Hypertrophy-Promoting Effects of Leucine Supplementation and Moderate Intensity Aerobic Exercise in Pre-Senescent Mice
Abstract
:1. Introduction
2. Experimental Section
2.1. Animals and Diets
2.2. Experimental Protocols
2.3. Blood Glucose, Serum Insulin, Serum Urea Nitrogen, and Plasma Free Amino Acids Measurement
2.4. Western Blot Analysis
2.5. Histological Examination
2.6. Statistical Analysis
3. Results
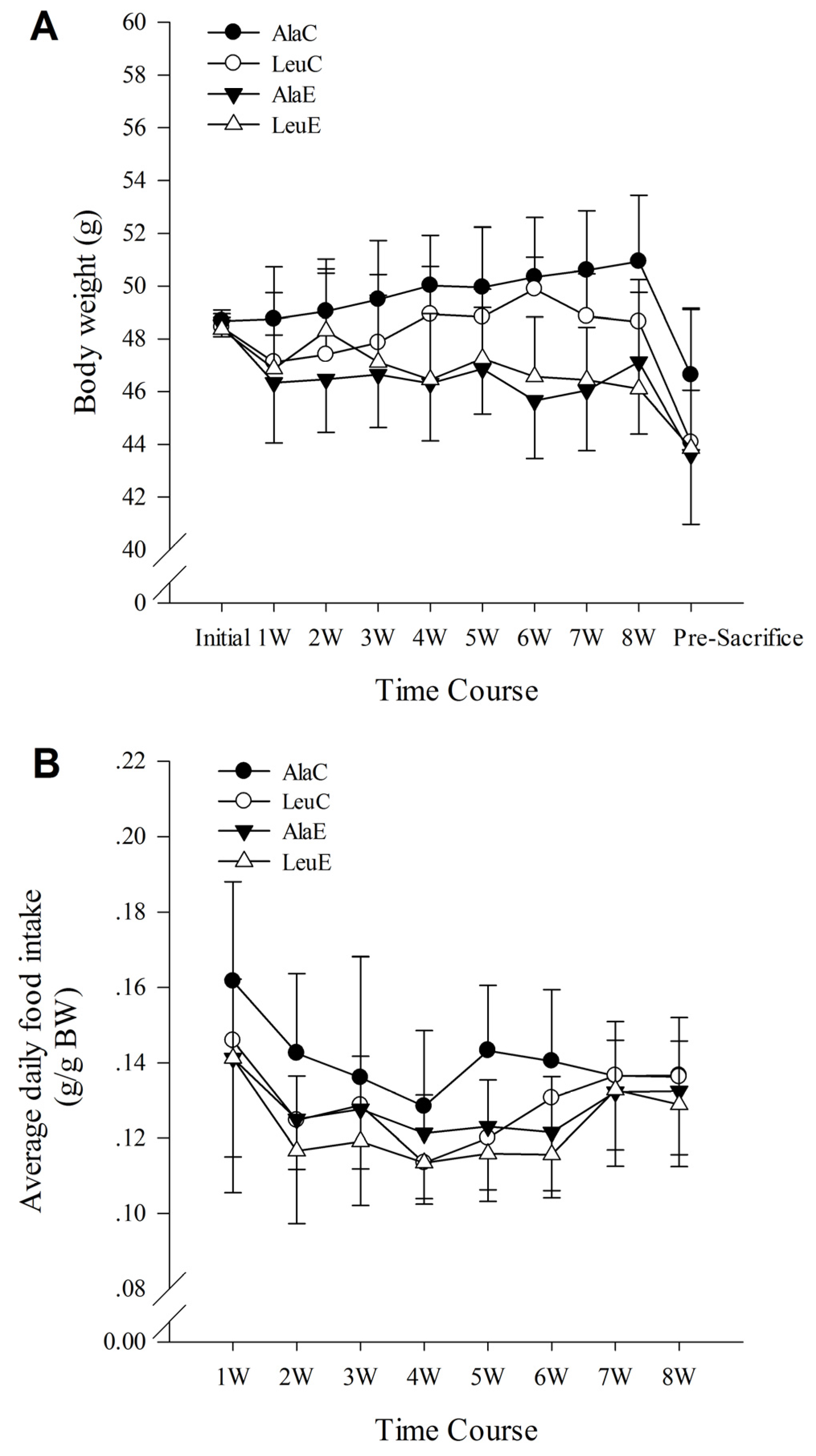
3.1. Body Weight
3.2. Food Intake
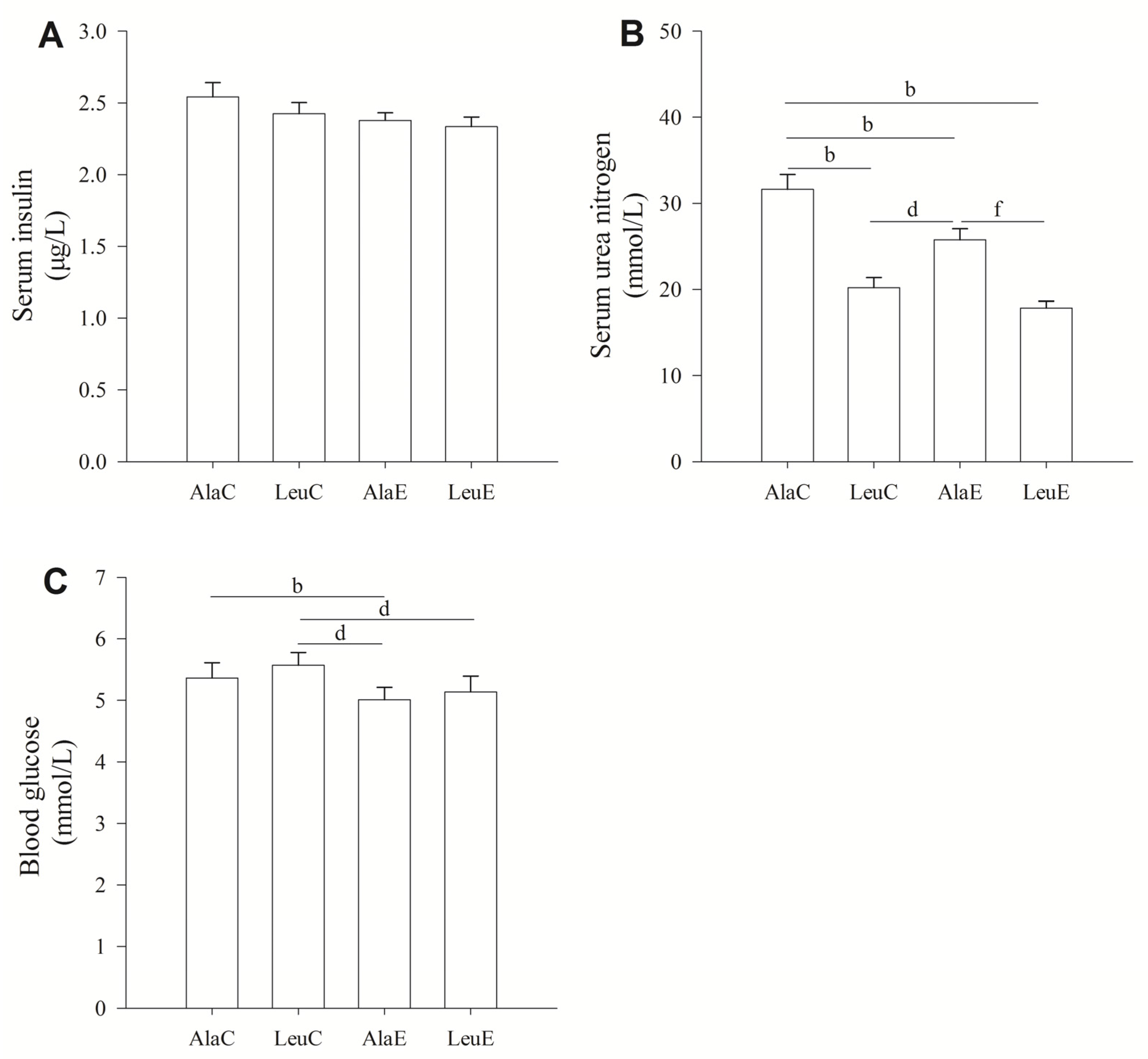
3.3. Concentrations of Serum Insulin, Urea Nitrogen, and Blood Glucose
3.4. Plasma Free Amino Acids Analysis
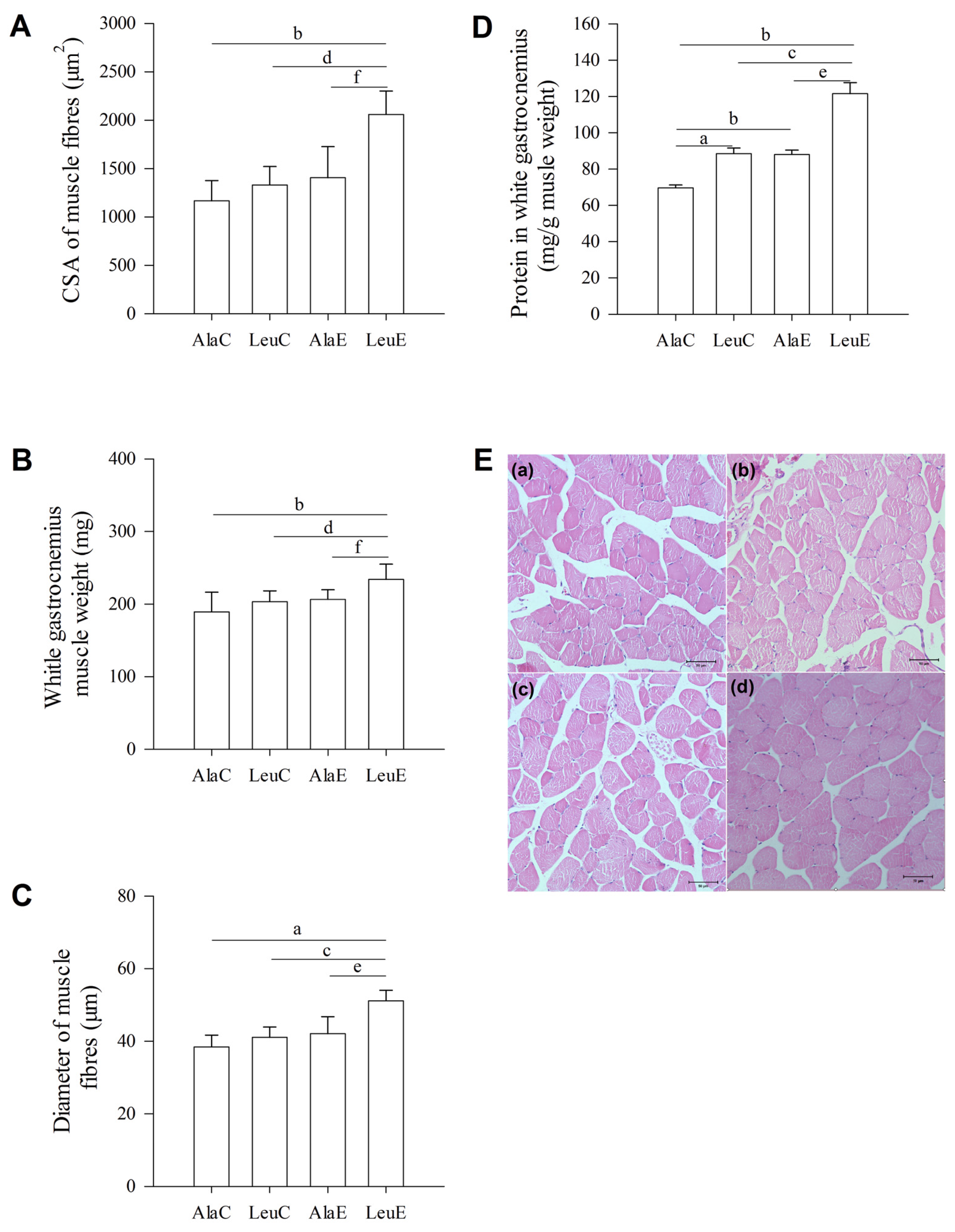
3.5. Histological Changes of Muscle Fibers
3.6. Total Protein in White Gastrocnemius Muscle
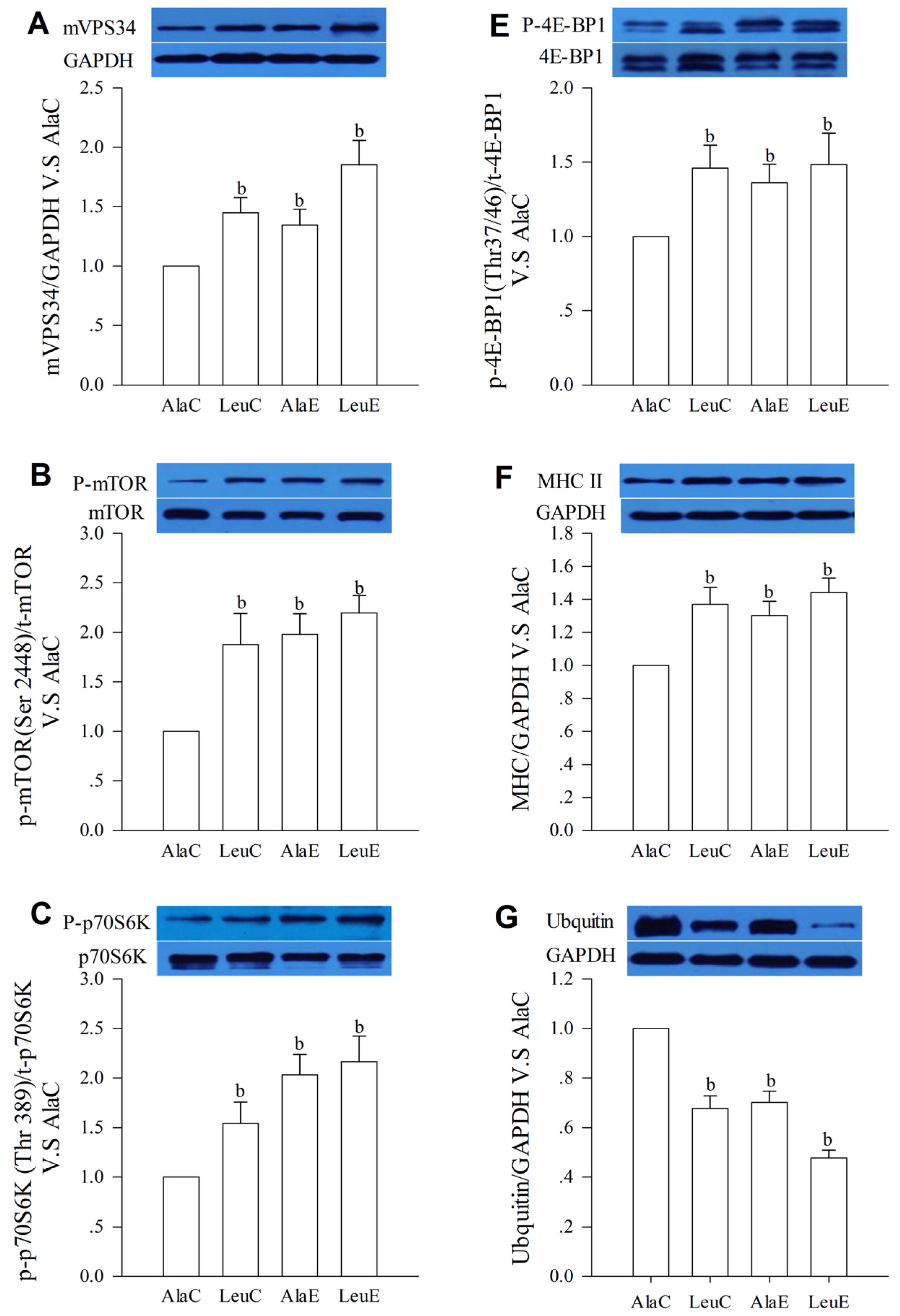
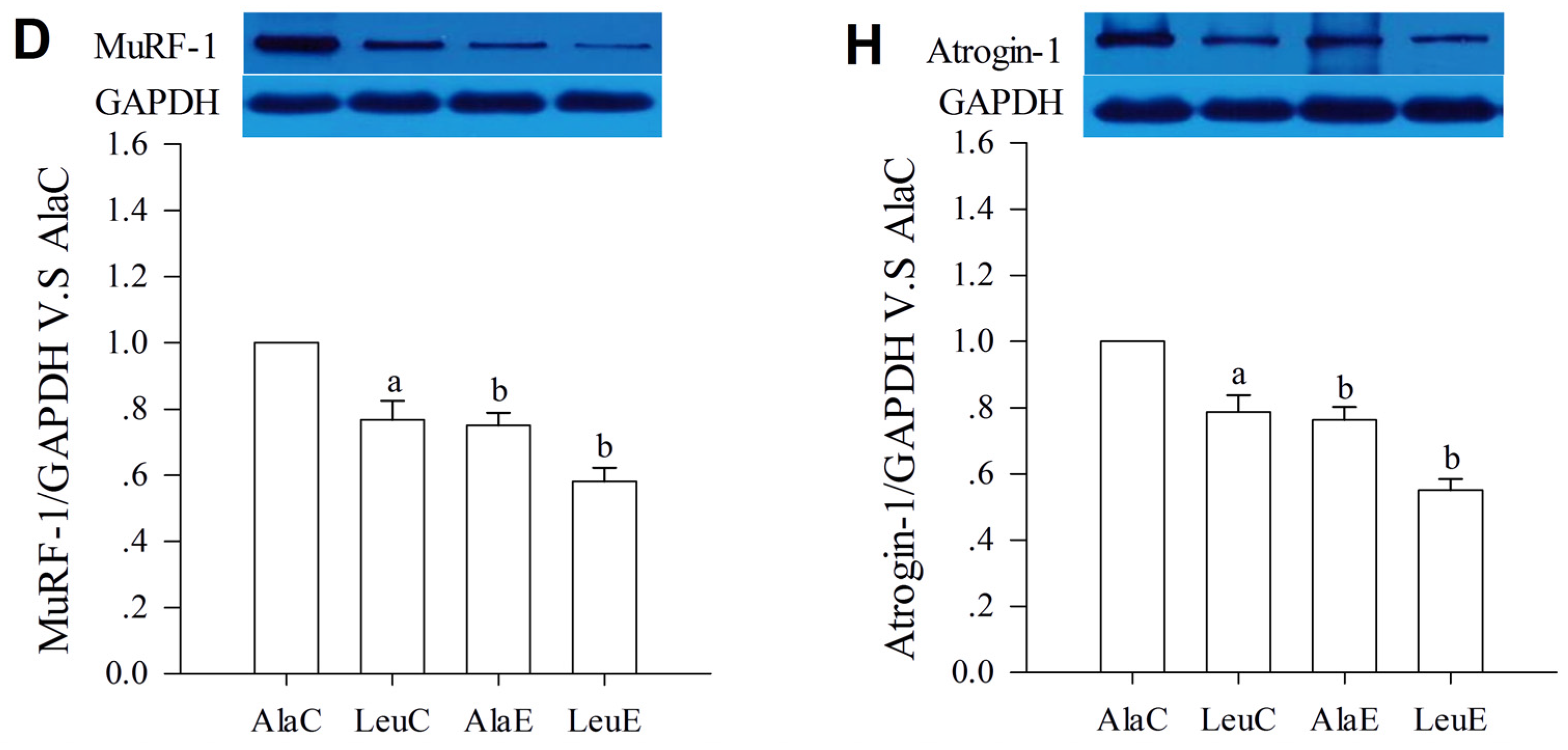
3.7. Protein Expression with Relation to Hypertrophy and Atrophy
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Roberts, S.B.; Rosenberg, I. Nutrition and aging: Changes in the regulation of energy metabolism with aging. Physiol. Rev. 2006, 86, 651–667. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M.A.; Hagberg, J.M.; Martin, W.H., 3rd; Ehsani, A.A.; Holloszy, J.O. Decline in VO2max with aging in master athletes and sedentary men. J. Appl. Physiol. 1990, 68, 2195–2199. [Google Scholar] [PubMed]
- Short, K.R.; Bigelow, M.L.; Kahl, J.; Singh, R.; Coenen-Schimke, J.; Raghavakaimal, S.; Nair, K.S. Decline in skeletal muscle mitochondrial function with aging in humans. Proc. Natl. Acad. Sci. USA 2005, 102, 5618–5623. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E.; Abbatecola, A.M.; Argiles, J.M.; Baracos, V.; Bauer, J.; Bhasin, S.; Cederholm, T.; Coats, A.J.; Cummings, S.R.; Evans, W.J.; et al. Sarcopenia with limited mobility: An international consensus. J. Am. Med. Dir. Assoc. 2011, 12, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Delos Reyes, A.D.; Bagchi, D.; Preuss, H.G. Overview of resistance training, diet, hormone replacement and nutritional supplements on age-related sarcopenia—A mini review. Res. Commun. Mol. Pathol. Pharmacol. 2003, 113–114, 159–170. [Google Scholar]
- Yarasheski, K.E. Exercise, aging, and muscle protein metabolism. J. Gerontol. A Biol. Sci. Med. Sci. 2003, 58, M918–M922. [Google Scholar] [CrossRef] [PubMed]
