The Effects of Phytosterols Extracted from Diascorea alata on the Antioxidant Activity, Plasma Lipids, and Hematological Profiles in Taiwanese Menopausal Women
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Saponification and Derivatization
2.3. Phytosterol Analysis
2.4. Quantification Analysis
2.5. Design
2.6. Intervention
2.7. Experimental Methods
2.8. Statistical Analysis
3. Results
Participants and Follow-Up
4. Discussion
4.1. Main Findings
4.2. Aging and Hematopoiesis
4.3. Effects of Phytosterols/Diascorea on Blood Cells in Menopause
4.4. Lipid Profiles in Postmenopausal Women
4.5. The Effects of Phytosterols on Lipid Profiles
4.6. Modulating Antioxidant Activity by Diascorea/Phytosterols
4.7. Strengths and Limitations
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pai, J.K.; Manson, J.E. Acceleration of cardiovascular risk during the late menopausal transition. Menopause 2013, 20, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Samsioe, G. Estrogen therapy for cardiovascular disease. In The Management of the Menopause, 3rd ed.; Studd, J., Ed.; The Parthenon Publishing Group: London, UK, 2003; pp. 17–26. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M. Role of free radicals and catalytic metal ions in human disease: An overview. Methods Enzymol. 1990, 186, 1–85. [Google Scholar] [PubMed]
- Ross, R. The pathogenesis of atherosclerosis: A perspective for the 1990s. Nature 1993, 362, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Ross, R. Atherosclerosis, an inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Akunuru, S.; Geiger, H. Aging, clonality, and rejuvenation of hematopoietic stem cells. Trends Mol. Med. 2016, 22, 701–712. [Google Scholar] [CrossRef] [PubMed]
- Chatta, G.S.; Dale, D.C. Aging and haemopoiesis: Implications for treatment with haemopoietic growth factors. Drugs Aging 1996, 9, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Mirand, E.A.; Gordon, A.S. Mechanism of estrogen action in erythropoeisis. Endocrinology 1966, 78, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Horiguchi, H.; Oguma, E.; Kayama, F. The effects of iron deficiency on estradiol-induced suppression of erythropoietin induction in rats: Implications of pregnancy-related anemia. Blood 2005, 106, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Bodis, J.M.; Koppan, K.; Zambo, G.K.; Torok, A. Estrogen: An instrument or the conductor of the orchestra? Hum. Reprod. 2003, 18, 1561–1563. [Google Scholar] [PubMed]
- Liu, S.Y.; Wang, J.Y.; Shyu, Y.T.; Song, L.M. Studies on yams (Diascorea spp.) in Taiwan. J. Chin. Med. 1995, 6, 111–126. [Google Scholar]
