Effect of Curcumin on the Diversity of Gut Microbiota in Ovariectomized Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Preparation of Specimens
2.3. Measurements of Estradiol in Serum
2.4. Fecal Collection and Bacterial DNA Extraction
2.5. PCR Amplification and Sequencing
2.6. Statistical Analysis
3. Results
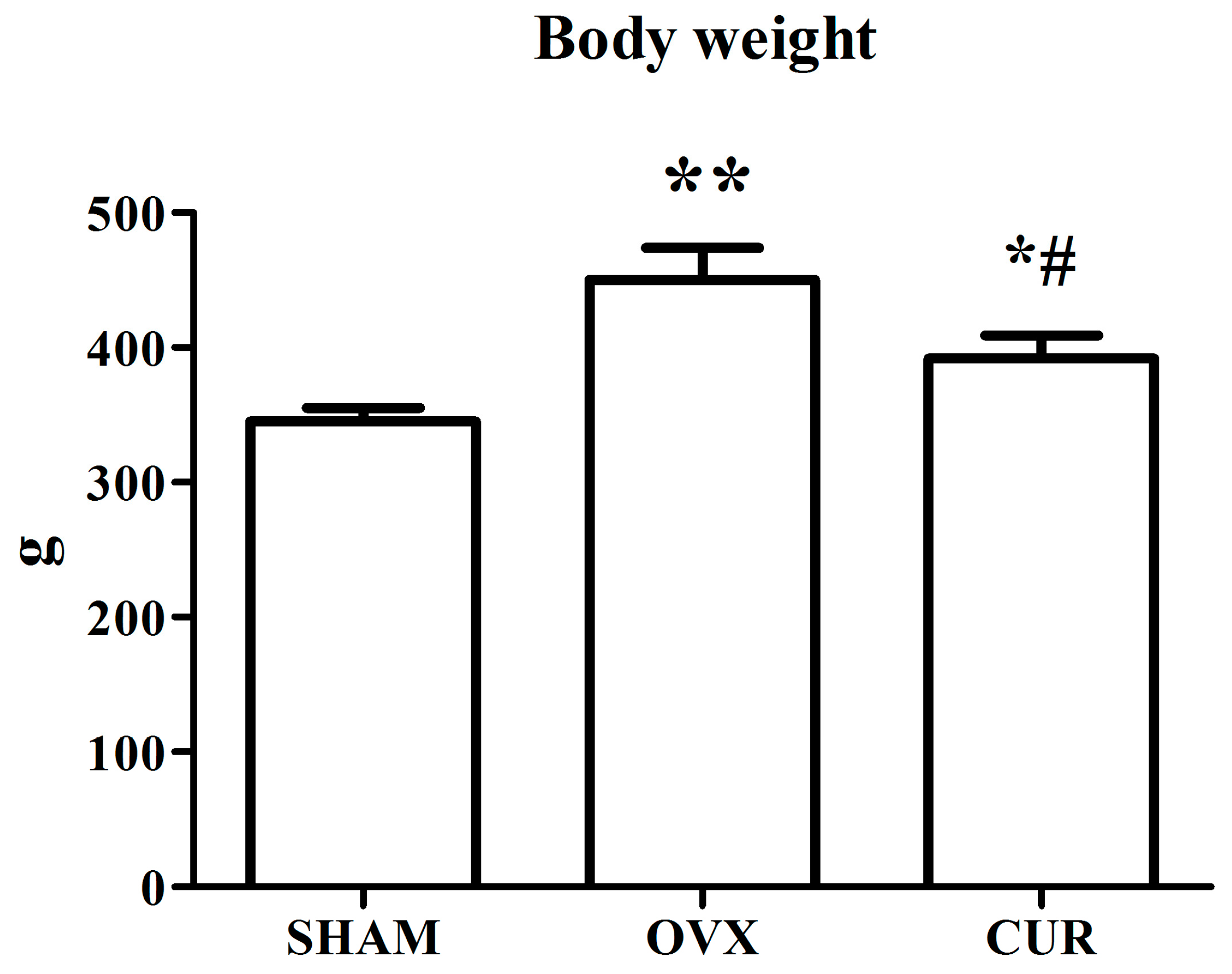
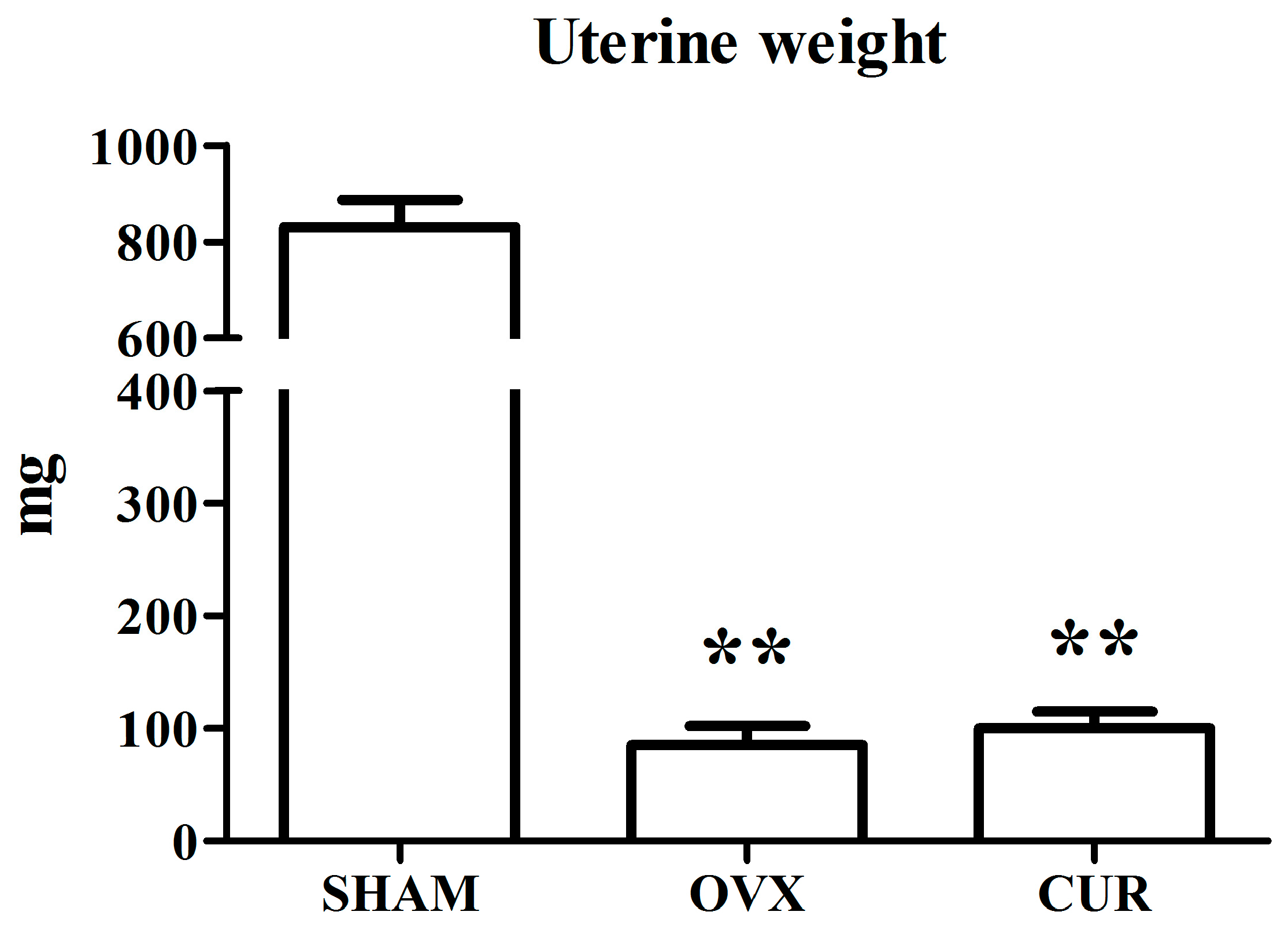
3.1. The Effect of Curcumin on the Weights of Bodies and Uteri
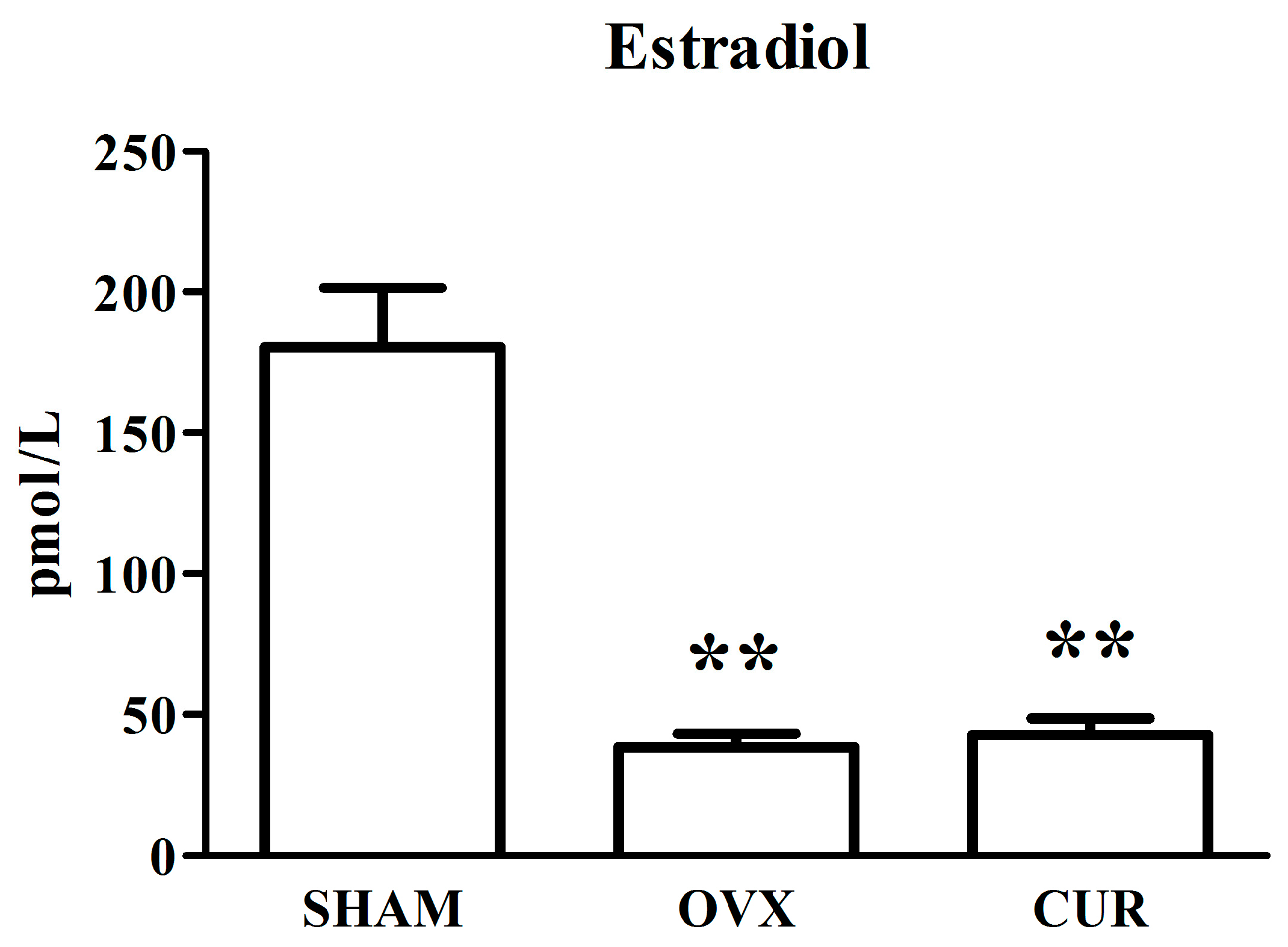
3.2. The Effect of Curcumin on the Level of Estradiol in Serum
3.3. Quality Evaluation and Filtering of Original Data
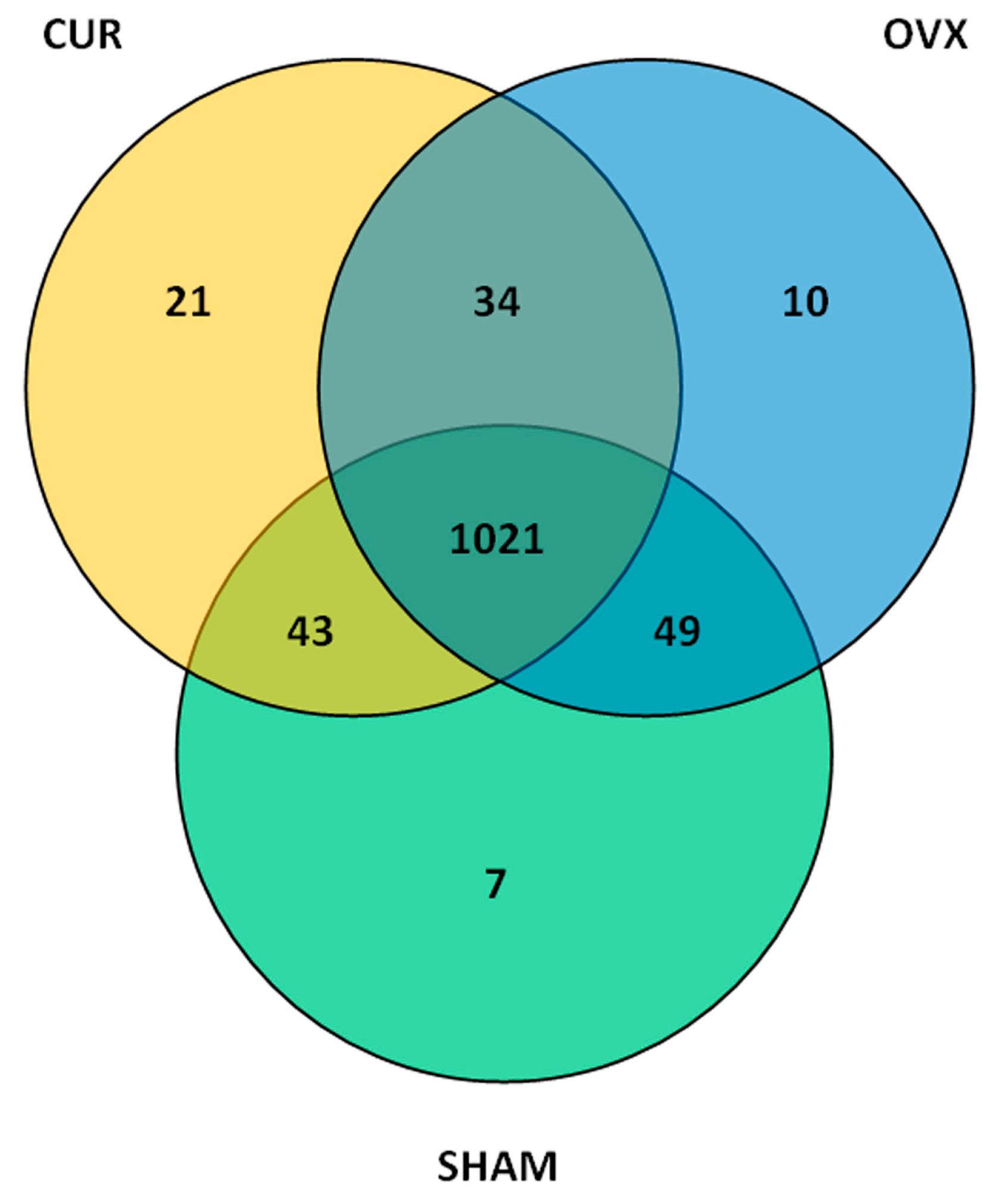
3.4. Operational Taxonomic Units
3.5. Alpha Diversity Analysis
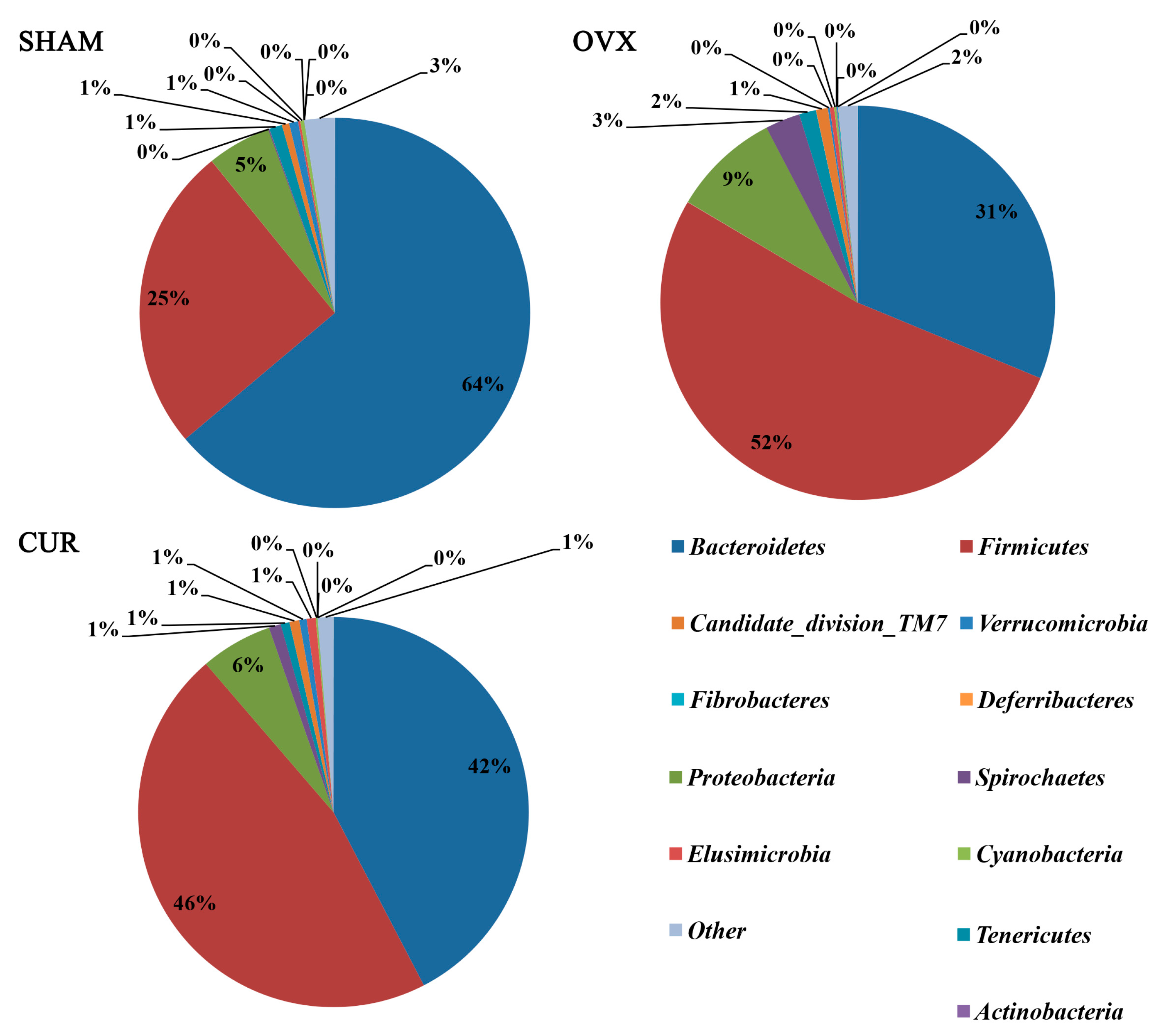
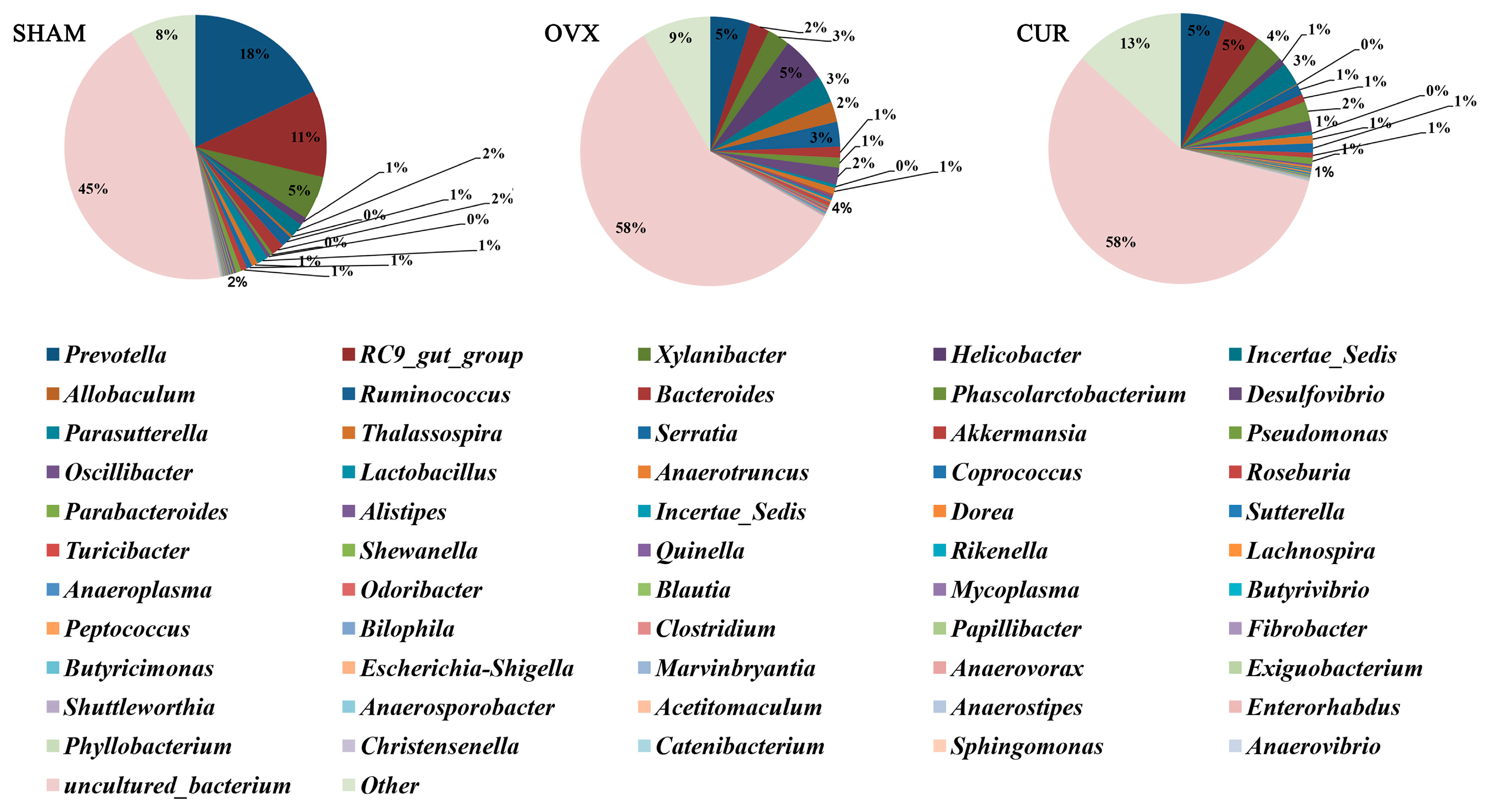
3.6. Analysis of Differential Gut Microbiota
3.7. Beta Diversity Analysis
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Oppermann, K.; Fuchs, S.C.; Donato, G.; Bastos, C.A.; Spritzer, P.M. Physical, psychological, and menopause-related symptoms and minor psychiatric disorders in a community-based sample of brazilian premenopausal, perimenopausal, and postmenopausal women. Menopause 2012, 19, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Boschitsch, E.P.; Durchschlag, E.; Dimai, H.P. Age-related prevalence of osteoporosis and fragility fractures: Real-world data from an austrian menopause and osteoporosis clinic. Climacteric 2017, 20, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, J.T.; Silveira, M.F.; Campos, M.C.; Costa, L.H. Overweight and obesity and factors associated with menopause. Cienc. Saude Coletiva 2016, 21, 1145–1156. [Google Scholar]
