Asthmatic Patients with Vitamin D Deficiency have Decreased Exacerbations after Vitamin Replacement
Abstract
:1. Introduction
2. Materials and Methods
- a cross-sectional retrospective study on a cohort of asthmatic patients
- intervention study in patients with vitamin D deficiency, consisting in one year vitamin D supplementation.
2.1 Phase 1–Cross-Sectional Study
2.2 Phase 2–Intervention Study
2.3 Statistical Analysis
3. Results
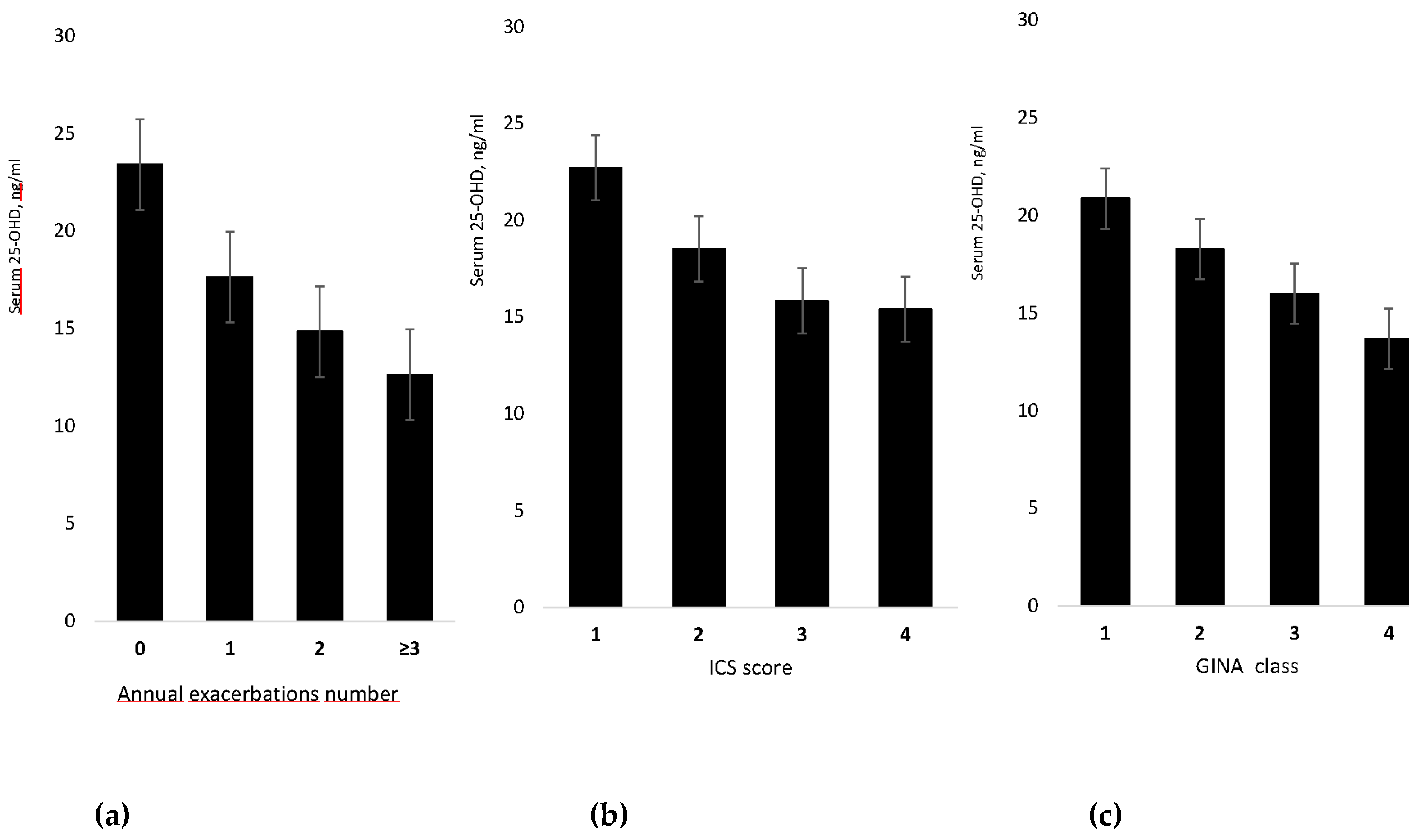
3.1 Cross-Sectional Study
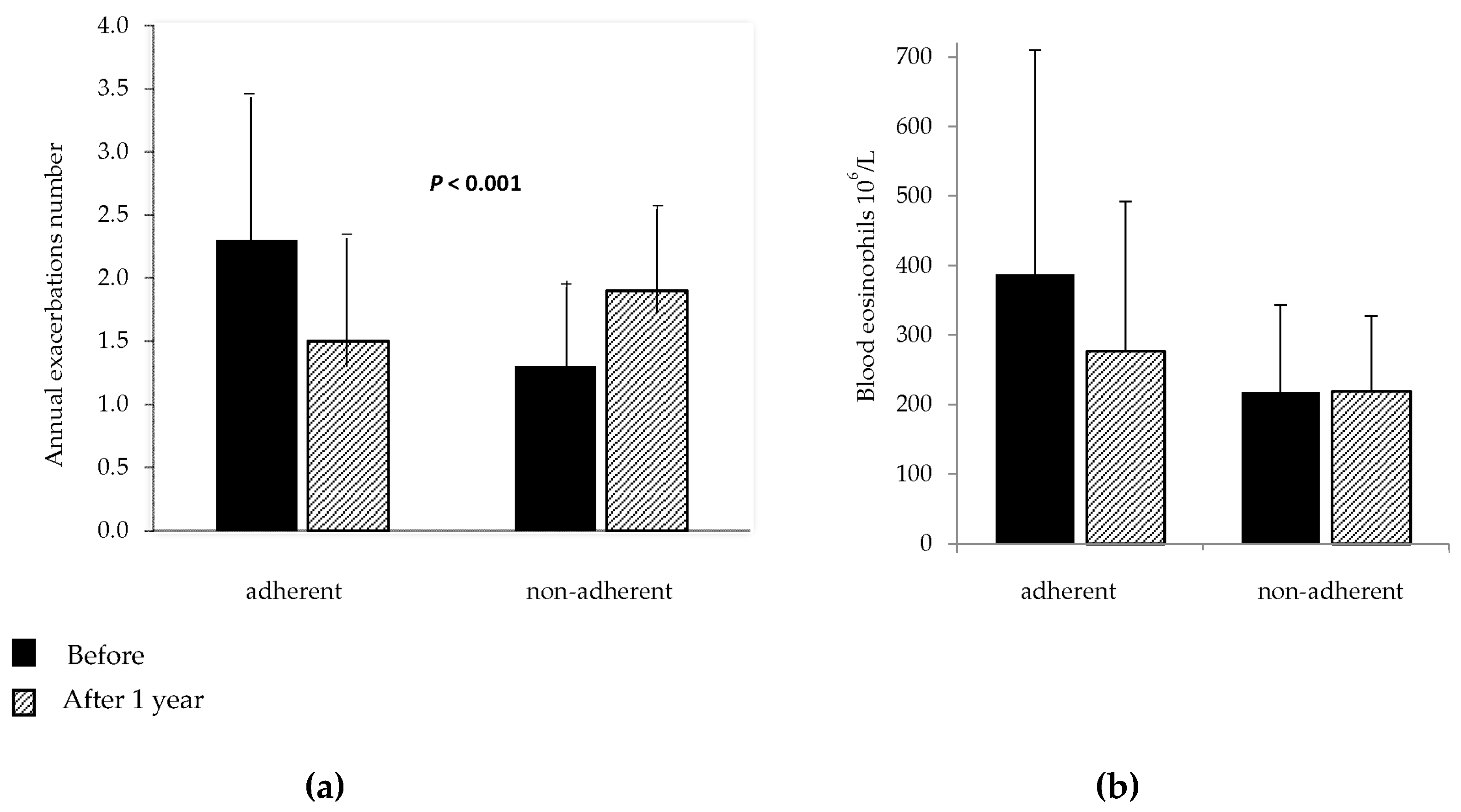
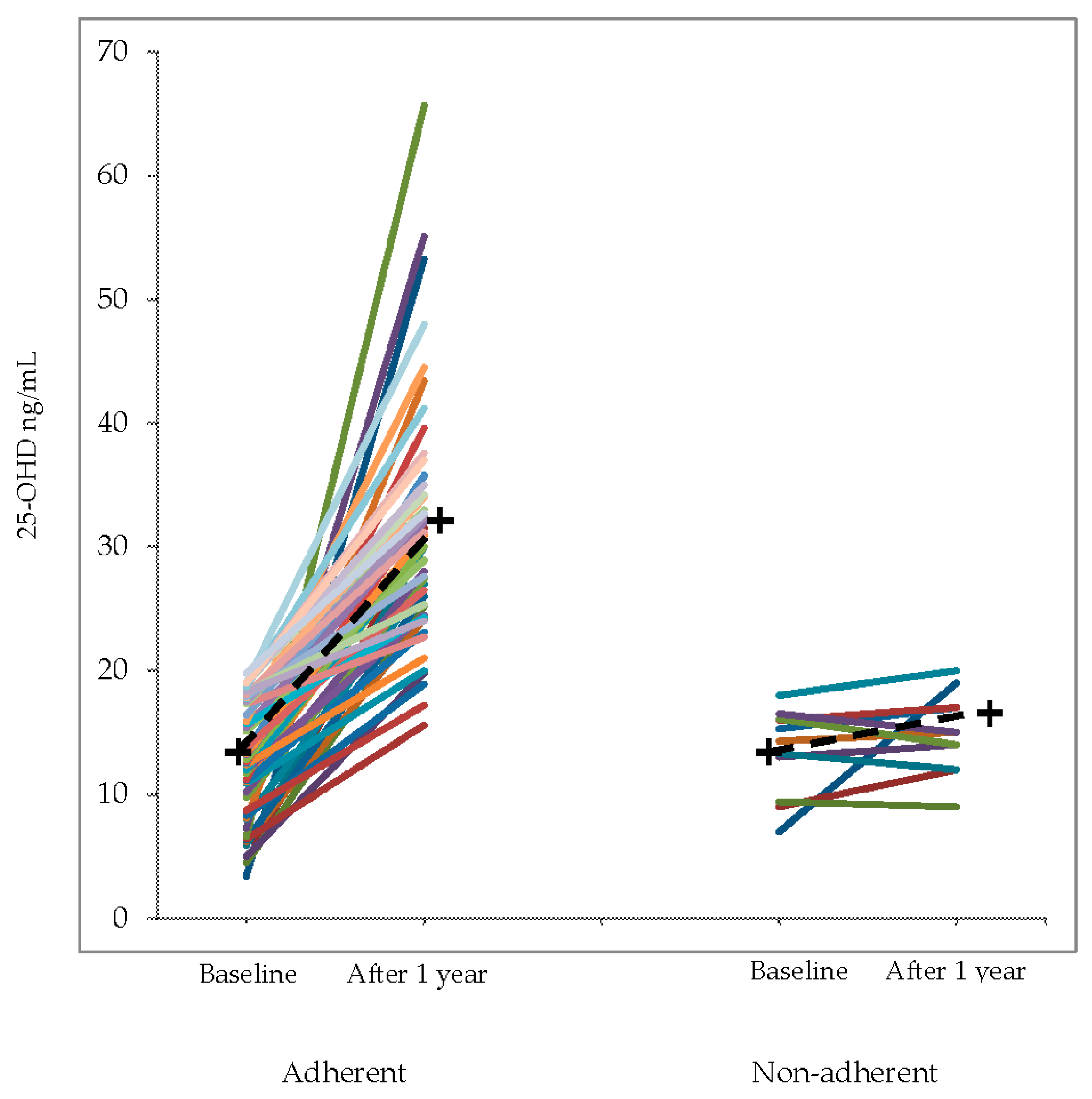
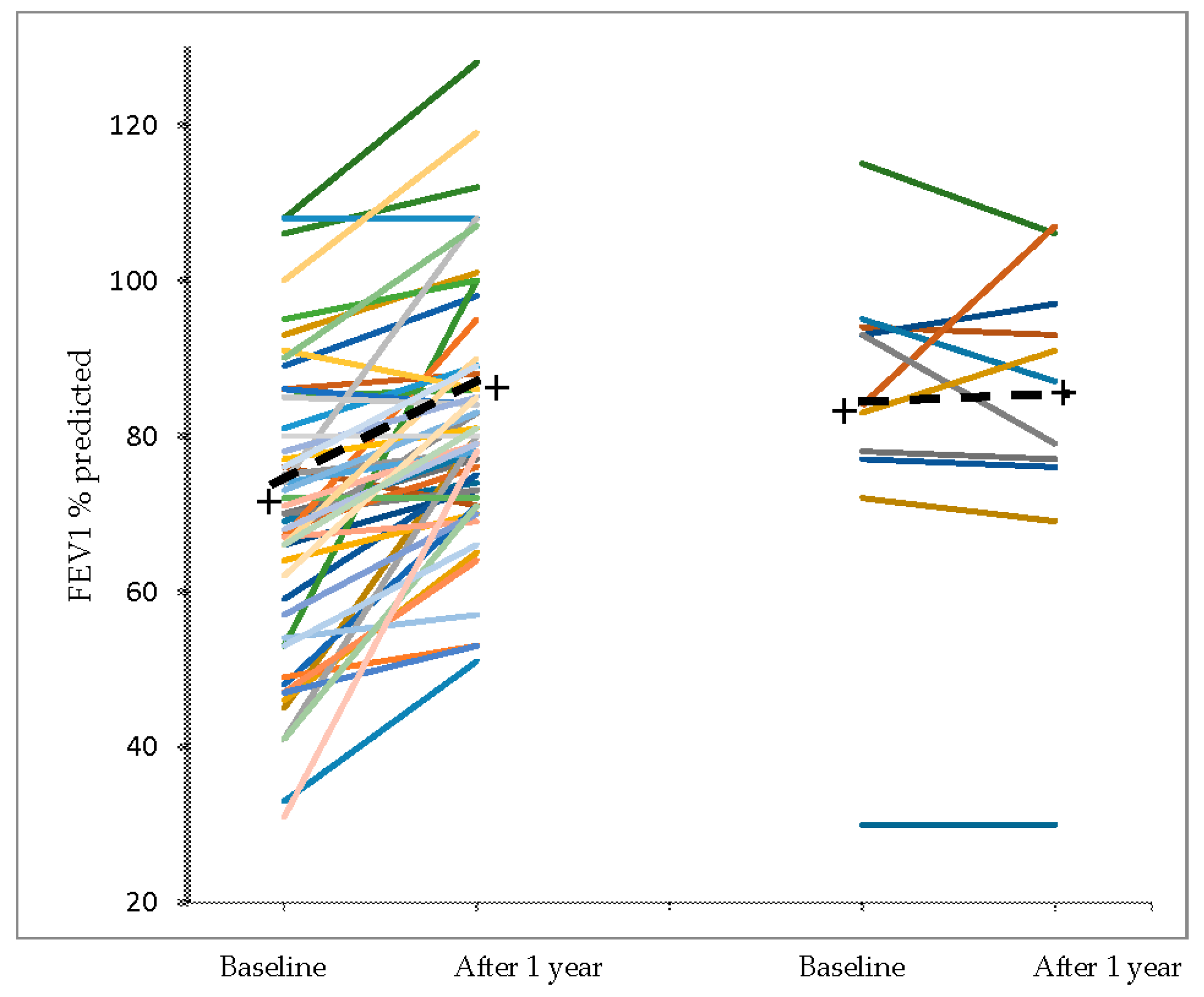
3.2 Phase 2
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Eder, W.; Ege, M.J.; von Mutius, E. The asthma epidemic. N. Engl. J. Med. 2006, 355, 2226–2235. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Aligne, C.A.; Auinger, P.; Byrd, R.S.; Weitzman, M. Risk factors for pediatric asthma. Contributions of poverty, race, and urban residence. Am. J. Respir. Crit. Care Med. 2000, 162, 873–877. [Google Scholar] [CrossRef] [PubMed]
- Clemens, T.L.; Adams, J.S.; Henderson, S.L.; Holick, M.F. Increased skin pigment reduces the capacity of skin to synthesise vitamin D3. Lancet 1982, 1, 74–76. [Google Scholar] [CrossRef] [PubMed]
- Masoli, M.; Fabian, D.; Holt, S.; Beasley, R. The global burden of asthma: Executive summary of the GINA Dissemination Committee report. Allergy 2004, 59, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Beuther, D.A.; Sutherland, E.R. Overweight, obesity, and incident asthma: A meta-analysis of prospective epidemiologic studies. Am. J. Respir. Crit. Care Med. 2007, 175, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Krstic, G. Asthma prevalence associated with geographical latitude and regional insolation in the United States of America and Australia. PLoS ONE 2011, 6, e18492. [Google Scholar] [CrossRef] [PubMed]
- Devgun, M.S.; Paterson, C.R.; Johnson, B.E.; Cohen, C. Vitamin D nutrition in relation to season and occupation. Am. J. Clin. Nutr. 1981, 34, 1501–1504. [Google Scholar] [PubMed]