- Wu, W.H.; Liu, L.Y.; Chung, C.J.; Jou, H.J.; Wang, T.A. Estrogenic effect of yam ingestion in healthy postmenopausal women. J. Am. Coll. Nutr. 2005, 24, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.C.; Kuo, H.C.; Chang, S.Y.; Wu, T.C.; Huang, K.E. The assessment of efficacy of Diascoreaalata for menopausal symptom treatment in Taiwanese women. Climacteric 2011, 14, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Barbaglia, G.; Macià, F.; Comas, M.; Sala, M.; del Mar Vernet, M.; Casamitjana, M.; Castells, X. Trends in hormone therapy use before and after publication of the Women’s Health Initiative trial: 10 years of follow-up. Menopause 2009, 16, 1061–1064. [Google Scholar] [CrossRef] [PubMed]
- Low Dog, T. Menopause: A review of botanical dietary supplements. Am. J. Med. 2005, 118, 98S–108S. [Google Scholar] [CrossRef] [PubMed]
- Awad, A.B.; Chan, K.C.; Downie, A.C.; Fink, C.S. Peanuts as a source of beta-sitosterol, a sterol with anticancer properties. Nutr. Cancer 2000, 36, 238–241. [Google Scholar] [CrossRef] [PubMed]
- Van Zant, G.; Liang, Y. The role of stem cells in aging. Exp. Hematol. 2003, 31, 659–672. [Google Scholar] [CrossRef]
- Balducci, L.; Hardy, C.L.; Lyman, G.H. Hemopoietic reserve in the older cancer patient: Clinical and economic considerations. Cancer Control 2000, 7, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Chatta, G.S.; Price, T.H.; Allen, R.C.; Dale, D.C. Effects of in vivo recombinant methionyl human granulocyte colony- stimulating factor on the neutrophil response and peripheral blood colony-forming cells in healthy young and elderly adult volunteers. Blood 1994, 84, 2923–2929. [Google Scholar] [PubMed]
- Morris, C.L.; Siegel, E.; Barlogie, B.; Cottler-Fox, M.; Lin, P.; Fassas, A.; Zangari, M.; Anaissie, E.; Tricot, G. Mobilization of CD34+ cells in elderly patients (≥70 years) with multiple myeloma: Influence of age, prior therapy, platelet count and mobilization regimen. Br. J. Haematol. 2003, 120, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Achie, L.N.; Olorunshola, K.V.; Mabrouk, M. A Study of Some Red Cell Indices in Menopausal Women in Zaria, Nigeria. Asian J. Med. Sci. 2011, 3, 154–157. [Google Scholar]
- Kim, H.L.; Cho, H.Y.; Park, I.Y.; Choi, J.M.; Kim, M.; Jang, H.J.; Hwang, S.M. The Positive Association between Peripheral Blood Cell Counts and Bone Mineral Density in Postmenopausal Women. Yonsei Med. J. 2011, 52, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Milman, N.; Kirchhoff, M.; Jorgensen, T. Iron status markers, serum ferritin and hemoglobin in 1359 Danish women in relation to menstruation, hormonal contraception, parity, and postmenopausal hormone treatment. Ann. Haematol. 1992, 65, 96–102. [Google Scholar] [CrossRef]
