The Association of Dietary l-Arginine Intake and Serum Nitric Oxide Metabolites in Adults: A Population-Based Study
Abstract
:1. Introduction
2. Methods
2.1. Study Population
2.2. Ethics, Consent and Permissions
2.3. Demographic and Anthropometric Measures
2.4. Biochemical Measures
2.5. Dietary Assessment
2.6. Definition of Terms
2.7. Statistical Methods
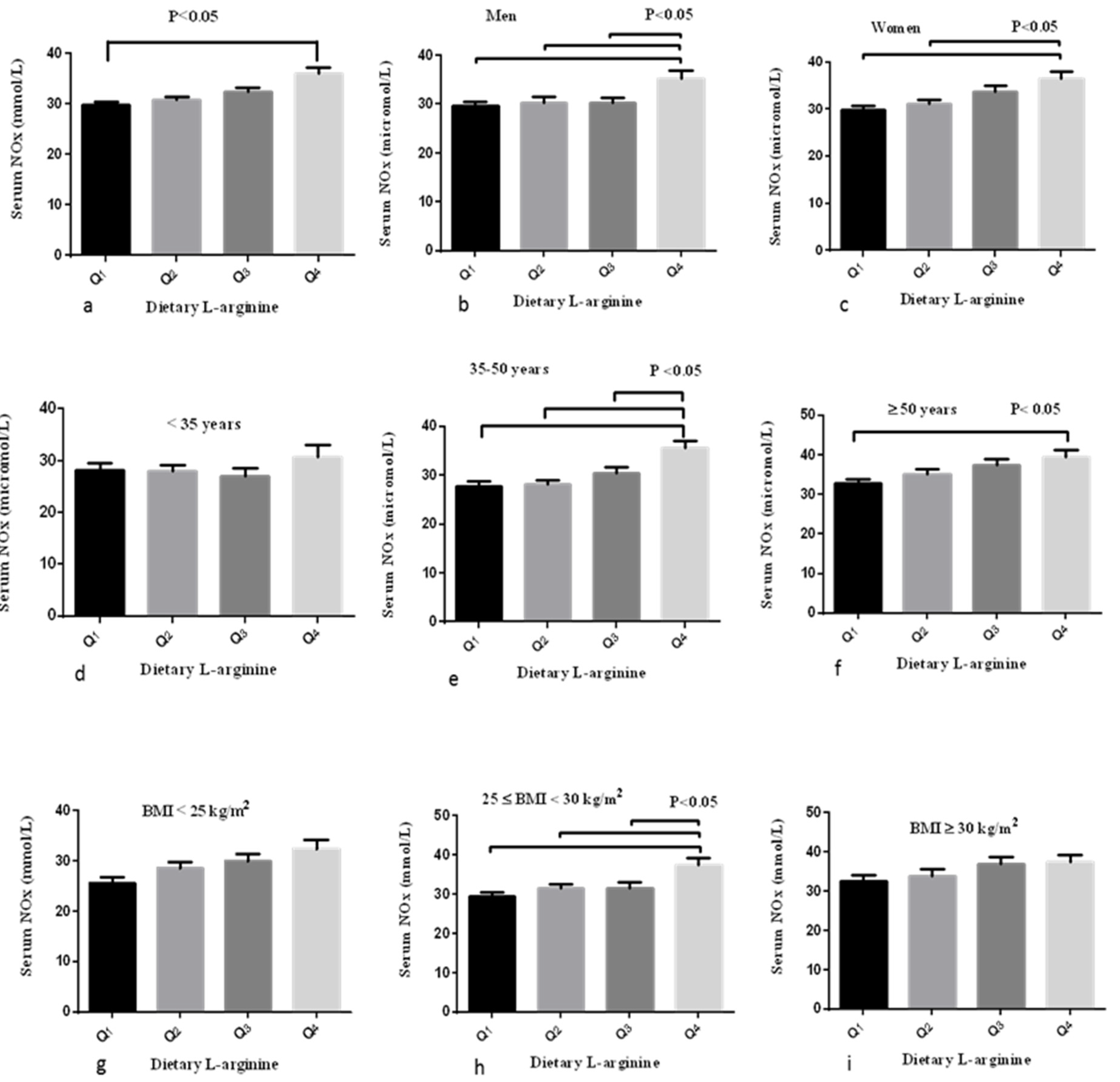
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wells, B.J.; Mainous, A.G.; Everett, C.J. Association between dietary arginine and C-reactive protein. Nutrition 2005, 21, 125–130. [Google Scholar] [CrossRef] [PubMed]
- King, D.E.; Mainous, A.G.; Geesey, M.E. Variation in l-arginine intake follow demographics and lifestyle factors that may impact cardiovascular disease risk. Nutr. Res. 2008, 28, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Venho, B.; Voutilainen, S.; Valkonen, V.P.; Virtanen, J.; Lakka, T.A.; Rissanen, T.H.; Ovaskainen, M.L.; Laitinen, M.; Salonen, J.T. Arginine intake, blood pressure, and the incidence of acute coronary events in men: The Kuopio Ischaemic Heart Disease Risk Factor Study. Am. J. Clin. Nutr. 2002, 76, 359–364. [Google Scholar] [PubMed]
- Morris, S.M. Arginine: Beyond protein. Am. J. Clin. Nutr. 2006, 83, 508S–512S. [Google Scholar] [PubMed]
- Litvinova, L.; Atochin, D.N.; Fattakhov, N.; Vasilenko, M.; Zatolokin, P.; Kirienkova, E. Nitric oxide and mitochondria in metabolic syndrome. Front. Physiol. 2015, 6, 20. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, A.; Zahediasl, S. Is nitric oxide a hormone? Iran. Biomed. J. 2011, 15, 59–65. [Google Scholar] [PubMed]
- Rajapakse, N.W.; Nanayakkara, S.; Kaye, D.M. Pathogenesis and treatment of the cardiorenal syndrome: Implications of l-arginine-nitric oxide pathway impairment. Pharmacol. Ther. 2015, 154, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Virarkar, M.; Alappat, L.; Bradford, P.G.; Awad, A.B. l-arginine and nitric oxide in CNS function and neurodegenerative diseases. Crit. Rev. Food Sci. Nutr. 2013, 53, 1157–1167. [Google Scholar] [CrossRef] [PubMed]
