Tooth Wear Is Frequent in Adult Patients with Celiac Disease
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ludvigsson, J.F.; Bai, J.C.; Biagi, F.; Card, T.R.; Ciacci, C.; Ciclitira, P.J.; Green, P.H.; Hadjivassiliou, M.; Holdoway, A.; van Heel, D.A.; et al. Diagnosis and management of adult coeliac disease: Guidelines from the british society of gastroenterology. Gut 2014, 63, 1210–1228. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Leffler, D.A.; Bai, J.C.; Biagi, F.; Fasano, A.; Green, P.H.; Hadjivassiliou, M.; Kaukinen, K.; Kelly, C.P.; Leonard, J.N.; et al. The Oslo definitions for coeliac disease and related terms. Gut 2013, 62, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Zingone, F.; West, J.; Crooks, C.J.; Fleming, K.M.; Card, T.R.; Ciacci, C.; Tata, L.J. Socioeconomic variation in the incidence of childhood coeliac disease in the UK. Arch. Dis. Child. 2015, 100, 466–473. [Google Scholar] [CrossRef] [PubMed]
- West, J.; Fleming, K.M.; Tata, L.J.; Card, T.R.; Crooks, C.J. Incidence and prevalence of celiac disease and dermatitis herpetiformis in the UK over two decades: Population-based study. Am. J. Gastroenterol. 2014, 109, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Kang, A.; Green, A.; Gwee, K.; Ho, K. Systematic review: Worldwide variation in the frequency of coeliac disease and changes over time. Aliment. Pharmacol. Ther. 2013, 38, 226–245. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.; Lebwohl, B.; Green, P.H. Screening for celiac disease in average-risk and high-risk populations. Ther. Adv. Gastroenterol. 2012, 5, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Krzywicka, B.; Herman, K.; Kowalczyk-Zajac, M.; Pytrus, T. Celiac disease and its impact on the oral health status—Review of the literature. Adv. Clin. Exp. Med. 2014, 23, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Trotta, L.; Biagi, F.; Bianchi, P.I.; Marchese, A.; Vattiato, C.; Balduzzi, D.; Collesano, V.; Corazza, G.R. Dental enamel defects in adult coeliac disease: Prevalence and correlation with symptoms and age at diagnosis. Eur. J. Intern. Med. 2013, 24, 832–834. [Google Scholar] [CrossRef] [PubMed]
