Phosphorus Supplementation Recovers the Blunted Diet-Induced Thermogenesis of Overweight and Obese Adults: A Pilot Study
Abstract
:1. Background
2. Experimental Section
2.1. Study Design and Participants
2.2. Experimental Protocol
2.3. Questionnaires
2.4. Statistics
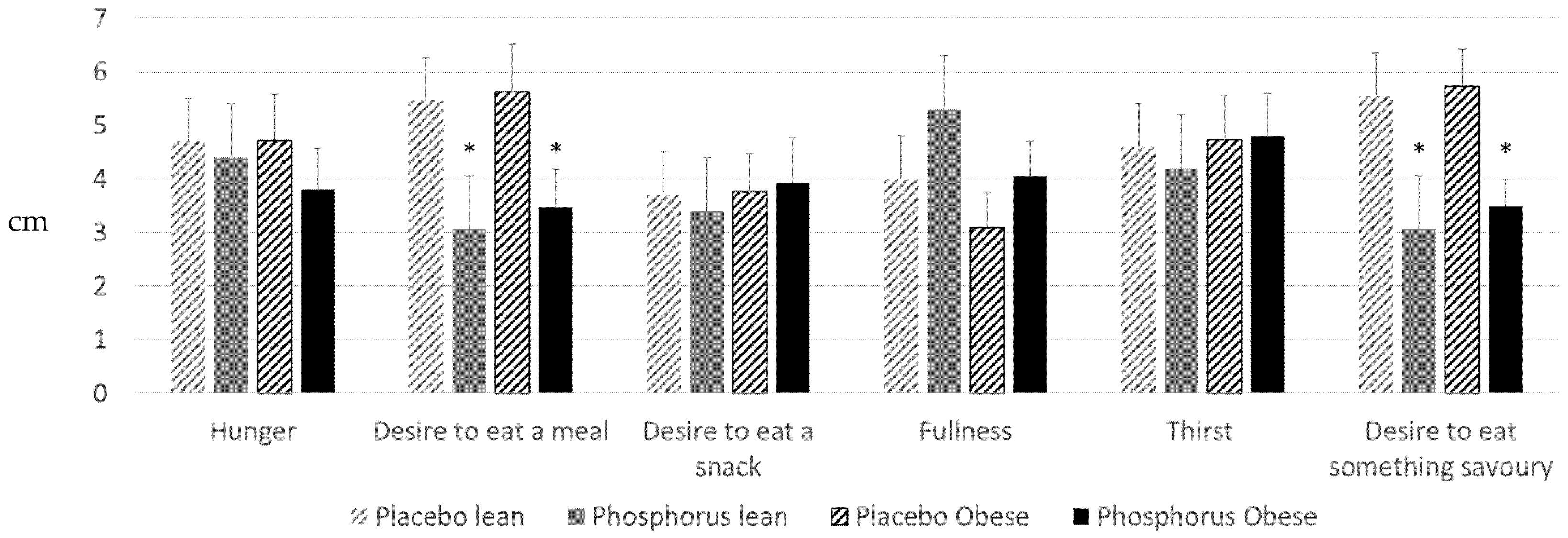
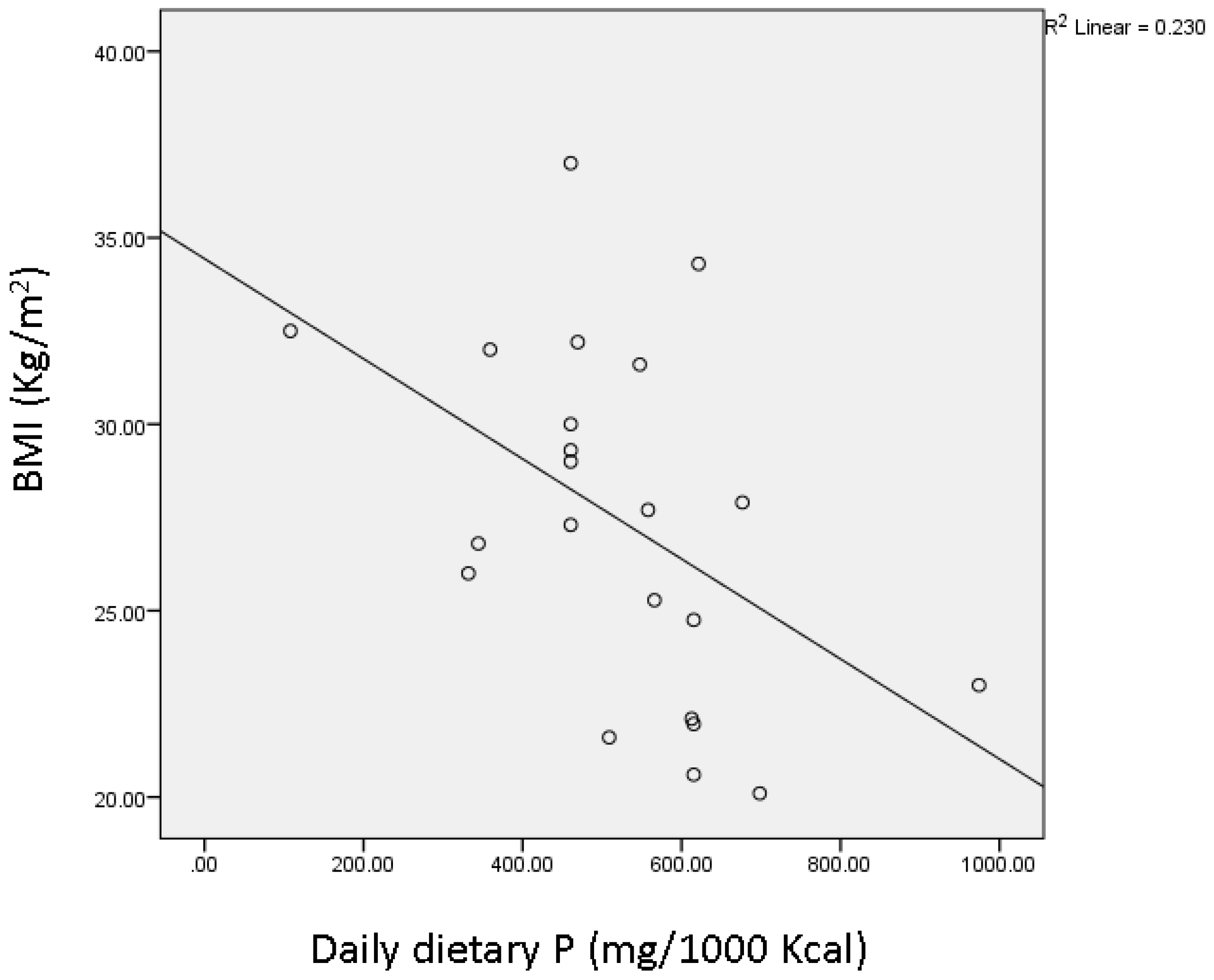
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schutz, Y.; Bessard, T.; Jequier, E. Diet-induced thermogenesis measured over a whole day in obese and nonobese women. Am. J. Clin. Nutr. 1984, 40, 545–552. [Google Scholar]
- Flat, J.P. The biochemistry of energy expenditure. In Recent Advances in Obesity Research; Bray, G.A., Ed.; Newman Publishing Ltd.: London, UK, 1978; pp. 211–228. [Google Scholar]
- Segal, K.R.; Gutin, B.; Nyman, A.M.; Pi-Sunyer, F.X. Thermic effect of food at rest, during exercise, and after exercise in lean and obese men of similar body weight. J. Clin. Investig. 1985, 76, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
- Segal, K.R.; Edano, A.; Blando, L.; Pi-Sunyer, F. Comparison of thermic effects of constant and relative caloric loads in lean and obese men. Am. J. Clin. Nutr. 1990, 51, 14–21. [Google Scholar] [PubMed]
- Segal, K.R.; Edano, A.; Tomas, M.B. Thermic effect of a meal over 3 and 6 hours in lean and obese men. Metabolism 1990, 39, 985–992. [Google Scholar] [CrossRef]
- Thorne, A.; Hallberg, D.; Wahren, J. Meal-induced thermogenesis in obese patients before and after weight reduction. Clin. Physiol. 1989, 9, 481–498. [Google Scholar] [CrossRef] [PubMed]
- Thorne, A.; Naslund, I.; Wahren, J. Meal-induced thermogenesis in previously obese patients. Clin. Physiol. 1990, 10, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Golay, A.; Schutz, Y.; Meyer, H.U.; Thiebaud, D.; Curchod, B.; Maeder, E.; Felber, J.P.; Jequier, E. Glucose-induced thermogenesis in nondiabetic and diabetic obese subjects. Diabetes 1982, 11, 1023–1028. [Google Scholar] [CrossRef]
- Felig, P. Insulin is the mediator of feeding-related thermogenesis: Insulin resistance and/or deficiency results in a thermogenic defect which contributes to the pathogenesis of obesity. Clin. Physiol. 1984, 4, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Campillo, J.E.; Aguayo, J.; Pages, I.; Castillo, M.; Osorio, C. Inorganic phosphate-insulin relationships in normal subjects and in patients with moderate glucose intolerance. Diabete Metab. 1982, 4, 289–293. [Google Scholar]
