Beverage Consumption: Are Alcoholic and Sugary Drinks Tipping the Balance towards Overweight and Obesity?
Abstract
:1. Introduction
1.1. Alcoholic Beverages (ABs)
1.2. Sugar Sweetened Beverages (SSBs)
2. Solid Food vs. Liquid Beverage: Consequences for Energy Intake
3. Energy Intake and Body Weight
3.1. Alcoholic Beverages (ABs)
3.1.1. Short-Term Intervention Studies
 ), 50en% from CHO substituted for alcohol; ADD (
), 50en% from CHO substituted for alcohol; ADD (  ), 50en% from CHO added as alcohol. Both addition and substitution of alcohol within a meal has an immediate suppressive effect on CHO and fat oxidation (from Murgatroyd et al., 1996 [46], with permission).
), 50en% from CHO added as alcohol. Both addition and substitution of alcohol within a meal has an immediate suppressive effect on CHO and fat oxidation (from Murgatroyd et al., 1996 [46], with permission).
 ), 50en% from CHO substituted for alcohol; ADD (
), 50en% from CHO substituted for alcohol; ADD (  ), 50en% from CHO added as alcohol. Both addition and substitution of alcohol within a meal has an immediate suppressive effect on CHO and fat oxidation (from Murgatroyd et al., 1996 [46], with permission).
), 50en% from CHO added as alcohol. Both addition and substitution of alcohol within a meal has an immediate suppressive effect on CHO and fat oxidation (from Murgatroyd et al., 1996 [46], with permission).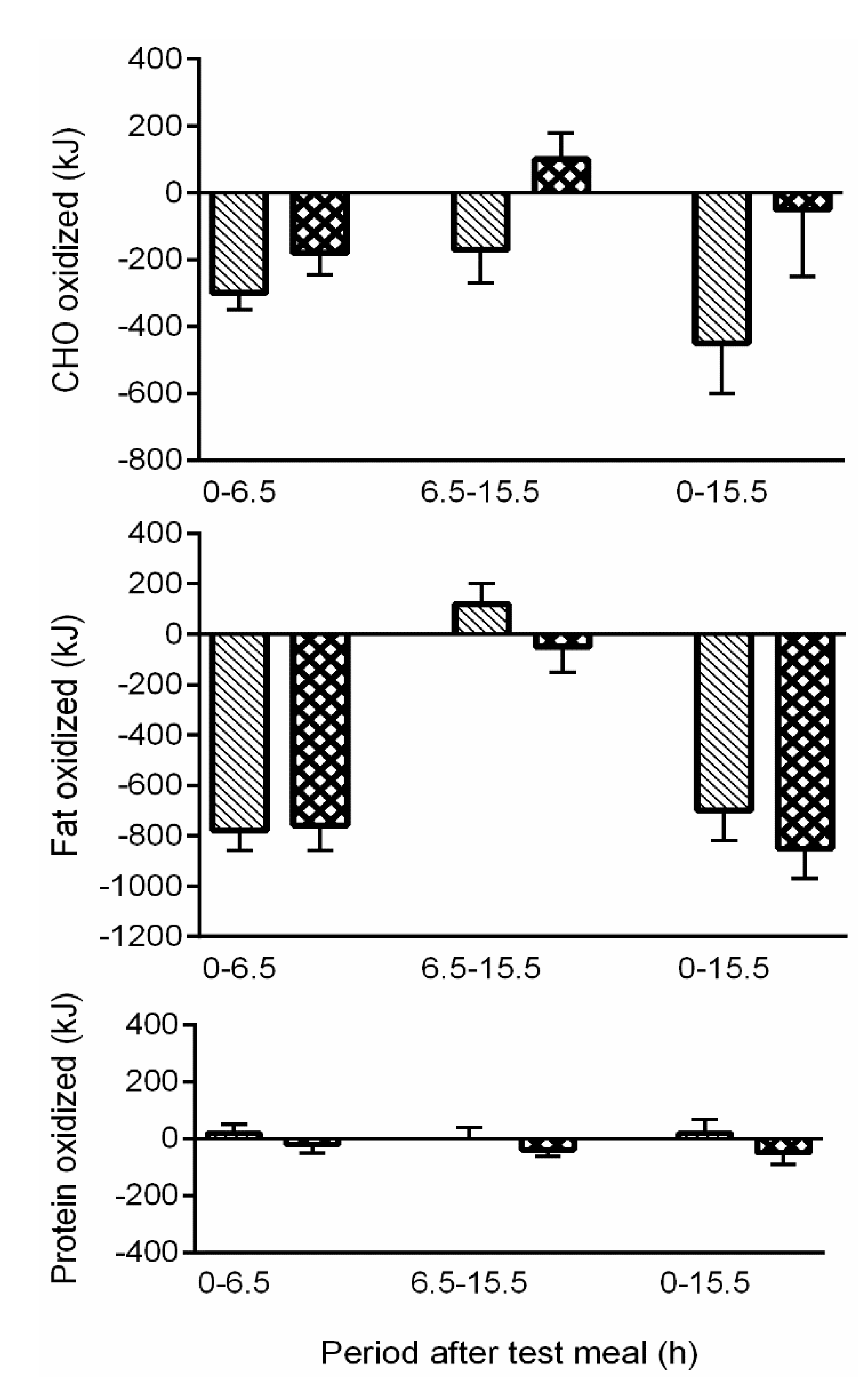
