Supplemental Protein during Heavy Cycling Training and Recovery Impacts Skeletal Muscle and Heart Rate Responses but Not Performance
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Preliminary Testing
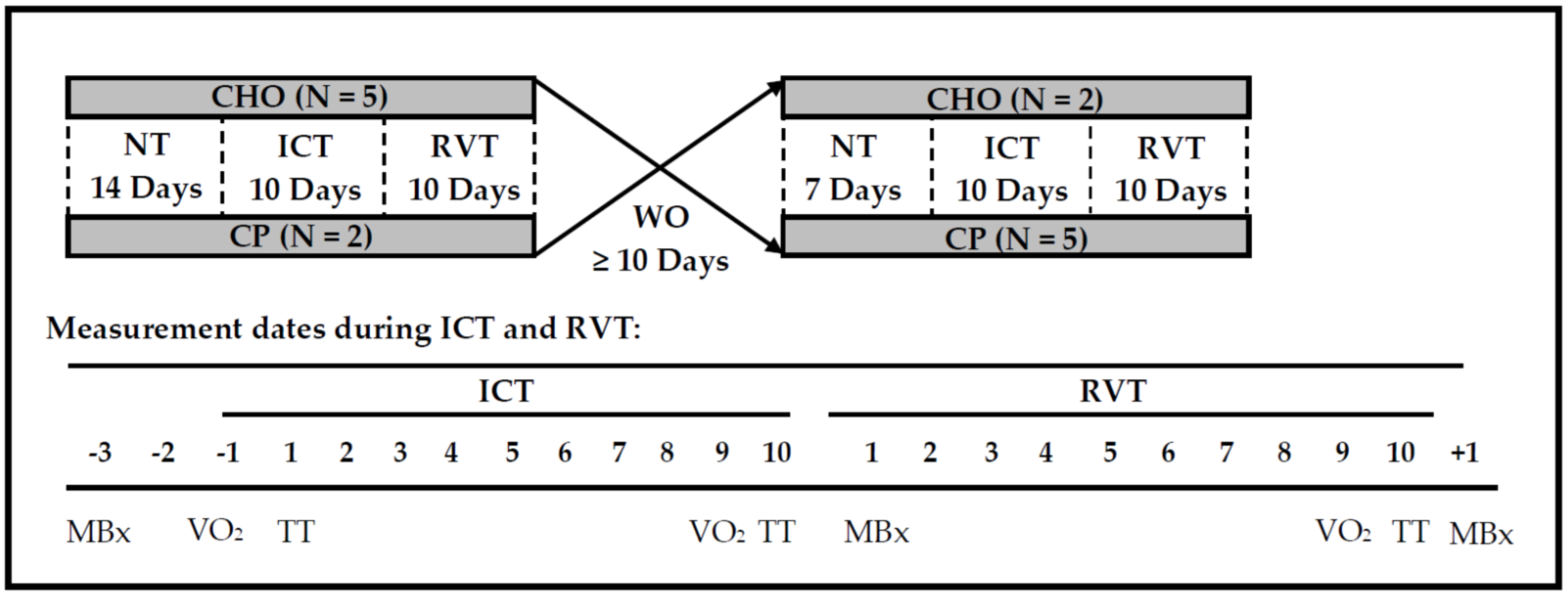
2.3. Experimental Design
2.3.1. Normal Training
2.3.2. Intensified Cycling Training
2.3.3. Reduced Volume Training
2.3.4. Washout (WO) Period
2.4. Treatment Beverages
2.5. Dietary Controls
2.6. Dependent Measurements
2.6.1. Cycling Performance and Responses to Constant-Load Exercise
Cardiorespiratory Responses to Constant-Load Exercise
Cycling Performance
2.6.2. VO2peak and Body Weight
2.6.3. Peak Isokinetic Torque
2.6.4. Muscle Soreness
2.6.5. Muscle Fiber Cross-Sectional Area
2.6.6. Biomarkers
2.6.7. Statistical Analyses
3. Results
3.1. Training Loads and Nutrient Intake
3.2. Dependent Measurements
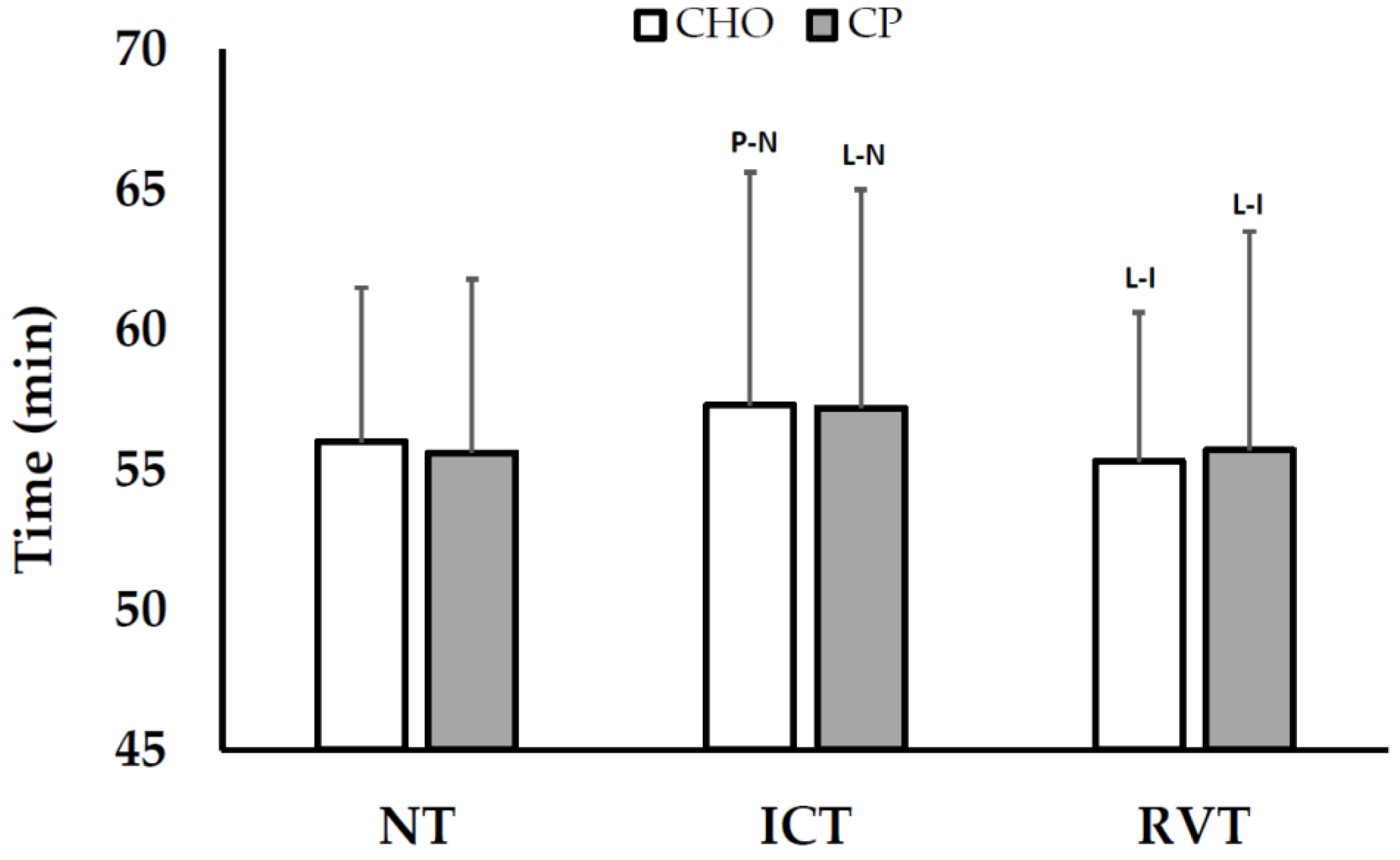
3.2.1. Cycling Time Trial (TT) Performance
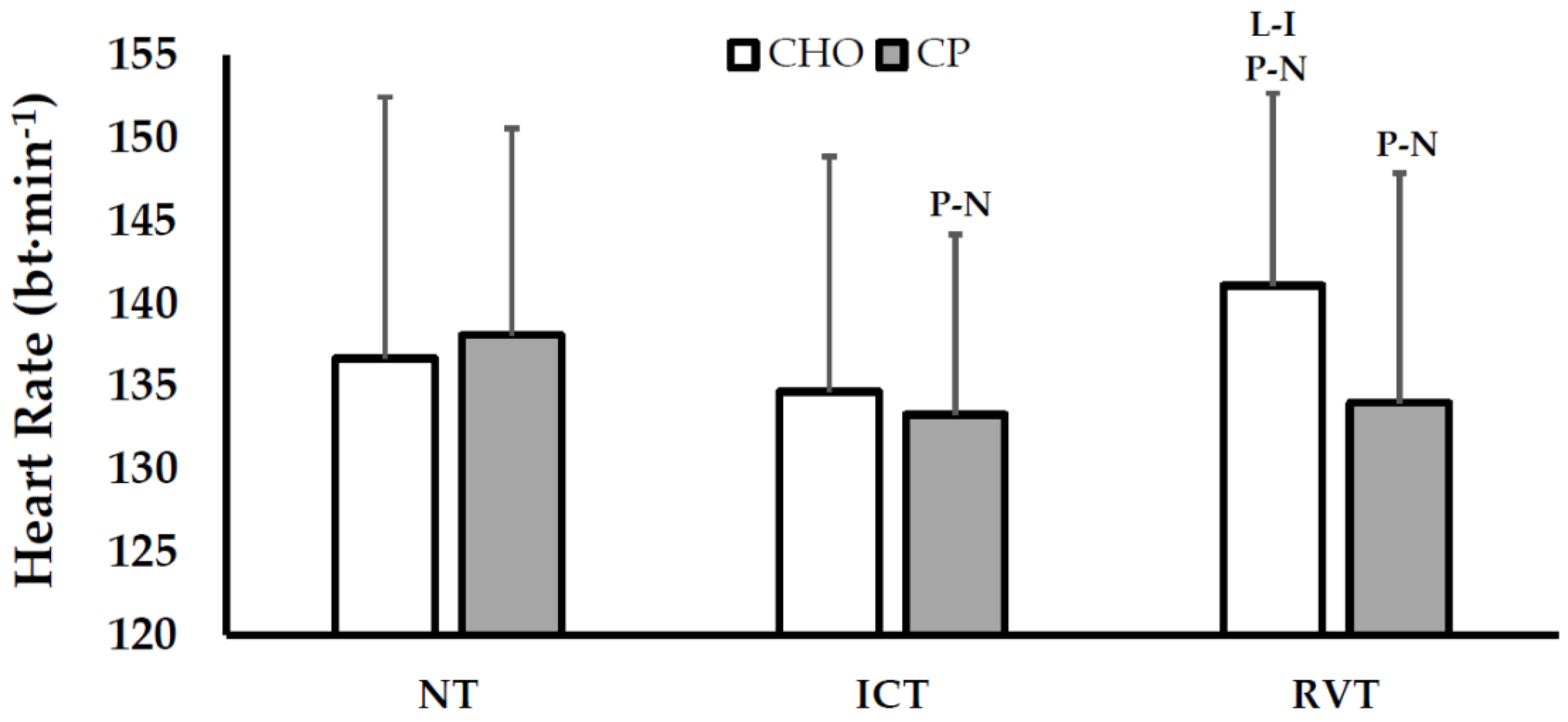
3.2.2. Responses during Constant-Load Exercise
3.2.3. VO2peak and Body Weight
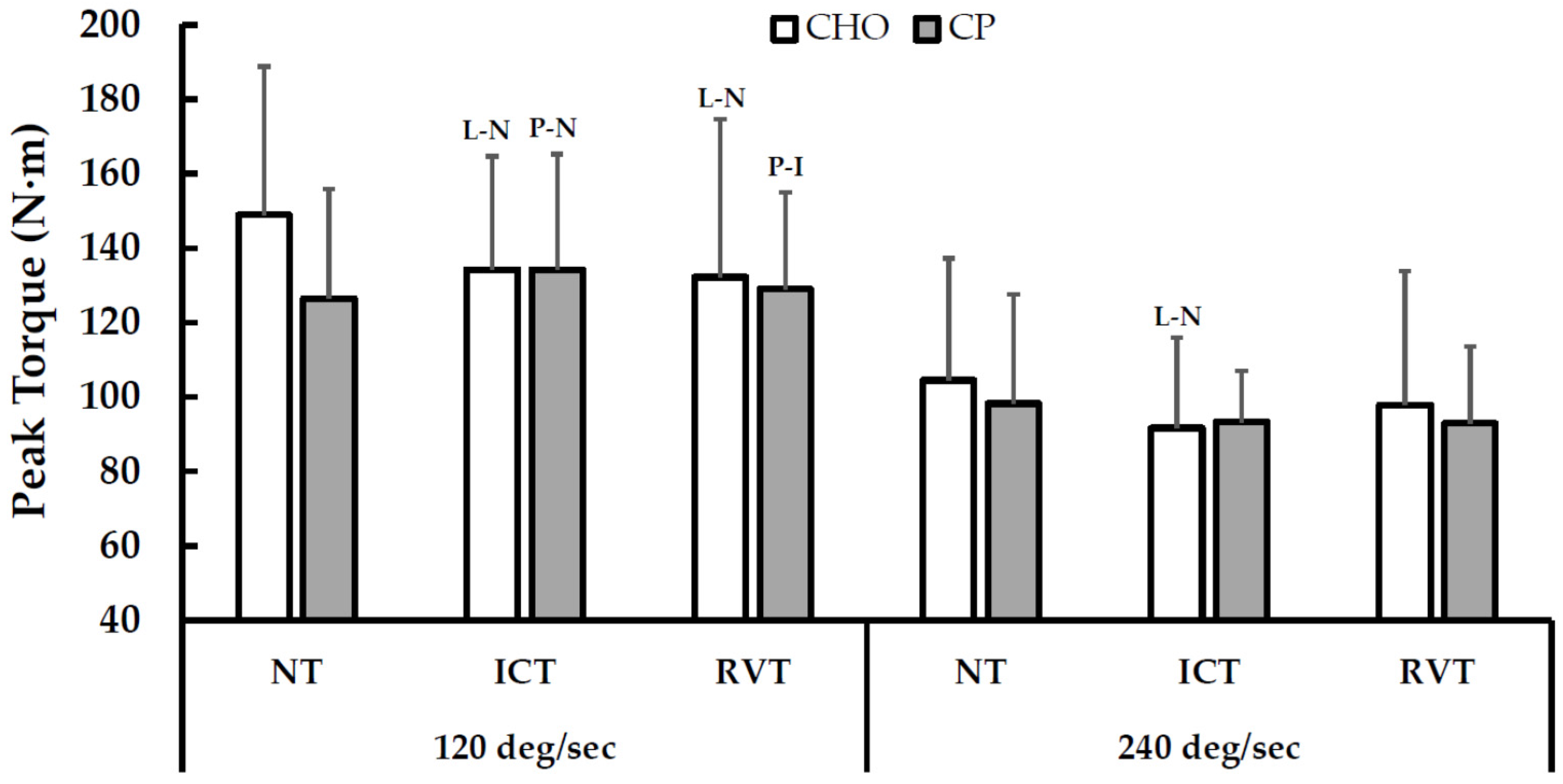
3.2.4. Muscle Function, Size and Soreness
3.2.5. Serum Biomarkers: Albumin, Creatine Kinase, and Cortisol
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jeukendrup, A.E.; Hesselink, M.K.; Snyder, A.C.; Kuipers, H.; Keizer, H.A. Physiological changes in male competitive cyclists after two weeks of intensified training. Int. J. Sports Med. 1992, 13, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Halson, S.L.; Bridge, M.W.; Meeusen, R.; Busschaert, B.; Gleeson, M.; Jones, D.A.; Jeukendrup, A.E. Time course of performance changes and fatigue markers during intensified training in trained cyclists. J. Appl. Physiol. 2002, 93, 947–956. [Google Scholar] [CrossRef] [PubMed]
- Achten, J.; Halson, S.L.; Moseley, L.; Rayson, M.P.; Casey, A.; Jeukendrup, A.E. Higher dietary carbohydrate content during intensified running training results in better maintenance of performance and mood state. J. Appl. Physiol. 2004, 96, 1331–1340. [Google Scholar] [CrossRef] [PubMed]
- Saunders, M.J.; Kane, M.D.; Todd, M.K. Effects of a carbohydrate-protein beverage on cycling endurance and muscle damage. Med. Sci. Sports Exerc. 2004, 36, 1233–1238. [Google Scholar] [CrossRef] [PubMed]