- Campisi, G.; Di Liberto, C.; Iacono, G.; Compilato, D.; Di Prima, L.; Calvino, F.; Di Marco, V.; Lo Muzio, L.; Sferrazza, C.; Scalici, C.; et al. Oral pathology in untreated coeliac [corrected] disease. Aliment. Pharmacol. Ther. 2007, 26, 1529–1536. [Google Scholar] [CrossRef] [PubMed]
- Cigic, L.; Galic, T.; Kero, D.; Simunic, M.; Medvedec Mikic, I.; Kalibovic Govorko, D.; Biocina Lukenda, D. The prevalence of celiac disease in patients with geographic tongue. J. Oral Pathol. Med. 2016, 45, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Corazza, G.R.; Villanacci, V. Coeliac disease. J. Clin. Pathol. 2005, 58, 573–574. [Google Scholar] [CrossRef] [PubMed]
- Jurge, S.; Kuffer, R.; Scully, C.; Porter, S.R. Mucosal Disease series. Number VI. Recurrent aphthous stomatitis. Oral Dis. 2006, 12, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Aine, L. Permanent tooth dental enamel defects leading to the diagnosis of coeliac disease. Br. Dent. J. 1994, 177, 253–254. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.G.; Knight, J.K. An index for measuring the wear of teeth. Br. Dent. J. 1984, 156, 435–438. [Google Scholar] [CrossRef] [PubMed]
- Ciacci, C.; Bucci, C.; Zingone, F.; Iovino, P.; Amato, M. Buccal localization of Crohn’s disease with long-term infliximab therapy: A case report. J. Med. Case Rep. 2014, 8, 397. [Google Scholar] [CrossRef] [PubMed]
- Mignogna, M.D.; Fortuna, G.; Leuci, S.; Amato, M. Oral Crohn’s disease: A favorable clinical response with delayed-release triamcinolone acetonide intralesional injections. Am. J. Gastroenterol. 2008, 103, 2954–2955. [Google Scholar] [CrossRef] [PubMed]
- Adamo, D.; Ruoppo, E.; Leuci, S.; Aria, M.; Amato, M.; Mignogna, M.D. Sleep disturbances, anxiety and depression in patients with oral lichen planus: A case-control study. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Lehner, T. Autoimmunity in oral diseases, with special reference to recurrent oral ulceration. Proc. R. Soc. Med. 1968, 61, 515–524. [Google Scholar] [PubMed]