- Round, J.L.; Mazmanian, S.K. The gut microbiota shapes intestinal immune responses during health and disease. Nat. Rev. Immunol. 2009, 9, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Sanz, Y.; Santacruz, A.; Gauffin, P. Gut microbiota in obesity and metabolic disorders. Proc. Nutr. Soc. 2010, 69, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Gordon, J.I. Honor thy symbionts. Proc. Natl. Acad. Sci. USA 2003, 100, 10452–10459. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.M.; Lampe, J.W.; Newton, K.M.; Gundersen, G.; Fuller, S.; Reed, S.D.; Frankenfeld, C.L. Being overweight or obese is associated with harboring a gut microbial community not capable of metabolizing the soy isoflavone daidzein to o-desmethylangolensin in peri- and post-menopausal women. Maturitas 2017, 99, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Cox-York, K.A.; Sheflin, A.M.; Foster, M.T.; Gentile, C.L.; Kahl, A.; Koch, L.G.; Britton, S.L.; Weir, T.L. Ovariectomy results in differential shifts in gut microbiota in low versus high aerobic capacity rats. Physiol. Rep. 2015, 3, e12488. [Google Scholar] [CrossRef] [PubMed]
- Guadamuro, L.; Delgado, S.; Redruello, B.; Florez, A.B.; Suarez, A.; Martinez-Camblor, P.; Mayo, B. Equol status and changes in fecal microbiota in menopausal women receiving long-term treatment for menopause symptoms with a soy-isoflavone concentrate. Front. Microbiol. 2015, 6, 777. [Google Scholar] [CrossRef] [PubMed]
