Association between Dietary Intakes of Nitrate and Nitrite and the Risk of Hypertension and Chronic Kidney Disease: Tehran Lipid and Glucose Study
Abstract
:1. Introduction
2. Methods
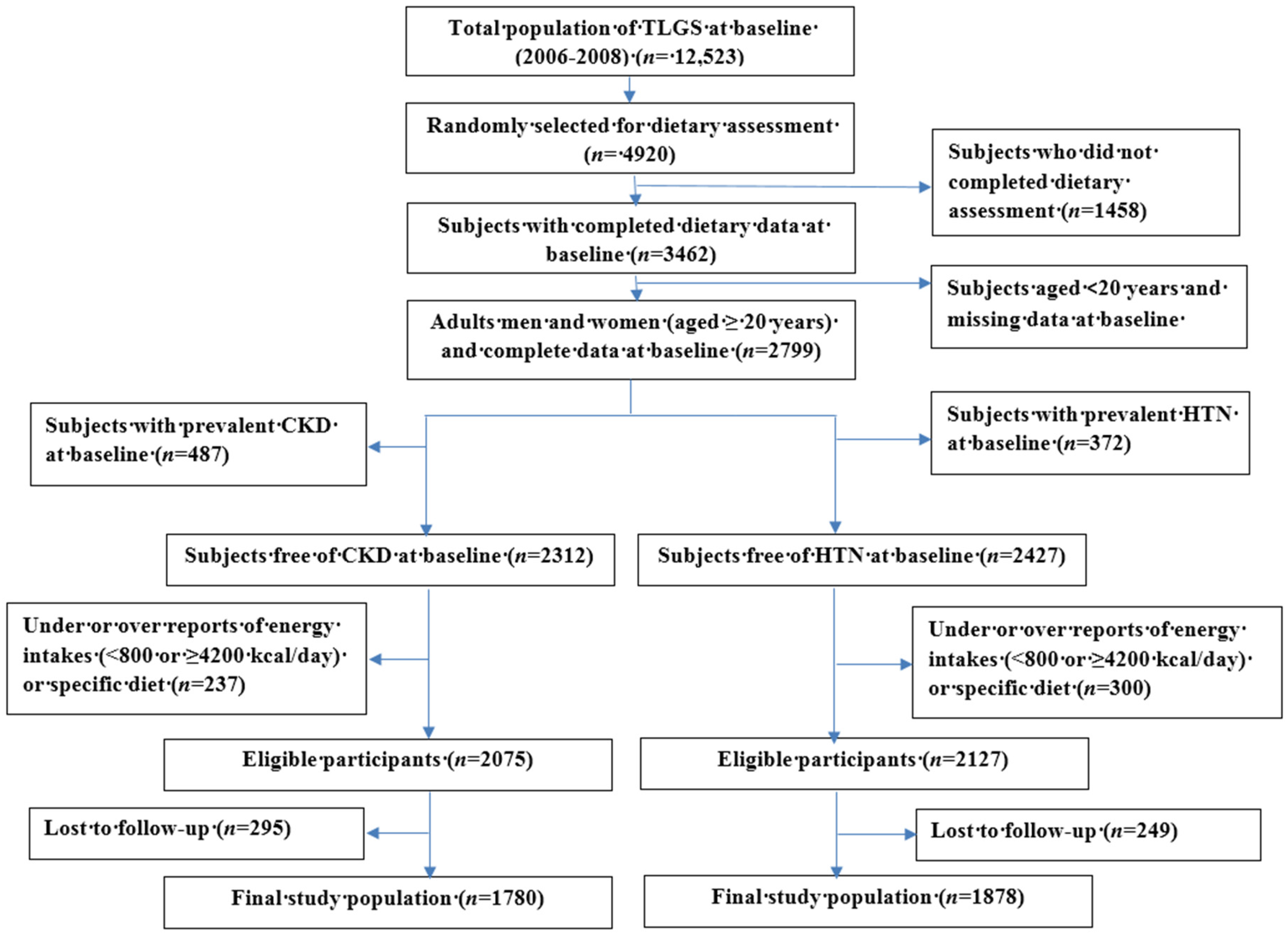
2.1. Study Population
2.2. Demographic, Anthropometric and Clinical Measures
2.3. Biochemical Measures
2.4. Dietary Assessment
2.5. Validity of NO3− and NO2− Estimation by FFQ
2.6. Definition of Terms and Outcomes
2.7. Statistical Analyses
3. Results
4. Discussion
5. Perspectives
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of interest
References
- Weiner, D.E.; Tighiouart, H.; Amin, M.G.; Stark, P.C.; MacLeod, B.; Griffith, J.L.; Salem, D.N.; Levey, A.S.; Sarnak, M.J. Chronic Kidney Disease as a Risk Factor for Cardiovascular Disease and All-Cause Mortality: A Pooled Analysis of Community-Based Studies. J. Am. Soc. Nephrol. 2004, 15, 1307–1315. [Google Scholar] [CrossRef] [PubMed]
- Lackland, D.T.; Weber, M.A. Global burden of cardiovascular disease and stroke: Hypertension at the core. Can. J. Cardiol. 2015, 31, 569–571. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Youn, J.Y.; Cai, H. Mechanisms and consequences of endothelial nitric oxide synthase dysfunction in hypertension. J. Hypertens. 2015, 33, 1128–1136. [Google Scholar] [CrossRef] [PubMed]
- Baylis, C. Nitric oxide deficiency in chronic kidney disease. Am. J. Physiol. Ren. Physiol. 2008, 294, F1–F9. [Google Scholar] [CrossRef] [PubMed]
- Korish, A.A. Oxidative stress and nitric oxide deficiency in inflammation of chronic renal failure. Possible preventive role of l-arginine and multiple antioxidants. Saudi Med. J. 2009, 30, 1150–1157. [Google Scholar] [PubMed]
- Klahr, S. The role of nitric oxide in hypertension and renal disease progression. Nephrol. Dial. Transp. 2001, 16 (Suppl. S1), 60–62. [Google Scholar] [CrossRef]
- Zatz, R.; Baylis, C. Chronic nitric oxide inhibition model six years on. Hypertension 1998, 32, 958–964. [Google Scholar] [CrossRef] [PubMed]
- Carlstrom, M.; Wilcox, C.S.; Arendshorst, W.J. Renal autoregulation in health and disease. Physiol. Rev. 2015, 95, 405–511. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, M.; Winyard, P.G.; Benjamin, N. Dietary nitrate—Good or bad? Nitric Oxide 2010, 22, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Bahadoran, Z.; Mirmiran, P.; Ghasemi, A.; Kabir, A.; Azizi, F.; Hadaegh, F. Is dietary nitrate/nitrite exposure a risk factor for development of thyroid abnormality? A systematic review and meta-analysis. Nitric Oxide 2015, 47, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Kapil, V.; Khambata, R.S.; Robertson, A.; Caulfield, M.J.; Ahluwalia, A. Dietary nitrate provides sustained blood pressure lowering in hypertensive patients: A randomized, phase 