- Marcotte, G.R.; West, D.W.; Baar, K. The molecular basis for load-induced skeletal muscle hypertrophy. Calcif. Tissue Int. 2015, 96, 196–210. [Google Scholar] [CrossRef] [PubMed]
- Murton, A.J.; Greenhaff, P.L. Resistance exercise and the mechanisms of muscle mass regulation in humans: Acute effects on muscle protein turnover and the gaps in our understanding of chronic resistance exercise training adaptation. Int. J. Biochem. Cell. Biol. 2013, 45, 2209–2214. [Google Scholar] [CrossRef] [PubMed]
- Battaglini, C.L.; Hackney, A.C.; Goodwin, M.L. Cancer cachexia: Muscle physiology and exercise training. Cancers (Basel) 2012, 4, 1247–1251. [Google Scholar] [CrossRef] [PubMed]
- Lira, F.S.; Neto, J.C.; Seelaender, M. Exercise training as treatment in cancer cachexia. Appl. Physiol. Nutr. Metab. 2014, 39, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Alves, C.R.; da Cunha, T.F.; da Paixao, N.A.; Brum, P.C. Aerobic exercise training as therapy for cardiac and cancer cachexia. Life Sci. 2015, 125, 9–14. [Google Scholar] [PubMed]
- O’Hagan, C.; de Vito, G.; Boreham, C.A. Exercise prescription in the treatment of type 2 diabetes mellitus: Current practices, existing guidelines and future directions. Sports Med. 2013, 43, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Mohajeri, S.; Riddell, M.C. Advances in exercise, physical activity, and diabetes mellitus. Diabetes Technol. Ther. 2015, 17, S88–S95. [Google Scholar] [CrossRef] [PubMed]
- Bell, K.E.; Séguin, C.; Parise, G.; Baker, S.K.; Phillips, S.M. Day-to-Day changes in muscle protein synthesis in recovery from resistance, aerobic, and high-intensity interval exercise in older men. J. Gerontol. A Biol. Sci. Med. Sci. 2015, 70, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Mascher, H.; Ekblom, B.; Rooyackers, O.; Blomstrand, E. Enhanced rates of muscle protein synthesis and elevated mTOR signalling following endurance exercise in human subjects. Acta. Physiol. (Oxf.) 2011, 202, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Harber, M.P.; Dickinson, J.M.; Crane, J.D.; Trappe, S.W.; Trappe, T.A. Influence of tracer selection on protein synthesis rates at rest and postexercise in multiple human muscles. Metabolism 2011, 60, 689–697. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Denison, H.J.; Cooper, C.; Sayer, A.A.; Robinson, S.M. Prevention and optimal management of sarcopenia: A review of combined exercise and nutrition interventions to improve muscle outcomes in older people. Clin. Interv. Aging 2015, 10, 859–869. [Google Scholar] [PubMed]