- Hoang, H.H.; Padgham, S.V.; Meininger, C.J. l-arginine, tetrahydrobiopterin, nitric oxide and diabetes. Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, N.W.; Mattson, D.L. Role of l-arginine in nitric oxide production in health and hypertension. Clin. Exp. Pharmacol. Physiol. 2009, 36, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Bahadoran, Z.; Mirmiran, P.; Tahmasebinejad, Z.; Azizi, F. Dietary l-arginine intake and the incidence of coronary heart disease: Tehran lipid and glucose study. Nutr. Metab. 2016, 13, 23. [Google Scholar] [CrossRef] [PubMed]
- Bahadoran, Z.; Mirmiran, P.; Ghasemi, A.; Azizi, F. Serum nitric oxide metabolites are associated with the risk of hypertriglyceridemic-waist phenotype in women: Tehran lipid and glucose study. Nitric Oxide 2015, 50, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Bahadoran, Z.; Mirmiran, P.; Tahmasebi Nejad, Z.; Ghasemi, A.; Azizi, F. Serum nitric oxide is associated with the risk of chronic kidney disease in women: Tehran lipid and glucose study. Scand. J. Clin. Lab. Investig. 2016, 76, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Castillo, L.; Beaumier, L.; Ajami, A.M.; Young, V.R. Whole body nitric oxide synthesis in healthy men determined from [15N] arginine-to-[15N]citrulline labeling. Proc. Natl. Acad. Sci. USA 1996, 93, 11460–11465. [Google Scholar] [CrossRef] [PubMed]
- Van Eijk, H.M.; Luiking, Y.C.; Deutz, N.E. Methods using stable isotopes to measure nitric oxide (NO) synthesis in the l-arginine/NO pathway in health and disease. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2007, 851, 172–185. [Google Scholar] [CrossRef] [PubMed]
- Mariotti, F.; Petzke, K.J.; Bonnet, D.; Szezepanski, I.; Bos, C.; Huneau, J.F.; Fouillet, H. Kinetics of the utilization of dietary arginine for nitric oxide and urea synthesis: Insight into the arginine-nitric oxide metabolic system in humans. Am. J. Clin. Nutr. 2013, 97, 972–979. [Google Scholar] [CrossRef] [PubMed]
- Baylis, C. Arginine, arginine analogs and nitric oxide production in chronic kidney disease. Nat. Clin. Pract. Nephrol. 2006, 2, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.M.; Billiar, T.R. New insights into the regulation of inducible nitric oxide synthesis. Am. J. Physiol. 1994, 266, E829–E839. [Google Scholar] [PubMed]
- Preli, R.B.; Klein, K.P.; Herrington, D.M. Vascular effects of dietary l-arginine supplementation. Atherosclerosis 2002, 162, 1–15. [Google Scholar] [CrossRef]
- Loscalzo, J. What we know and don’t know about l-arginine and NO. Circulation 2000, 101, 2126–2129. [Google Scholar] [CrossRef] [PubMed]