2, double-blind, placebo-controlled study. Hypertension 2015, 65, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Bondonno, C.P.; Liu, A.H.; Croft, K.D.; Ward, N.C.; Yang, X.; Considine, M.J.; Puddey, I.B.; Woodman, R.J.; Hodgson, J.M. Short-term effects of nitrate-rich green leafy vegetables on blood pressure and arterial stiffness in individuals with high-normal blood pressure. Free Radic. Biol. Med. 2014, 77, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.H.; Bondonno, C.P.; Croft, K.D.; Puddey, I.B.; Woodman, R.J.; Rich, L.; Ward, N.C.; Vita, J.A.; Hodgson, J.M. Effects of a nitrate-rich meal on arterial stiffness and blood pressure in healthy volunteers. Nitric Oxide 2013, 35, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Rammos, C.; Hendgen-Cotta, U.B.; Pohl, J.; Totzeck, M.; Luedike, P.; Schulze, V.T.; Kelm, M.; Rassaf, T. Modulation of circulating macrophage migration inhibitory factor in the elderly. Biomed. Res. Int. 2014, 2014, 582586. [Google Scholar] [CrossRef] [PubMed]
- Ingram, T.E.; Fraser, A.G.; Bleasdale, R.A.; Ellins, E.A.; Margulescu, A.D.; Halcox, J.P.; James, P.E. Low-dose sodium nitrite attenuates myocardial ischemia and vascular ischemia-reperfusion injury in human models. J. Am. Coll. Cardiol. 2013, 61, 2534–2541. [Google Scholar] [CrossRef] [PubMed]
- Rammos, C.; Hendgen-Cotta, U.B.; Sobierajski, J.; Bernard, A.; Kelm, M.; Rassaf, T. Dietary nitrate reverses vascular dysfunction in older adults with moderately increased cardiovascular risk. J. Am. Coll. Cardiol. 2014, 63, 1584–1585. [Google Scholar] [CrossRef] [PubMed]
- Calvert, J.W.; Lefer, D.J. Clinical translation of nitrite therapy for cardiovascular diseases. Nitric Oxide 2010, 22, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Pattillo, C.B.; Bir, S.; Rajaram, V.; Kevil, C.G. Inorganic nitrite and chronic tissue ischaemia: A novel therapeutic modality for peripheral vascular diseases. Cardiovasc. Res. 2011, 89, 533–541. [Google Scholar] [CrossRef] [PubMed]
- Bahadoran, Z.; Ghasemi, A.; Mirmiran, P.; Azizi, F.; Hadaegh, F. Beneficial effects of inorganic nitrate/nitrite in type 2 diabetes and its complications. Nutr. Metab. 2015, 12, 16. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, A.I.; Wilkerson, D.P.; Dobson, L.; Kelly, J.; Winyard, P.G.; Jones, A.M.; Benjamin, N.; Shore, A.C.; Gilchrist, M. The effect of dietary nitrate supplementation on the oxygen cost of cycling, walking performance and resting blood pressure in individuals with chronic obstructive pulmonary disease: A double blind placebo controlled, randomised control trial. Nitric Oxide 2015, 48, 31–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berry, M.J.; Justus, N.W.; Hauser, J.I.; Case, A.H.; Helms, C.C.; Basu, S.; Rogers, Z.; Lewis, M.T.; Miller, G.D. Dietary