- Van Til, A.J.; Naumann, E.; Cox-Claessens, I.J.; Kremer, S.; Boelsma, E.; de van der Schueren, M.A. Effects of the daily consumption of protein enriched bread and protein enriched drinking yoghurt on the total protein intake in older adults in a rehabilitation centre: A single blind randomised controlled trial. J. Nutr. Health Aging 2015, 19, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Koh, F.; Charlton, K.; Walton, K.; McMahon, A.T. Role of dietary protein and thiamine intakes on cognitive function in healthy older people: A systematic review. Nutrients 2015, 7, 2415–2439. [Google Scholar] [CrossRef] [PubMed]
- Paddon-Jones, D.; Campbell, W.W.; Jacques, P.F.; Kritchevsky, S.B.; Moore, L.L.; Rodriguez, N.R.; van Loon, L.J. Protein and healthy aging. Am. J. Clin. Nutr. 2015, 101, 1339S–1345S. [Google Scholar] [CrossRef] [PubMed]
- Breen, L.; Stokes, K.A.; Churchward-Venne, T.A.; Moore, D.R.; Baker, S.K.; Smith, K.; Atherton, P.J.; Phillips, S.M. Two weeks of reduced activity decreases leg lean mass and induces “anabolic resistance” of myofibrillar protein synthesis in healthy elderly. J. Clin. Endocrinol. Metab. 2013, 98, 2604–2612. [Google Scholar] [CrossRef] [PubMed]
- Vary, T.C.; Lynch, C.J. Meal feeding stimulates phosphorylation of multiple effector proteins regulating protein synthetic processes in rat hearts. J. Nutr. 2006, 136, 2284–2290. [Google Scholar] [PubMed]
- Dickinson, J.M.; Gundermann, D.M.; Walker, D.K.; Reidy, P.T.; Borack, M.S.; Drummond, M.J.; Arora, M.; Volpi, E.; Rasmussen, B.B. Leucine-enriched amino acid ingestion after resistance exercise prolongs myofibrillar protein synthesis and amino acid transporter expression in older men. J. Nutr. 2014, 144, 1694–1702. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.L.; Irving, B.A.; Lanza, I.R.; Vendelbo, M.H.; Konopka, A.R.; Robinson, M.M.; Henderson, G.C.; Klaus, K.A.; Morse, D.M.; Heppelmann, C.; et al. Differential effect of endurance training on mitochondrial protein damage, degradation, and acetylation in the context of aging. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 70, 1386–1393. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.M.; Yu, M.X.; Shen, Z.Y.; Guo, C.Y.; Zhuang, S.Q.; Qiu, X.S. Leucine promotes proliferation and differentiation of primary preterm rat satellite cells in part through mTORC1 signaling pathway. Nutrients 2015, 7, 3387–3400. [Google Scholar] [CrossRef] [PubMed]