3.1.2. Observational Studies
3.2. Sugar Sweetened Beverages (SSBs)
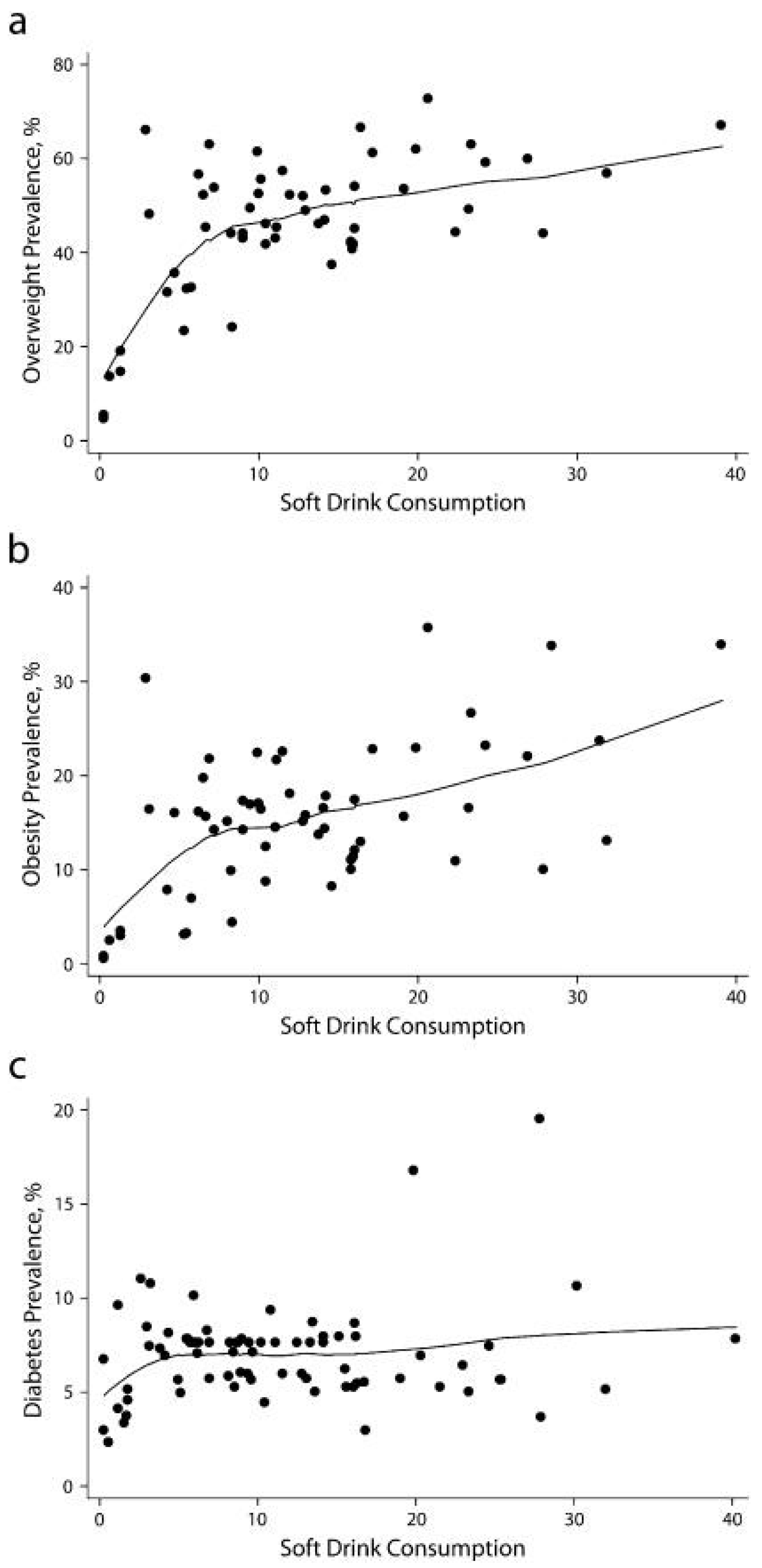
3.2.1. Observational Studies
3.2.2. Intervention Studies
4. Children and Adolescents
4.1. Preschool Children
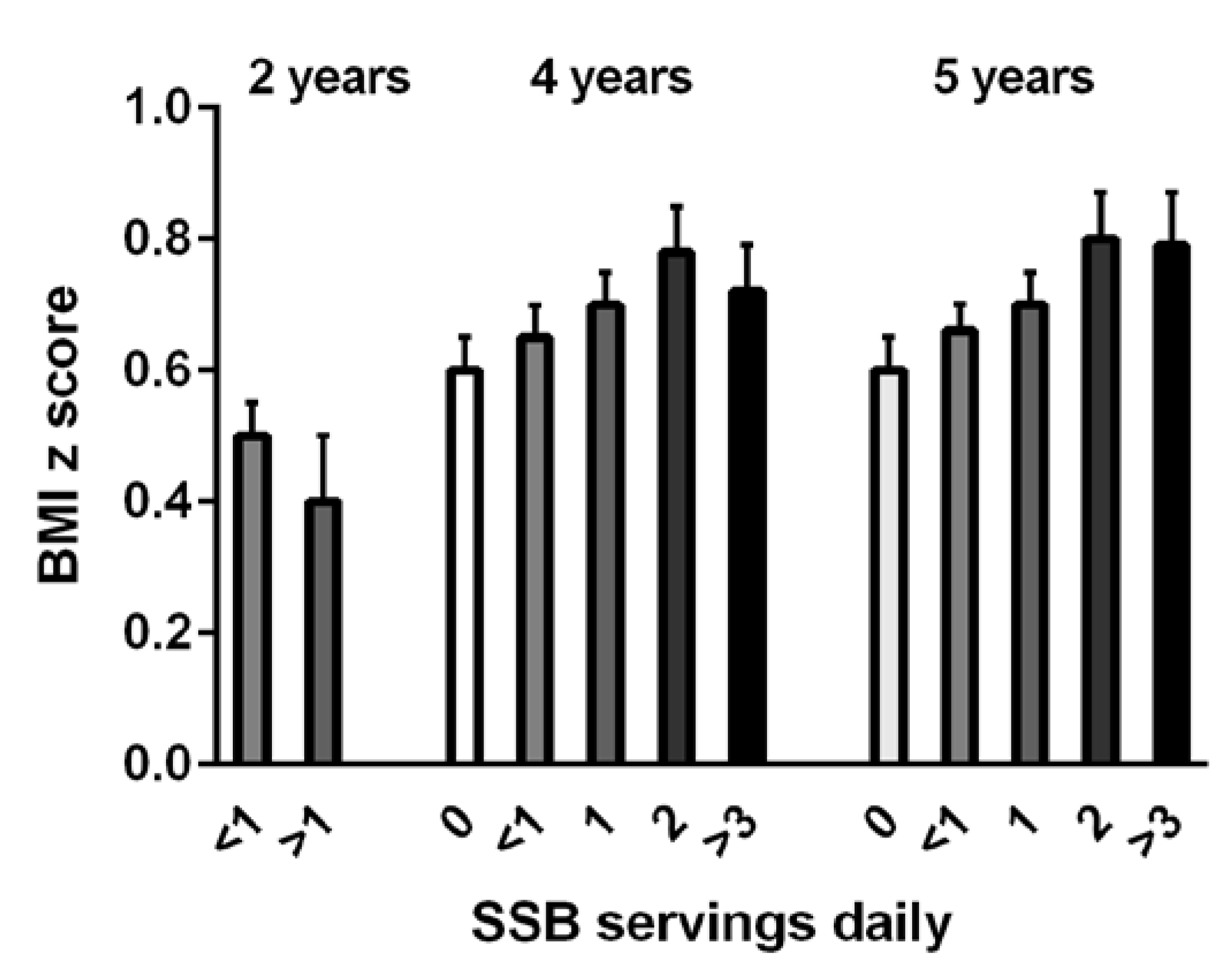
4.2. Milk as a Beverage
5. Are Beverages Tipping the Balance?
Acknowledgments
Conflicts of Interest
References
- International Council of Beverages Association (ICBA). ICBA Guidelines for the Composition, Labelling and Responsible Marketing of Energy Drinks. Available online: http://www.icba-net.org/files/resources/energy-drink-guidelines.pdf (accessed on 14 May 2015).
- US Beverage Information Group. US Beverage Alcohol Trends. Available online: http://www.usdrinksconference.com/assets/files/agenda/US%20Beverage%20Alcohol%20Trends.pdf (accessed on 18 February 2015).
- Traversy, G.; Chaput, J.P. Alcohol consumption and obesity: An update. Curr. Obes. Rep. 2015, 4, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Malik, V.S.; Pan, A.; Willett, W.C.; Hu, F.B. Sugar-sweetened beverages and weight gain in children and adults: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2013, 98, 1084–1102. [Google Scholar] [CrossRef] [PubMed]
- Norrie, P.A. The History of Wine as a Medicine; Taylor and Francis: London, UK; New York, NY, USA, 2003. [Google Scholar]