nitrate supplementation improves exercise performance and decreases blood pressure in COPD patients. Nitric Oxide 2015, 48, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Bryan, N.S.; Fernandez, B.O.; Bauer, S.M.; Garcia-Saura, M.F.; Milsom, A.B.; Rassaf, T.; Maloney, R.E.; Bharti, A.; Rodriguez, J.; Feelisch, M. Nitrite is a signaling molecule and regulator of gene expression in mammalian tissues. Nat. Chem. Biol. 2005, 1, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Jiang, H.; Bryan, N.S. Nitrite and nitrate: Cardiovascular risk-benefit and metabolic effect. Curr. Opin. Lipidol. 2011, 22, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Carlstrom, M.; Persson, A.E.; Larsson, E.; Hezel, M.; Scheffer, P.G.; Teerlink, T.; Weitzberg, E.; Lundberg, J.O. Dietary nitrate attenuates oxidative stress, prevents cardiac and renal injuries, and reduces blood pressure in salt-induced hypertension. Cardiovasc. Res. 2011, 89, 574–585. [Google Scholar] [CrossRef] [PubMed]
- Hezel, M.; Peleli, M.; Liu, M.; Zollbrecht, C.; Jensen, B.L.; Checa, A.; Giulietti, A.; Wheelock, C.E.; Lundberg, J.O.; Weitzberg, E.; et al. Dietary nitrate improves age-related hypertension and metabolic abnormalities in rats via modulation of angiotensin II receptor signaling and inhibition of superoxide generation. Free Radic. Biol. Med. 2016, 99, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O.; Carlstrom, M.; Larsen, F.J.; Weitzberg, E. Roles of dietary inorganic nitrate in cardiovascular health and disease. Cardiovasc. Res. 2011, 89, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O. Cardiovascular prevention by dietary nitrate and nitrite. Am. J. Physiol. Heart Circ. Physiol. 2009, 296, H1221–H1223. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O.; Weitzberg, E.; Gladwin, M.T. The nitrate–nitrite–nitric oxide pathway in physiology and therapeutics. Nat. Rev. Drug Discov. 2008, 7, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Lidder, S.; Webb, A.J. Vascular effects of dietary nitrate (as found in green leafy vegetables and beetroot) via the nitrate-nitrite-nitric oxide pathway. Br. J. Clin. Pharmacol. 2013, 75, 677–696. [Google Scholar] [CrossRef] [PubMed]
- Golzarand, M.; Bahadoran, Z.; Mirmiran, P.; Zadeh-Vakili, A.; Azizi, F. Consumption of nitrate-containing vegetables is inversely associated with hypertension in adults: A prospective investigation from the Tehran Lipid and Glucose Study. J. Nephrol. 2016, 29, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Nitrate and Nitrite in Drinking-Water; Background Document for Development of WHO Guidelines for Drinking-Water Quality. Available online: http://www.who.int/water_sanitation_health/dwq/chemicals/fluoride.pdf (accessed on 3 December 2015).