- Castro, O.L.; Haddy, T.B.; Rana, S.R.; Worrell, K.D.; Scott, R.B. Electronically determined red blood cell values in a large number of healthy black adults. Subpopulations with low hemoglobin and red blood cell indices. Am. J. Epidemiol. 1985, 121, 930–936. [Google Scholar] [CrossRef] [PubMed]
- Cruikshank, J.M. Some variations in the normal haemoglobin concentration. Br. J. Haematol. 2008, 18, 523–530. [Google Scholar] [CrossRef]
- Bain, B.J. Blood Cells: A practical Guide, 4th ed.; Blackwell: Oxford, UK, 2006; pp. 177–187. [Google Scholar]
- Bottero, V.; Withoff, S.; Verma, I.M. NF-kappaB and the regulation of hematopoiesis. Cell Death Differ. 2006, 13, 785–797. [Google Scholar] [CrossRef] [PubMed]
- Valerio, M.; Awad, A.B. β-Sitosterol down-regulates some pro-inflammatory signal transduction pathways by increasing the activity of tyrosine phosphatase SHP-1 in J774A.1 murine macrophages. Int. Immunopharmacol. 2011, 11, 1012–1017. [Google Scholar] [CrossRef] [PubMed]
- Stein, S.J.; Baldwin, A.S. Deletion of the NF-κB subunit p65/RelA in the hematopoietic compartment leads to defects in hematopoietic stem cell function. Blood 2013, 121, 5015–5024. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Xiu, Y.; Ashton, J.; Xing, L.; Morita, Y.; Jordan, C.T.; Boyce, B.F. Noncanonical NF-κB signaling regulates hematopoietic stem cell self-renewal and microenvironment interactions. Stem Cells 2012, 30, 709–718. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, D.M.; Levi, A.J.; Chanarin, I.; North, W.R.S.; Meade, T.W. Mean cell volume in a working population: The effects of age, smoking, alcohol and oral contraception. Br. J. Haematol. 1979, 43, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Laurencet, F. Qualitative changes of hematopoiesis. In Blood Disorders in the Elderly; Balducci, L., Ershler, W., de Gaetano, G., Eds.; Cambridge University Press: Cambridge, UK, 2008; pp. 95–119. [Google Scholar]
- Wagner, E.C.; Prevolsek, J.S.; Wynne-Edwards, K.E.; Williams, T.D. Hematological changes associated with egg production: Estrogen dependence and repeatability. J. Exp. Biol. 2008, 211, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Alexanderson, P.; Toussaint, A.; Christiansen, C.; Devogelaer, J.P.; Roux, C.; Fechtenbaum, J.; Gennari, C.; Reginster, J.Y. Ipriflavone Multicenter European Fracture Study. Ipriflavone in the treatment of postmenopausal osteoporosis: A randomized controlled trial. JAMA 2001, 285, 1482–1488. [Google Scholar] [CrossRef]