- Berardi, J.M.; Noreen, E.E.; Lemon, P.W. Recovery from a cycling time trial is enhanced with carbohydrate-protein supplementation vs. Isoenergetic carbohydrate supplementation. J. Int. Soc. Sports Nutr. 2008, 5, 24. [Google Scholar] [CrossRef] [PubMed]
- Ferguson-Stegall, L.; McCleave, E.L.; Ding, Z.; Doerner, P.G., 3rd; Wang, B.; Liao, Y.H.; Kammer, L.; Liu, Y.; Hwang, J.; Dessard, B.M.; et al. Postexercise carbohydrate-protein supplementation improves subsequent exercise performance and intracellular signaling for protein synthesis. J. Strength Cond. Res. 2011, 25, 1210–1224. [Google Scholar] [CrossRef] [PubMed]
- Lunn, W.R.; Pasiakos, S.M.; Colletto, M.R.; Karfonta, K.E.; Carbone, J.W.; Anderson, J.M.; Rodrigues, N.R. Chcolate milk and endurance exercise recovery: Protein balance, glycogen, and performance. Med. Sci. Sports Exerc. 2012, 44, 682–691. [Google Scholar] [CrossRef] [PubMed]
- Rustad, P.I.; Sailer, M.; Cumming, K.T.; Jeppesen, P.B.; Kolnes, K.J.; Sollie, O.; Franch, J.; Ivy, J.L.; Daniel, H.; Jensen, J. Intake of protein plus carbohydrate during the first two hours after exhaustive cycling improves performance the following day. PLoS ONE 2016, 11, e0153229. [Google Scholar] [CrossRef] [PubMed]
- Rowlands, D.S.; Rossler, K.; Thorp, R.M.; Graham, D.F.; Timmons, B.W.; Stannard, S.R.; Tarnopolsky, M.A. Effect of dietary protein content during recovery from high-intensity cycling on subsequent performance and markers of stress, inflammation, and muscle damage in well-trained men. Appl. Physiol. Nutr. Metab. 2008, 33, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Thomson, J.S.; Ali, A.; Rowlands, D.S. Leucine-protein supplemented recovery feeding enhances subsequent cycling performance in well-trained men. Appl. Physiol. Nutr. Metab. 2011, 36, 242–253. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.; Bangsbo, J.; Jensen, J.; Bibby, B.M.; Madsen, K. Effect of whey protein hydrolysate on performance and recovery of top-class orienteering runners. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Luden, N.D.; Saunders, M.J.; Todd, M.K. Postexercise carbohydrate-protein- antioxidant ingestion decreases plasma creatine kinase and muscle soreness. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, 109–123. [Google Scholar] [CrossRef] [PubMed]
- Gilson, S.F.; Saunders, M.J.; Moran, C.W.; Moore, R.W.; Womack, C.J.; Todd, M.K. Effects of chocolate milk consumption on markers of muscle recovery following soccer training: A randomized cross-over study. J. Int. Soc. Sports Nutr. 2010, 7, 19. [Google Scholar] [CrossRef] [PubMed]
