasanu, C.A. Vitamin B12 deficiency in patients undergoing bariatric surgery: Preventive strategies and key recommendations. Surg. Obes. Relat. Dis. 2013, 9, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Food Standards Agency. Safe Upper Levels for Vitamins and Minerals Expert Group on Vitamins and Minerals; Food Standards Agency: London, UK, 2003.
- Ladipo, O.A. Nutrition in pregnancy: Mineral and vitamin supplements. Am. J. Clin. Nutr. 2000, 72, 280S–290S. [Google Scholar] [PubMed]
- Frank, L.L. Thiamin in Clinical Practice. J. Parenter. Enter. Nutr. 2015, 39, 503–520. [Google Scholar] [CrossRef] [PubMed]
- Einarson, T.R.; Piwko, C.; Koren, G. Quantifying the global rates of nausea and vomiting of pregnancy: A meta analysis. J. Popul. Ther. Clin. Pharmacol. 2013, 20, e171–e183. [Google Scholar] [PubMed]
- Jacquemyn, Y.; Meesters, J. Pregnancy as a risk factor for undertreatment after bariatric surgery. BMJ Case Rep. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Pilone, V.; Di Micco, R.; Monda, A.; Villamaina, E.; Gentile, M.; Forestieri, P. LAGB in pregnancy: Slippage after hyperemesis gravidarum. Report of a case. Ann. Ital. Chir. 2012, 83, 429–432. [Google Scholar] [PubMed]
- Saab, R.O.; El Khoury, M.I.; Jabbour, R.A. Wernicke encephalopathy after Roux-en-Y gastric bypass and hyperemesis gravidarum. Surg. Obes. Relat. Dis. 2013, 9, e105–e107. [Google Scholar] [CrossRef] [PubMed]
- Tack, J.; Arts, J.; Caenepeel, P.; De Wulf, D.; Bisschops, R. Pathophysiology, diagnosis and management of postoperative dumping syndrome. Nat. Rev. Gastroenterol. Hepatol. 2009, 6, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Adam, S.; Ammori, B.; Soran, H.; Syed, A.A. Pregnancy after bariatric surgery: Screening for gestational diabetes. BMJ 2017, 356, j533. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence Dyspepsia-Pregnancy-Associated. Available online: https://cks.nice.org.uk/dyspepsia-pregnancy-associated#!scenario (accessed on 1 November 2017).
- Miller, A.D.; Smith, K.M. Medication and nutrient administration considerations after bariatric surgery. Am. J. Health-Syst. Pharm. 2006, 63, 1852–1857. [Google Scholar] [CrossRef] [PubMed]
| Recommendation | Comments | |
|---|---|---|
| Multivitamin and mineral supplement | 1–2 daily | Avoid retinol-based vitamin A during pregnancy and lactation; safe to continue beta-carotene |
| Calcium | 800–1500 mg daily | Calcium citrate may have better bioavailability |
| Vitamin D | 800 units daily | Higher doses may be necessary if pre-existing deficiency |
| Iron | 45–60 mg daily | 100 mg elemental iron is recommended for menstruating women |
| Vitamin B12 | 1000 micrograms orally daily or 1000 micrograms intramuscular injection 4–12 weekly | |
| Thiamine (B1) | As contained in Multivitamin or 12–50 mg daily | Additional 200–300 mg if prolonged vomiting is experienced |
| Folic Acid | As contained in Multivitamin or 400–800 microgram daily | 5 mg preconception to 12 weeks of gestation |
| Vitamin A | As contained in Multivitamin or 5000–1000 IU daily | Additional screening in BPD/DS * or if Steatorrhoea. Increased requirements in pregnancy—avoid retinol and retinyl esters. |
| Vitamin E | As contained in Multivitamin or 15 mg daily | Additional screening in BPD/DS * or if Steatorrhoea |
| Vitamin K | As contained in Multivitamin or 90–300 micrograms daily | Additional screening in BPD/DS * or if Steatorrhoea |
| Zinc | As contained in Multivitamin to meet 100–200% RDA † | Maintain Ratio of 8–15 mg Zinc per 1 mg Copper |
| Copper | As contained in Multivitamin to meet 100–200% RDA † | Maintain Ratio of 8–15 mg Zinc per 1 mg Copper |
| Selenium | As contained in Multivitamin |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Slater, C.; Morris, L.; Ellison, J.; Syed, A.A. Nutrition in Pregnancy Following Bariatric Surgery. Nutrients 2017, 9, 1338. https://doi.org/10.3390/nu9121338
Slater C, Morris L, Ellison J, Syed AA. Nutrition in Pregnancy Following Bariatric Surgery. Nutrients. 2017; 9(12):1338. https://doi.org/10.3390/nu9121338
Chicago/Turabian StyleSlater, Christopher, Lauren Morris, Jodi Ellison, and Akheel A. Syed. 2017. "Nutrition in Pregnancy Following Bariatric Surgery" Nutrients 9, no. 12: 1338. https://doi.org/10.3390/nu9121338
APA StyleSlater, C., Morris, L., Ellison, J., & Syed, A. A. (2017). Nutrition in Pregnancy Following Bariatric Surgery. Nutrients, 9(12), 1338. https://doi.org/10.3390/nu9121338