- Bode-Boger, S.M.; Scalera, F.; Ignarro, L.J. The l-arginine paradox: Importance of the l-arginine/asymmetrical dimethylarginine ratio. Pharmacol. Ther. 2007, 114, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Alvares, T.S.; Conte-Junior, C.A.; Silva, J.T.; Paschoalin, V.M. Acute l-Arginine supplementation does not increase nitric oxide production in healthy subjects. Nutr. Metab. 2012, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- Petrella, E.; Pignatti, L.; Neri, I.; Facchinetti, F. The l-arginine/nitric oxide pathway is impaired in overweight/obese pregnant women. Pregnancy Hypertens. 2014, 4, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Azizi, F.; Rahmani, M.; Emami, H.; Mirmiran, P.; Hajipour, R.; Madjid, M.; Ghanbili, J.; Ghanbarian, A.; Mehrabi, J.; Saadat, N.; et al. Cardiovascular risk factors in an Iranian urban population: Tehran lipid and glucose study (phase 1). Soz. Praventivmed. 2002, 47, 408–426. [Google Scholar] [CrossRef] [PubMed]
- Miranda, K.M.; Espey, M.G.; Wink, D.A. A rapid, simple spectrophotometric method for simultaneous detection of nitrate and nitrite. Nitric Oxide 2001, 5, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, A.H.M.; Biabani, H. Protein precipitation methods evaluated for determination of serum nitric oxide end products by the Griess assay. JMSR 2007, 2, 43–46. [Google Scholar]
- Ghasemi, A.; Zahediasl, S. Preanalytical and analytical considerations for measuring nitric oxide metabolites in serum or plasma using the Griess method. Clin. Lab. 2012, 58, 615–624. [Google Scholar] [PubMed]
- Ghasemi, A.; Zahediasl, S.; Azizi, F. Elevated nitric oxide metabolites are associated with obesity in women. Arch. Iran. Med. 2013, 16, 521–525. [Google Scholar] [PubMed]
- Hosseini-Esfahani, F.; Jessri, M.; Mirmiran, P.; Bastan, S.; Azizi, F. Adherence to dietary recommendations and risk of metabolic syndrome: Tehran lipid and glucose study. Metabolism 2010, 59, 1833–1842. [Google Scholar] [CrossRef] [PubMed]
- Mirmiran, P.; Esfahani, F.H.; Mehrabi, Y.; Hedayati, M.; Azizi, F. Reliability and relative validity of an FFQ for nutrients in the Tehran lipid and glucose study. Public Health Nutr. 2010, 13, 654–662. [Google Scholar] [CrossRef] [PubMed]
- US. Food Composition Table. Available online: http://ndb.nal.usda.gov/ndb/search (accessed on 10 February 2014).