- Ji, J.; Hemminki, K.; Sundquist, K.; Sundquist, J. Seasonal and regional variations of asthma and association with osteoporosis: Possible role of vitamin D in asthma. J. Asthma Off. J. Assoc. Care Asthma 2010, 47, 1045–1048. [Google Scholar] [CrossRef] [PubMed]
- Litonjua, A.A.; Weiss, S.T. Is vitamin D deficiency to blame for the asthma epidemic? J. Allergy Clin. Immunol. 2007, 120, 1031–1035. [Google Scholar] [CrossRef] [PubMed]
- Kerley, C.P.; Elnazir, B.; Faul, J.; Cormican, L. Vitamin D as an adjunctive therapy in asthma. Part 1: A review of potential mechanisms. Pulm. Pharmacol. Ther. 2015, 32, 60–74. [Google Scholar] [CrossRef] [PubMed]
- Mann, E.H.; Chambers, E.S.; Pfeffer, P.E.; Hawrylowicz, C.M. Immunoregulatory mechanisms of vitamin D relevant to respiratory health and asthma. Ann. N. Y. Acad. Sci. 2014, 1317, 57–69. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, T.; Gupta, G.K.; Agrawal, D.K. Calcitriol decreases expression of importin 3 and attenuates RelA translocation in human bronchial smooth muscle cells. J. Clin. Immunol. 2012, 32, 1093–1103. [Google Scholar] [CrossRef] [PubMed]
- Kerley, C.P.; Elnazir, B.; Faul, J.; Cormican, L. Vitamin D as an adjunctive therapy in asthma. Part 2: A review of human studies. Pulm. Pharmacol. Ther. 2015, 32, 75–92. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Liu, D.; Liu, C.T. Can Vitamin D Supplementation in Addition to Asthma Controllers Improve Clinical Outcomes in Patients With Asthma? A Meta-Analysis. Medicine 2015, 94, e2185. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.R.; Cates, C.J.; Urashima, M.; Jensen, M.; Griffiths, A.P.; Nurmatov, U.; Sheikh, A.; Griffiths, C.J. Vitamin D for the management of asthma. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Searing, D.A.; Zhang, Y.; Murphy, J.R.; Hauk, P.J.; Goleva, E.; Leung, D.Y. Decreased serum vitamin D levels in children with asthma are associated with increased corticosteroid use. J. Allergy Clin. Immunol. 2010, 125, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Brehm, J.M.; Schuemann, B.; Fuhlbrigge, A.L.; Hollis, B.W.; Strunk, R.C.; Zeiger, R.S.; Weiss, S.T.; Litonjua, A.A. Childhood Asthma Management Program Research Group. Serum vitamin D levels and severe asthma exacerbations in the Childhood Asthma Management Program study. J. Allergy Clin. Immunol. 2010, 126, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Salas, N.M.; Luo, L.; Harkins, M.S. Vitamin D deficiency and adult asthma exacerbations. J. Asthma 2014, 51, 950–955. [Google Scholar] [CrossRef] [PubMed]