- Xie, W.; Tran, L.; Finegood, D.; Werve, G. Dietary Pi deprivation in rats affects liver CAMP, glycogen, key steps of gluconeogenesis and glucose production. Biochem. J. 2000, 352, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Park, W.; Kim, B.S.; Lee, J.E.; Huh, J.K.; Kim, B.J.; Sung, K.C.; Kang, J.H.; Lee, M.H.; Park, J.R.; Rhee, E.J.; et al. Serum phosphate levels and the risk of cardiovascular disease and metabolic syndrome: A double-edged sword. Diabetes Res. Clin. Pract. 2009, 83, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Haap, M.; Heller, E.; Thamer, C.; Tschritter, O.; Stefan, N.; Fritsche, A. Association of serum phosphate levels with glucose tolerance, insulin sensitivity and insulin secretion in non-diabetic subjects. Eur. J. Clin. Nutr. 2006, 60, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Paula, F.J.; Plens, A.E.; Foss, M.C. Effects of hypophosphatemia on glucose tolerance and insulin secretion. Horm. Metab. Res. 1998, 30, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Eller, P.; Eller, K.; Kirsch, A.H.; Patsch, J.J.; Wolf, A.M.; Tagwerker, A.; Stanzl, U.; Kaindl, R.; Kahlenberg, V.; Mayer, G.; et al. A murine model of phosphate nephropathy. Am. J. Pathol. 2011, 175, 1999–2006. [Google Scholar] [CrossRef] [PubMed]
- Abuduli, M.; Ohminami, H.; Otani, T.; Kubo, H.; Ueda, H.; Kawai, Y.; Masuda, M.; Yamanaka-Okumura, H.; Sakaue, H.; Yamamoto, H.; et al. Effects of dietary phosphate on glucose and lipid metabolism. Am. J. Physiol. Endocrinol. Metab. 2016, 310, E526–E538. [Google Scholar] [CrossRef] [PubMed]
- Khattab, M.; Abi-Rashed, C.; Ghattas, H.; Hlais, S.; Obeid, O. Phosphorus ingestion improves oral glucose tolerance of healthy male subjects: A crossover experiment. Nut J. 2015, 14, 112. [Google Scholar] [CrossRef] [PubMed]
- Kaciuba-Uściłko, H.; Nazar, K.; Chwalbińska-Moneta, J.; Ziemba, A.; Kruk, B.; Szczepanik, J.; Titow-Stupnicka, E.; Bicz, B. Effect of phosphate supplementation on metabolic and neuroendocrine responses to exercise and oral glucose load in obese women during weight reduction. J. Physiol. Pharmacol. 1993, 4, 425–440. [Google Scholar]
- Nazar, K.; Kaciuba-Uściłko, H.; Szczepanik, J.; Zemba, A.W.; Kruk, B.; Chwalbińska-Moneta, J.; Titow-Stupnicka, E.; Bicz, B.; Krotkiewski, M. Phosphate supplementation prevents a decrease of triiodothyronine and increases resting metabolic rate during low energy diet. J. Physiol. Pharmacol. 1996, 47, 373–383. [Google Scholar] [PubMed]
- Jaedig, S.; Henningsen, N.C. Increased metabolic rate in obese women after ingestion of potassium, magnesium- and phosphate-enriched orange juice or injection of ephedrine. Int. J. Obes. 1991, 15, 429–436. [Google Scholar] [PubMed]
