A Healthy Dietary Pattern Reduces Lung Cancer Risk: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Selection Criteria
2.3. Data Extraction and Quality Assessment
2.4. Statistical Methods
3. Results
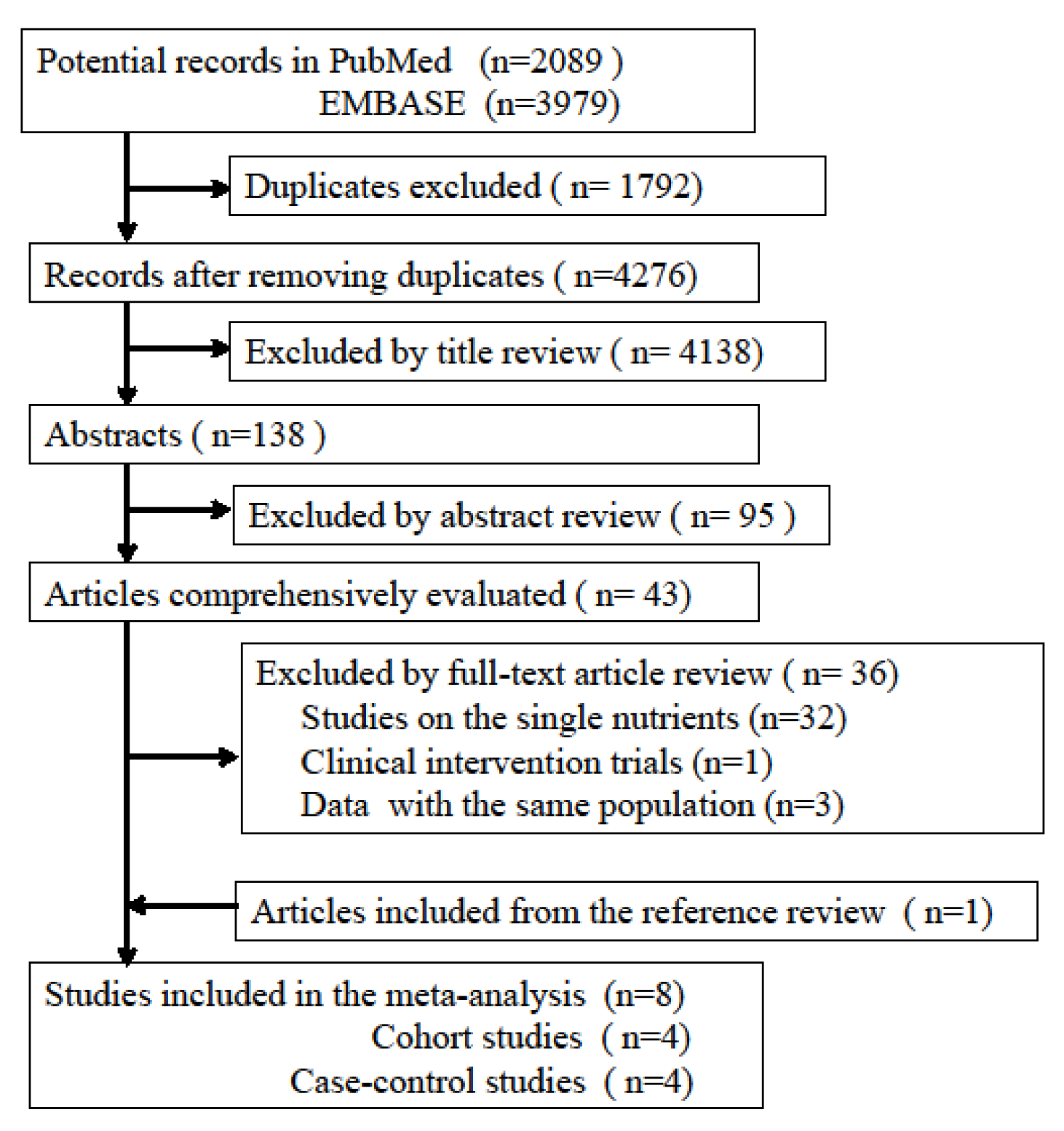
3.1. Literature Search and Study Characteristics
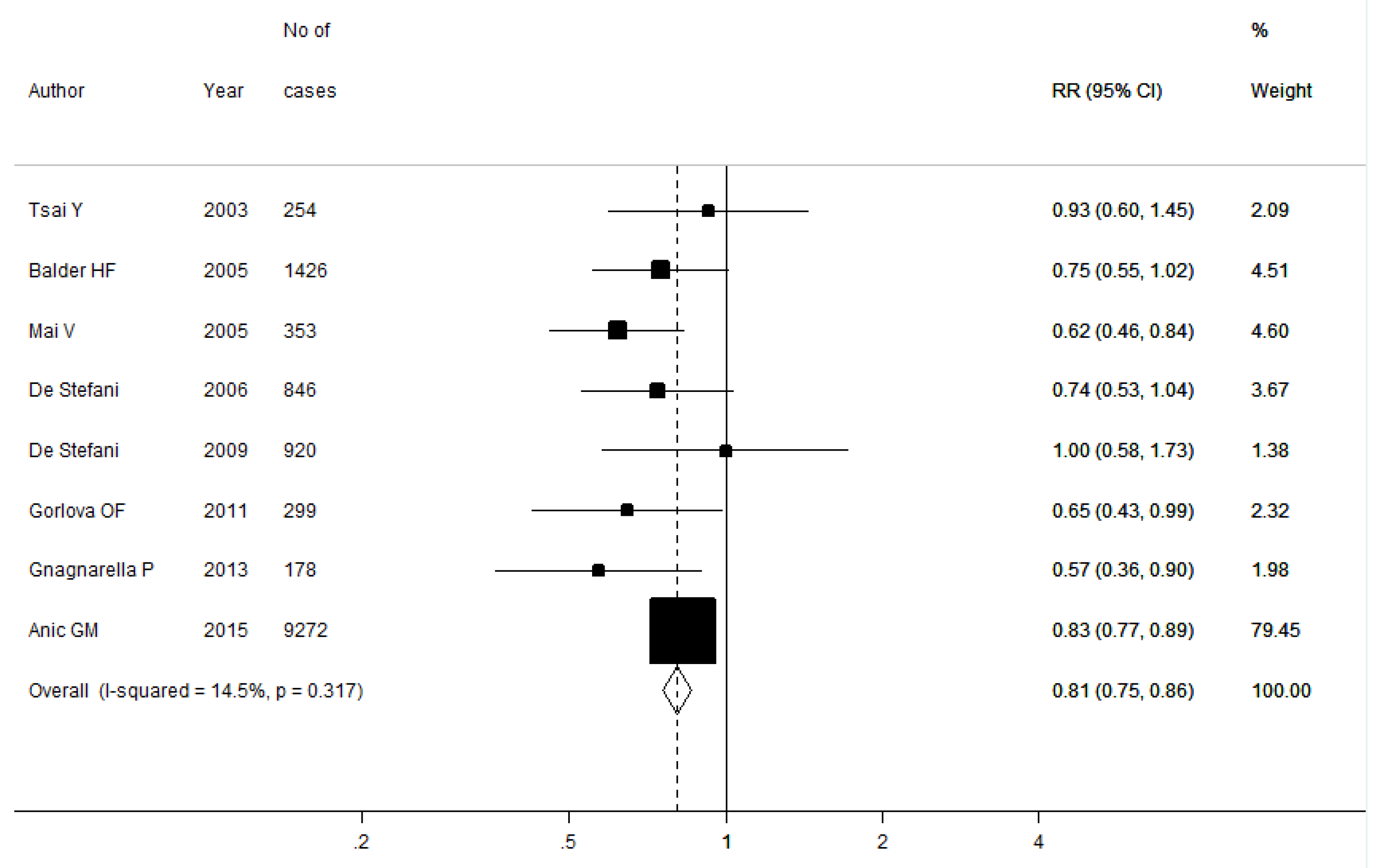
3.2. Meta-Analysis
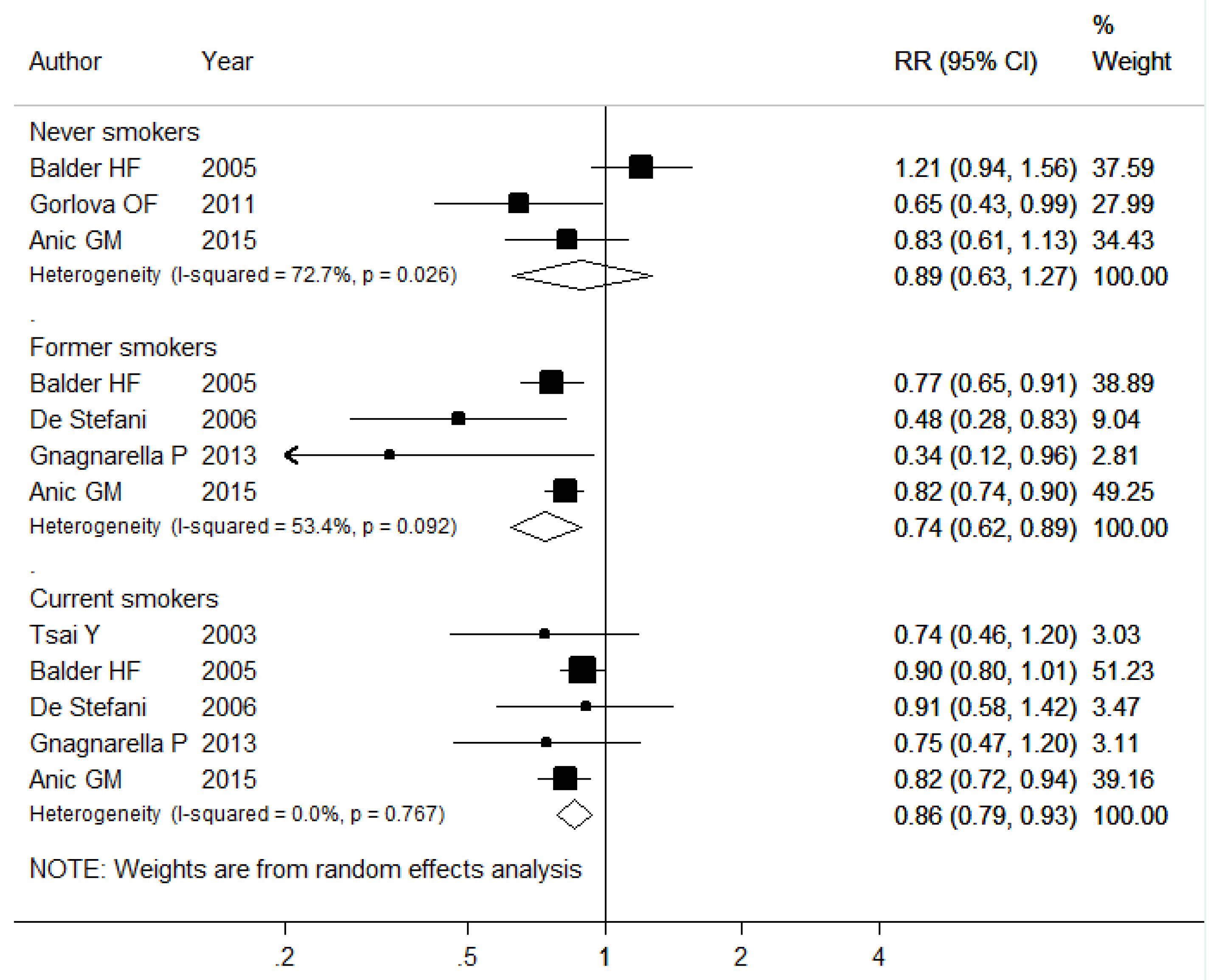
3.3. Subgroup Analyses
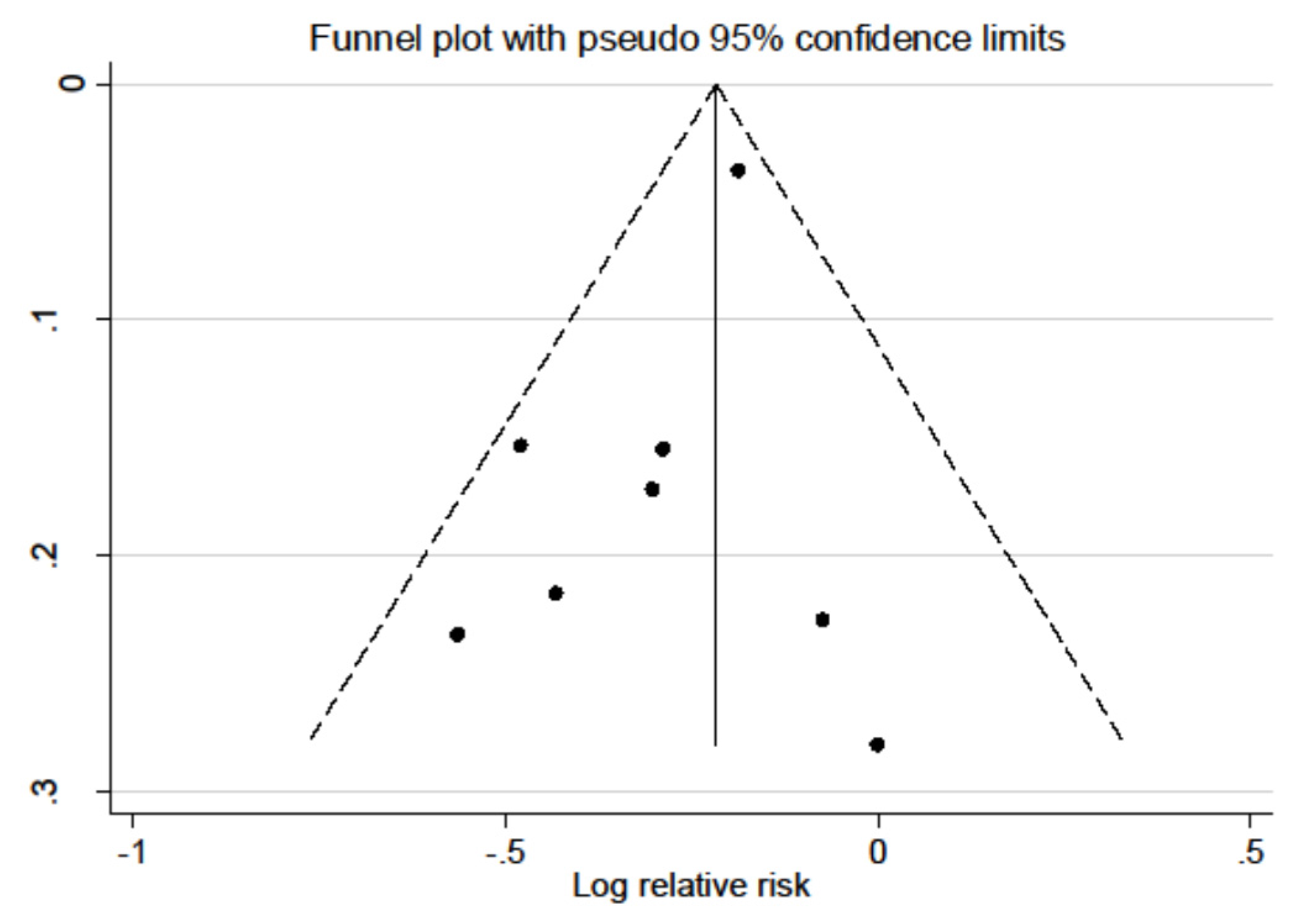
3.4. Sensitivity Analysis and Publication Bias
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Siegel, R.; Ma, J.; Zou, Z.; Jemal, A. Cancer statistics, 2014. CA Cancer J. Clin. 2014, 64, 9–29. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Center, M.M.; DeSantis, C.; Ward, E.M. Global patterns of cancer incidence and mortality rates and trends. Cancer Epidemiol. Biomark. Prev. 2010, 19, 1893–1907. [Google Scholar] [CrossRef] [PubMed]
- Alberg, A.J.; Brock, M.V.; Ford, J.G.; Samet, J.M.; Spivack, S.D. Epidemiology of lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013, 143, e1S–e29S. [Google Scholar] [CrossRef] [PubMed]
- DeSantis, C.E.; Lin, C.C.; Mariotto, A.B.; Siegel, R.L.; Stein, K.D.; Kramer, J.L.; Alteri, R.; Robbins, A.S.; Jemal, A. Cancer treatment and survivorship statistics, 2014. CA Cancer J. Clin. 2014, 64, 252–271. [Google Scholar] [CrossRef] [PubMed]