- White, J. A Great and Monstrous Thing; Harvard University Press: Cambridge, MA, USA, 2013. [Google Scholar]
- Poppitt, S.D. Alcohol: Absorption, metabolism and physiological effects. In Encyclopedia of Human Nutrition; Sadler, M.C.B., Strain, S., Eds.; Academic Press: London, UK, 1998. [Google Scholar]
- WHO. Alcohol. Available online: http://www.who.int/substance_abuse/facts/alcohol/en/ (accessed on 18 February 2015).
- Leon, D.A.; McCambridge, J. Liver cirrhosis mortality rates in Britain from 1950 to 2002: An analysis of routine data. Lancet 2006, 367, 52–56. [Google Scholar] [CrossRef]
- Westerterp, K.R.; Prentice, A.M.; Jequier, E. Alcohol and body weigh. In Health Issues Related to Alcohol Consumption, 2nd ed.; McDonald, I., Ed.; ILSI Europe: Brussels, Belgium, 1999; pp. 103–123. [Google Scholar]
- Suter, P.M.; Hasler, E.; Vetter, W. Effects of alcohol on energy metabolism and body weight regulation: Is alcohol a risk factor for obesity? Nutr. Rev. 1997, 55, 155–171. [Google Scholar] [CrossRef]
- Leibel, R.L.; Dufour, M.; Hubbard, V.S.; Lands, W.E. Alcohol and calories: A matter of balance. Alcolhol 1993, 10, 429–434. [Google Scholar] [CrossRef]
- American Medical Association (AMA). Obesity as a disease. Available online: http://www.ama-assn.org/ama/pub/news/news/2013/2013-06-18-new-ama-policies-annual-meeting.page (accessed on 18 February 2015).