- Majorana, A.; Sapelli, P.L.; Malagoli, A.; Meini, A.; Pillan, M.N.; Duse, M.; Ugazio, A.G. Celiac disease and recurrent aphthous stomatitis. The clinical and immunogenetic aspects. Minerva Stomatol. 1992, 41, 33–40. [Google Scholar] [PubMed]
- Procaccini, M.; Campisi, G.; Bufo, P.; Compilato, D.; Massaccesi, C.; Catassi, C.; Lo Muzio, L. Lack of association between celiac disease and dental enamel hypoplasia in a case-control study from an Italian central region. Head Face Med. 2007, 3, 25. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Malahias, T.; Brar, P.; Minaya, M.T.; Green, P.H. The association between celiac disease, dental enamel defects, and aphthous ulcers in a United States cohort. J. Clin. Gastroenterol. 2010, 44, 191–194. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, F.K.; de Queiroz, A.M.; Bezerra da Silva, R.A.; Sawamura, R.; Bachmann, L.; Bezerra da Silva, L.A.; Nelson-Filho, P. Oral aspects in celiac disease children: Clinical and dental enamel chemical evaluation. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2015, 119, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Cantekin, K.; Arslan, D.; Delikan, E. Presence and distribution of dental enamel defects, recurrent aphthous lesions and dental caries in children with celiac disease. Pak. J. Med. Sci. 2015, 31, 606–609. [Google Scholar] [CrossRef] [PubMed]
- Bucci, P.; Carile, F.; Sangianantoni, A.; D’Angio, F.; Santarelli, A.; Lo Muzio, L. Oral aphthous ulcers and dental enamel defects in children with coeliac disease. Acta. Paediatr. 2006, 95, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Costacurta, M.; Maturo, P.; Bartolino, M.; Docimo, R. Oral manifestations of coeliac disease: A clinical-statistic study. Oral Implantol. 2010, 3, 12–19. [Google Scholar]
