Sugar- and Intense-Sweetened Drinks in Australia: A Systematic Review on Cardiometabolic Risk
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Search Strategy and Study Selection
2.3. Data Extraction
3. Results
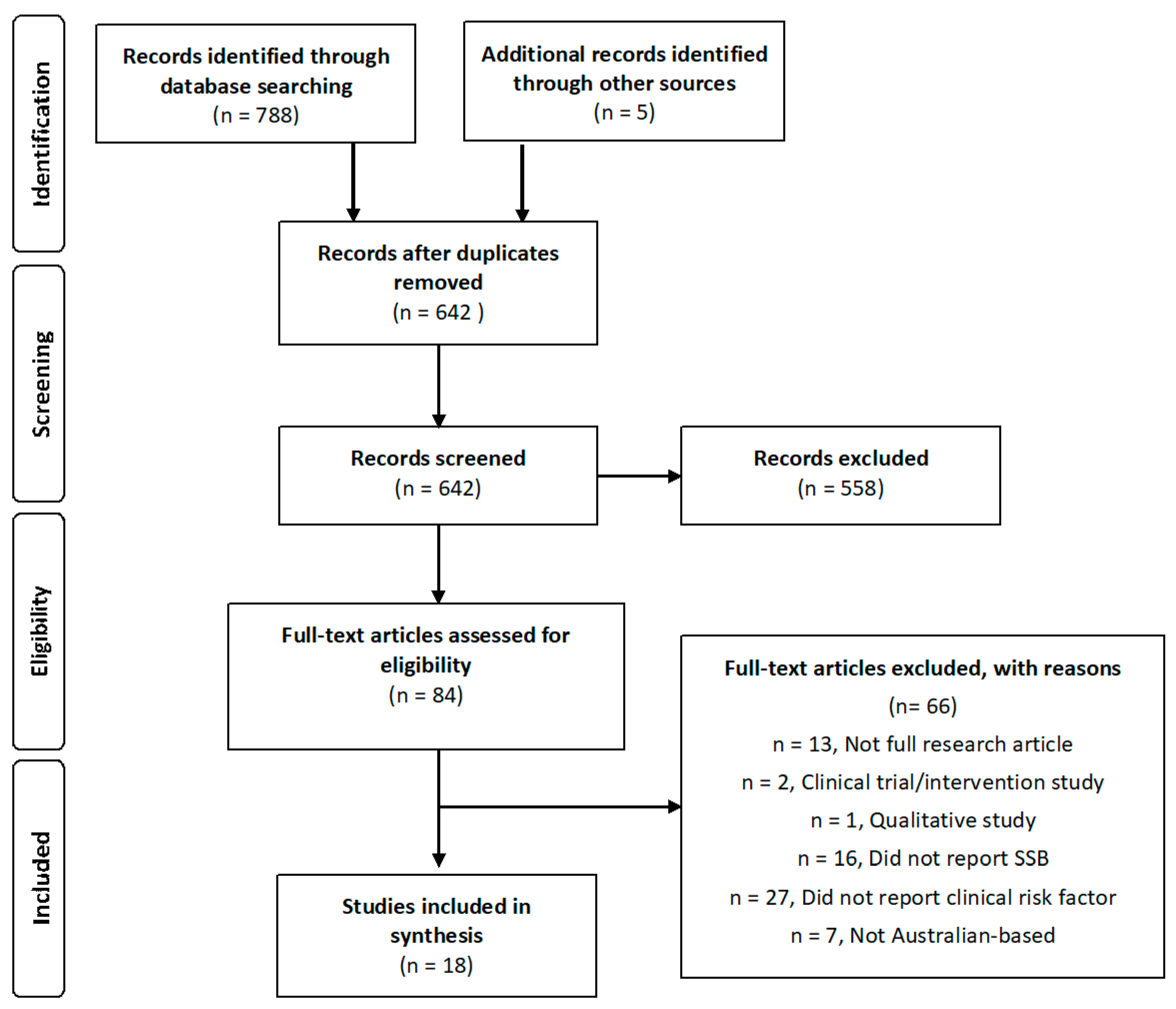
3.1. Search Results
3.2. Outcomes
3.2.1. Adiposity-Related Risk Factors
3.2.2. Blood Pressure
3.2.3. Other Cardiovascular and Metabolic Risk Factors
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Marriott, B.P.; Olsho, L.; Hadden, L.; Connor, P. Intake of added sugars and selected nutrients in the United States, National Health and Nutrition Examination Survey (NHANES) 2003–2006. Crit. Rev. Food Sci. Nutr. 2010, 50, 228–258. [Google Scholar] [CrossRef] [PubMed]
- Malik, V.S.; Pan, A.; Willett, W.C.; Hu, F.B. Sugar-sweetened beverages and weight gain in children and adults: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2013, 98, 1084–1102. [Google Scholar] [CrossRef] [PubMed]
- Vartanian, L.R.; Schwartz, M.B.; Brownell, K.D. Effects of soft drink consumption on nutrition and health: A systematic review and meta-analysis. Am. J. Public Health 2007, 97, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Australian Government. Australian New Zealand Food Standards Code—Standard 1.3.1—Food Additives; Legislation, F.R., Ed.; Australian Government: Canberra, Australia, 2016.