- Azizi, F.; Ghanbarian, A.; Momenan, A.A.; Hadaegh, F.; Mirmiran, P.; Hedayati, M.; Mehrabi, Y.; Zahedi-Asl, S. Prevention of non-communicable disease in a population in nutrition transition: Tehran Lipid and Glucose Study phase II. Trials 2009, 10, 5. [Google Scholar] [CrossRef] [PubMed]
- Hosseini-Esfahani, F.; Jessri, M.; Mirmiran, P.; Bastan, S.; Azizi, F. Adherence to dietary recommendations and risk of metabolic syndrome: Tehran Lipid and Glucose Study. Metab. Clin. Exp. 2010, 59, 1833–1842. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, A.H.M.; Biabani, H. Protein precipitation methods evaluated for determination of serum nitric oxide end products by the Griess assay. JMSR 2007, 2, 43–46. [Google Scholar]
- Ghasemi, A.; Zahediasl, S. Preanalytical and analytical considerations for measuring nitric oxide metabolites in serum or plasma using the Griess method. Clin. Lab. 2012, 58, 615–624. [Google Scholar] [PubMed]
- Miranda, K.M.; Espey, M.G.; Wink, D.A. A Rapid, Simple Spectrophotometric Method for Simultaneous Detection of Nitrate and Nitrite. Nitric Oxide 2001, 5, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Mirmiran, P.; Esfahani, F.H.; Mehrabi, Y.; Hedayati, M.; Azizi, F. Reliability and relative validity of an FFQ for nutrients in the Tehran lipid and glucose study. Public Health Nutr. 2010, 13, 654–662. [Google Scholar] [CrossRef] [PubMed]
- Fewtrell, L. Drinking-water nitrate, methemoglobinemia, and global burden of disease: A discussion. Environ. Health Perspect. 2004, 112, 1371–1374. [Google Scholar] [CrossRef] [PubMed]
- Bahadoran, Z.; Mirmiran, P.; Jeddi, S.; Azizi, F.; Ghasemi, A.; Hadaegh, F. Nitrate and nitrite content of vegetables, fruits, grains, legumes, dairy products, meats and processed meats. J. Food Compos. Anal. 2016, 51, 93–105. [Google Scholar] [CrossRef]
- Mancia, G.; De Backer, G.; Dominiczak, A.; Cifkova, R.; Fagard, R.; Germano, G.; Grassi, G.; Heagerty, A.M.; Kjeldsen, S.E.; Laurent, S.; et al. 2007 Guidelines for the Management of Arterial Hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J. Hypertens. 2007, 25, 1105–1187. [Google Scholar] [CrossRef] [PubMed]
- Bolton, K.; Culleton, B.; Harvey, K. K/DOQI clinical practice guidelines for chronic kidney disease: Evaluation, classification, and stratification. Am. J. Kidney Dis. 2002, 39 (Suppl. S1), S1–S266. [Google Scholar]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F.; Feldman, H.I.; Kusek, J.W.; Eggers, P.; van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Bozorgmanesh, M.; Hadaegh, F.; Mehrabi, Y.; Azizi, F. A point-score system superior to blood pressure measures alone for predicting incident hypertension: Tehran Lipid and Glucose Study. J. Hypertens. 2011, 29, 1486–1493. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Standards of medical care in diabetes 2014. Diabetes Care 2014, 37 (Suppl. S1), S14–S80. [Google Scholar]