- Panda, A.K.; Chakraborty, D.; Sarkar, I.; Khan, T.; Sa, G. New insights into therapeutic activity and anticancer properties of curcumin. J. Exp. Pharmacol. 2017, 9, 31–45. [Google Scholar] [CrossRef] [PubMed]
- Egan, M.E.; Pearson, M.; Weiner, S.A.; Rajendran, V.; Rubin, D.; Glockner-Pagel, J.; Canny, S.; Du, K.; Lukacs, G.L.; Caplan, M.J. Curcumin, a major constituent of turmeric, corrects cystic fibrosis defects. Science 2004, 304, 600–602. [Google Scholar] [CrossRef] [PubMed]
- Bengmark, S. Curcumin, an atoxic antioxidant and natural nfkappab, cyclooxygenase-2, lipooxygenase, and inducible nitric oxide synthase inhibitor: A shield against acute and chronic diseases. J. Parenter. Enter. Nutr. 2006, 30, 45–51. [Google Scholar] [CrossRef]
- Punithavathi, D.; Venkatesan, N.; Babu, M. Protective effects of curcumin against amiodarone-induced pulmonary fibrosis in rats. Br. J. Pharmacol. 2003, 139, 1342–1350. [Google Scholar] [CrossRef] [PubMed]
- Duvoix, A.; Blasius, R.; Delhalle, S.; Schnekenburger, M.; Morceau, F.; Henry, E.; Dicato, M.; Diederich, M. Chemopreventive and therapeutic effects of curcumin. Cancer Lett. 2005, 223, 181–190. [Google Scholar] [CrossRef] [PubMed]
- French, D.L.; Muir, J.M.; Webber, C.E. The ovariectomized, mature rat model of postmenopausal osteoporosis: An assessment of the bone sparing effects of curcumin. Phytomedicine 2008, 15, 1069–1078. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.K.; Ke, K.; Sul, O.J.; Kim, H.J.; Kim, S.H.; Lee, M.H.; Kim, H.J.; Kim, S.Y.; Chung, H.T.; Choi, H.S. Curcumin protects against ovariectomy-induced bone loss and decreases osteoclastogenesis. J. Cell. Biochem. 