- Lukasiewi, E.; Mennen, L.I.; Bertrai, S.; Arnault, N.; Preziosi, P.; Galan, P.; Hercberg, S. Alcohol intake in relation to body mass index and waist-to-hip ratio: The importance of type of alcoholic beverage. Public Health Nutr. 2005, 8, 315–320. [Google Scholar]
- Wannamethee, S.G.; Shaper, A.G.; Whincup, P.H. Alcohol and adiposity: Effects of quantity and type of drink and time relation with meals. Int. J. Obes. 2005, 29, 1436–1444. [Google Scholar] [CrossRef] [PubMed]
- Bleich, S.N.; Wang, Y.C.; Wang, Y.; Gortmaker, S.L. Increasing consumption of sugar-sweetened beverages among US adults: 1988–1994 to 1999–2004. Am. J. Clin. Nutr. 2009, 89, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; Bleich, S.N.; Gortmaker, S.L. Increasing caloric contribution from sugar sweetened beverages and 100% fruit juices among US children and adolescents, 1988–2004. Pediatrics 2008, 121, e1604–e1614. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B.; Malik, V.S. Sugar-sweetened beverages and risk of obesity and type 2 diabetes: Epidemiological evidence. Physiol. Behav. 2010, 100, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Ogden, C.L.; Kit, B.K.; Carroll, M.D.; Park, S. Consumption of Sugar Drinks in the United States, 2005–2008. Available online: www.cdc.gov/nchs/data/databriefs/db71.htm (accessed on 10 April 2012).