- Yilmaz, B.; Sahin, K.; Bilen, H.; Bahcecioglu, I.H.; Bilir, B.; Ashraf, S.; Halazun, K.J.; Kucuk, O. Carotenoids and non-alcoholic fatty liver disease. Hepatobiliary Surg. Nutr. 2015, 4, 161–171. [Google Scholar] [PubMed]
- Tohidi, M.; Hasheminia, M.; Mohebi, R.; Khalili, D.; Hosseinpanah, F.; Yazdani, B.; Nasiri, A.A.; Azizi, F.; Hadaegh, F. Incidence of Chronic Kidney Disease and Its Risk Factors, Results of Over 10 Year Follow Up in an Iranian Cohort. PLoS ONE 2012, 7, e45304. [Google Scholar] [CrossRef] [PubMed]
- McNally, B.; Griffin, J.L.; Roberts, L.D. Dietary inorganic nitrate: From villain to hero in metabolic disease? Mol. Nutr. Food Res. 2016, 60, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Yang, T.; Liu, M.; Peleli, M.; Zollbrecht, C.; Weitzberg, E.; Lundberg, J.O.; Persson, A.E.G.; Carlström, M. NADPH Oxidase in the Renal Microvasculature Is a Primary Target for Blood Pressure–Lowering Effects by Inorganic Nitrate and Nitrite. Hypertension 2015, 65, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, C.R. Nitrate, nitrite, and nitric oxide find a home in the kidney by offsetting angiotensin II-mediated hypertension. Hypertension 2015, 65, 31–33. [Google Scholar] [CrossRef] [PubMed]
- Milsom, A.B.; Patel, N.S.; Mazzon, E.; Tripatara, P.; Storey, A.; Mota-Filipe, H.; Sepodes, B.; Webb, A.J.; Cuzzocrea, S.; Hobbs, A.J.; et al. Role for endothelial nitric oxide synthase in nitrite-induced protection against renal ischemia-reperfusion injury in mice. Nitric Oxide 2010, 22, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Tripatara, P.; Patel, N.S.; Webb, A.; Rathod, K.; Lecomte, F.M.; Mazzon, E.; Cuzzocrea, S.; Yaqoob, M.M.; Ahluwalia, A.; Thiemermann, C. Nitrite-derived nitric oxide protects the rat kidney against ischemia/reperfusion injury in vivo: Role for xanthine oxidoreductase. J. Am. Soc. Nephrol. 2007, 18, 570–580. [Google Scholar] [CrossRef] [PubMed]
- Carpentier, A.; Stragier, S.; Brejeon, C.; Poortmans, J.R. Nitrate Supplementation, Exercise, and Kidney Function: Are There Detrimental Effects? Med. Sci. Sports Exerc. 2015, 47, 1519–1522. [Google Scholar] [CrossRef] [PubMed]
- Mirmiran, P.; Bahadoran, Z.; Golzarand, M.; Asghari, G.; Azizi, F. Consumption of nitrate containing vegetables and the risk of chronic kidney disease: Tehran Lipid and Glucose Study. Ren. Fail. 2016, 38, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Aschebrook-Kilfoy, B.; Shu, X.-O.; Gao, Y.-T.; Ji, B.-T.; Yang, G.; Li, H.L.; Rothman, N.; Chow, W.H.; Zheng, W.; Ward, M.H. Thyroid cancer risk and dietary nitrate and nitrite intake in the Shanghai Women’s Health Study. Int. J. Cancer 2013, 132, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Ward, M.H.; Kilfoy, B.A.; Weyer, P.J.; Anderson, K.E.; Folsom, A.R.; Cerhan, J.R. Nitrate intake and the risk of thyroid cancer and thyroid disease. Epidemiology 2010, 21, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.; Benford, D.; Cockburn, A.; Cravedi, J.; Dogliotti, E.; Di Domenico, A.; Fernandez-Cruz, M.; Fink-Gremmels, J.; Fürst, P.; Galli, C.; et al. Opinion of the Scientific Panel on Contaminants in the Food chain on a request from the European Commission to perform a scientific risk assessment on nitrate in vegetables. EFSA J. 2008, 689, 1–79. [Google Scholar]