- Urashima, M.; Segawa, T.; Okazaki, M.; Kurihara, M.; Wada, Y.; Ida, H. Randomized trial of vitamin D supplementation to prevent seasonal influenza A in schoolchildren. Am. J. Clin. Nutr. 2010, 91, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- Majak, P.; Olszowiec-Chlebna, M.; Smejda, K.; Stelmach, I. Vitamin D supplementation in children may prevent asthma exacerbation triggered by acute respiratory infection. J. Allergy Clin. Immunol. 2011, 127, 1294–1296. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.; King, T.S.; Kunselman, S.J.; Cabana, M.D.; Denlinger, L.; Holguin, F.; Kazani, S.D.; Moore, W.C.; Moy, J.; Sorkness, C.A.; et al. National Heart, Lung, and Blood Institute’s AsthmaNet. Effect of Vitamin D3 on Asthma Treatment Failures in Adults With Symptomatic Asthma and Lower Vitamin D Levels The VIDA Randomized Clinical Trial. JAMA 2014, 311, 2083–2091. [Google Scholar] [CrossRef] [PubMed]
- Maguire, J.L.; Birken, C.S.; Loeb, M.B.; Mamdani, M.; Thorpe, K.; Hoch, J.S.; Mazzulli, T.; Borkhoff, C.M.; Macarthur, C.; Parkin, P.C. DO IT Trial: Vitamin D Outcomes and Interventions in Toddlers–A TARGet Kids! Randomized controlled trial. BMC Pediatr. 2014, 14, 37. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef]
- Global Initiative for Asthma: GINA Report, Global Strategy for Asthma Management and Prevention—Updated 2016. Available online: http://www.ginasthma.org (accessed on 10 November 2017).
- Bousquet, J.; Heinzerling, L.; Bachert, C.; Papadopoulos, N.G.; Bousquet, P.J.; Burney, P.G.; Canonica, G.W.; Carlsen, K.H.; Cox, L.; Haahtela, T.; et al. Practical guide to skin prick tests in allergy to aeroallergens. Allergy 2012, 67, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Quanjer, P.H.; Tammeling, G.J.; Cotes, J.E.; Pedersen, O.F.; Peslin, R.; Yernault, J.C. Lung volumes and forced ventilatory flows: Report Working Party Standardization of Lung Function Tests, European Community for Steel and Coal [official statement of the European Respiratory Society]. Eur. Respir. J. Suppl. 1993, 16, 5–40. [Google Scholar] [CrossRef] [PubMed]