- Soung, D.Y.; Patade, A.; Khalil, D.A.; Lucas, E.A.; Devareddy, L.; Greaves, K.A.; Arjmandi, B.H. Soy protein supplementation does not cause lymphocytopenia in postmenopausal women. Nutr. J. 2006, 5, 12. [Google Scholar] [CrossRef] [PubMed]
- Collins, P.; Rosano, G.; Casey, C.; Daly, C.; Gambacciani, M.; Hadji, P.; Kaaja, R.; Mikkola, T.; Palacios, S.; Preston, R.; et al. Management of cardiovascular risk in the perimenopausal women: A consensus statement of European cardiologists and gynecologists. Climacteric 2007, 10, 508–526. [Google Scholar] [CrossRef] [PubMed]
- Matthews, K.A.; Wing, R.R.; Kuller, L.H.; Meilahn, E.N.; Plantinga, P. Influence of the perimenopause on cardiovascular risk factors and symptoms of middle-aged healthy women. Arch. Intern. Med. 1994, 154, 2349–2355. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.J.; Wu, C.H.; Yao, W.J.; Yang, Y.C.; Wu, J.S.; Lu, F.H. Relationships of age, menopause and central obesity on cardiovascular disease risk factors in Chinese women. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 1699–1704. [Google Scholar] [CrossRef] [PubMed]
- Derby, C.A.; Crawford, S.L.; Pasternak, R.C.; Sowers, M.; Sternfeld, B.; Matthews, K.A. Lipid changes during the menopause transition in relation to age and weight: The Study of Women’s Health Across the Nation. Am. J. Epidemiol. 2009, 169, 1352–1361. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Chang, Y.; Kim, B.K.; Kang, D.; Kwon, M.J.; Kim, C.W.; Jeong, C.; Ahn, Y.; Park, H.Y.; Ryu, S.; et al. Menopausal stages and serum lipid and lipoprotein abnormalities in middle-aged women. Maturitas 2015, 80, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Ostlund, R.E., Jr.; Racette, S.B.; Okeke, A.; Stenson, W.F. Phytosterolsthat are naturally present in commercial corn oil significantly reduce cholesterol absorption in humans. Am. J. Clin. Nutr. 2002, 75, 1000–1004. [Google Scholar] [PubMed]
- Marangoni, F.; Poli, A. Phytosterols and cardiovascular health. Pharmacol. Res. 2010, 61, 193–199. [Google Scholar] [CrossRef] [PubMed]
- De Graaf, J.; De Sauvage Nolting, P.R.; Van Dam, M.; Belsey, E.M.; Kastelein, J.J.; Haydn Pritchard, P.; Stalenhoef, A.F. Consumption of tail-oil derivedphytosterols in a chocolate matrix significantly decreases plasma total and low-densitylipoprotein-cholesterol levels. Br. J. Nutr. 2002, 88, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Vanstone, C.A.; Raeini-Sarjaz, M.; Parsons, W.E.; Jones, P.J. Unesterified plant sterols and stanolslower LDL-cholesterol concentrations equivalentlyin hypercholesterolemic persons. Am. J. Clin. Nutr. 2002, 76, 1272–1278. [Google Scholar] [PubMed]