- Ziaaldini, M.M.; Koltai, E.; Csende, Z.; Goto, S.; Boldogh, I.; Taylor, A.W.; Radak, Z. Exercise training increases anabolic and attenuates catabolic and apoptotic processes in aged skeletal muscle of male rats. Exp. Gerontol. 2015, 67, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Lynch, C.J.; Adams, S.H. Branched-chain amino acids in metabolic signalling and insulin resistance. Nat. Rev. Endocrinol. 2014, 10, 723–736. [Google Scholar] [CrossRef] [PubMed]
- Garlick, P.J.; Grant, I. Amino acid infusion increases the sensitivity of muscle protein synthesis in vivo to insulin. Effect of branched-chain amino acids. Biochem. J. 1988, 254, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Dardevet, D.; Sornet, C.; Bayle, G.; Prugnaud, J.; Pouyet, C.; Grizard, J. Postprandial stimulation of muscle protein synthesis in old rats can be restored by a leucine-supplemented meal. J. Nutr. 2002, 132, 95–100. [Google Scholar] [PubMed]
- Rieu, I.; Sornet, C.; Bayle, G.; Prugnaud, J.; Pouyet, C.; Balage, M.; Papet, I.; Grizard, J.; Dardevet, D. Leucine-supplemented meal feeding for ten days beneficially affects postprandial muscle protein synthesis in old rats. J. Nutr. 2003, 133, 1198–1205. [Google Scholar] [PubMed]
- Combaret, L.; Dardevet, D.; Rieu, I.; Pouch, M.N.; Bechet, D.; Taillandier, D.; Grizard, J.; Attaix, D. A leucine-supplemented diet restores the defective postprandial inhibition of proteasome-dependent proteolysis in aged rat skeletal muscle. J. Physiol. 2005, 569, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Gobatto, C.A.; Mello, M.A.; Souza, C.T.; Ribeiro, I.A. The monosodium glutamate (MSG) obese rat as a model for the study of exercise in obesity. Res. Commun. Mol. Pathol. Pharmacol. 2002, 111, 89–101. [Google Scholar] [PubMed]
- Bartolomeu, R.F.; Barbosa, T.M.; Morais, J.E.; Lopes, V.P.; Bragada, J.A.; Costa, M.J. The aging influence on cardiorespiratory, metabolic and energy expenditure adaptations in head-out aquatic exercises: Differences between young and elderly women. Women Health 2016, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, Y.; Zhang, Q.W.; Sugimoto, T.; Furuhata, Y.; Sakai, R.; Mori, M.; Takahashi, M.; Kimura, T. Network analysis of plasma and tissue amino acids and the generation of an amino index for potential diagnostic use. Am. J. Clin. Nutr. 2006, 83, 513S–519S. [Google Scholar] [PubMed]