- Garraway, L.A.; Verweij, J.; Ballman, K.V. Precision oncology: An overview. J. Clin. Oncol. 2013, 31, 1803–1805. [Google Scholar] [CrossRef] [PubMed]
- Kushi, L.H.; Doyle, C.; McCullough, M.; Rock, C.L.; Demark-Wahnefried, W.; Bandera, E.V.; Gapstur, S.; Patel, A.V.; Andrews, K.; Gansler, T. American Cancer Society Guidelines on nutrition and physical activity for cancer prevention: Reducing the risk of cancer with healthy food choices and physical activity. CA Cancer J. Clin. 2012, 62, 30–67. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, F.; Wang, Z.; Qiu, T.; Shen, Y.; Wang, M. Fruit and vegetable consumption and risk of lung cancer: A dose-response meta-analysis of prospective cohort studies. Lung Cancer 2015, 88, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Su, H.; Wang, B.L.; Zhou, Y.Y.; Guo, L.L. Fish consumption and lung cancer risk: Systematic review and meta-analysis. Nutr. Cancer 2014, 66, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.S.; Va, P.; Wong, M.Y.; Zhang, H.L.; Xiang, Y.B. Soy intake is associated with lower lung cancer risk: Results from a meta-analysis of epidemiologic studies. Am. J. Clin. Nutr. 2011, 94, 1575–1583. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.S.; Wong, M.Y.; Vogtmann, E.; Tang, R.Q.; Xie, L.; Yang, Y.S.; Wu, Q.J.; Zhang, W.; Xiang, Y.B. Meat consumption and risk of lung cancer: Evidence from observational studies. Ann. Oncol. 2012, 23, 3163–3170. [Google Scholar] [CrossRef] [PubMed]
- Gnagnarella, P.; Maisonneuve, P.; Bellomi, M.; Rampinelli, C.; Bertolotti, R.; Spaggiari, L.; Palli, D.; Veronesi, G. Red meat, Mediterranean diet and lung cancer risk among heavy smokers in the COSMOS screening study. Ann. Oncol. 2013, 24, 2606–2611. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.; Hunter, D.J.; Spiegelman, D.; Albanes, D.; Beeson, W.L.; van den Brandt, P.A.; Colditz, G.A.; Feskanich, D.; Folsom, A.R.; Fraser, G.E.; et al. Intakes of vitamins A, C and E and folate and multivitamins and lung cancer: A pooled analysis of 8 prospective studies. Int. J. Cancer 2006, 118, 970–978. [Google Scholar] [CrossRef]
- Gallicchio, L.; Boyd, K.; Matanoski, G.; Tao, X.G.; Chen, L.; Lam, T.K.; Shiels, M.; Hammond, E.; Robinson, K.A.; Caulfield, L.E.; et al. Carotenoids and the risk of developing lung cancer: A systematic review. Am. J. Clin. Nutr. 2008, 88, 372–383. [Google Scholar] [PubMed]
- The Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. N. Engl. J. Med. 1994, 330, 1029–1035. [Google Scholar]