- Jaedig, S.; Lindgärde, F.; Arborelius, M. Increased postprandial energy expenditure in obese women after peroral K- and Mg-phosphate. Miner. Electrolyte Metab. 1994, 20, 147–152. [Google Scholar] [PubMed]
- Friedman, M.I. Obesity and the hepatic control of feeding behavior. Drug News Perspect. 2007, 20, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Graczyk-Milbrandt, G.; Friedman, M.I. Metabolic inhibitors synergistically decrease hepatic energy status and increase food intake. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000, 278, 1579–1582. [Google Scholar]
- Oberhaensli, R.D.; Galloway, G.J.; Taylor, D.J.; Bore, P.J.; Radda, G.K. Assessment of human liver metabolism by phosphorus-31-magnetic resonance spectroscopy. Br. J. Radiol. 1986, 59, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Rawson, N.E.; Freidman, M.I. Phosphate loading prevents the decrease in ATP and increase in food intake produced by 2,5-anhydro-d-mannitol. Am. J. Physiol. Regul. Integr. Comp. Physiol. 1994, 266, 1792–1796. [Google Scholar]
- Riquelme, P.T.; Wernette-Hammond, M.E.; Kneer, N.M.; Lardy, H.A. Mechanism of action of 2,5-anhydro-d-mannitol in hepatocytes. J. Biol. Chem. 1984, 259, 5115–5123. [Google Scholar] [PubMed]
- Wlodek, D.; Gonzales, M. Decreased energy levels can cause and sustain obesity. J. Theor. Biol. 2003, 225, 33–44. [Google Scholar] [CrossRef]
- Obeid, O.A.; Dimachkie, S.; Hlais, S. Increased phosphorus content of preload suppresses ad libitum energy intake at subsequent meal. Int. J. Obes. 2010, 34, 1446–1448. [Google Scholar] [CrossRef] [PubMed]
- Håglin, L. Hypophosphataemia: Cause of the disturbed metabolism in the metabolic syndrome. Med. Hypotheses 2001, 56, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Haglin, L.; Lindblad, A.; Bygren, L.O. Hypophosphataemia in the metabolic syndrome. Gender differences in body weight and blood glucose. Eur. J. Clin. Nutr. 2001, 55, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Kalaitzidis, R.; Tsimihodimos, V.; Bairaktari, E.; Siamopoulos, K.C.; Elisaf, M. Disturbances of phosphate metabolism: Another feature of metabolic syndrome. Am. J. Kidney Dis. 2005, 45, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Lind, L.; Lithell, H.; Hvarfner, A.; Pollare, T.; Ljunghall, S. On the relationships between mineral metabolism, obesity and fat distribution. Eur. J. Clin. Investig. 1993, 23, 307–310. [Google Scholar] [CrossRef]