- Santamaria, P. Nitrate in vegetables: Toxicity, content, intake and EC regulation. J. Sci. Food Agric. 2006, 86, 10–17. [Google Scholar] [CrossRef]
- American Diabetes Association. Standards of medical care in diabetes—2014. Diabetes Care 2014, 37 (Suppl. S1), S14–S80. [Google Scholar]
- Chobanian, A.V.; Bakris, G.L.; Black, H.R.; Cushman, W.C.; Green, L.A.; Izzo, J.L.; Jones, D.W.; Materson, B.J.; Oparil, S.; Wright, J.T.; et al. The seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure: The JNC 7 report. JAMA 2003, 289, 2560–2572. [Google Scholar] [CrossRef] [PubMed]
- Bozorgmanesh, M.; Hadaegh, F.; Azizi, F. Predictive performances of lipid accumulation product vs. adiposity measures for cardiovascular diseases and all-cause mortality, 8.6-years follow-up: Tehran lipid and glucose study. Lipids Health Dis. 2010, 9, 100. [Google Scholar] [CrossRef] [PubMed]
- Nwagha, U.I.; Ikekpeazu, E.J.; Ejezie, F.E.; Neboh, E.E.; Maduka, I.C. Atherogenic index of plasma as useful predictor of cardiovascular risk among postmenopausal women in Enugu, Nigeria. Afr. Health Sci. 2010, 10, 248–252. [Google Scholar] [PubMed]
- Willett, W.C.; Howe, G.R.; Kushi, L.H. Adjustment for total energy intake in epidemiologic studies. Am. J. Clin. Nutr. 1997, 65, 1220S–1228S. [Google Scholar] [PubMed]
- Bondonno, C.P.; Liu, A.H.; Croft, K.D.; Ward, N.C.; Puddey, I.B.; Woodman, R.J.; Hodgson, J.M. Short-term effects of a high nitrate diet on nitrate metabolism in healthy individuals. Nutrients 2015, 7, 1906–1915. [Google Scholar] [CrossRef] [PubMed]
- Crawford, N.M.; Guo, F.Q. New insights into nitric oxide metabolism and regulatory functions. Trends Plant Sci. 2005, 10, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O.; Weitzberg, E.; Gladwin, M.T. The nitrate-nitrite-nitric oxide pathway in physiology and therapeutics. Nat. Rev. Drug Discov. 2008, 7, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Faldetta, M.C.; Desideri, G.; Bravi, M.C.; De Luca, O.; Marinucci, M.C.; De Mattia, G.; Ferri, C. l-arginine infusion decreases plasma total homocysteine concentrations through increased nitric oxide production and decreased oxidative status in Type II diabetic patients. Diabetologia 2002, 45, 1120–1127. [Google Scholar] [CrossRef] [PubMed]
- Jabłecka, A.; Chęciński, P.; Krauss, H.; Micker, M.; Ast, J. The influence of two different doses of l-arginine oral supplementation on nitric oxide (NO) concentration and total antioxidant status (TAS) in atherosclerotic patients. Med. Sci. Monit. 2004, 10, 29–32. [Google Scholar]