- Brand-Miller, J.C.; Barclay, A.W. Declining consumption of added sugars and sugar-sweetened beverages in Australia: A challenge for obesity prevention. Am. J. Clin. Nutr. 2017, 105, 854–863. [Google Scholar] [CrossRef] [PubMed]
- Forshee, R.A.; Anderson, P.A.; Storey, M.L. Sugar-sweetened beverages and body mass index in children and adolescents: A meta-analysis. Am. J. Clin. Nutr. 2008, 87, 1662–1671. [Google Scholar] [PubMed]
- Gibson, S. Sugar-sweetened soft drinks and obesity: A systematic review of the evidence from observational studies and interventions. Nutr. Res. Rev. 2008, 21, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Imamura, F.; O’Connor, L.; Ye, Z.; Mursu, J.; Hayashino, Y.; Bhupathiraju, S.N.; Forouhi, N.G. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: Systematic review, meta-analysis, and estimation of population attributable fraction. BMJ 2015. [Google Scholar] [CrossRef] [PubMed]
- Malik, V.S.; Popkin, B.M.; Bray, G.A.; Després, J.-P.; Willett, W.C.; Hu, F.B. Sugar-sweetened beverages and risk of metabolic syndrome and type 2 diabetes a meta-analysis. Diabetes Care 2010, 33, 2477–2483. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, D.; Threapleton, D.; Evans, C.; Cleghorn, C.; Nykjaer, C.; Woodhead, C.; Burley, V. Association between sugar-sweetened and artificially sweetened soft drinks and type 2 diabetes: Systematic review and dose-response meta-analysis of prospective studies. Br. J. Nutr. 2014, 112, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Yu, M.; Fang, L.; Hu, R.Y. Association between sugar-sweetened beverages and type 2 diabetes: A meta-analysis. J. Diabetes Investig. 2015, 6, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Popkin, B.M.; Hawkes, C. Sweetening of the global diet, particularly beverages: Patterns, trends, and policy responses. Lancet Diabetes Endocrinol. 2016, 4, 174–186. [Google Scholar] [CrossRef]
- Pollard, C.M.; Meng, X.; Hendrie, G.A.; Hendrie, D.; Sullivan, D.; Pratt, I.S.; Kerr, D.A.; Scott, J.A. Obesity, socio-demographic and attitudinal factors associated with sugar-sweetened beverage consumption: Australian evidence. Aust. N. Z. J. Public Health 2016, 40, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Millar, L.; Rowland, B.; Nichols, M.; Swinburn, B.; Bennett, C.; Skouteris, H.; Allender, S. Relationship between raised BMI and sugar sweetened beverage and high fat food consumption among children. Obesity 2014, 22, E96–E103. [Google Scholar] [CrossRef] [PubMed]
- Ambrosini, G.L.; Oddy, W.H.; Huang, R.C.; Mori, T.A.; Beilin, L.J.; Jebb, S.A. Prospective associations between sugar-sweetened beverage intakes and cardiometabolic risk factors in adolescents. Am. J. Clin. Nutr. 2013, 98, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Talley, N.J. National health summit on obesity calls for Australia to take action to stem the pandemic. Med. J. Aust. 2017, 206, 106–107. [Google Scholar] [CrossRef] [PubMed]
- Avogaro, A. Postprandial glucose: Marker or risk factor? Diabetes Care 2011, 34, 2333–2335. [Google Scholar] [CrossRef] [PubMed]
- Jameel, F.; Phang, M.; Wood, L.G.; Garg, M.L. Acute effects of feeding fructose, glucose and sucrose on blood lipid levels and systemic inflammation. Lipids Health Dis. 2014, 13, 195. [Google Scholar] [CrossRef] [PubMed]
- Barr, E.L.; Zimmet, P.Z.; Welborn, T.A.; Jolley, D.; Magliano, D.J.; Dunstan, D.W.; Cameron, A.J.; Dwyer, T.; Taylor, H.R.; Tonkin, A.M. Risk of cardiovascular and all-cause mortality in individuals with diabetes mellitus, impaired fasting glucose, and impaired glucose tolerance the Australian diabetes, obesity, and lifestyle study (AusDiab). Circulation 2007, 116, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Varsamis, P.; Larsen, R.N.; Dunstan, D.W.; Jennings, G.L.; Owen, N.; Kingwell, B.A. The sugar content of soft drinks in Australia, Europe and the United States. Med. J. Aust. 2017, 206, 454–455. [Google Scholar] [CrossRef] [PubMed]
- Bantle, J.P.; Raatz, S.K.; Thomas, W.; Georgopoulos, A. Effects of dietary fructose on plasma lipids in healthy subjects. Am. J. Clin. Nutr. 2000, 72, 1128–1134. [Google Scholar] [PubMed]