2011, 112, 3159–3166. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.G.; Bai, D.; Liu, M.J.; Li, Y.; Pan, J.H.; Liu, H.; Wang, W.L.; Xiang, L.H.; Xiao, G.G.; Ju, D.H. Therapeutic effect of aqueous extract from ecliptae herba on bone metabolism of ovariectomized rats. Menopause 2013, 20, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Masella, A.P.; Bartram, A.K.; Truszkowski, J.M.; Brown, D.G.; Neufeld, J.D. Pandaseq: Paired-end assembler for illumina sequences. BMC Bioinform. 2012, 13, 31. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. Qiime allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Folwarczna, J.; Zych, M.; Trzeciak, H.I. Effects of curcumin on the skeletal system in rats. Pharmacol. Rep. 2010, 62, 900–909. [Google Scholar] [CrossRef]
- Cho, D.C.; Kim, K.T.; Jeon, Y.; Sung, J.K. A synergistic bone sparing effect of curcumin and alendronate in ovariectomized rat. Acta Neurochir. 2012, 154, 2215–2223. [Google Scholar] [CrossRef] [PubMed]
- Kozakowski, J.; Gietka-Czernel, M.; Leszczynska, D.; Majos, A. Obesity in menopause—Our negligence or an unfortunate inevitability? Przeglad Menopauzalny = Menopause Rev. 2017, 16, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Song, C.; Fu, X.; Liu, M.; Li, Y.; Pan, J.; Liu, H.; Wang, S.; Xiang, L.; Xiao, G.G.; et al. High-dose diosgenin reduces bone loss in ovariectomized rats via attenuation of the rankl/opg ratio. Int. J. Mol. Sci. 2014, 15, 17130–17147. [Google Scholar] [CrossRef] [PubMed]