- Crapo, R.O.; Casaburi, R.; Coates, A.L.; Enright, P.L.; Hankinson, J.L.; Irvin, C.G.; MacIntyre, N.R.; McKay, R.T.; Wanger, J.S.; Anderson, S.D.; et al. Guidelines for methacholine and exercise challenge testing-1999. Am. J. Respir. Crit. Care Med. 2000, 161, 309–329. [Google Scholar] [CrossRef] [PubMed]
- American Thoracic Society; European Respiratory Society. ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005. Am. J. Respir. Crit. Care Med. 2005, 171, 912–930. [Google Scholar] [CrossRef]
- Reddel, H.K.; Taylor, D.R.; Bateman, E.D.; Boulet, L.P.; Boushey, H.A.; Busse, W.W.; Casale, T.B.; Chanez, P.; Enright, P.L.; Gibson, P.G.; et al. An official American Thoracic Society/European respiratory society statement: Asthma control and exacerbations: Standardizing endpoints for clinical asthma trials and clinical practice. Am. J. Respir. Crit. Care Med. 2009, 180, 59–99. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Endocrine Society. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Andersen, R.; Brot, C.; Jakobsen, J.; Mejborn, H.; Mølgaard, C.; Skovgaard, L.T.; Trolle, E.; Tetens, I.; Ovesen, L. Seasonal changes in vitamin D status among Danish adolescent girls and elderly women: The influence of sun exposure and vitamin D intake. Eur. J. Clin. Nutr. 2013, 67, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Burgaz, A.; Akesson, A.; Oster, A.; Michaëlsson, K.; Wolk, A. Associations of diet, supplement use, and ultraviolet B radiation exposure with vitamin D status in Swedish women during winter. Am. J. Clin. Nutr. 2007, 86, 1399–1404. [Google Scholar] [PubMed]
- Brehm, J.M.; Acosta-Perez, E.; Klei, L.; Roeder, K.; Barmada, M.; Boutaoui, N.; Forno, E.; Kelly, R.; Paul, K.; Sylvia, J.; et al. Vitamin D insufficiency and severe asthma exacerbations in Puerto Rican children. Am. J. Respir. Crit. Care Med. 2012, 186, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Beyhan-Sagmen, S.; Baykan, O.; Balcan, B.; Ceyhan, B. Association Between Severe Vitamin D Deficiency, Lung Function and Asthma Control. Arch. Bronconeumol. 2017, 53, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Confino-Cohen, R.; Brufman, I.; Goldberg, A.; Feldman, B.S. Vitamin D, asthma prevalence and asthma exacerbations: A large adult population-based study. Allergy 2014, 69, 1673–1680. [Google Scholar] [CrossRef] [PubMed]