- Nelson, A.R.; Phillips, S.M.; Stellingwerff, T.; Rezzi, S.; Bruce, S.J.; Breton, I.; Thorimbert, A.; Guy, P.A.; Clarke, J.; Broadbent, S.; et al. A protein-leucine supplement increases branched-chain amino acid and nitrogen turnover but not performance. Med. Sci. Sports Exerc. 2012, 44, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Witard, O.C.; Jackman, S.R.; Kies, A.K.; Jeukendrup, A.E.; Tipton, K.D. Effect of increased dietary protein on tolerance to intensified training. Med. Sci. Sports Exerc. 2011, 43, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.; Bangsbo, J.; Jensen, J.; Krause-Jensen, M.; Bibby, B.M.; Sollie, O.; Hall, U.A.; Madsen, K. Protein intake during training sessions has no effect on performance and recovery during a strenuous training camp for elite cyclists. J. Int. Soc. Sports Nutr. 2016, 13, 9. [Google Scholar] [CrossRef] [PubMed]
- Rowlands, D.S.; Thomson, J.S.; Timmons, B.W.; Raymond, F.; Fuerholz, A.; Mansourian, R.; Zwahlen, M.C.; Metairon, S.; Glover, E.; Stellingwerff, T.; et al. Transcriptome and translational signaling following endurance exercise in trained skeletal muscle: Impact of dietary protein. Physiol. Genom. 2011, 43, 1004–1020. [Google Scholar] [CrossRef] [PubMed]
- Greer, B.K.; Woodard, J.L.; White, J.P.; Arguello, E.M.; Haymes, E.M. Branched-chain amino acid supplementation and indicators of muscle damage after endurance exercise. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, 595–607. [Google Scholar] [CrossRef] [PubMed]
- Valentine, R.J.; Saunders, M.J.; Todd, M.K.; St Laurent, T.G. Influence of carbohydrate-protein beverage on cycling endurance and indices of muscle disruption. Int. J. Sport Nutr. Exerc. Metab. 2008, 18, 363–378. [Google Scholar] [CrossRef] [PubMed]
- Flakoll, P.J.; Judy, T.; Flinn, K.; Carr, C.; Flinn, S. Postexercise protein supplementation improves health and muscle soreness during basic military training in marine recruits. J. Appl. Physiol. 2004, 96, 951–956. [Google Scholar] [CrossRef] [PubMed]
- Skillen, R.A.; Testa, M.; Applegate, E.A.; Heiden, E.A.; Fascetti, A.J.; Casazza, G.A. Effects of an amino acid carbohydrate drink on exercise performance after consecutive-day exercise bouts. Int. J. Sport Nutr. Exerc. Metab. 2008, 18, 473–492. [Google Scholar] [CrossRef] [PubMed]