- Erriu, M.; Abbate, G.M.; Pili, F.M.; Novara, F.; Orru, G.; Montaldo, C.; Piras, V.; Levrini, L. Oral signs and HLA-DQB1 *02 haplotypes in the celiac paediatric patient: A preliminary study. Autoimmun. Dis. 2013, 2013, 389590. [Google Scholar] [CrossRef] [PubMed]
- Pastore, L.; Campisi, G.; Compilato, D.; Lo Muzio, L. Orally based diagnosis of celiac disease: Current perspectives. J. Dent. Res. 2008, 87, 1100–1107. [Google Scholar] [CrossRef] [PubMed]
- Mariani, P.; Mazzilli, M.C.; Margutti, G.; Lionetti, P.; Triglione, P.; Petronzelli, F.; Ferrante, E.; Bonamico, M. Coeliac disease, enamel defects and HLA typing. Acta Paediatr. 1994, 83, 1272–1275. [Google Scholar] [CrossRef] [PubMed]
- Nikiforuk, G.; Fraser, D. The etiology of enamel hypoplasia: A unifying concept. J. Pediatr. 1981, 98, 888–893. [Google Scholar] [CrossRef]
- Petrecca, S.; Giammaria, G.; Giammaria, A.F. Oral cavity changes in the child with celiac disease. Minerva Stomatol. 1994, 43, 137–140. [Google Scholar] [PubMed]
- Sonora, C.; Arbildi, P.; Rodriguez-Camejo, C.; Beovide, V.; Marco, A.; Hernandez, A. Enamel organ proteins as targets for antibodies in celiac disease: Implications for oral health. Eur. J. Oral Sci. 2016, 124, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, J.M.; Rodriguez, R.; Oribe, D.; Vitoria, J.C. Dental enamel defects in celiac patients. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1997, 84, 646–650. [Google Scholar] [CrossRef]
- Ortega Paez, E.; Junco Lafuente, P.; Baca Garcia, P.; Maldonado Lozano, J.; Llodra Calvo, J.C. Prevalence of dental enamel defects in celiac patients with deciduous dentition: A pilot study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2008, 106, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Wierink, C.D.; van Diermen, D.E.; Aartman, I.H.; Heymans, H.S. Dental enamel defects in children with coeliac disease. Int. J. Paediatr. Dent. 2007, 17, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Bossu, M.; Bartoli, A.; Orsini, G.; Luppino, E.; Polimeni, A. Enamel hypoplasia in coeliac children: A potential clinical marker of early diagnosis. Eur. J. Paediatr. Dent. 2007, 8, 31–37. [Google Scholar] [PubMed]