- Bahreynian, M.; Esmaillzadeh, A. Quantity and quality of carbohydrate intake in Iran: A target for nutritional intervention. Arch. Iran. Med. 2012, 15, 648–649. [Google Scholar] [PubMed]
- Akhavan, S.; Zare Abyaneh, H.; Bayat Varkeshi, M. A Systematic Review on Nitrate Concentration in Water Resources of Iran. Iran. J. Health Environ. 2014, 7, 205–228. [Google Scholar]
- Ferreira-Pêgo, C.; Guelinckx, I.; Moreno, L.A.; Kavouras, S.A.; Gandy, J.; Martinez, H.; Bardosono, S.; Abdollahi, M.; Nasseri, E.; Jarosz, A.; et al. Total fluid intake and its determinants: Cross-sectional surveys among adults in 13 countries worldwide. Eur. J. Nutr. 2015, 54 (Suppl. S2), 35–43. [Google Scholar] [CrossRef] [PubMed]
| Dietary Nitrate (mg/Day) | Dietary Nitrite (mg/Day) | |||||
|---|---|---|---|---|---|---|
| Tertile 1 | Tertile 2 | Tertile 3 | Tertile 1 | Tertile 2 | Tertile 3 | |
| <359 | 359–505 | ≥505 | <7.5 | 7.5–10.6 | ≥10.6 | |
| Age (years) | 36.3 ± 12.1 | 37.3 ± 12.2 | 37.7 ± 12.8 | 37.8 ± 12.5 | 37.5 ± 12.2 | 35.8 ± 12.3 |
| Men (%) | 39.9 | 45.7 | 43.1 | 37.5 | 41.7 | 49.5 * |
| Smoking (%) | 13.1 | 13.9 | 8.9 * | 10.6 | 13.1 | 12.4 |
| Lipid lowering drugs (%) | 1.1 | 2.7 | 2.5 | 1.6 | 2.9 | 2.2 |
| Aspirin (%) | 3.7 | 5.9 | 5.4 | 5.3 | 4.3 | 5.4 |
| Body mass index (kg/m2) | 26.0 ± 4.6 | 26.3 ± 4.5 | 26.5 ± 4.8 | 26.1 ± 4.8 | 26.3 ± 4.6 | 26.4 ± 4.7 |
| Waist circumference (cm) | 86.1 ± 13.3 | 78.5 ± 12.4 | 88.1 ± 13.1 | 86.2 ± 13.2 | 87.4 ± 12.2 | 88.1 ± 13.4 |
| SBP (mmHg) | 106 ± 12.0 | 107 ± 11.3 | 107 ± 11.9 | 106 ± 11.3 | 107 ± 12.5 | 107 ± 11.5 |
| DBP (mmHg) | 70.2 ± 9.0 | 71.2 ± 8.6 | 71.1 ± 8.8 | 70.3 ± 8.8 | 71.1 ± 8.7 | 71.0 ± 8.9 |
| FPG (mmol/L) | 86.9 ± 13.1 | 88.1 ± 14.6 | 88.0 ± 16.7 | 88.3 ± 17.8 | 87.7 ± 13.8 | 86.2 ± 12.8 |
| TG to HDL-C ratio | 3.2 ± 2.6 | 3.4 ± 2.6 | 3.3 ± 2.5 | 3.2 ± 2.4 | 3.3 ± 2.4 | 3.4 ± 2.8 |
| Serum creatinine (μmol/L) | 88.6 ± 12.6 | 89.4 ± 12.2 | 90.0 ± 14.4 | 88.1 ± 14.0 | 89.1 ± 13.0 | 90.0 ± 12.0 |
| eGFR (mL/min/1.73 m2) | 81.2 ± 13.0 | 81.0 ± 13.0 | 80.0 ± 13.7 | 80.3 ± 13.2 | 80.6 ± 13.7 | 81.6 ± 12.7 |
| Dietary NO3− (mg/day) | 276 ± 58.7 | 428 ± 41.9 | 660 ± 162 * | 314 ± 106 | 447 ± 117 | 604 ± 196 * |
| Dietary NO2− (mg/day) | 6.7 ± 2.4 | 9.2 ± 2.2 | 12.4 ± 3.5 * | 5.9 ± 1.1 | 9.0 ± 0.8 | 13.5 ± 2.8 * |
| Incident case of HTN after 5.8 years (%) | 16.7 | 18.6 | 17.0 | 17.1 | 14.7 | 14.7 |
| Tertile 1 | Tertile 2 | Tertile 3 | |
|---|---|---|---|
| <359 mg/Day | 359–505 mg/Day | ≥505 mg/Day | |
| Energy (kcal/day) | 1708 ± 473 | 2273 ± 536 | 2809 ± 640 * |