- Al-Safi, Z.A.; Polotsky, A.J. Obesity and menopause. Best Pract. Res. Clin. Obstet. Gynaecol. 2015, 29, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Backhed, F.; Turnbaugh, P.; Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity alters gut microbial ecology. Proc. Natl. Acad. Sci. USA 2005, 102, 11070–11075. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.M.; Al-Nakkash, L.; Herbst-Kralovetz, M.M. Estrogen-gut microbiome axis: Physiological and clinical implications. Maturitas 2017, 103, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.P.; Dumas, M.E.; Wang, Y.; Legido-Quigley, C.; Yap, I.K.; Tang, H.; Zirah, S.; Murphy, G.M.; Cloarec, O.; Lindon, J.C.; et al. A top-down systems biology view of microbiome-mammalian metabolic interactions in a mouse model. Mol. Syst. Biol. 2007, 3, 112. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.; Shen, C.J.; Jia, G.; Wang, K.N. Effect of dietary bacillus subtilis on proportion of bacteroidetes and firmicutes in swine intestine and lipid metabolism. Genet. Mol. Res. 2013, 12, 1766–1776. [Google Scholar] [CrossRef] [PubMed]
- Koliada, A.; Syzenko, G.; Moseiko, V.; Budovska, L.; Puchkov, K.; Perederiy, V.; Gavalko, Y.; Dorofeyev, A.; Romanenko, M.; Tkach, S.; et al. Association between body mass index and firmicutes/bacteroidetes ratio in an adult ukrainian population. BMC Microbiol. 2017, 17, 120. [Google Scholar] [CrossRef] [PubMed]
- Parnell, J.A.; Reimer, R.A. Prebiotic fibres dose-dependently increase satiety hormones and alter bacteroidetes and firmicutes in lean and obese jcr:La-cp rats. Br. J. Nutr. 2012, 107, 601–613. [Google Scholar] [CrossRef] [PubMed]