- Korn, S.; Hübner, M.; Jung, M.; Blettner, M.; Buhl, R. Severe and uncontrolled adult asthma is associated with vitamin D insufficiency and deficiency. Respir. Res. 2013, 14, 25. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, E.R.; Goleva, E.; Jackson, L.P.; Stevens, A.D.; Leung, D.Y. Vitamin D levels, lung function, and steroid response in adult asthma. Am. J. Respir. Crit. Care Med. 2010, 181, 699–704. [Google Scholar] [CrossRef] [PubMed]
- Lan, N.; Luo, G.; Yang, X.; Cheng, Y.; Zhang, Y.; Wang, X.; Wang, X.; Xie, T.; Li, G.; Liu, Z.; et al. 25-Hydroxyvitamin D3-Deficiency Enhances Oxidative Stress and Corticosteroid Resistance in Severe Asthma Exacerbation. PLoS ONE 2014, 9, e111599. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Sjoukes, A.; Richards, D.; Banya, W.; Hawrylowicz, C.; Bush, A.; Saglani, S. Relationship between serum vitamin D, disease severity, and airway remodeling in children with asthma. Am. J. Respir. Crit. Care Med. 2011, 184, 1342–1349. [Google Scholar] [CrossRef] [PubMed]
- Afzal, S.; Lange, P.; Bojesen, S.E.; Freiberg, J.J.; Nordestgaard, B.G. Plasma 25-hydroxyvitamin D, lung function and risk of chronic obstructive pulmonary disease. Thorax 2014, 69, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Black, P.N.; Scragg, R. Relationship between serum 25-hydroxyvitamin D and pulmonary function in the third national health and nutrition examination survey. Chest 2005, 128, 3792–3798. [Google Scholar] [CrossRef] [PubMed]
- Larose, T.L.; Langhammer, A.; Chen, Y.; Camargo, C.A., Jr.; Romundstad, P.; Mai, X.M. Serum 25-hydroxyvitamin D levels and lung function in adults with asthma: The HUNT Study. Eur. Respir. J. 2015, 45, 1019–1026. [Google Scholar] [CrossRef] [PubMed]