- Welsh, J.A.; Sharma, A.J.; Grellinger, L.; Vos, M.B. Consumption of added sugars is decreasing in the United States. Am. J. Clin. Nutr. 2011, 94, 726–734. [Google Scholar] [CrossRef] [PubMed]
- Libuda, L.; Kersting, M. Soft drinks and body weight development in childhood: Is there a relationship? Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 596–600. [Google Scholar] [CrossRef] [PubMed]
- Ervin, R.B.; Kit, B.K.; Carroll, M.D.; Ogden, C.L. Consumption of added sugar among U.S. children and adolescents, 2005–2008. NCHS Data Brief 2012, 87, 1–8. [Google Scholar] [PubMed]
- Nielsen, S.J.; Popkin, B.M. Changes in beverage intake between 1977 and 2001. Am. J. Prev. Med. 2004, 27, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; McKee, M.; Galea, G.; Stuckler, D. Relationship of soft drink consumption to global overweight, obesity, and diabetes: A cross-national analysis of 75 countries. Am. J. Public Health 2013, 103, 2071–2077. [Google Scholar] [CrossRef] [PubMed]
- Massougbodji, J.; Bodo, Y.L.; Fratu, R.; Wals, P.D. Reviews examining sugar-sweetened beverages and body weight: Correlates of their quality and conclusions. Am. J. Clin. Nutr. 2014, 99, 1096–1104. [Google Scholar] [CrossRef] [PubMed]
- Gornall, J. Sugar: Spinning a web of influence. BMJ 2015, 350, h231. [Google Scholar] [CrossRef] [PubMed]
- Gornall, J. Sugar’s web of influence 3: Why the responsibility deal is a “dead duck” for sugar reduction. BMJ 2015, 350, h219. [Google Scholar] [CrossRef] [PubMed]
- Te Morenga, L.; Mallard, S.; Mann, J. Dietary sugars and body weight: Systematic review and meta-analyses of randomised controlled trials and cohort studies. BMJ 2012, 346, e7492. [Google Scholar] [CrossRef] [PubMed]
- WHO. Diet, nutrition and the prevention of chronic diseases: Report of a Joint WHO/FAO Expert Consultation. In WHO Technical Report Series, No 916; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Almiron-Roig, E.; Palla, L.; Guest, K.; Ricchiuti, C.; Vint, N.; Jebb, S.A.; Drewnowski, A. Factors that determine energy compensation: A systematic review of preload studies. Nutr. Rev. 2013, 2013, 458–473. [Google Scholar] [CrossRef] [PubMed]
- Mattes, R.D. Beverages and positive energy balance: The menace is the medium. Int. J. Obes. 2006, 30, S60–S65. [Google Scholar] [CrossRef]
- Cassady, B.A.; Considine, R.V.; Mattes, R.D. Beverage consumption, appetite, and energy intake: What did you expect? Am. J. Clin. Nutr. 2012, 95, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Mourao, D.; Bressan, J.; Campbell, W.W.; Mattes, R.D. Effects of food form on appetite and energy intake in lean and obese young adults. Int. J. Obes. 2007, 31, 1688–1695. [Google Scholar] [CrossRef] [PubMed]
- Crapo, P.A.; Henry, R.R. Postprandial metabolic responses to the influence of food form. Am. J. Clin. Nutr. 1998, 48, 560–564. [Google Scholar]
- Mattes, R.D.; Campbell, W.W. Effects of food form and timing of ingestion on appetite and energy intake in lean and obese young adults. J. Am. Diet. Assoc. 2009, 109, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Poppitt, S.D.; Eckhardt, J.W.; McGonagle, J.; Murgatroyd, P.R.; Prentice, A.M. Short-term effects of alcohol consumption on appetite and energy intake. Physiol. Behav. 1996, 60, 1063–1070. [Google Scholar] [CrossRef]