- Fortmann, S.P.; Whitlock, E.P.; Burda, B.U. Vitamin and mineral supplements in the primary prevention of cardiovascular disease and cancer. Ann. Intern. Med. 2014, 160, 656. [Google Scholar] [CrossRef] [PubMed]
- Tucker, K.L. Dietary patterns, approaches, and multicultural perspective. Appl. Physiol. Nutr. Metab. 2010, 35, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Reedy, J.; Wirfalt, E.; Flood, A.; Mitrou, P.N.; Krebs-Smith, S.M.; Kipnis, V.; Midthune, D.; Leitzmann, M.; Hollenbeck, A.; Schatzkin, A.; et al. Comparing 3 dietary pattern methods-cluster analysis, factor analysis, and index analysis--With colorectal cancer risk: The NIH-AARP Diet and Health Study. Am. J. Epidemiol. 2010, 171, 479–487. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, A.; Gibney, M.J.; Brennan, L. Dietary intake patterns are reflected in metabolomic profiles: Potential role in dietary assessment studies. Am. J. Clin. Nutr. 2011, 93, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Heidemann, C.; Schulze, M.B.; Franco, O.H.; van Dam, R.M.; Mantzoros, C.S.; Hu, F.B. Dietary patterns and risk of mortality from cardiovascular disease, cancer, and all causes in a prospective cohort of women. Circulation 2008, 118, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Bertuccio, P.; Rosato, V.; Andreano, A.; Ferraroni, M.; Decarli, A.; Edefonti, V.; La Vecchia, C. Dietary patterns and gastric cancer risk: A systematic review and meta-analysis. Ann. Oncol. 2013, 24, 1450–1458. [Google Scholar] [CrossRef] [PubMed]