- Stokes, P.J.; Rimmer, J. The relationship between serum vitamin D and chronic rhinosinusitis: A systematic review. Am. J. Rhinol. Allergy 2016, 30, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Price, D.; Wilson, A.M.; Chisholm, A.; Rigazio, A.; Burden, A.; Thomas, M.; King, C. Predicting frequent asthma exacerbations using blood eosinophil count and other patient data routinely available in clinical practice. J. Asthma Allergy 2016, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- De Groot, J.C.; van Roon, E.N.H.; Storm, H.; Veeger, N.J.; Zwinderman, A.H.; Hiemstra, P.S.; Bel, E.H.; ten Brinke, A. Vitamin D reduces eosinophilic airway inflammation in nonatopic asthma. J. Allergy Clin. Immunol. 2015, 135, 670–675. [Google Scholar] [CrossRef] [PubMed]
- DiPietro, N.A. Methods in epidemiology: Observational study designs. Pharmacotherapy 2010, 30, 973–984. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Zilov, A.; Soewondo, P.; Bech, O.M.; Sekkal, F.; Home, P.D. Observational studies: Going beyond the boundaries of randomized controlled trials. Diabetes Res. Clin. Pract. 2010, 88 (Suppl. 1), S3–S9. [Google Scholar] [CrossRef]

| Women/Men, n | 94/25 |
| Age, years (range) | 52 (13–80) |
| Never smokers, n (%) | 94 (79) |
| Ex-smokers, n (%) | 11 (9) |
| Current smokers, n (%) | 14 (12) |
| Vitamin D status: | |
| Normal, n (%) | 8 (7) |
| insufficient, n (%) | 21 (17) |
| deficient, n (%) | 90 (76) |
| BMI, kg/m2 | 24 ± 0.4 |
| Atopy, n (%) | 83 (70) |
| VC% predicted | 86 ± 1 |
| FEV1% predicted | 78 ± 2 |
| FEV1/VC% | 67 ± 1 |
| FENO, ppb | 42.9 ± 3.9 |
| GINA class: | |
| 1 | 11 (9) |
| 2 | 36 (30) |
| 3 | 41 (35) |
| 4 | 31 (26) |
| N. exacerbations/year | |
| 0 | 12 (10) |
| 1 | 37 (31) |
| 2 | 31 (26) |
| ≥ 3 | 39 (33) |
| ICS dose: | |
| No, n (%) | 8 (7) |
| Low, n (%) | 20 (17) |
| Medium, n (%) | 30 (25) |
| High, n (%) | 61 (51) |
| ICS alone, n (%) | 3 (3) |
| ICS + LABA, n (%) | 108 (90) |
| ICS + LABA + LAMA, n (%) | 23 (19) |
| Oral corticosteroid, n (%) (*) | 55 (46) |
| Group 1 25-OHD > 20 ng/mL | Group 2 25-OHD < 20 ng/mL 24 Discontinued, 11 Non-adherent | Group 3 25-OHD < 20 ng/mL Adherent to Vitamin D Supplement | p | |
|---|---|---|---|---|
| Number | 29 | 35 | 55 | |
| Age (years) | 44 (38–50) c | 50 (45–56) c | 57 (54–61) | 0.001 |