- Lindgärde, F.; Trell, E. serum inorganic phosphate in middle-aged men. I. Inverse relation to body weight. Acta Med. Scand. 1977, 202, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Baecke, J.A.; Burema, J.; Frijters, J.E. A short questionnaire for the measurement of habitual physical activity in epidemiological studies. Am. J. Clin. Nutr. 1982, 36, 936–942. [Google Scholar] [PubMed]
- Jequier, E.; Acheson, K.; Schutz, Y. Assessment of energy expenditure and fuel utilization in man. Annu. Rev. Nutr. 1987, 7, 187–208. [Google Scholar] [CrossRef] [PubMed]
- Flint, A.; Raben, A.; Blundell, J.; Astrup, A. Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Nowicki, M.; Fliser, D.; Fode, P.; Ritz, E. Changes in plasma phosphate levels influence insulin sensitivity under euglycemic conditions. J. Clin. Endocrinol. Metab. 1996, 1, 156–159. [Google Scholar]
- Langhans, W.; Scharrer, E. Metabolic control of eating. World Rev. Nut Diet. 1992, 70, 1–67. [Google Scholar]
- Morris, R.C., Jr.; Nigon, K.; Reed, E.B. Evidence that the severity of depletion of inorganic phosphate determines the severity of the disturbance of adenine nucleotide metabolism in the liver and renal cortex of the fructose-loaded rat. J. Clin. Investig. 1978, 61, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Muzik, O.; Mangner, T.J.; Leonard, W.R.; Kumar, A.; Janisse, J.; Granneman, J.G. 15O PET measurement of blood flow and oxygen consumption in cold-activated human brown fat. J. Nucl. Med. 2013, 54, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Ayoub, J.J.; Samra, M.J.A.; Hlais, S.A.; Bassil, M.S.; Obeid, O.A. Effect of phosphorus supplementation on weight gain and waist circumference of overweight/obese adults: A randomized clinical trial. Nutr. Diabetes 2015, 5, e189. [Google Scholar] [CrossRef] [PubMed]
- Sweeny, J.M.; Seibert, H.E.; Woda, C.; Schulkin, J.; Haramati, A.; Mulroney, S.E. Evidence for induction of a phosphate appetite in juvenile rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 1998, 275, R1358–R1365. [Google Scholar]
- Ohnishi, R.; Segawa, H.; Kawakami, E.; Furutani, J.; Ito, M.; Tatsumi, S.; Kuwahata, M.; Miyamoto, K.I. Control of phosphate appetite in young rats. J. Med. Investig. 2007, 54, 366–369. [Google Scholar] [CrossRef]
- De Boer, I.H.; Rue, T.C.; Kestenbaum, B. Serum phosphorus concentrations in the third National Health and Nutrition Examination Survey (NHANES III). Am. J. Kidney Dis. 2009, 53, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Halton, T.H.; Hu, F.B. The effects of high protein diets on thermogenesis, satiety and weight loss: A critical review. J. Am. Coll. Nutr. 2004, 23, 373–385. [Google Scholar] [CrossRef] [PubMed]