- Basciano, H.; Federico, L.; Adeli, K. Fructose, insulin resistance, and metabolic dyslipidemia. Nutr. Metab. 2005, 2, 5. [Google Scholar] [CrossRef] [PubMed]
- Chong, M.F.; Fielding, B.A.; Frayn, K.N. Mechanisms for the acute effect of fructose on postprandial lipemia. Am. J. Clin. Nutr. 2007, 85, 1511–1520. [Google Scholar] [PubMed]
- Australian Bureau of Statistics. Australian Health Survey 2011–2012; Australian Bureau of Statistics: Canberra, Australia, 2012.
- Hafekost, K.; Mitrou, F.; Lawrence, D.; Zubrick, S.R. Sugar sweetened beverage consumption by Australian children: Implications for public health strategy. BMC Public Health 2011, 11, 950. [Google Scholar] [CrossRef] [PubMed]
- French, S.; Rosenberg, M.; Wood, L.; Maitland, C.; Shilton, T.; Pratt, I.S.; Buzzacott, P. Soft drink consumption patterns among western Australians. J. Nutr. Educ. Behav. 2013, 45, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Australian Bureau of Statistics. Australian Health Survey: Nutrition First Results—Foods and Nutrients, 2011–2012; Australian Bureau of Statistics: Canberra, Australia, 2014.
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Thurber, K.A.; Dobbins, T.; Neeman, T.; Banwell, C.; Banks, E. Body mass index trajectories of Indigenous Australian children and relation to screen time, diet, and demographic factors. Obesity 2017, 25, 747–756. [Google Scholar] [CrossRef] [PubMed]
- Clifton, P.M.; Chan, L.; Moss, C.L.; Miller, M.D.; Cobiac, L. Beverage intake and obesity in Australian children. Nutr. Metab. 2011, 8, 87. [Google Scholar] [CrossRef] [PubMed]
- Grimes, C.A.; Riddel, L.J.; Campbell, K.J.; Nowson, C.A. Dietary Salt Intake, sugar-sweetened beverage consumption, and obesity risk. Pediatrics 2013, 131, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Hardy, L.L.; Grunseit, A.; Khambalia, A.; Bell, C.; Wolfenden, L.; Milat, A.J. Co-occurrence of obesogenic risk factors among adolescents. J. Adolesc. Health 2012, 51, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Jensen, B.W.; Nichols, M.; Allender, S.; de Silva-Sanigorski, A.; Millar, L.; Kremer, P.; Lacy, K.; Swinburn, B. Inconsistent associations between sweet drink intake and 2-year change in BMI among Victorian children and adolescents. Pediatr. Obes. 2013, 8, 271–283. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.; Barclay, A.W.; Brand-Miller, J.C.; Louie, J.C.Y. Dietary glycaemic index and glycaemic load among Australian children and adolescents: Results from the 2011–2012 Australian Health Survey. Br. J. Nutr. 2016, 116, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Sanigorski, A.M.; Bell, A.C.; Swinburn, B.A. Association of key foods and beverages with obesity in Australian schoolchildren. Public Health Nutr. 2007, 10, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Allman-Farinelli, M.; Heitmann, B.L.; Toelle, B.; Marks, G.; Cowell, C.; Rangan, A. Liquid versus solid energy intake in relation to body composition among Australian children. J. Hum. Nutr. Diet. 2015, 28, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Sluyter, J.D.; Scragg, R.K.R.; Plank, L.D.; Waqa, G.D.; Fotu, K.F.; Swinburn, B.A. Sizing the association between lifestyle behaviours and fatness in a large, heterogeneous sample of youth of multiple ethnicities from 4 countries. Int. J. Behav. Nutr. Phys. Act. 2013, 10, 115–139. [Google Scholar] [CrossRef] [PubMed]
- Wheaton, N.; Millar, L.; Allender, S.; Nichols, M. The stability of weight status through the early to middle childhood years in Australia: A longitudinal study. BMJ Open 2015, 5, e006963. [Google Scholar] [CrossRef] [PubMed]
- Trapp, G.S.A.; Allen, K.L.; O’Sullivan, T.; Robinson, M.; Jacoby, P.; Oddy, W.H. Energy drink consumption among young Australian adults: Associations with alcohol and illicit drug use. Drug Alcohol Depend. 2014, 134, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Pase, M.P.; Grima, N.; Cockerell, R.; Pipingas, A. Habitual intake of fruit juice predicts central blood pressure. Appetite 2015, 84, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Gearon, E.; Backholer, K.; Hodge, A.; Peeters, A. The mediating role of dietary factors and leisure time physical activity on socioeconomic inequalities in body mass index among Australian adults. BMC Public Health 2013, 13, 1214. [Google Scholar] [CrossRef] [PubMed]