- Brennan, S.F.; Cantwell, M.M.; Cardwell, C.R.; Velentzis, L.S.; Woodside, J.V. Dietary patterns and breast cancer risk: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2010, 91, 1294–1302. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, X.; Lin, S.; Yuan, J.; Yu, I.T. Dietary patterns and oesophageal squamous cell carcinoma: A systematic review and meta-analysis. Br. J. Cancer 2014, 110, 2785–2795. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.Y.; McGlynn, K.A.; Hu, Y.; Cassidy, A.B.; Arnold, J.; Engstrom, P.F.; Buetow, K.H. Genetic susceptibility and dietary patterns in lung cancer. Lung Cancer 2003, 41, 269–281. [Google Scholar] [CrossRef]
- Mai, V.; Kant, A.K.; Flood, A.; Lacey, J.V., Jr.; Schairer, C.; Schatzkin, A. Diet quality and subsequent cancer incidence and mortality in a prospective cohort of women. Int. J. Epidemiol. 2005, 34, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Gnagnarella, P.; Maisonneuve, P.; Bellomi, M.; Rampinelli, C.; Bertolotti, R.; Spaggiari, L.; Palli, D.; Veronesi, G. Nutrient intake and nutrient patterns and risk of lung cancer among heavy smokers: Results from the COSMOS screening study with annual low-dose CT. Eur. J. Epidemiol. 