- Kaidonis, J.A. Tooth wear: The view of the anthropologist. Clin. Oral Investig. 2008, 12 (Suppl. 1), S21–S26. [Google Scholar] [CrossRef] [PubMed]
- Ahlberg, J.; Lobbezoo, F.; Ahlberg, K.; Manfredini, D.; Hublin, C.; Sinisalo, J.; Kononen, M.; Savolainen, A. Self-reported bruxism mirrors anxiety and stress in adults. Med. Oral Patol. Oral Cir. Bucal 2013, 18, e7–e11. [Google Scholar] [CrossRef] [PubMed]
- Serra-Negra, J.M.; Scarpelli, A.C.; Tirsa-Costa, D.; Guimaraes, F.H.; Pordeus, I.A.; Paiva, S.M. Sleep bruxism, awake bruxism and sleep quality among Brazilian dental students: A cross-sectional study. Braz. Dent. J. 2014, 25, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Bruno, V.; Amato, M.; Catapano, S.; Iovino, P. Dental erosion in patients seeking treatment for gastrointestinal complaints: A case series. J. Med. Case Rep. 2015, 9, 250. [Google Scholar] [CrossRef] [PubMed]
- Zingone, F.; Siniscalchi, M.; Capone, P.; Tortora, R.; Andreozzi, P.; Capone, E.; Ciacci, C. The quality of sleep in patients with coeliac disease. Aliment. Pharmacol. Ther. 2010, 32, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- Zingone, F.; Swift, G.L.; Card, T.R.; Sanders, D.S.; Ludvigsson, J.F.; Bai, J.C. Psychological morbidity of celiac disease: A review of the literature. United Eur. Gastroenterol. J. 2015, 3, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Passananti, V.; Siniscalchi, M.; Zingone, F.; Bucci, C.; Tortora, R.; Iovino, P.; Ciacci, C. Prevalence of eating disorders in adults with celiac disease. Gastroenterol. Res. Pract. 2013, 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- De Rosa, A.; Troncone, A.; Vacca, M.; Ciacci, C. Characteristics and quality of illness behavior in celiac disease. Psychosomatics 2004, 45, 336–342. [Google Scholar] [CrossRef] [PubMed]
| CD Patients (49) n (%) | Controls (51) n (%) | p | |
|---|---|---|---|
| RAS | 26 (53) | 13 (25.5) | 0.005 |
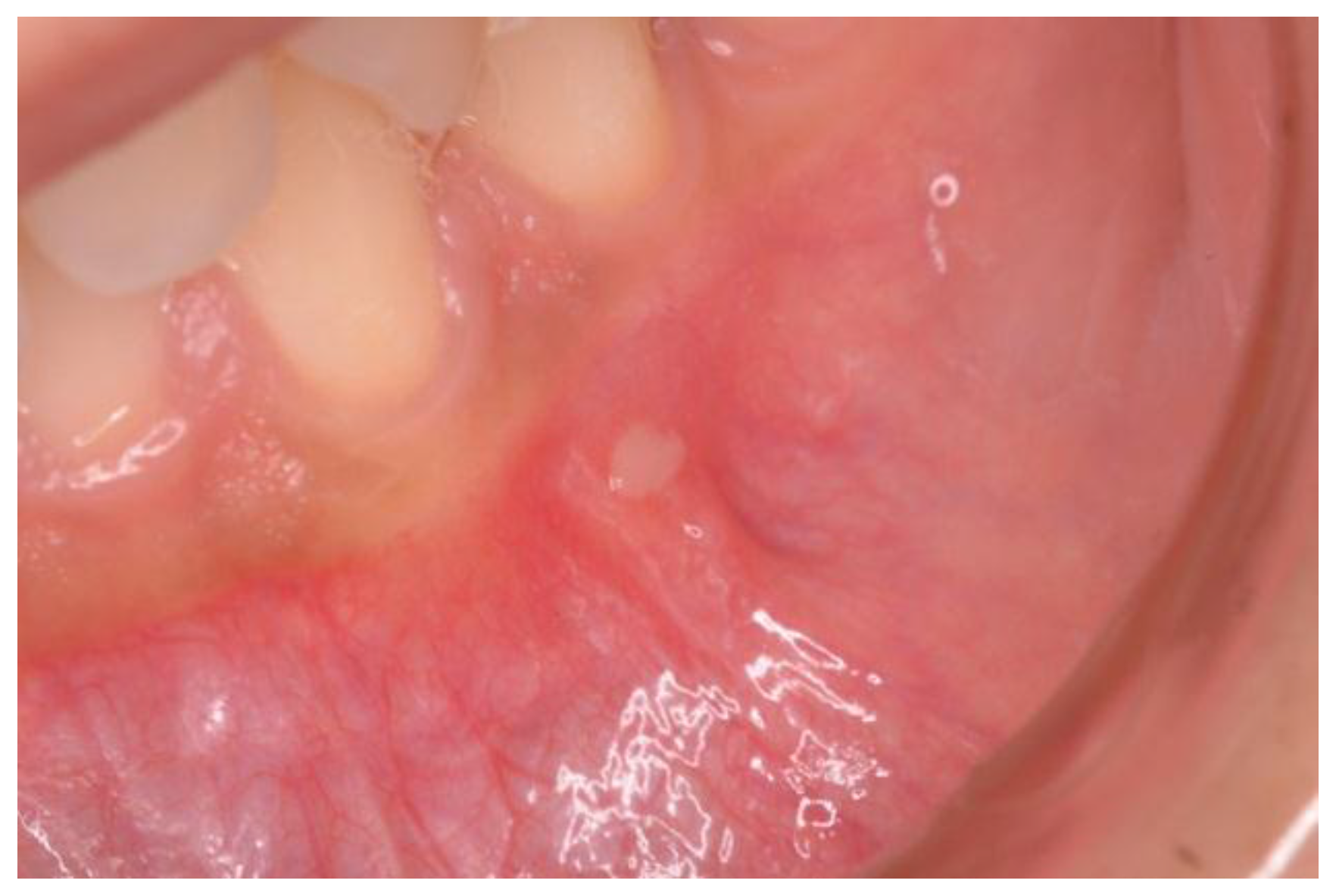
| Aphthosis during visit | 0 | 0 | |