- Jabłecka, A.B.P.; Balcer, N.; Cieślewicz, A.; Skołuda, A.; Musialik, K. The effect of oral l-arginine supplementation on fasting glucose, HbA1c, nitric oxide and total antioxidant status in diabetic patients with atherosclerotic peripheral arterial disease of lower extremities. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 342–350. [Google Scholar] [PubMed]
- Tangphao, O.; Chalon, S.; Coulston, A.M.; Moreno, H.; Chan, J.R.; Cooke, J.P.; Hoffman, B.B.; Blaschke, T.F. l-arginine and nitric oxide-related compounds in plasma: Comparison of normal and arginine-free diets in a 24-h crossover study. Vasc. Med. 1999, 4, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Boger, R.H. The pharmacodynamics of l-arginine. J. Nutr. 2007, 137, 1650S–1655S. [Google Scholar] [PubMed]
- Olszanecka-Glinianowicz, M.; Zahorska-Markiewicz, B.; Janowska, J.; Zurakowski, A. Serum concentrations of nitric oxide, tumor necrosis factor (TNF)-alpha and TNF soluble receptors in women with overweight and obesity. Metabolism 2004, 53, 1268–1273. [Google Scholar] [CrossRef] [PubMed]
- Kocak, H.; Oner-Iyidogan, Y.; Gurdol, F.; Oner, P.; Esin, D. Serum asymmetric dimethylarginine and nitric oxide levels in obese postmenopausal women. J. Clin. Lab. Anal. 2011, 25, 174–178. [Google Scholar] [CrossRef] [PubMed]
- Koc, F.; Tokac, M.; Erdem, S.; Kaya, C.; Unlu, A.; Karabag, T.; Vatankulu, M.A.; Demir, K.; Ayhan, S.; Kaya, A. Serum asymmetric dimethylarginine levels in normotensive obese individuals. Med. Sci. Monit. 2010, 16, CR536–CR539. [Google Scholar] [PubMed]
- Taddei, S.; Virdis, A.; Mattei, P.; Ghiadoni, L.; Sudano, I.; Salvetti, A. Defective l-arginine-nitric oxide pathway in offspring of essential hypertensive patients. Circulation 1996, 94, 1298–1303. [Google Scholar] [CrossRef] [PubMed]
- Masini, E.; Mannaioni, P.F.; Pistelli, A.; Salvemini, D.; Vane, J. Impairment of the l-arginine-nitric oxide pathway in mast cells from spontaneously hypertensive rats. Biochem. Biophys. Res. Commun. 1991, 177, 1178–1182. [Google Scholar] [CrossRef]
- Kielstein, J.T.; Bode-Boger, S.M.; Frolich, J.C.; Ritz, E.; Haller, H.; Fliser, D. Asymmetric dimethylarginine, blood pressure, and renal perfusion in elderly subjects. Circulation 2003, 107, 1891–1895. [Google Scholar] [CrossRef] [PubMed]