2013, 28, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Gorlova, O.Y.; Weng, S.F.; Hernandez, L.; Spitz, M.R.; Forman, M.R. Dietary patterns affect lung cancer risk in never smokers. Nutr. Cancer 2011, 63, 842–849. [Google Scholar] [CrossRef] [PubMed]
- Anic, G.M.; Park, Y.; Subar, A.F.; Schap, T.E.; Reedy, J. Index-based dietary patterns and risk of lung cancer in the NIH-AARP diet and health study. Eur. J. Clin. Nutr. 2015. [Google Scholar] [CrossRef] [PubMed]
- Balder, H.F.; Goldbohm, R.A.; van den Brandt, P.A. Dietary patterns associated with male lung cancer risk in the Netherlands Cohort Study. Cancer Epidemiol. Biomark. Prev. 2005, 14, 483–490. [Google Scholar] [CrossRef] [PubMed]
- De Stefani, E.; Ronco, A.L.; Boffetta, P.; Deneo-Pellegrini, H.; Correa, P.; Acosta, G.; Gutiérrez, L.P.; Mendilaharsu, M. Nutritional patterns and lung cancer risk in Uruguayan men. Cancer Ther. 2006, 4, 153–162. [Google Scholar]
- De Stefani, E.; Deneo-Pellegrini, H.; Boffetta, P.; Ronco, A.L.; Aune, D.; Acosta, G.; Mendilaharsu, M.; Brennan, P.; Ferro, G. Dietary patterns and risk of cancer: A factor analysis in Uruguay. Int. J. Cancer 2009, 124, 1391–1397. [Google Scholar] [CrossRef] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 1 October 2015).