| Men n (%) | 6 (21) | 7 (20) | 12 (22) | 0.97 |
| BMI (Kg/m2) mean (95% CI) | 24 (22–25) | 24 (22–26) | 25 (24–26) | 0.29 |
| Smokers n (%) | 4 (14) | 1 (3) | 9 (16) | 0.14 |
| GINA | ||||
| 1. Intermittent n (%) | 5 (17) | 4 (11) | 2 (4) | |
| 2. Mild persistent n (%) | 14 (48) | 13 (37) | 9 (16) | 0.002 |
| 3. Moderate persistent n (%) | 8 (28) | 10 (29) | 23 (42) | |
| 4. Severe persistent n (%) | 2 (7) | 8 (23) | 21 (38) | |
| VC% pred. mean (95% CI) | 94 (87–101) c | 87 (83–91) c | 80 (77–84) | <0.001 |
| FEV1% pred. mean (95% CI) | 88 (81–96) c | 82 (75–89) c | 70 (65–75) | <0.001 |
| FEV1/VC% mean (95% CI) | 71 (67–75) c | 70 (65–75) c | 63 (59–66) | 0.007 |
| FENO ppb mean (95% CI) | 47 (22–72) | 40 (36–52) | 45 (20–60) | 0.83 |
| Exacerbations n mean (95% CI) | 1 (0.6–1.5) | 2.2 (1.7–2.6) a | 2.6 (2.2–2.9) a,b | <0.001 |
| Eosinophils 106/L mean (95% CI) | 320 (190–460) | 330 (220–440) | 380 (290–470) | 0.66 |
| Vitamin D, ng/mL mean (95% CI) | 26.6 (25–29) b,c | 13.6 (12–15) a | 13.0 (12–14) a | <0.001 |
| Atopy n (%) | 22 (76) | 21 (60) | 40 (73) | 0.31 |
| Rhinitis n (%) | 22 (76) | 25 (71) | 42 (76) | 0.86 |
| Rhinosinusitis n (%) | 8 (28) | 17 (49) | 32 (58) | 0.03 |
| Hypertension n (%) | 11 (38) | 13 (37) | 37 (67) | 0.005 |
| Heart disease n (%) | 10 (37) | 10 (29) | 25 (46) | 0.25 |
| Depression n (%) | 7 (26) | 12 (35) | 9 (16) | 0.15 |
| Thyroid disease n (%) | 8 (30) | 5 (15) | 16 (29) | 0.25 |
| OSA n (%) | 3 (11) | 1 (3) | 4 (7) | 0.51 |
| COPD n (%) | 7 (26) | 6 (18) | 8 (14) | 0.55 |
| Osteoporosis n (%) | 4 (15) | 7 (21) | 26 (47) | 0.002 |
| GERD n (%) | 8 (30) | 9 (27) | 22 (42) | 0.29 |
| ICS + LABA n (%) | 24 (83) | 32 (91) | 55 (100) | 0.009 |
| LAMA n (%) | 6 (21) | 5 (14) | 18 (33) | 0.12 |
| LTRA n (%) | 4 (14) | 7 (20) | 28 (51) | <0.001 |
| Oral corticosteroid n (%) (*) | 7 (25) | 13 (37) | 35 (64) | 0.001 |
| ICS: No n (%) | 5 (17) | 3 (9) | 0 (0) | 0.04 |
| Low n (%) | 7 (24) | 9 (26) | 4 (7) | |
| Medium n (%) | 6 (21) | 8 (23) | 16 (29) | |
| High n (%) | 11 (38) | 15 (27) | 35 (64) |
| Variable | Before Supplement | After Supplement | Mean Difference (95% CI) | p Value |
|---|---|---|---|---|
| Patients adherent | 55 | 55 | ||