- Marliss, E.B.; Chevalier, S.; Gougeon, R.; Morais, J.A.; Lamarche, M.; Adegoke, O.A.J.; Wu, G. Elevations of plasma methylarginines in obesity and ageing are related to insulin sensitivity and rates of protein turnover. Diabetologia 2006, 49, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Ryu, H.; Ferrante, R.J.; Morris, S.M.; Ratan, R.R. Translational control of inducible nitric oxide synthase expression by arginine can explain the arginine paradox. Proc. Natl. Acad. Sci. USA 2003, 100, 4843–4848. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Meininger, C.J. Nitric oxide and vascular insulin resistance. Biofactors 2009, 35, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Luiking, Y.C.; Engelen, M.P.; Deutz, N.E. Regulation of nitric oxide production in health and disease. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Kim-Shapiro, D.B.; Gladwin, M.T. Pitfalls in measuring NO bioavailability using NOx. Nitric Oxide 2015, 44, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Tsikas, D. Circulating and excretory nitrite and nitrate: Their value as measures of nitric oxide synthesis, bioavailability and activity is inherently limited. Nitric Oxide 2015, 45, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Tsikas, D. Methods of quantitative analysis of the nitric oxide metabolites nitrite and nitrate in human biological fluids. Free Radic. Res. 2005, 39, 797–815. [Google Scholar] [CrossRef] [PubMed]
- Moshage, H.; Kok, B.; Huizenga, J.R.; Jansen, P.L. Nitrite and nitrate determinations in plasma: A critical evaluation. Clin. Chem. 1995, 41, 892–896. [Google Scholar] [PubMed]
- Baylis, C.; Vallance, P. Measurement of nitrite and nitrate levels in plasma and urine—What does this measure tell us about the activity of the endogenous nitric oxide system? Curr. Opin. Nephrol. Hypertens. 1998, 7, 59–62. [Google Scholar] [CrossRef] [PubMed]
| Dietary l-Arginine | |||||
|---|---|---|---|---|---|
| Q1 (n = 692) | Q2 (n = 693) | Q3 (n = 693) | Q4 (n = 693) | p * | |
| l-arginine (g/day) | |||||
| Range | <3.06 | 3.06–3.92 | 3.92–5.20 | ≥5.20 | |
| Median | 2.55 | 3.49 | 4.46 | 6.33 | |
| Age (years) | 45.5 ± 16.2 | 46.1 ± 15.8 | 45.9 ± 15.5 | 46.0 ± 16.2 | 0.90 |
| Men (%) | 39.4 | 40.0 | 39.2 | 38.2 | 0.91 |
| Smoking (%) | 9.5 | 9.4 | 8.7 | 7.9 | 0.69 |
| Body mass index (kg/m2) | 27.6 ± 4.9 | 27.1 ± 4.8 | 27.7 ± 4.7 | 27.6 ± 4.7 | 0.13 |
| Waist circumferences (cm) | 91.8 ± 13.2 | 90.8 ± 12.9 | 92.1 ± 13.0 | 91.8 ± 12.9 | 0.27 |
| Systolic blood pressure (mmHg) | 116 ± 19.3 | 119 ± 17.2 | 117 ± 18.2 | 117 ± 19.4 | 0.56 |
| Diastolic blood pressure (mmHg) | 72.5 ± 10.3 | 72.9 ± 10.5 | 73.6 ± 10.2 | 72.7 ± 10.7 | 0.24 |
| Fasting blood glucose (mg/dL) | 97.7 ± 31.3 | 98.0 ± 30.1 | 97.2 ± 30.4 | 98.9 ± 33.8 | 0.79 |
| Total cholesterol (mg/dL) | 191 ± 38.0 | 193 ± 42.3 | 194 ± 34.0 | 193 ± 41.0 | 0.49 |