| Carbohydrate (g/day) | 234 ± 63.9 | 322 ± 77.9 | 414 ± 108 * |
| Total fats (g/day) | 64.1 ± 26.7 | 81.3 ± 28.2 | 93.8 ± 31.6 * |
| Proteins (g/day) | 57.7 ± 18.9 | 75.9 ± 19.8 | 97.1 ± 27.1 * |
| Total fiber (g/day) | 24.1 ± 12.5 | 36.5 ± 15.7 | 49.0 ± 21.2 * |
| Sodium (g/day) | 4.1 ± 0.6 | 4.5 ± 1.0 | 5.4 ± 1.2 * |
| Potassium (g/day) | 2.5 ± 0.7 | 3.6 ± 1.4 | 5.1 ± 1.8 * |
| Vegetables (g/day) | 90.2 ± 42.1 | 155 ± 57.9 | 269 ± 142 * |
| Fruits (g/day) | 184 ± 130 | 319 ± 196 | 501 ± 320 * |
| Dairy (g/day) | 343 ± 248 | 450 ± 261 | 525 ± 331 * |
| Legumes (g/day) | 10.9 ± 12.5 | 15.7 ± 19.8 | 20.7 ± 27.4 * |
| Grains (g/day) | 297 ± 127 | 402 ± 174 | 491 ± 282 * |
| Meats (g/day) | 42.7 ± 35.2 | 52.0 ± 32.4 | 67.1 ± 60.4 * |
| Processed meats (g/day) | 9.5 ± 11.2 | 11.2 ± 10.9 | 13.8 ± 17.1 * |
| Tertile 2 | Tertile 3 | |
|---|---|---|
| Dietary NO3− (mg/day) | 359–505 | ≥505 |
| Crude | 1.13 (0.83–1.53) | 1.02 (0.75–1.39) |
| Model 1 | 1.06 (0.73–1.43) | 0.81 (0.58–1.17) |
| Model 2 | 1.02 (0.68–1.51) | 0.81 (0.48–1.38) |
| Dietary NO2− (mg/day) | 7.58–10.6 | ≥10.6 |
| Crude | 0.85 (0.61–1.18) | 0.86 (0.56–1.33) |
| Model 1 | 0.66 (0.45–1.00) | 0.58 (0.34–0.99) |
| Model 2 | 0.66 (0.44–1.00) | 0.58 (0.33–0.98) |
| Tertile 2 | Tertile 3 | |
|---|---|---|
| Dietary NO3− (mg/day) | 365–511 | ≥511 |
| Crude | 0.83 (0.61–1.14) | 0.76 (0.52–1.12) |
| Model 1 | 1.07 (0.71–1.60) | 0.78 (0.48–1.28) |
| Model 2 | 1.04 (0.68–1.57) | 0.76 (0.43–1.24) |
| Dietary NO2− (mg/day) | 7.69–10.7 | ≥10.7 |
| Crude | 0.95 (0.69–1.31) | 0.87 (0.57–1.33) |
| Model 1 | 0.79 (0.52–1.18) | 0.55 (0.32–0.93) |
| Model 2 | 0.76 (0.50–1.13) | 0.50 (0.24–0.89) |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bahadoran, Z.; Mirmiran, P.; Ghasemi, A.; Carlström, M.; Azizi, F.; Hadaegh, F. Association between Dietary Intakes of Nitrate and Nitrite and the Risk of Hypertension and Chronic Kidney Disease: Tehran Lipid and Glucose Study. Nutrients 2016, 8, 811. https://doi.org/10.3390/nu8120811
Bahadoran Z, Mirmiran P, Ghasemi A, Carlström M, Azizi F, Hadaegh F. Association between Dietary Intakes of Nitrate and Nitrite and the Risk of Hypertension and Chronic Kidney Disease: Tehran Lipid and Glucose Study. Nutrients. 2016; 8(12):811. https://doi.org/10.3390/nu8120811
Chicago/Turabian StyleBahadoran, Zahra, Parvin Mirmiran, Asghar Ghasemi, Mattias Carlström, Fereidoun Azizi, and Farzad Hadaegh. 2016. "Association between Dietary Intakes of Nitrate and Nitrite and the Risk of Hypertension and Chronic Kidney Disease: Tehran Lipid and Glucose Study" Nutrients 8, no. 12: 811. https://doi.org/10.3390/nu8120811
APA StyleBahadoran, Z., Mirmiran, P., Ghasemi, A., Carlström, M., Azizi, F., & Hadaegh, F. (2016). Association between Dietary Intakes of Nitrate and Nitrite and the Risk of Hypertension and Chronic Kidney Disease: Tehran Lipid and Glucose Study. Nutrients, 8(12), 811. https://doi.org/10.3390/nu8120811