- Yates, A.A.; Schlicker, S.A.; Suitor, C.W. Dietary reference intakes for calcium, phosphorus, magnesium, vitamin D and fluoride. J. Am. Diet. Assoc. 1998, 699–706. [Google Scholar] [CrossRef]
- Obeid, O.A. Low phosphorus status might contribute to the onset of obesity. Obes. Rev. 2013, 14, 659–664. [Google Scholar] [CrossRef] [PubMed]

| Lean (n = 10) | Obese (n = 13) | |
|---|---|---|
| Age (years) | 21 ± 1 | 22 ± 1 |
| Sex (M/F) | 5/5 | 5/8 |
| Weight (kg) | 71.6 ± 6.5 | 89.0 ± 1.9 * |
| Height (cm) | 173.8 ± 4.8 | 170.8 ± 2.2 |
| BMI (kg/m2) | 23.0 ± 0.9 | 30.6 ± 0.8 * |
| FFM (kg) | 57.5 ± 5.3 | 56.5 ± 2.5 |
| Fat mass percentage (%) | 20.0 ± 2.3 | 36.3 ± 2.1 * |
| Baseline RMR (kcal/min) | 1.25 ± 0.08 | 1.44 ± 0.05 * |
| Daily energy intake (kcal) † | 2256 ± 163 | 2521 ± 160 |
| Daily dietary phosphorus (mg/1000 kcal) ‡ | 615.3 ± 56.49 | 460.5 ± 39.04 * |
| Daily dietary calcium (mg/1000 kcal) † | 415.4 ± 58.7 | 438.4 ± 31.2 |
| Lean | Obese | |||
|---|---|---|---|---|
| Placebo | Phosphorus | Placebo | Phosphorus | |
| Carbohydrate oxidation | ||||
| Baseline (mg/min) | 86.5 ± 12.4 | 77.4 ± 4.8 | 90.6 ± 5.9 | 87.6 ± 3.1 |
| Postprandial (mg/min) | 177.9 ± 13.3 | 182.0 ± 14.4 | 178.1 ± 10.5 | 207.6 ± 10.4 † |
| % Change from baseline | 121 ± 17 | 134 ± 10 | 107 ± 10 | 139 ± 12 |
| Fat oxidation | ||||
| Baseline (mg/min) | 100.3 ± 8.5 | 106.5 ± 6.6 | 125.2 ± 6.4 * | 123.7 ± 3.7 * |
| Postprandial (mg/min) | 73.5 ± 4.6 | 75.3 ± 5.3 | 79.6 ± 4.5 | 85.3 ± 3.9 |
| % Change from baseline | −36 ± 7 | −30 ± 1 | −58 ± 9 * | −31 ± 2 † |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bassil, M.S.; Obeid, O.A. Phosphorus Supplementation Recovers the Blunted Diet-Induced Thermogenesis of Overweight and Obese Adults: A Pilot Study. Nutrients 2016, 8, 801. https://doi.org/10.3390/nu8120801
Bassil MS, Obeid OA. Phosphorus Supplementation Recovers the Blunted Diet-Induced Thermogenesis of Overweight and Obese Adults: A Pilot Study. Nutrients. 2016; 8(12):801. https://doi.org/10.3390/nu8120801
Chicago/Turabian StyleBassil, Maya S., and Omar A. Obeid. 2016. "Phosphorus Supplementation Recovers the Blunted Diet-Induced Thermogenesis of Overweight and Obese Adults: A Pilot Study" Nutrients 8, no. 12: 801. https://doi.org/10.3390/nu8120801
APA StyleBassil, M. S., & Obeid, O. A. (2016). Phosphorus Supplementation Recovers the Blunted Diet-Induced Thermogenesis of Overweight and Obese Adults: A Pilot Study. Nutrients, 8(12), 801. https://doi.org/10.3390/nu8120801