| 25-OHD, ng/mL | 13 (4.8) | 31.7 (9.3) | 18.7 (15.9–21.4) | <0.001 |
| Asthma Exacerbations, n | 2.6 (1.2) | 1.6 (1.1) | −1 (−0.7–−1.3) | <0.001 |
| Oral corticosteroid, N (%) (*) | 35 (64) | 20 (40) | 0.007 | |
| FEV1, l | 1,86 (0.7) | 2.11 (0.8) | 0.25 (0.16–0.35) | <0.001 |
| FEV1/VC% | 63 (13) | 67 (9) | 4 (1–6) | 0.002 |
| Blood eosinophils 106/L (**) | 390 (330) | 270 (200) | −120 (−50–−200) | 0.002 |
| Patients non-adherent | 11 | 11 | ||
| 25-OHD, ng/mL | 13.7 (3.6) | 15.7 (3.9) | 1.9 (1.0–4.8) | 0.18 |
| Asthma Exacerbations, n | 2.0 (0.9) | 2.0 (0.9) | 0 (−0.7–0.7) | 1.00 |
| Oral corticosteroid N (%) (*) | 5 (45) | 6 (55) | 1.00 | |
| FEV1, l | 2.26 (0.75) | 2.19 (0.77) | 0.07 (−0.25–0.12) | 0.80 |
| FEV1/VC% | 69 (16) | 69 (17) | 0.1 (−3.2–3.4) | 0.94 |
| Blood eosinophils 106/L | 270 (210) | 260 (180) | −6 (−56–40) | 0.81 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solidoro, P.; Bellocchia, M.; Aredano, I.; Mattei, A.; Pivetta, E.; Patrucco, F.; Boita, M.; De Blasio, F.; Brussino, L.; Rolla, G.; et al. Asthmatic Patients with Vitamin D Deficiency have Decreased Exacerbations after Vitamin Replacement. Nutrients 2017, 9, 1234. https://doi.org/10.3390/nu9111234
Solidoro P, Bellocchia M, Aredano I, Mattei A, Pivetta E, Patrucco F, Boita M, De Blasio F, Brussino L, Rolla G, et al. Asthmatic Patients with Vitamin D Deficiency have Decreased Exacerbations after Vitamin Replacement. Nutrients. 2017; 9(11):1234. https://doi.org/10.3390/nu9111234
Chicago/Turabian StyleSolidoro, Paolo, Michela Bellocchia, Ilaria Aredano, Alessio Mattei, Emanuele Pivetta, Filippo Patrucco, Monica Boita, Francesca De Blasio, Luisa Brussino, Giovanni Rolla, and et al. 2017. "Asthmatic Patients with Vitamin D Deficiency have Decreased Exacerbations after Vitamin Replacement" Nutrients 9, no. 11: 1234. https://doi.org/10.3390/nu9111234
APA StyleSolidoro, P., Bellocchia, M., Aredano, I., Mattei, A., Pivetta, E., Patrucco, F., Boita, M., De Blasio, F., Brussino, L., Rolla, G., & Bucca, C. (2017). Asthmatic Patients with Vitamin D Deficiency have Decreased Exacerbations after Vitamin Replacement. Nutrients, 9(11), 1234. https://doi.org/10.3390/nu9111234