- Zeanandin, G.; Balage, M.; Schneider, S.M.; Dupont, J.; Hebuterne, X.; Mothe-Satney, I.; Dardevet, D. Differential effect of long-term leucine supplementation on skeletal muscle and adipose tissue in old rats: An insulin signaling pathway approach. Age (Dordr.) 2012, 34, 371–387. [Google Scholar] [CrossRef] [PubMed]
- Ohira, T.; Kawano, F.; Shibaguchi, T.; Okabe, H.; Goto, K.; Ogita, F.; Sudoh, M.; Roy, R.R.; Edgerton, V.R.; Cancedda, R.; et al. Effects of gravitational loading levels on protein expression related to metabolic and/or morphologic properties of mouse neck muscles. Physiol. Rep. 2014, 2, e00183. [Google Scholar] [CrossRef] [PubMed]
- Adams, G.R.; Haddad, F. The relationships among IGF-1, DNA content, and protein accumulation during skeletal muscle hypertrophy. J. Appl. Physiol. 1996, 81, 2509–2516. [Google Scholar] [PubMed]
- Martone, A.M.; Lattanzio, F.; Abbatecola, A.M.; Carpia, D.L.; Tosato, M.; Marzetti, E.; Calvani, R.; Onder, G.; Landi, F. Treating sarcopenia in older and oldest old. Curr. Pharm. Des. 2015, 21, 1715–1722. [Google Scholar] [CrossRef] [PubMed]
- Howarth, K.R.; Burgomaster, K.A.; Phillips, S.M.; Gibala, M.J. Exercise training increases branched-chain oxoacid dehydrogenase kinase content in human skeletal muscle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 293, R1335–R1341. [Google Scholar] [CrossRef] [PubMed]
- She, P.; Zhou, Y.; Zhang, Z.; Griffin, K.; Gowda, K.; Lynch, C.J. Disruption of BCAA metabolism in mice impairs exercise metabolism and endurance. J. Appl. Physiol. 2010, 108, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Melnik, B.C. Leucine signaling in the pathogenesis of type 2 diabetes and obesity. World J. Diabetes 2012, 3, 38–53. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Liang, X.; Shan, T.; Jiang, Q.; Deng, C.; Zheng, R.; Kuang, S. mTOR is necessary for proper satellite cell activity and skeletal muscle regeneration. Biochem. Biophys. Res. Commun. 