- Cleland, V.J.; Schmidt, M.D.; Dwyer, T.; Venn, A.J. Television viewing and abdominal obesity in young adults: Is the association mediated by food and beverage consumption during viewing time or reduced leisure-time physical activity? Am. J. Clin. Nutr. 2008, 87, 1148–1155. [Google Scholar] [PubMed]
- Frary, C.D.; Johnson, R.K.; Wang, M.Q. Children and adolescents’ choices of foods and beverages high in added sugars are associated with intakes of key nutrients and food groups. J. Adolesc. Health 2004, 34, 56–63. [Google Scholar] [CrossRef]
- Arsenault, B.J.; Lamarche, B.; Després, J.-P. Targeting Overconsumption of sugar-sweetened beverages vs. overall poor diet quality for cardiometabolic diseases risk prevention: Place your bets! Nutrients 2017, 9, 600. [Google Scholar] [CrossRef] [PubMed]
- Han, E.; Powell, L.M. Consumption patterns of sugar-sweetened beverages in the United States. J. Acad. Nutr. Diet. 2013, 113, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Hulsegge, G.; Spijkerman, A.M.; van der Schouw, Y.T.; Bakker, S.J.; Gansevoort, R.T.; Smit, H.A.; Verschuren, W.M. Trajectories of metabolic risk factors and biochemical markers prior to the onset of cardiovascular disease—The doetinchem cohort study. PLoS ONE 2016, 11, e0155978. [Google Scholar] [CrossRef] [PubMed]
- Varsamis, P.; Larsen, R.N.; Owen, N.; Dunstan, D.W.; Jennings, G.L.; Kingwell, B.A. A comparison of the composition of sugars in popular soft drinks in Australia, Europe and the USA. J. Nutr. Intermed. Metab. 2017, 8, 104. [Google Scholar] [CrossRef]
- Dobbins, M.; Husson, H.; DeCorby, K.; LaRocca, R.L. School-based physical activity programs for promoting physical activity and fitness in children and adolescents aged 6 to 18. Cochrane Database Syst. Rev. 2013, 2. [Google Scholar] [CrossRef]
- Australian Beverage Council. The Role of Beverages in the Australian Diet; Australian Beverage Council: Waterloo, Australia, 2015. [Google Scholar]
- Brown, R.J.; De Banate, M.A.; Rother, K.I. Artificial sweeteners: A systematic review of metabolic effects in youth. Int. J. Pediatr. Obes. 2010, 5, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Suez, J.; Korem, T.; Zeevi, D.; Zilberman-Schapira, G.; Thaiss, C.A.; Maza, O.; Israeli, D.; Zmora, N.; Gilad, S.; Weinberger, A. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature 2014, 514, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Fox, C.S.; Jacques, P.F.; Speliotes, E.K.; Hoffmann, U.; Smith, C.E.; Saltzman, E.; McKeown, N.M. Sugar-sweetened beverage, diet soda, and fatty liver disease in the framingham heart study cohorts. J. Hepatol. 2015, 63, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Abdelmalek, M.F.; Day, C. Sugar sweetened beverages and fatty liver disease: Rising concern and call to action. J. Hepatol. 2015, 63, 306–308. [Google Scholar] [CrossRef] [PubMed]
- International Business Times. To Lower Australia’s Rising Obesity Rates, Tax Soft Drinks; International Business Times: Sydney, Australia, 2011. [Google Scholar]
- Roberto, C.A.; Swinburn, B.; Hawkes, C.; Huang, T.T.; Costa, S.A.; Ashe, M.; Zwicker, L.; Cawley, J.H.; Brownell, K.D. Patchy progress on obesity prevention: Emerging examples, entrenched barriers, and new thinking. Lancet 2015, 385, 2400–2409. [Google Scholar] [CrossRef]
- Marwick, T. The campaign against Heart Disease Must Keep Ticking. Available online: https://myaccount.news.com.au/theaustralian/subscribe?pkgDef=TA_SDO_P0415A_W04&directSubscribe=true&b=true&sourceCode=TAWEB_WRE170_a&mode=premium&dest=http://www.theaustralian.com.au/opinion/the-campaign-against-heart-disease-must-keep-ticking/news-story/6409de46044b887558d73621b9d6d067&memtype=anonymous (accessed on 27 September 2017).