- De Stefani, E.; Boffetta, P.; Ronco, A.L.; Deneo-Pellegrini, H.; Acosta, G.; Gutierrez, L.P.; Mendilaharsu, M. Nutrient patterns and risk of lung cancer: A factor analysis in Uruguayan men. Lung Cancer 2008, 61, 283–291. [Google Scholar] [CrossRef] [PubMed]
- De Stefani, E.; Ronco, A.L.; Deneo-Pellegrini, H.; Correa, P.; Boffetta, P.; Acosta, G.; Mendilaharsu, M. Dietary patterns and risk of adenocarcinoma of the lung in males: A factor analysis in Uruguay. Nutr. Cancer 2011, 63, 699–706. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, B.; Peleteiro, B.; Lunet, N. Dietary patterns and colorectal cancer: Systematic review and meta-analysis. Eur. J. Cancer Prev. 2012, 21, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Shu, L.; Wang, X.Q.; Wang, S.F.; Wang, S.; Mu, M.; Zhao, Y.; Sheng, J.; Tao, F.B. Dietary patterns and stomach cancer: A meta-analysis. Nutr. Cancer 2013, 65, 1105–1115. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.J.; Xie, L.; Zheng, W.; Vogtmann, E.; Li, H.L.; Yang, G.; Ji, B.T.; Gao, Y.T.; Shu, X.O.; Xiang, Y.B. Cruciferous vegetables consumption and the risk of female lung cancer: A prospective study and a meta-analysis. Ann. Oncol. 2013, 24, 1918–1924. [Google Scholar] [CrossRef] [PubMed]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R.; et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Louis, P.; Hold, G.L.; Flint, H.J. The gut microbiota, bacterial metabolites and colorectal cancer. Nat. Rev. Microbiol. 2014, 12, 661–672. [Google Scholar] [CrossRef] [PubMed]
| Author Year | Duration | Design | Location | Study Population | Sex | No. of Case | Cohort Size/Control | Dietary Assessment | Dietary Patterns Identification Method | Dietary Pattern Characteristics | Dietary Patterns Identified | RR (Highest to Lowest) | Adjusted Variables | Quality |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tsai Y., 2003 [24] | 1995–1996 | Case-control | USA | Patients seen at Fox Chase Cancer Center Network | Both | 254 | 184 | FFQ (food frequency questionnaire)-61 items, validated, self-reported | Posteriori, cluster analysis | A relatively greater intake of carbohydrates and dietary fiber and a lower intake of animal fat and protein. | Healthy pattern | 0.93 (0.59–1.44) | Gender, age, and smoking | 7 |
| Balder H.F., 2005 [30] | 1986–1995 | Cohort | Europe | The Netherlands Cohort Study on Diet and Cancer | Male | 1426 | 58,279 | FFQ-150 items, validated, interviewed | Posteriori, principal components analysis | High factor loadings on several vegetable items, several fruit items, pasta, rice, poultry, fish, and oil | Salad vegetables | 0.75 (0.55–1.01) | All other dietary patterns and age at baseline, total energy intake, current cigarette smoker, number of cigarettes smoked per day, years of smoking cigarettes, higher vocational or university education, family history of lung cancer, physical activity | 9 |
| Mai V., 2005 [32] | 1987–1998 | Cohort | USA | The Breast Cancer Detection Demonstration Project cohort | Female | 353 | 42,254 | FFQ-62 items, validated, mailed | Priori | Increasing consumption of fruits, vegetables, whole grains, lean meats or meat alternatives, and low-fat dairy. | High RFS (the Recommended Foods Score) dietary pattern | 0.62 (0.46–0.84) | Energy intake, smoking, NSAID (nonsteroidal anti-inflammatory drug) use, and BMI (body mass index), smoking duration and cigarettes/day | 8 |