2015, 463, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Konopka, A.R.; Harber, M.P. Skeletal muscle hypertrophy after aerobic exercise training. Exerc. Sport Sci. Rev. 2014, 42, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Yoon, M.S.; Chen, J. Distinct amino acid-sensing mTOR pathways regulate skeletal myogenesis. Mol. Biol. Cell 2013, 24, 3754–3763. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, M.G.; Hamilton, D.L.; Murray, J.T.; Taylor, P.M.; Baar, K. mVps34 is activated following high-resistance contractions. J. Physiol. 2009, 587, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, M.G.; Hamilton, D.L.; Murray, J.T.; Baar, K. mVps34 is activated by an acute bout of resistance exercise. Biochem. Soc. Trans. 2007, 35, 1314–1316. [Google Scholar] [CrossRef] [PubMed]
- Torgan, C.E.; Daniels, M.P. Regulation of myosin heavy chain expression during rat skeletal muscle development in vitro. Mol. Biol. Cell 2001, 12, 1499–1508. [Google Scholar] [CrossRef] [PubMed]
- Steinacker, J.M.; Opitz-Gress, A.; Baur, S.; Lormes, W.; Bolkart, K.; Sunder-Plassmann, L.; Liewald, F.; Lehmann, M.; Liu, Y. Expression of myosin heavy chain isoforms in skeletal muscle of patients with peripheral arterial occlusive disease. J. Vasc. Surg. 2000, 31, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Short, K.R.; Vittone, J.L.; Bigelow, M.L.; Proctor, D.N.; Coenen-Schimke, J.M.; Rys, P.; Nair, K.S. Changes in myosin heavy chain mRNA and protein expression in human skeletal muscle with age and endurance exercise training. J. Appl. Physiol. 2005, 99, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.S.; Cheung, W.H.; Qin, L.; Tang, N.; Leung, K.S. Age-associated decrease of type IIa/b human skeletal muscle fibers. Clin. Orthop. Relat. Res. 2006, 450, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Paul, P.K.; Kumar, A. TRAF6 coordinates the activation of autophagy and ubiquitin-proteasome systems in atrophying skeletal muscle. Autophagy 2011, 7, 555–556. [Google Scholar] [CrossRef] [PubMed]
- Gumucio, J.P.; Mendias, C.L. Atrogin-1, MuRF-1, and sarcopenia. Endocrine 2013, 43, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Lecker, S.H.; Goldberg, A.L.; Mitch, W.E. Protein degradation by the ubiquitin-proteasome pathway in normal and disease states. J. Am. Soc. Nephrol. 2006, 17, 1807–1819. [Google Scholar] [CrossRef] [PubMed]
- Suryawan, A.; Davis, T.A. Regulation of protein degradation pathways by amino acids and insulin in skeletal muscle of neonatal pigs. J. Anim. Sci. Biotechnol. 2014, 5, 8. [Google Scholar] [CrossRef] [PubMed]