- Shim, J.-S.; Oh, K.; Kim, H.C. Dietary assessment methods in epidemiologic studies. Epidemiol. Health 2014, 36, e2014009. [Google Scholar] [CrossRef] [PubMed]
| Study (Author/Year) | n | BMI/Overweight/Obesity | Waist Circumference | T2DM | Blood Pressure | Insulin Resistance | Glucose | Glycaemic Index | Triglycerides | Cholesterol | Metabolic Syndrome | Controlled for Physical Activity | Controlled for Diet | Other | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Obj | Sub | ||||||||||||||
| Ambrosini et al. (2013) [15] | 1433 | √ | √ | √ | √ | √ | √ | √ | √ | √ | √ | √ 1 | |||
| Cleland et al. (2008) [42] | 2001 | √ | √ 2 | ||||||||||||
| Clifton et al. (2011) [30] | 4400 | √ | |||||||||||||
| French et al. (2013) [26] | 1015 | √ | |||||||||||||
| Gearon et al. (2013) [41] | 30,630 | √ | √ 3 | ||||||||||||
| Grimes et al. (2013) [31] | 4283 | √ | √ | √ | √ 4 | ||||||||||
| Hardy et al. (2012) [32] | 1568 | √ | |||||||||||||
| Jensen et al. (2013) [33] | 1465 | √ | √ 5 | ||||||||||||
| Jones et al. (2016) [34] | 1876 | √ | |||||||||||||
| Millar et al. (2014) [14] | 4164 | √ | |||||||||||||
| Pase et al. (2015) [40] | 160 | √ | √ 6 | ||||||||||||
| Pollard et al. (2016) [13] | 13,596 | √ | |||||||||||||
| Sanigorski et al. (2007) [35] | 1944 | √ | |||||||||||||
| Sluyter et al. (2013) [37] | 1673 | √ | √ 7 | ||||||||||||
| Trapp et al. (2014) [39] | 1234 | √ | |||||||||||||
| Thurber et al. (2017) [29] | 887 | √ | |||||||||||||
| Wheaton et al. (2015) [38] | 4169 | √ | √ 8 | ||||||||||||
| Zheng et al. (2015) [36] | 158 | √ | √ | √ 9 | |||||||||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoare, E.; Varsamis, P.; Owen, N.; Dunstan, D.W.; Jennings, G.L.; Kingwell, B.A. Sugar- and Intense-Sweetened Drinks in Australia: A Systematic Review on Cardiometabolic Risk. Nutrients 2017, 9, 1075. https://doi.org/10.3390/nu9101075
Hoare E, Varsamis P, Owen N, Dunstan DW, Jennings GL, Kingwell BA. Sugar- and Intense-Sweetened Drinks in Australia: A Systematic Review on Cardiometabolic Risk. Nutrients. 2017; 9(10):1075. https://doi.org/10.3390/nu9101075
Chicago/Turabian StyleHoare, Erin, Pia Varsamis, Neville Owen, David W. Dunstan, Garry L. Jennings, and Bronwyn A. Kingwell. 2017. "Sugar- and Intense-Sweetened Drinks in Australia: A Systematic Review on Cardiometabolic Risk" Nutrients 9, no. 10: 1075. https://doi.org/10.3390/nu9101075
APA StyleHoare, E., Varsamis, P., Owen, N., Dunstan, D. W., Jennings, G. L., & Kingwell, B. A. (2017). Sugar- and Intense-Sweetened Drinks in Australia: A Systematic Review on Cardiometabolic Risk. Nutrients, 9(10), 1075. https://doi.org/10.3390/nu9101075