- Cathcart, A.J.; Murgatroyd, S.R.; McNab, A.; Whyte, L.J.; Easton, C. Combined carbohydrate-protein supplementation improves competitive endurance exercise performance in the heat. Eur. J. Appl. Physiol. 2011, 111, 2051–2061. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, K.; Hayase, H.; Ichinose, T.; Mitono, H.; Doi, T.; Nose, H. Protein and carbohydrate supplementation after exercise increases plasma volume and albumin content in older and young men. J. Appl. Physiol. 2009, 107, 770–779. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, K.; Ichinose, T.; Mitono, H.; Chen, M.; Masuki, S.; Endoh, H.; Hayase, H.; Doi, T.; Nose, H. Impact of protein and carbohydrate supplementation on plasma volume expansion and thermoregulatory adaptation by aerobic training in older men. J. Appl. Physiol. 2009, 107, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Goto, M.; Okazaki, K.; Kamijo, Y.; Ikegawa, S.; Masuki, S.; Miyagawa, K.; Nose, H. Protein and carbohydrate supplementation during 5-day aerobic training enhanced plasma volume expansion and thermoregulatory adaptation in young men. J. Appl. Physiol. 2010, 109, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Baur, D.A.; Schroer, A.B.; Luden, N.D.; Womack, C.J.; Smyth, S.A.; Saunders, M.J. Glucose-fructose enhances performance versus isocaloric, but not moderate, glucose. Med. Sci. Sports Exerc. 2014, 46, 1778–1786. [Google Scholar] [CrossRef] [PubMed]
- Costill, D.L.; Flynn, M.G.; Kirwan, J.P.; Houmard, J.A.; Mitchell, J.B.; Thomas, R.; Park, S.H. Effects of repeated days of intensified training on muscle glycogen and swimming performance. Med. Sci. Sports Exerc. 1988, 20, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Halson, S.L.; Lancaster, G.I.; Achten, J.; Gleeson, M.; Jeukendrup, A.E. Effects of carbohydrate supplementation on performance and carbohydrate oxidation after intensified cycling training. J. Appl. Physiol. 2004, 97, 1245–1253. [Google Scholar] [CrossRef] [PubMed]
- Babcock, L.; Escano, M.; D’Lugos, A.; Todd, K.; Murach, K.; Luden, N. Concurrent aerobic exercise interferes with the satellite cell response to acute resistance exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2012, 302, R1458–R1465. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, W.G.; Marshall, S.W.; Batterham, A.M.; Hanin, J. Progressive statistics for studies in sports medicine and exercise science. Med. Sci. Sports Exerc. 2009, 41, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, W. A spreadsheet for deriving a confidence interval, mechanistic inference and clinical inference from a p value. Sportscience 2007, 1, 16–20. [Google Scholar]