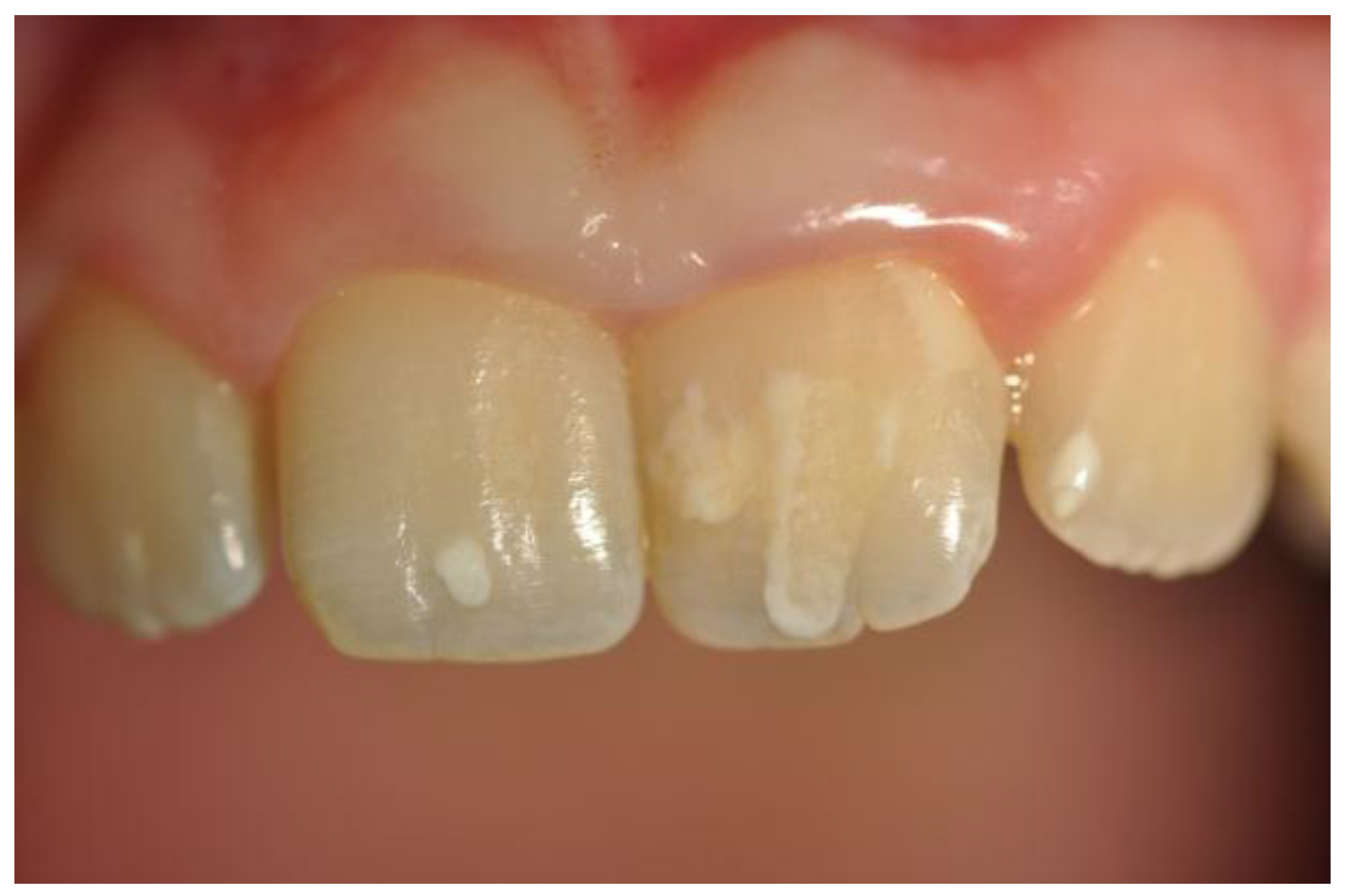
| Enamel hypoplasia | 7 (14.3) | 0 | 0.002 |
| Aine grade 1 | 4 | ||
| Aine grade 2 | 3 | ||
| Aine grade 3 | 0 | ||
| Aine grade 4 | 0 | ||
| Atrophic glossitis | 0 | 0 | |
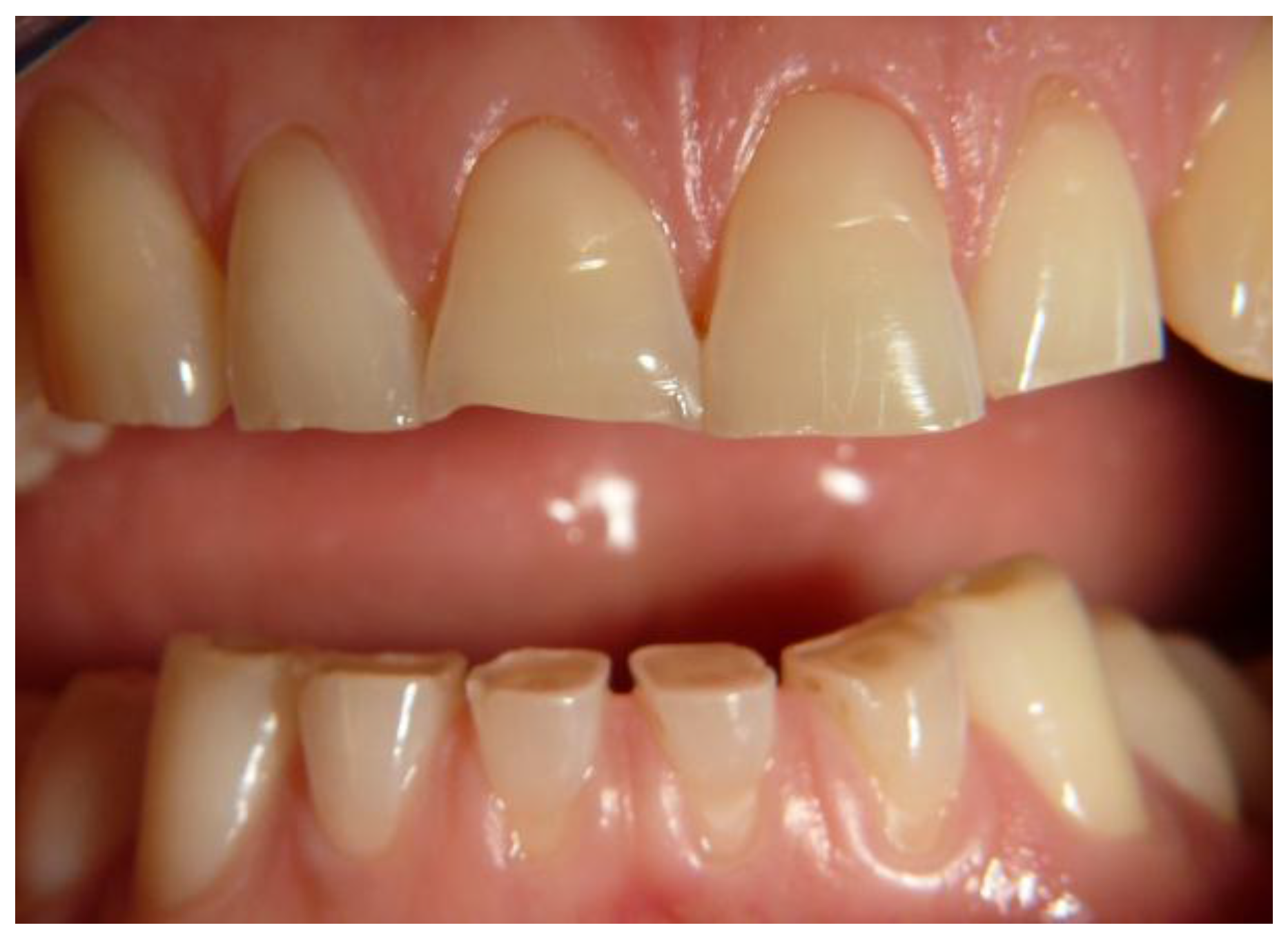
| Non-specific enamel wear | 9 (18.3) | 3 (5.9) | 0.05 |
| Smith and Knight index grade 1 | 4 | 3 | |
| Smith and Knight index grade 2 | 3 | 0 | |
| Smith and Knight index grade 3 | 2 | 0 | |
| Smith and Knight index grade 4 | 0 | 0 |
| Author | Country | Number of Subjects | Population studied | Prevalence of RAS Detected |
|---|---|---|---|---|
| Bucci 2006 [24] | Italy | CD Patients: 72 Controls: 162 | Children | CD Patients: 33.3% Controls: 23.4% |
| Campisi 2007 [9] | Italy | CD Patients: (90/107) Controls: (180/233) | Adults and Children | Adult CD Patients: 21% Adult Controls: 0.5% Pediatric CD Patients: 17% Pediatric Controls: 1% |
| Procaccini 2007 [20] | Italy | CD Patients: 50 Controls: 50 | Children/young adult | CD Patients: 36% Controls: 12% |
| Cheng 2010 [21] | USA | CD Patients: 67 Controls: 69 | Adults and Children | CD Patients: 42.4% Controls: 23.2% |
| Costacurta 2010 [25] | Italy | CD Patients: 300 Controls: 300 | Children | CD Patients: 8.3% Controls: 3% |
| Erriu 2013 [26] | Italy | CD Patients: 44 | Children | CD Patients: 18.2% |
| de Carvalho 2015 [22] | Brazil | CD Patients: 52 Controls: 52 | Children | CD Patients: 40.3% Controls: 17.3% |
| Cantekin 2015 [23] | Turkey | CD Patients: 25 Controls: 25 | Children | CD Patients: 44% Controls: 0% |
| This Study 2017 | Italy | CD Patients: 49 Controls: 51 | Adults | CD Patients: 53% Controls: 25.5% |
| Author | Country | Number of Subjects | Population studied | Prevalence of Enamel Defects |
|---|---|---|---|---|
| Bucci 2006 [24] | Italy | CD Patients: 72 Controls: 162 | Children | CD Patients: 20% Controls: 5.6% |
| Campisi 2007 [9] | Italy | CD Patients: (90/107) Controls: (180/233) | Adults and Children | Adult CD Patients: 23% Adult Controls: 8% Pediatric CD Patients: 23% Pediatric Controls: 9% |