- Armougom, F.; Raoult, D. Use of pyrosequencing and DNA barcodes to monitor variations in firmicutes and bacteroidetes communities in the gut microbiota of obese humans. BMC Genom. 2008, 9, 576. [Google Scholar] [CrossRef] [PubMed]
- Golubeva, A.V.; Crampton, S.; Desbonnet, L.; Edge, D.; O’Sullivan, O.; Lomasney, K.W.; Zhdanov, A.V.; Crispie, F.; Moloney, R.D.; Borre, Y.E.; et al. Prenatal stress-induced alterations in major physiological systems correlate with gut microbiota composition in adulthood. Psychoneuroendocrinology 2015, 60, 58–74. [Google Scholar] [CrossRef] [PubMed]
- Zinkernagel, M.S.; Zysset-Burri, D.C.; Keller, I.; Berger, L.E.; Leichtle, A.B.; Largiader, C.R.; Fiedler, G.M.; Wolf, S. Association of the intestinal microbiome with the development of neovascular age-related macular degeneration. Sci. Rep. 2017, 7, 40826. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, M.; Garcia, A.; Rogers, A.B.; Ge, Z.; Taylor, N.S.; Xu, S.; Watanabe, K.; Marini, R.P.; Whary, M.T.; Wang, T.C.; et al. Protective role of 17 beta-estradiol against the development of Helicobacter pylori-induced gastric cancer in ins-gas mice. Carcinogenesis 2007, 28, 2597–2604. [Google Scholar] [CrossRef] [PubMed]
- Saqui-Salces, M.; Rocha-Gutierrez, B.L.; Barrios-Payan, J.A.; Ruiz-Palacios, G.; Camacho-Arroyo, I.; Gamboa-Dominguez, A. Effects of estradiol and progesterone on gastric mucosal response to early Helicobacter pylori infection in female gerbils. Helicobacter 2006, 11, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.M.; Lopes, T.; Oleastro, M.; Gato, I.V.; Floch, P.; Benejat, L.; Chaves, P.; Pereira, T.; Seixas, E.; Machado, J.; et al. Curcumin inhibits gastric inflammation induced by Helicobacter pylori infection in a mouse model. Nutrients 2015, 7, 306–320. [Google Scholar] [CrossRef] [PubMed]