- Law, M. Plant Sterol and Stanol Margarines and Health. Br. Med. J. 2000, 320, 861–864. [Google Scholar] [CrossRef]
- Ostlund, R.E., Jr. Phytosterols in human nutrition. Annu. Rev. Nutr. 2002, 22, 533–549. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, M.P.; Jones, P.J.H. Phytosterols and Human Lipid Metabolism: Efficacy, Safety, and Novel Foods. Lipids 2003, 38, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.J.; Lee, J.H.; Jang, Y.; Lee-Kim, Y.C.; Park, E.; Kim, K.M.; Chung, B.C.; Chung, N. Micellar Phytosterols Effectively Reduce Cholesterol Absorption at Low Doses. Ann. Nutr. Metab. 2005, 49, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Normen, L.; Dutta, P.; Lia, A.; Andersson, H. Soysterol esters and beta-sitostanol ester as inhibitorsof cholesterol absorption in human small bowel. Am. J. Clin. Nutr. 2000, 71, 908–913. [Google Scholar] [PubMed]
- Ostlund, R.E., Jr.; Spilburg, C.A.; Stenson, W.F. Sitostanol Administered in Lecithin Micelles Potently Reduces Cholesterol Absorption in Humans. Am. J. Clin. Nutr. 1999, 70, 826–831. [Google Scholar] [PubMed]
- Genser, B.; Silbernagel, G.; Backer, G.D.; Bruckert, E.; Carmena, R.; Chapman, M.J.; Deanfield, J.; Descamps, O.S.; Rietzschel, E.R.; Dias, K.C.; et al. Plant sterols and cardiovascular disease: A systematic review and meta-analysis. Eur. Heart J. 2012, 33, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Niki, E. Antioxidant Effects of Phytosterol and Its Components. J. Nutr. Sci. Vitaminol. 2003, 49, 277–280. [Google Scholar] [CrossRef] [PubMed]
- Vivancos, M.; Moreno, J.J. β-Sitosterol modulates antioxidant enzyme response in RAW 264.7 macrophages. Free Radic. Biol. Med. 2005, 39, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, G.; Bacchetti, T.; Masciangelo, S.; Bicchiega, V. Effect of phytosterols on copper lipid peroxidation of human low-density lipoproteins. Nutrition 2010, 26, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Hu, H.; Li, C. Antioxidant activities of novel galloyl phytosterols evaluated by human erythrocytes with the aid of confocal microscopy imaging. J. Funct. Foods 2016, 22, 224–231. [Google Scholar] [CrossRef]
- Strehlow, K.; Rotter, S.; Wassmann, S.; Adam, O.; Grohe, C.; Laufs, K.; Bohm, M.; Nickenig, G. Modulation of antioxidant enzyme expression and function by estrogen. Circ. Res. 2003, 93, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Gutendorf, B.; Westendorf, J. Comparison of an array of in vitro assays for the assessment of the estrogenic potential of natural and synthetic estrogens, phytoestrogens and xenoestrogens. Toxicology 2001, 166, 79–89. [Google Scholar] [CrossRef]