- Paton, C.D.; Hopkins, W.G. Variation in performance of elite cyclists from race to race. Eur. J. Sport Sci. 2006, 6, 25–31. [Google Scholar] [CrossRef]
- Atkinson, G. Estimating sample size for magnitude-based inferences. Sportscience 2006, 10, 63–70. [Google Scholar]
- Coutts, A.; Reaburn, P.; Piva, T.J.; Murphy, A. Changes in selected biochemical, muscular strength, power, and endurance measures during deliberate overreaching and tapering in rugby league players. Int. J. Sports Med. 2007, 28, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Rosler, K.; Conley, K.E.; Howald, H.; Gerber, C.; Hoppeler, H. Specificity of leg power changes to velocities used in bicycle endurance training. J. Appl. Physiol. 1986, 61, 30–36. [Google Scholar] [PubMed]
- Harber, M.P.; Gallagher, P.M.; Creer, A.R.; Minchev, K.M.; Trappe, S.W. Single muscle fiber contractile properties during a competitive season in male runners. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 287, R1124–R1131. [Google Scholar] [CrossRef] [PubMed]
- Fitts, R.H.; Costill, D.L.; Gardetto, P.R. Effect of swim exercise training on human muscle fiber function. J. Appl. Physiol. 1989, 66, 465–475. [Google Scholar] [PubMed]
- Kohn, T.A.; Essen-Gustavsson, B.; Myburgh, K.H. Specific muscle adaptations in type ii fibers after high-intensity interval training of well-trained runners. Scand. J. Med. Sci. Sports 2011, 21, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Howarth, K.R.; Moreau, N.A.; Phillips, S.M.; Gibala, M.J. Coingestion of protein with carbohydrate during recovery from endurance exercise stimulates skeletal muscle protein synthesis in humans. J. Appl. Physiol. 2009, 106, 1394–1402. [Google Scholar] [CrossRef] [PubMed]
- Levenhagen, D.K.; Carr, C.; Carlson, M.G.; Maron, D.J.; Borel, M.J.; Flakoll, P.J. Postexercise protein intake enhances whole-body and leg protein accretion in humans. Med. Sci. Sports Exerc. 2002, 34, 828–837. [Google Scholar] [CrossRef] [PubMed]