- Bodine, S.C.; Baehr, L.M. Skeletal muscle atrophy and the E3 ubiquitin ligases MuRF1 and MAFbx/atrogin-1. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E469–E484. [Google Scholar] [CrossRef] [PubMed]
- Foletta, V.C.; White, L.J.; Larsen, A.E.; Leger, B.; Russell, A.P. The role and regulation of MAFbx/atrogin-1 and MuRF1 in skeletal muscle atrophy. Pflugers. Archiv. 2011, 461, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Zanchi, N.E.; de Siqueira Filho, M.A.; Lira, F.S.; Rosa, J.C.; Yamashita, A.S.; de Oliveira Carvalho, C.R.; Seelaender, M.; Lancha, A.H., Jr. Chronic resistance training decreases MuRF-1 and Atrogin-1 gene expression but does not modify Akt, GSK-3beta and p70S6K levels in rats. Eur. J. Appl. Physiol. 2009, 106, 415–423. [Google Scholar] [CrossRef] [PubMed]
| Feed Composition | Basal Diet | Leucine Diet | Alanine Diet |
|---|---|---|---|
| Corn | 22.8 | 17.8 | 19.4 |
| Wheat | 34.0 | 34.0 | 34.0 |
| Wheat bran | 10.0 | 10.0 | 10.0 |
| Soybean | 13.0 | 13.0 | 13.0 |
| Soya bean meal | 5.0 | 5.0 | 5.0 |
| Rice bran | 4.0 | 4.0 | 4.0 |
| Fish meal | 7.0 | 7.0 | 7.0 |
| Calcium hydrogen phosphate I | 2.4 | 2.4 | 2.4 |
| Calcium carbonate | 0.6 | 0.6 | 0.6 |
| Additives and Microelements | 1.2 | 1.2 | 1.2 |
| Leucine | ~ | 5.0 | ~ |
| Alanine | ~ | ~ | 3.4 |
| Amino Acid Composition | Basal Diet | Leucine Diet | Alanine Diet |
|---|---|---|---|
| Aspartic acid | 1.81 | 1.72 | 1.72 |
| Threonine | 0.71 | 0.70 | 0.71 |
| Serine | 0.97 | 0.94 | 0.94 |
| Glutamic acid | 4.06 | 4.05 | 4.01 |
| Glycine | 0.97 | 0.92 | 0.93 |
| Alanine | 1.11 | 1.04 | 4.83 |
| Cystine | 0.11 | 0.20 | 0.17 |
| Valine | 0.78 | 0.79 | 0.82 |
| Methionine | 0.45 | 0.54 | 0.50 |
| Isoleucine | 0.64 | 0.62 | 0.62 |
| Leucine | 1.45 | 6.51 | 1.42 |
| Tyrosine | 0.39 | 0.47 | 0.46 |
| Phenylalanine | 0.79 | 0.86 | 0.86 |
| Lysine | 1.17 | 1.10 | 1.15 |
| Histidine | 0.69 | 0.62 | 0.71 |
| Argnine | 1.13 | 1.06 | 1.04 |
| Proline | 1.39 | 1.30 | 1.38 |
| AlaC (n = 8) | LeuC (n = 7) | AlaE (n = 10) | LeuE (n = 8) | |
|---|---|---|---|---|
| Essential amino acids | ||||
| Leucine | 127.3 ± 7.0 | 158.8 ± 5.5 b | 148.0 ± 9.2 b,c | 168.5 ± 11.1 b,f |
| Isoleucine | 74.6 ± 2.7 | 73.2 ± 3.2 | 78.7 ± 2.7 | 77.7 ± 6.0 |
| Valine | 281.0 ± 9.2 | 268.2 ± 30.7 | 279.7 ± 2.6 | 269.2 ± 16.4 |