- Bu, D.X.; Erl, W.; de Martin, R.; Hansson, G.K.; Yan, Z.Q. IKKbeta-dependent NF-kappaB pathway controls vascular inflammation and intimal hyperplasia. FASEB J. 2005, 19, 1293–1295. [Google Scholar] [PubMed]
- Hussain, N.; Mohamad, R. Effect of Different Solvents on Phytosterols and Antioxidant Activity of Cocoa Beans. Int. J. Food Eng. 2015, 1, 18–22. [Google Scholar]
- Wang, C.H.; Chen, M.J.; Tseng, C.Y.; Wei, K.M.; Chen, W. Structural characterization of functional compositions isolated from Dioscorea Purpurea (Cultivar of Ming-Chien) by Raman Spectroscopy. Nat. Prod. Chem. Res. 2013, 1, 3. [Google Scholar] [CrossRef]
- Chen, Y.T.; Kao, W.T.; Lin, K.W. Effects of pH on the total phenolic compound, antioxidative ability and the stability of dioscorin of various yam cultivars. Food Chem. 2008, 107, 250–257. [Google Scholar] [CrossRef]
- Hsu, F.L.; Lin, Y.H.; Lee, M.H.; Lin, C.L.; Hou, W.C. Both dioscorin, the tuber storage protein of yam (Dioscoreaalata cv. Tainong No. 1), and its peptic hydrolysates exhibited angiotensin converting enzyme inhibitory activities. J. Agric. Food Chem. 2002, 50, 6109–6113. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Kuan, T.C.; Hsieh, Y.Y.; Ho, Y.J.; Sun, Y.L.; Lin, C.S. Effects of diosgenin on myometrial matrix metalloproteinase-2 and -9 activity and expression in ovariectomized rats. Int. J. Biol. Sci. 2011, 7, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Wojcikowski, K.; Wohlmuth, H.; Johnson, D.W.; Gobe, G. Dioscorea villosa (wild yam) induces chronic kidney injury via pro-fibrotic pathways. Food Chem. Toxicol. 2008, 46, 3122–3123. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C.; Tsai, C.C.; Chen, J.C.; Lin, J.G.; Lin, C.C.; Hu, M.L.; Lu, S. Effects of “Chinese yam” on hepato-nephrotoxicity of acetaminophen in rats. Acta. Pharmacol. Sin. 2002, 23, 503–508. [Google Scholar] [PubMed]
| Diascorea (n = 25) | Placebo (n = 25) | F (0 and 6) | F (0 and 12) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Month 0 | Month 6 | Month 12 | Month 0 | Month 6 | Month 12 | |||||||||||
| Body weight (kg) | 56.40 | (8.32) | 56.40 | (8.22) | 55.80 | (8.03) | 58.85 | (8.24) | 58.60 | (8.46) | 58.65 | (8.54) | 0.98 | 0.37 | ||
| BMI (kg/m2) | 23.37 | (3.93) | 23.12 | (3.59) | 22.81 | (3.16) | 24.19 | (3.13) | 23.95 | (2.99) | 24.20 | (3.24) | 0.22 | 6.69 | * | |
| Systolic blood pressure (mmHg) | 118.68 | (13.48) | 111.92 | (12.11) | 113.12 | (14.72) | 125.16 | (18.67) | 119.76 | (13.43) | 116.12 | (13.96) | 2.59 | 0.12 | ||