- Rowlands, D.S.; Nelson, A.R.; Phillips, S.M.; Faulkner, J.A.; Clarke, J.; Burd, N.A.; Moore, D.; Stellingwerff, T. Protein-leucine fed dose effects on muscle protein synthesis after endurance exercise. Med. Sci. Sports Exerc. 2015, 47, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, S.; Halliwill, J.R.; Sawka, M.N.; Minson, C.T. Heat acclimation improves exercise performance. J. Appl. Physiol. 2010, 109, 1140–1147. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, M.; Schnee, W. Decreased nocturnal catecholamine excretion parameter for an overtraining syndrom in athletes. Int. J. Sports Med. 1992, 13, 236–242. [Google Scholar] [CrossRef] [PubMed]
| Variable | NT | ICT | RVT | ||||||
|---|---|---|---|---|---|---|---|---|---|
| CHO | CP | CP-CHO Effects | CHO | CP | CP-CHO Effects | CHO | CP | CP-CHO Effects | |
| Duration (min/day) | 61.5 ± 8.3 | 63.0 ± 6.0 | 1.5 ± 4.1 | 137.1 ± 14.8 | 139.9 ± 16.9 | 2.7 ± 3.9 | 41.2 ± 4.9 | 42.2 ± 5.2 | 1.1 ± 1.8 |
| 52/39/10 | 20/79/1 | 40/58/2 | |||||||
| Unclear | Likely trivial | Possible | |||||||
| Power output (W) | 189 ± 31 | 182 ± 30 | −8 ± 13 | 165 ± 32 | 167 ± 26 | 2 ± 6 | 193 ± 37 | 193 ± 36 | 0 ± 5 |
| 7/21/72 | 30/66/3 | 4/93/3 | |||||||
| Unclear | Possible | Likely trivial | |||||||
| Heart Rate (bt·min−1) | 150 ± 16 | 146 ± 14 | −4 ± 5 | 139 ± 13 | 137 ± 13 | −2 ± 6 | 150 ± 13 | 146 ± 11 | −4 ± 5 |
| 2/32/65 | 5/60/35 | 1/28/72 | |||||||
| Possible | Unclear | Possible | |||||||
| Dietary Macronutrients (Day) | During-Exercise | Post-Exercise | Total Macronutrient Intake (Day) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| CHO | CP | CP-CHO Effects | CHO | CP | CHO | CP | CHO | CP | CP-CHO Effects | |
| ICT Period | ||||||||||
| Carbohydrate (g) | 337 ± 100 | 331 ± 114 | −6 ± 15 | 112 ± 11 | 112 ± 11 | 86 ± 14 | 86 ± 14 | 535 ± 110 | 529 ± 122 | −6 ± 15 |
| 1/88/11 | 1/92/7 | |||||||||
| Likely trivial | Likely trivial | |||||||||
| Protein (g) | 120 ± 38 | 118 ± 38 | −3 ± 11 | 0 ± 0 | 38 ± 4 | 0 ± 0 | 29 ± 5 | 120 ± 38 | 184 ± 44 | 64 ± 12 |
| 9/46/45 | 100/0/0 | |||||||||
| Unclear | Most likely | |||||||||
| Fat (g) | 118 ± 29 | 118 ± 34 | 0 ± 7 | 0 ± 0 | 2 ± 0 | 7 ± 1 | 8 ± 1 | 124 ± 30 | 127 ± 35 | 3 ± 7 |
| 1/97/2 | 13/84/3 | |||||||||
| V.L. trivial | Likely trivial | |||||||||
| Calories (kcal) | 2948 ± 779 | 2899 ± 878 | −49 ± 122 | 447 ± 46 | 612 ± 61 | 404 ± 65 | 532 ± 85 | 3799 ± 841 | 4043 ± 951 | 244 ± 122 |
| 1/79/20 | 91/9/0 | |||||||||