| Triglycerides (mg/dL) | 153 ± 104 | 158 ± 106 | 156 ± 92.0 | 149 ± 93.0 | 0.41 |
| HDL-C (mg/dL) | 42.8 ± 10.6 | 42.5 ± 10.2 | 42.3 ± 9.7 | 43.3 ± 10.1 | 0.29 |
| LDL-C (mg/dL) | 119 ± 33.0 | 119 ± 32.8 | 121 ± 33.1 | 119 ± 32.3 | 0.46 |
| Serum creatinine (μmol/L) | 93.0 ± 18.6 | 91.4 ± 16.8 | 91.5 ± 16.1 | 91.0 ± 15.0 | 0.15 |
| Hypertension (%) | 16.0 | 18.1 | 14.3 | 18.0 | 0.19 |
| History of cardiovascular disease (%) | 2.9 | 3.9 | 4.1 | 4.7 | 0.40 |
| Dietary l-Arginine | ||||
|---|---|---|---|---|
| Q1 (n = 692) | Q2 (n = 693) | Q3 (n = 693) | Q4 (n = 693) | |
| Dietary l-arginine (g/day) | ||||
| Range | <3.06 | 3.06–3.92 | 3.92–5.20 | ≥5.20 |
| Median | 2.55 | 3.49 | 4.46 | 6.33 |
| Energy intake (kcal/day) | 296151.0 | 2178 ± 32.0 | 2099 ± 29.8 | 2638 ± 48.6 |
| Carbohydrate (% energy) | 55.8 ± 0.3 | 57.9 ± 0.3 | 58.8 ± 0.2 | 56.5 ± 0.3 |
| Protein (% energy) | 11.4 ± 0.1 | 12.8 ± 0.1 | 14.1 ± 0.1 | 16.1 ± 0.1 |
| Total fats (% energy) | 35.1 ± 0.3 | 31.7 ± 0.2 | 29.7 ± 0.2 | 30.0 ± 0.3 * |
| Total vegetables (g/day) | 275 ± 7.1 | 270 ± 6.9 | 305 ± 6.9 | 342 ± 6.9 * |
| High-nitrate vegetables (g/day) | 35.4 ± 1.4 | 25.1 ± 1.5 | 42.0 ± 1.5 | 43.4 ± 1.5 * |
| Medium-nitrate vegetables (g/day) | 31.1 ± 1.3 | 31.2 ± 1.3 | 33.5 ± 1.3 | 41.3 ± 1.3 * |
| Low-nitrate vegetables (g/day) | 280 ± 9.0 | 273 ± 9.1 | 275 ± 8.8 | 262 ± 9.2 * |
| Grains (g/day) | 15.5 ± 1.9 | 20.4 ± 1.9 | 23.4 ± 1.9 | 33.9 ± 1.9 * |
| Processed meats | 7.2 ± 0.6 | 7.9 ± 0.5 | 8.1 ± 0.5 | 11.8 ± 0.5 * |
| Dietary l-Arginine (mg/Day) | |||||
|---|---|---|---|---|---|
| l-Arginine as Continuous | Q2 (3.06–3.92) | Q3 (3.92–5.20) | Q4 (≥5.20) | p for Trend 3 | |
| Total | 0.96 (0.63, 1.29) 1 1.67 (1.03, 2.32) 2 | 1.07 (−1.28, 3.43) 1 1.24 (−1.24, 3.73) 2 | 2.63 (0.27, 4.99) 1 2.89 (−0.42, 5.37) 2 | 6.28 (3.92, 8.64) 1 6.63 (4.14, 9.12) 2 | 0.003 0.001 |
| Men (n = 1086) | 0.22 (−0.31, 0.74) 1 0.35 (−0.54, 1.24) 2 | 0.78 (−2.61, 4.18) 1 1.22 (−2.48, 4.92) 2 | 0.69 (−2.72, 4.11) 1 1.61 (−2.19, 5.40) 2 | 5.64 (2.20, 9.08) 1 4.36 (0.57−8.14) 2 | 0.121 0.024 |
| Women (n = 1685) | 1.29 (0.87, 1.71) 1 2.69 (1.78, 3.61) 2 | 1.27 (−1.93, 4.48) 1 0.93 (−2.72, 4.57) 2 | 3.89 (0.68, 7.09) 1 3.49 (−0.16, 7.16) 2 | 6.67 (3.49, 9.86) 1 6.12 (6.52, 9.72) 2 | 0.016 0.001 |
| Age-categories | |||||
| <35 years (n = 788) | 1.50 (0.95, 2.04) 1 0.17 (−0.46, 0.81) 2 | 0.11 (−3.75, 3.97) 1 0.55 (−3.87, 4.33) 2 | 0.91 (−3.01, 4.83) 1 1.74 (−2.31, 6.32) 2 | 3.68 (−0.13, 7.50) 1 6.07 (0.44, 11.7) 2 | 0.327 0.051 |