| Diastolic blood pressure (mmHg) | 74.68 | (9.75) | 73.72 | (10.92) | 73.56 | (11.08) | 74.52 | (15.22) | 72.36 | (10.45) | 71.48 | (11.57) | 0.36 | 0.51 | ||
| Glucose fasting (mg/dL) | 82.48 | (6.47) | 76.80 | (4.76) | 90.36 | (8.06) | 99.40 | (18.77) | 84.28 | (19.73) | 94.56 | (28.36) | 0.19 | 2.78 | ||
| Uric acid (mg/dL) | 5.04 | (1.35) | 5.41 | (1.39) | 5.08 | (1.46) | 5.43 | (1.49) | 5.70 | (1.30) | 5.41 | (1.48) | 0.01 | 0.02 | ||
| Creatinine (mg/dL) | 0.78 | (0.08) | 0.78 | (0.07) | 0.74 | (0.09) | 0.84 | (0.13) | 0.84 | (0.10) | 0.80 | (0.10) | 2.19 | 1.30 | ||
| SGOT (U/L) | 25.68 | (6.45) | 26.64 | (6.30) | 21.96 | (5.96) | 27.24 | (7.89) | 25.56 | (5.93) | 25.28 | (10.05) | 1.58 | 1.39 | ||
| SGPT (U/L) | 18.68 | (5.61) | 12.56 | (6.41) | 18.48 | (5.39) | 20.88 | (9.62) | 16.68 | (6.92) | 23.44 | (13.77) | 3.65 | 1.97 | ||
| BMD (g/cm2) | 0.91 | (0.15) | 0.90 | (0.15) | 0.89 | (0.15) | 0.95 | (0.14) | 0.94 | (0.14) | 0.94 | (0.14) | 1.08 | 3.89 | ||
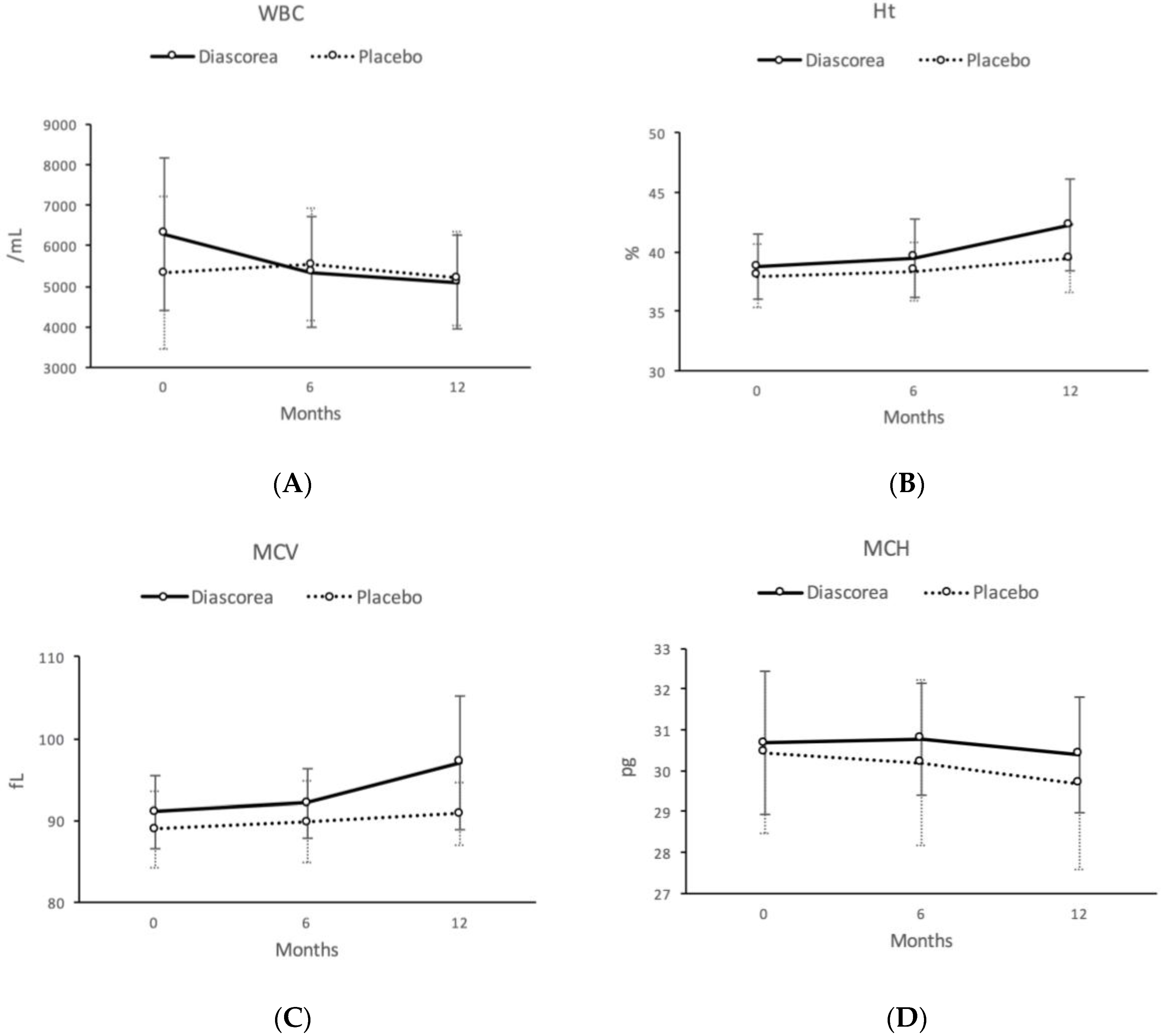
| WBC (/mL) | 6288.0 | (1870.5) | 5352.0 | (1380.9) | 5098.8 | (1151.6) | 5322.4 | (1200.1) | 5536.0 | (1235.8) | 5200.0 | (1208.9) | 14.22 | ** | 5.49 | * |
| RBC (109/dL) | 426.68 | (37.46) | 429.88 | (43.46) | 436.92 | (42.82) | 428.48 | (41.07) | 428.68 | (39.36) | 435.60 | (43.59) | 0.33 | 0.28 | ||
| Hb (g/dL) | 13.05 | (0.92) | 13.20 | (1.08) | 13.23 | (0.90) | 13.00 | (1.05) | 12.90 | (0.96) | 12.87 | (0.91) | 2.22 | 3.59 | ||
| Ht (%) | 38.73 | (2.73) | 39.48 | (3.27) | 42.25 | (3.89) | 38.00 | (2.72) | 38.36 | (2.46) | 39.44 | (2.88) | 1.00 | 8.57 | ** | |
| MCV (fL) | 91.01 | (4.49) | 92.09 | (4.23) | 97.08 | (8.23) | 88.96 | (4.64) | 89.79 | (5.03) | 90.80 | (3.90) | 0.52 | 10.13 | ** | |
| MCH (pg) | 30.68 | (1.76) | 30.78 | (1.38) | 30.40 | (1.41) | 30.44 | (1.98) | 30.20 | (2.03) | 29.69 | (2.10) | 3.84 | 6.30 | * | |
| MCHC (g/L) | 33.70 | (0.82) | 33.44 | (0.79) | 31.45 | (2.07) | 34.20 | (1.03) | 33.64 | (1.00) | 32.67 | (1.36) | 1.15 | 2.70 | ||
| Platelet (109/L) | 24.54 | (4.78) | 23.07 | (4.35) | 23.50 | (5.04) | 23.42 | (4.62) | 24.25 | (4.52) | 22.98 | (4.69) | 6.73 | * | 0.23 | |
| Diascorea (n = 25) | Placebo (n = 25) | F (0 and 6) | F (0 and 12) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Month 0 | Month 6 | Month 12 | Month 0 | Month 6 | Month 12 | ||||||||||
| TAS (mmole/L) | 8.56 | (4.87) | 10.64 | (6.67) | 10.39 | (6.36) | 11.65 | (5.49) | 9.22 | (5.17) | 9.09 | (4.76) | 2.36 | 2.51 | |
| MDA (nmol/mg) | 36.03 | (8.19) | 18.81 | (4.25) | 12.49 | (2.31) | 30.95 | (10.44) | 14.58 | (3.46) | 12.92 | (2.03) | 16.47 | *** | 0.16 |
| Cholesterol (mg/dL) | 212.28 | (36.58) | 213.28 | (38.05) | 200.88 | (36.84) | 229.16 | (38.41) | 222.08 | (31.62) | 215.48 | (37.15) | 0.15 | 0.06 | |