| Likely trivial | Likely | |||||||||
| RVT Period | ||||||||||
| Carbohydrate (g) | 317 ± 89 | 292 ± 104 | −25 ± 31 | 33 ± 4 | 33 ± 4 | 50 ± 9 | 50 ± 9 | 400 ± 94 | 376 ± 106 | −25 ± 31 |
| 3/27/70 | 3/27/70 | |||||||||
| Possible | Possible | |||||||||
| Protein (g) | 112 ± 22 | 110 ± 21 | −2 ± 10 | 0 ± 0 | 11 ± 1 | 0 ± 0 | 17 ± 3 | 112 ± 22 | 138 ± 24 | 26 ± 11 |
| 18/48/35 | 100/0/0 | |||||||||
| Unclear | Most likely | |||||||||
| Fat (g) | 105 ± 20 | 97 ± 13 | −8 ± 8 | 0 ± 0 | 1 ± 0 | 4 ± 1 | 5 ± 1 | 108 ± 21 | 102 ± 14 | −6 ± 8 |
| 0/68/31 | 2/30/69 | |||||||||
| Possible | Possible | |||||||||
| Calories (kcal) | 2706 ± 569 | 2527 ± 543 | −178 ± 198 | 132 ± 15 | 180 ± 20 | 236 ± 43 | 310 ± 56 | 3074 ± 608 | 3018 ± 577 | 56 ± 188 |
| 2/26/72 | 6/68/25 | |||||||||
| Possible | Unclear | |||||||||
| Variable | NT | ICT | RVT | Treatment Differences | |||||
|---|---|---|---|---|---|---|---|---|---|
| CHO | CP | CHO | CP | CHO | CP | NT-ICT | ICT-RVT | NT-RVT | |
| VO2 (mL·min−1) | 2767 ± 368 | 2704 ± 496 | 2679 ± 404 (P-N) | 2563 ± 418 (L-N) | 2671 ± 389 | 2587 ± 501 (L-N) | −53 ± 190 | 32 ± 204 | −20 ± 275 |
| 13/47/40 | 33/46/21 | 27/36/37 | |||||||
| Unclear | Unclear | Unclear | |||||||
| Heart Rate (bt·min−1) | 137 ± 16 | 138 ± 13 | 135 ± 12 | 133 ± 14 (P-N) | 141 ± 15 (P-N) | 134 ± 13 (P-N) | −3 ± 9 | −6 ± 6 | −9 ± 9 |
| 10/39/51 | 2/24/74 | 2/12/85 | |||||||
| Unclear | Possible | Likely | |||||||
| RER | 0.81 ± 0.04 | 0.83 ± 0.02 | 0.84 ± 0.11 | 0.80 ± 0.04 (L-N) | 0.87 ± 0.02 (ML-N) | 0.84 ± 0.03 (L-I) | −0.05 ± 0.05 | 0.01 ± 0.08 | −0.04 ± 0.03 |
| 4/3/93 | 49/12/39 | 2/2/96 | |||||||
| Likely | Unclear | Very likely | |||||||
| Glucose (mg·dL−1) | 87 ± 8 | 80 ± 6 | 93 ± 7 (L-N) | 83 ± 11 | 91 ± 5 | 86 ± 7 (L-N) | −3 ± 16 | 5 ± 16 | 2 ± 9 |
| 30/13/57 | 66/11/23 | 56/23/21 | |||||||
| Unclear | Unclear | Unclear | |||||||
| Lactate (mmol·L−1) | 1.1 ± 0.5 | 1.0 ± 0.5 | 0.9 ± 0.1 | 1.0 ± 0.4 | 1.1 ± 0.6 | 0.9 ± 0.2 | 0.1 ± 0.8 | −0.3 ± 0.7 | −0.2 ± 0.8 |
| 46/20/35 | 18/24/58 | 31/20/50 | |||||||
| Unclear | Unclear | Unclear | |||||||
| RPE (6–20) | 12.0 ± 1.0 | 11.7 ± 2.3 | 12.3 ± 1.9 | 12.0 ± 2.2 (P-N) | 11.4 ± 2.2 (L-I) | 11.6 ± 2.2 (P-I) | 0.0 ± 1.1 | 0.4 ± 0.6 | 0.4 ± 1.3 |
| 44/28/28 | 73/24/3 | 66/18/16 | |||||||
| Unclear | Possible | Unclear | |||||||
| Variable | NT | ICT | RVT | Treatment Differences | |||||