| Wierink 2007 [34] | Netherlands | CD Patients: 53 Controls: 28 | Children | CD Patients: 55% Controls: 18% |
| Procaccini 2007 [20] | Italy | CD Patients: 50 Controls: 50 | Children/young adult | CD Patients: 26% Controls: 16% |
| Ortega Paez 2008 [33] | Spain | CD Patients: 30 Controls: 30 | Children | CD Patients: 83.3% Controls: 53.3% |
| Cheng 2010 [21] | USA | CD Patients: 44/23 Controls: 45/24 | Adults and Children | Adult CD Patients:32% Adult Controls: 29% Pediatric CD Patients: 87% Pediatric Controls: 33% |
| Costacurta 2010 [25] | Italy | CD Patients: 300 Controls: 300 | Children | CD Patients: 33% Controls: 11% |
| Erriu 2013 [26] | Italy | CD Patients: 44 | Children | CD Patients: 38.6% |
| Trotta 2014 [8] | Italy | CD Patients: 54 | Adults | CD Patients: 85.2% |
| de Carvalho 2015 [22] | Brazil | CD Patients: 52 Controls: 52 | Children | CD Patients: 61.5% Controls: 21.1% |
| Cantekin 2015 [23] | Turkey | CD Patients: 25 Controls: 25 | Children | CD Patients: 48% Controls: 16% |
| This Study 2017 | Italy | CD Patients: 49 Controls: 51 | Adults | CD Patients: 14.3% Controls: 0% |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amato, M.; Zingone, F.; Caggiano, M.; Iovino, P.; Bucci, C.; Ciacci, C. Tooth Wear Is Frequent in Adult Patients with Celiac Disease. Nutrients 2017, 9, 1321. https://doi.org/10.3390/nu9121321
Amato M, Zingone F, Caggiano M, Iovino P, Bucci C, Ciacci C. Tooth Wear Is Frequent in Adult Patients with Celiac Disease. Nutrients. 2017; 9(12):1321. https://doi.org/10.3390/nu9121321
Chicago/Turabian StyleAmato, Massimo, Fabiana Zingone, Mario Caggiano, Paola Iovino, Cristina Bucci, and Carolina Ciacci. 2017. "Tooth Wear Is Frequent in Adult Patients with Celiac Disease" Nutrients 9, no. 12: 1321. https://doi.org/10.3390/nu9121321
APA StyleAmato, M., Zingone, F., Caggiano, M., Iovino, P., Bucci, C., & Ciacci, C. (2017). Tooth Wear Is Frequent in Adult Patients with Celiac Disease. Nutrients, 9(12), 1321. https://doi.org/10.3390/nu9121321