| Triglyceride (mg/dL) | 159.00 | (138.82) | 144.12 | (134.39) | 118.0 | (82.52) | 123.56 | (78.51) | 131.64 | (73.64) | 123.80 | (66.43) | 0.73 | 2.23 | |
| HDL (mg/dL) | 68.60 | (15.82) | 56.73 | (12.44) | 55.31 | (10.80) | 72.12 | (20.55) | 58.81 | (15.78) | 59.46 | (18.06) | 0.07 | 0.51 | |
| VLDL (mg/dL) | 19.02 | (13.93) | 14.83 | (12.54) | 9.12 | (5.80) | 17.77 | (18.53) | 16.26 | (10.54) | 9.36 | (5.29) | 0.54 | 0.13 | |
| LDL (mg/dL) | 124.65 | (21.78) | 141.74 | (27.23) | 136.44 | (33.78) | 139.39 | (26.74) | 147.00 | (23.83) | 146.68 | (37.07) | 0.77 | 0.49 | |
| T.CHO/HDL | 3.21 | (0.72) | 3.90 | (0.98) | 3.74 | (0.95) | 3.40 | (1.14) | 4.01 | (1.18) | 3.94 | (1.39) | 0.00 | 0.03 | |
| LDL/HDL | 1.90 | (0.50) | 2.60 | (0.69) | 2.56 | (0.84) | 2.08 | (0.71) | 2.69 | (0.90) | 2.75 | (1.25) | 0.01 | 0.00 | |
| Apolipo.Al (mg/dL) | 162.26 | (28.84) | 153.68 | (14.63) | 152.52 | (15.08) | 159.06 | (44.69) | 159.64 | (25.63) | 152.18 | (24.21) | 1.53 | 0.01 | |
| Apolipo.B (mg/dL) | 95.04 | (21.84) | 100.45 | (21.00) | 87.60 | (20.24) | 99.70 | (23.75) | 105.37 | (24.12) | 94.28 | (26.18) | 0.12 | 0.51 | |
| Number of breast cyst | 1.04 | (1.17) | 0.52 | (1.05) | 0.76 | (1.30) | 0.20 | (0.41) | 0.12 | (0.33) | 0.32 | (0.85) | 0.72 | 0.45 | |
| Total size of breast cyst (cm) | 0.61 | (0.75) | 0.34 | (0.80) | 0.39 | (0.74) | 0.11 | (0.24) | 0.06 | (0.15) | 0.17 | (0.49) | 0.42 | 0.81 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsu, C.-C.; Kuo, H.-C.; Huang, K.-E. The Effects of Phytosterols Extracted from Diascorea alata on the Antioxidant Activity, Plasma Lipids, and Hematological Profiles in Taiwanese Menopausal Women. Nutrients 2017, 9, 1320. https://doi.org/10.3390/nu9121320
Hsu C-C, Kuo H-C, Huang K-E. The Effects of Phytosterols Extracted from Diascorea alata on the Antioxidant Activity, Plasma Lipids, and Hematological Profiles in Taiwanese Menopausal Women. Nutrients. 2017; 9(12):1320. https://doi.org/10.3390/nu9121320
Chicago/Turabian StyleHsu, Chao-Chin, Hsin-Chih Kuo, and Ko-En Huang. 2017. "The Effects of Phytosterols Extracted from Diascorea alata on the Antioxidant Activity, Plasma Lipids, and Hematological Profiles in Taiwanese Menopausal Women" Nutrients 9, no. 12: 1320. https://doi.org/10.3390/nu9121320
APA StyleHsu, C.-C., Kuo, H.-C., & Huang, K.-E. (2017). The Effects of Phytosterols Extracted from Diascorea alata on the Antioxidant Activity, Plasma Lipids, and Hematological Profiles in Taiwanese Menopausal Women. Nutrients, 9(12), 1320. https://doi.org/10.3390/nu9121320