| De Stefani, 2006 [28] | 1995–2001 | Case-control | Uruguay | Patients from four major hospitals located in Montevideo, Uruguay | Male | 846 | 846 | FFQ-64 items, unvalidated, interviewed | Posteriori, principal component analysis | High correlations of white meat, fresh vegetables, cooked vegetables, citrus fruits and non-citrus fruits | Healthy pattern | 0.74 (0.53–1.04) | Age, residence, urban/rural status, education, family history of lung cancer among first-degree relatives, body mass index, cigarettes per day, years since quit and total energy intake. | 7 |
| De Stefani, 2009 [29] | 1996–2004 | Case-control | Uruguay | Patients from four major hospitals in Montevideo, Uruguay | Both | 920 | 2532 | FFQ-64 items, unvalidated, interviewed | Posteriori, principal component analysis | High positive loadings for poultry, fish, fresh vegetables, and total fruits. | Prudent pattern | 1.00 (0.58–1.74) | Age, residence, urban/rural status, education, body mass index, smoking status, years since stopping, number of cigarettes/day, among current smokers, total energy intake and all the dietary patterns | 7 |
| Gorlova O.F., 2011 [27] | 1995–2008 | Case-control | USA | Patients in MD Anderson Cancer Center | Both | 299 | 317 | FFQ-214 items, unvalidated, interviewed | Posteriori, a principal component-based factor analysis | High intake vegetables, fruits, and low fat products. | Healthy pattern | 0.65 (0.42–0.98) | Age, gender, caloric intake, and education | 6 |
| Gnagnarella P., 2013 [31] | 2004–2010 | Cohort | Italy | The COSMOS screening study, current smokers or former smokers | Both | 178 | 4336 | FFQ-188 items, validated, self-administered | Posteriori, principal component analysis | High intake of vegetables, fruits, nuts, cereals, legumes and fish; low consumption in red and processed meat | Vitamins and fiber | 0.57 (0.36–0.90) | Age, sex, smoking history, asbestos exposure and total energy | 7 |
| Anic G.M., 2015 [26] | 1995–2006 | Cohort | USA | The NIH–AARP Diet and Health Study | Both | 9272 | 460 770 | FFQ-124 items, validated, mailed | Apriori, HEI(healthy eating index)-2010 score | High intake of total vegetables, greens and beans, total fruits, whole fruits, seafood, whole grains and low-fat dairy | Healthy eating pattern | 0.83 (0.77–0.89) | Age, sex, race, education, body mass index, physical activity, total energy, smoking status, cigarettes per day, time since quitting smoking and regular use of cigars/pipes | 8 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Y.; Li, Z.; Li, J.; Li, Z.; Han, J. A Healthy Dietary Pattern Reduces Lung Cancer Risk: A Systematic Review and Meta-Analysis. Nutrients 2016, 8, 134. https://doi.org/10.3390/nu8030134
Sun Y, Li Z, Li J, Li Z, Han J. A Healthy Dietary Pattern Reduces Lung Cancer Risk: A Systematic Review and Meta-Analysis. Nutrients. 2016; 8(3):134. https://doi.org/10.3390/nu8030134
Chicago/Turabian StyleSun, Yanlai, Zhenxiang Li, Jianning Li, Zengjun Li, and Jianjun Han. 2016. "A Healthy Dietary Pattern Reduces Lung Cancer Risk: A Systematic Review and Meta-Analysis" Nutrients 8, no. 3: 134. https://doi.org/10.3390/nu8030134
APA StyleSun, Y., Li, Z., Li, J., Li, Z., & Han, J. (2016). A Healthy Dietary Pattern Reduces Lung Cancer Risk: A Systematic Review and Meta-Analysis. Nutrients, 8(3), 134. https://doi.org/10.3390/nu8030134