| Arginine | 71.5 ± 9.2 | 69.9 ± 5.6 | 78.1 ± 4.3 | 76.5 ± 2.5 |
| Histidine | 125.1 ± 6.6 | 126.8 ± 8.4 | 132.1 ± 3.4 | 132.4 ± 4.5 |
| Lysine | 269.2 ± 6.4 | 270.6 ± 12.0 | 277.1 ± 14.3 | 282.5 ± 12.5 |
| Methionine | 50.7 ± 8.5 | 54.1 ± 1.0 | 56.1 ± 9.3 | 59.1 ± 9.2 |
| Phenylalanine | 252.2 ± 14.8 | 261.5 ± 16.1 | 266.4 ± 12.8 | 268.0 ± 7.0 |
| Threonine | 140.9 ± 7.1 | 141.2 ± 12.0 | 148.9 ± 11.4 | 152.6 ± 4.9 |
| Non-essential amino acids | ||||
| Alanine | 306.3 ± 33.1 | 256.0 ± 23.3 b | 415.7 ± 31.9 b,d | 378.2 ± 21.1 b,d,e |
| Aspartic acid | 147.7 ± 13.5 | 151.4 ± 11.8 | 148.4 ± 6.6 | 158.3 ± 14.3 |
| Cystine | 47.5 ± 5.3 | 47.8 ± 9.3 | 51.2 ± 2.4 | 53.0 ± 5.3 |
| Glutamic acid | 465.2 ± 28.5 | 390.8 ± 20.2 b | 549.3 ± 38.0 b,d | 406.1 ± 26.4 b,f |
| Glycine | 380.6 ± 25.4 | 349.3 ± 13.8 a | 442.0 ± 24.5 b,d | 365.7 ± 15.7 f |
| Proline | 103.3 ± 3.6 | 107.5 ± 6.1 | 100.2 ± 4.9 | 107.1 ± 8.1 |
| Serine | 202.2 ± 13.2 | 198.9 ± 7.5 | 210.6 ± 9.1 | 210.4 ± 11.2 |
| Tyrosine | 94.1 ± 5.7 | 89.4 ± 6.8 | 148.5 ± 8.8 b,d | 120.5 ± 10.8 b,d,f |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xia, Z.; Cholewa, J.; Zhao, Y.; Yang, Y.-Q.; Shang, H.-Y.; Guimarães-Ferreira, L.; Naimo, M.A.; Su, Q.-S.; Zanchi, N.E. Hypertrophy-Promoting Effects of Leucine Supplementation and Moderate Intensity Aerobic Exercise in Pre-Senescent Mice. Nutrients 2016, 8, 246. https://doi.org/10.3390/nu8050246
Xia Z, Cholewa J, Zhao Y, Yang Y-Q, Shang H-Y, Guimarães-Ferreira L, Naimo MA, Su Q-S, Zanchi NE. Hypertrophy-Promoting Effects of Leucine Supplementation and Moderate Intensity Aerobic Exercise in Pre-Senescent Mice. Nutrients. 2016; 8(5):246. https://doi.org/10.3390/nu8050246
Chicago/Turabian StyleXia, Zhi, Jason Cholewa, Yan Zhao, Yue-Qin Yang, Hua-Yu Shang, Lucas Guimarães-Ferreira, Marshall Alan Naimo, Quan-Sheng Su, and Nelo Eidy Zanchi. 2016. "Hypertrophy-Promoting Effects of Leucine Supplementation and Moderate Intensity Aerobic Exercise in Pre-Senescent Mice" Nutrients 8, no. 5: 246. https://doi.org/10.3390/nu8050246
APA StyleXia, Z., Cholewa, J., Zhao, Y., Yang, Y.-Q., Shang, H.-Y., Guimarães-Ferreira, L., Naimo, M. A., Su, Q.-S., & Zanchi, N. E. (2016). Hypertrophy-Promoting Effects of Leucine Supplementation and Moderate Intensity Aerobic Exercise in Pre-Senescent Mice. Nutrients, 8(5), 246. https://doi.org/10.3390/nu8050246