| 35–50 years (n = 911) | 0.77 (0.19, 1.35) 1 0.58 (−0.10, 1.27)2 | 0.29 (−3.69, 4.27) 1 0.73 (−3.90, 5.67) 2 | 1.91 (−1.98, 5.80) 1 2.27 (−2.96, 6.38) 2 | 8.27 (4.26, 12.2) 1 9.12 (3.99, 13.61) 2 | 0.003 0.001 |
| ≥50 years (n = 1072) | 0.73 (0.20, 1.27) 1 0.91 (0.30, 1.52) 2 | 2.28 (−1.83, 6.41) 1 3.39 (−0.89, 7.69) 2 | 4.54 (0.37, 8.71) 1 7.07 (2.46, 11.6) 2 | 6.62 (2.48, 10.7) 1 12.1 (6.48, 17.7) 2 | 0.167 0.050 |
| BMI-categories | |||||
| <25 kg/m2 (n = 808) | 0.12 (−0.46, 0.70) 1 0.36 (−1.02, 1.70) 2 | 1.07 (−2.65, 4.79) 1 0.98 (−2.87, 4.83) 2 | 2.49 (−1.37, 6.36) 1 3.57 (−0.66, 7.81) 2 | 4.87 (1.02, 8.72) 1 8.12 (−2.83, 13.4) 2 | 0.888 0.091 |
| 25–30 kg/m2 (n = 1147) | 0.26 (−0.46, 0.99) 1 1.02 (−0.83, 2.87) 2 | 1.84 (−1.95, 5.65) 1 2.38 (−1.57, 6.33) 2 | 2.09 (−1.76, 5.94) 1 3.36 (−0.82, 7.54) 2 | 7.85 (3.98, 11.7) 1 10.7 (5.43, 16.0) 2 | 0.160 0.024 |
| ≥30 kg/m2 (n = 816) | 1.06 (0.57, 1.55) 1 2.12 (1.30, 2.94) 2 | 1.17 (−3.83, 6.17) 1 2.78 (−2.34, 7.91) 2 | 4.29 (−0.36, 8.95) 1 7.39 (2.05, 12.7) 2 | 4.85 (0.21, 9.49) 1 11.0 (4.29, 17.5) 2 | 0.027 0.001 |
| Body Mass Index (kg/m2) | |||
|---|---|---|---|
| <25 (n = 808) | 25–30 (n = 1147) | ≥30 (n = 816) | |
| Systolic blood pressure (mmHg) | 110 ± 18.2 | 116 ± 18.0 | 124 ± 18.8 * |
| Diastolic blood pressure (mmHg) | 68.9 ± 9.6 | 73.3 ± 9.7 | 77.4 ± 10.2 * |
| Serum creatinine (μmol/L) | 91.7 ± 16.5 | 91.4 ± 16.5 | 92.5 ± 17.5 |
| Atherogenic index of plasma | 0.37 ± 0.27 | 0.53 ± 0.29 | 0.61 ± 0.27 * |
| Diabetes (%) | 8.5 | 11.2 | 19.0 * |
| Hypertension (%) | 8.9 | 15.0 | 28.8 * |
| History of cardiovascular disease (%) | 2.5 | 4.3 | 4.8 * |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mirmiran, P.; Bahadoran, Z.; Ghasemi, A.; Azizi, F. The Association of Dietary l-Arginine Intake and Serum Nitric Oxide Metabolites in Adults: A Population-Based Study. Nutrients 2016, 8, 311. https://doi.org/10.3390/nu8050311
Mirmiran P, Bahadoran Z, Ghasemi A, Azizi F. The Association of Dietary l-Arginine Intake and Serum Nitric Oxide Metabolites in Adults: A Population-Based Study. Nutrients. 2016; 8(5):311. https://doi.org/10.3390/nu8050311
Chicago/Turabian StyleMirmiran, Parvin, Zahra Bahadoran, Asghar Ghasemi, and Fereidoun Azizi. 2016. "The Association of Dietary l-Arginine Intake and Serum Nitric Oxide Metabolites in Adults: A Population-Based Study" Nutrients 8, no. 5: 311. https://doi.org/10.3390/nu8050311
APA StyleMirmiran, P., Bahadoran, Z., Ghasemi, A., & Azizi, F. (2016). The Association of Dietary l-Arginine Intake and Serum Nitric Oxide Metabolites in Adults: A Population-Based Study. Nutrients, 8(5), 311. https://doi.org/10.3390/nu8050311