|---|---|---|---|---|---|---|---|---|---|
| CHO | CP | CHO | CP | CHO | CP | NT-ICT | ICT-RVT | NT-RVT | |
| Peak Torque at 120 deg/s (N·m) | 110 ± 29 | 93 ± 22 | 99 ± 23 (L-N) | 99 ± 23 (P-N) | 98 ± 31 (L-N) | 95 ± 19 | 17 ± 12 95/5/1 Likely | −2 ± 23 32/35/33 Unclear | 14 ± 18 84/12/4 Likely positive |
| ADJ = 6 ± 16 59/31/10 Unclear | |||||||||
| Peak Torque at 240 deg/s (N·m) | 77 ± 24 | 72 ± 22 | 68 ± 18 (L-N) | 69 ± 10 | 72 ± 27 | 69 ± 15 | 6 ± 16 64/25/11 Unclear | −5 ± 12 10/42/48 Unclear | 1 ± 18 47/29/24 Unclear |
| ADJ = −11 ± 11 2/14/84 Likely | ADJ = −6 ± 16 16/29/55 Unclear | ||||||||
| MHC I CSA (μm2) | 5273 ± 1355 | 4823 ± 1319 | 5356 ± 897 | 5401 ± 1119 (VL-N) | 5925 ±1680 | N/A | 496 ± 856 80/16/4 Likely | N/A | N/A |
| MHC II CSA (μm2) | 5605 ± 1512 | 5105 ± 2004 | 5685 ± 1457 | 5840 ± 1822 (L-N) | 6131 ± 1852 | N/A | 655 ± 1276 76/17/7 Unclear | N/A | N/A |
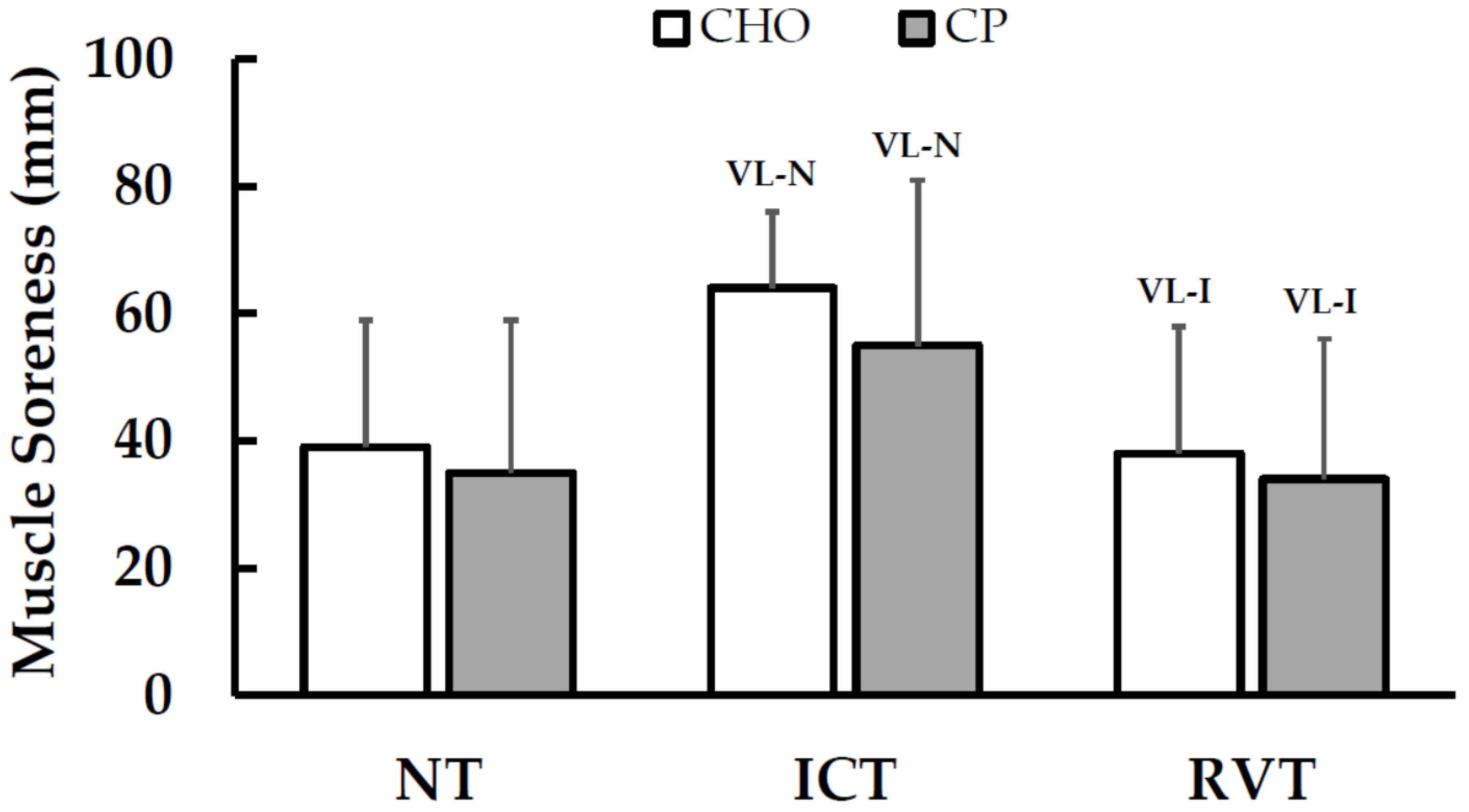
| Muscle Soreness (mm, 0–100) | 39 ± 20 | 35 ± 24 | 65 ± 12 (VL-N) | 55 ± 26 (VL-N) | 38 ± 20 (VL-I) | 34 ± 22 (VL-I) | −6 ± 21 52/26/22 Unclear | 5 ± 20 54/19/27 Unclear | 0 ± 17 61/13/26 Unclear |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Lugos, A.C.; Luden, N.D.; Faller, J.M.; Akers, J.D.; McKenzie, A.I.; Saunders, M.J. Supplemental Protein during Heavy Cycling Training and Recovery Impacts Skeletal Muscle and Heart Rate Responses but Not Performance. Nutrients 2016, 8, 550. https://doi.org/10.3390/nu8090550
D’Lugos AC, Luden ND, Faller JM, Akers JD, McKenzie AI, Saunders MJ. Supplemental Protein during Heavy Cycling Training and Recovery Impacts Skeletal Muscle and Heart Rate Responses but Not Performance. Nutrients. 2016; 8(9):550. https://doi.org/10.3390/nu8090550
Chicago/Turabian StyleD’Lugos, Andrew C., Nicholas D. Luden, Justin M. Faller, Jeremy D. Akers, Alec I. McKenzie, and Michael J. Saunders. 2016. "Supplemental Protein during Heavy Cycling Training and Recovery Impacts Skeletal Muscle and Heart Rate Responses but Not Performance" Nutrients 8, no. 9: 550. https://doi.org/10.3390/nu8090550
APA StyleD’Lugos, A. C., Luden, N. D., Faller, J. M., Akers, J. D., McKenzie, A. I., & Saunders, M. J. (2016). Supplemental Protein during Heavy Cycling Training and Recovery Impacts Skeletal Muscle and Heart Rate Responses but Not Performance. Nutrients, 8(9), 550. https://doi.org/10.3390/nu8090550