- Khonche, A.; Biglarian, O.; Panahi, Y.; Valizadegan, G.; Soflaei, S.S.; Ghamarchehreh, M.E.; Majeed, M.; Sahebkar, A. Adjunctive therapy with curcumin for peptic ulcer: A randomized controlled trial. Drug Res. 2016, 66, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, S.F.; Yamamoto, T.; Refaat, A.; Ahmed, K.; Sakurai, H.; Saiki, I.; Kondo, T.; Usmanghani, K.; Kadowaki, M.; Sugiyama, T. Modulation of activation-induced cytidine deaminase by curcumin in Helicobacter pylori-infected gastric epithelial cells. Helicobacter 2009, 14, 588–595. [Google Scholar] [CrossRef] [PubMed]
- Foryst-Ludwig, A.; Neumann, M.; Schneider-Brachert, W.; Naumann, M. Curcumin blocks nf-kappab and the motogenic response in Helicobacter pylori-infected epithelial cells. Biochem. Biophys. Res. Commun. 2004, 316, 1065–1072. [Google Scholar] [CrossRef] [PubMed]

| Group | Chao | Ace | Shannon | Simpson |
|---|---|---|---|---|
| SHAM | 776 | 878.71 | 7.00 | 0.972 |
| OVX | 746 | 825.70 | 7.16 | 0.980 |
| CUR | 762 | 847.71 | 7.00 | 0.975 |
| Taxa | SHAM | OVX | p Value |
|---|---|---|---|
| Firmicutes | 0.261144 ± 0.054002 | 0.535624 ± 0.069105 | 0.009583 |
| Bacteroidetes | 0.659073 ± 0.068158 | 0.321806 ± 0.111124 | 0.036333 |
| Taxa | SHAM | OVX | p Value |
|---|---|---|---|
| Incertae_Sedis | 0.043671 ± 0.001527 | 0.095096 ± 0.005247 | 0 |
| Anaerovorax | 0.000233 ± 0.000094 | 0.001323 ± 0.000079 | 0.00175 |
| Anaerotruncus | 0.003101 ± 0.000127 | 0.006812 ± 0.00127 | 0.019571 |
| Helicobacter | 0.027859 ± 0.012836 | 0.148457 ± 0.051536 | 0.044554 |
| Taxa | OVX | CUR | p Value |
|---|---|---|---|
| Serratia | 0.000475 ± 0.00017 | 0.033828 ± 0.003534 | 0.0022 |
| Anaerotruncus | 0.006812 ± 0.00127 | 0.002321 ± 0.000981 | 0.004073 |
| Shewanella | 0 ± 0 | 0.004168 ± 0.000632 | 0.005945 |
| Pseudomonas | 0.000178 ± 0.00009 | 0.023137 ± 0.005138 | 0.013782 |
| Papillibacter | 0.000467 ± 0.000072 | 0.001236 ± 0.000227 | 0.028545 |
| Exiguobacterium | 0.000017 ± 0.000017 | 0.000988 ± 0.000314 | 0.032164 |
| Helicobacter | 0.148457 ± 0.051536 | 0.027151 ± 0.015317 | 0.049273 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.; Chen, Y.; Xiang, L.; Wang, Z.; Xiao, G.G.; Hu, J. Effect of Curcumin on the Diversity of Gut Microbiota in Ovariectomized Rats. Nutrients 2017, 9, 1146. https://doi.org/10.3390/nu9101146
Zhang Z, Chen Y, Xiang L, Wang Z, Xiao GG, Hu J. Effect of Curcumin on the Diversity of Gut Microbiota in Ovariectomized Rats. Nutrients. 2017; 9(10):1146. https://doi.org/10.3390/nu9101146
Chicago/Turabian StyleZhang, Zhiguo, Yanjing Chen, Lihua Xiang, Zhen Wang, Gary Guishan Xiao, and Jingqing Hu. 2017. "Effect of Curcumin on the Diversity of Gut Microbiota in Ovariectomized Rats" Nutrients 9, no. 10: 1146. https://doi.org/10.3390/nu9101146
APA StyleZhang, Z., Chen, Y., Xiang, L., Wang, Z., Xiao, G. G., & Hu, J. (2017). Effect of Curcumin on the Diversity of Gut Microbiota in Ovariectomized Rats. Nutrients, 9(10), 1146. https://doi.org/10.3390/nu9101146