- DellaValle, D.M.; Roe, L.S.; Rolls, B.J. Does the consumption of caloric and non-caloric beverages with a meal affect energy intake? Appetite 2005, 44, 187–193. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, M.P.; Rubiano, F.; DeNino, W.F.; Jones, A., Jr.; Greenfield, D.; Ferguson, P.W.; Akrabawi, S.; Heymsfield, S.B. Added thermogenic and satiety effects of a mixed nutrient vs. a sugar-only beverage. Int. J. Obes. 2004, 28, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Bowen, J.; Noakes, M.; Clifton, P.M. Appetite hormones and energy intake in obese men after consumption of fructose, glucose and whey protein beverages. Int. J. Obes. 2007, 31, 1696–1703. [Google Scholar] [CrossRef] [PubMed]
- Poppitt, S.D.; Proctor, J.; McGill, A.T.; Wiessing, K.R.; Falk, S.; Xin, L.; Budgett, S.C.; Darragh, A.; Hall, R.S. Low-dose whey protein-enriched water beverages alter satiety in a study of overweight women. Appetite 2011, 56, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Di Meglio, D.P.; Mattes, R.D. Liquid versus solid carbohydrate: Effects on food intake and body weight. Int. J. Obes. 2000, 24, 794–800. [Google Scholar] [CrossRef]
- Houchins, J.A.; Burgess, J.R.; Campbell, W.W.; Daniel, J.R.; Ferruzzi, M.G.; McCabe, G.P.; Mattes, R.D. Beverage vs. solid fruits and vegetables: Effects on energy intake and body weight. Obesity 2012, 20, 1844–1850. [Google Scholar] [CrossRef] [PubMed]
- Houchins, J.A.; Tan, S.Y.; Campbell, W.W.; Mattes, R.D. Effects of fruit and vegetable, consumed in solid vs. beverage forms, on acute and chronic appetitive responses in lean and obese adults. Int. J. Obes. 2013, 37, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Shelton, N.J.; Knott, C.S. Association between alcohol calorie intake and overweight and obesity in English adults. Am. J. Public Health 2014, 104, 629–631. [Google Scholar] [CrossRef] [PubMed]
- Sonko, B.J.; Prentice, A.M.; Murgatroyd, P.R.; Goldberg, G.R.; van de Ven, M.L.; Coward, W.A. Effect of alcohol on post meal fat storage. Am. J. Clin. Nutr. 1994, 59, 619–625. [Google Scholar] [PubMed]
- Murgatroyd, P.R.; van de Ven, M.L.; Goldberg, G.R.; Prentice, A.M. Alcohol and the regulation of energy balance: Overnight effects on diet-induced thermogenesis and fuel storage. Br. J. Nutr. 1996, 75, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, A.; Wouters, E.; Wenker, M.; St-Pierre, S.; Bouchard, C.; Després, J.P. Alcohol and a high-fat diet: A combination favoring overfeeding. Am. J. Clin. Nutr. 1995, 62, 639–644. [Google Scholar] [PubMed]
- Yeomans, M.R. Alcohol, appetite and energy balance: Is alcohol intake a risk factor for obesity? Physiol. Behav. 2010, 100, 82–89. [Google Scholar] [CrossRef] [PubMed]
- O’Keefe, J.H.; Bhatti, S.K.; Bajwa, A.; Di Nicolantonio, J.J.; Lavie, C.J. Alcohol and cardiovascular health: The dose makes the poison…or the remedy. Mayo Clin. Proc. 2014, 89, 382–393. [Google Scholar] [CrossRef] [PubMed]
- Suter, P.M. Is alcohol consumption a risk factor for weight gain and obesity? Crit. Rev. Clin. Lab. Sci. 2005, 42, 197–227. [Google Scholar] [CrossRef] [PubMed]
- Sayon-Orea, C.; Martinez-Gonzalez, M.A.; Bes-Rastrollo, M. Alcohol consumption and body weight: A systematic review. Nutr. Rev. 2011, 69, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Stockwell, T.; Zhao, J.; Macdonald, S. Who under-reports their alcohol consumption in telephone surveys and by how much? An application of the ‘yesterday method’ in a national Canadian substance use survey. Addiction 2014, 109, 1657–1666. [Google Scholar] [CrossRef] [PubMed]
- Lieber, C.S. Perspectives: Do alcohol calories count? Am. J. Clin. Nutr. 1991, 54, 976–982. [Google Scholar] [PubMed]
- Jucker, B.M.; Barnard, M.L.; Shulman, R.G. NMR investigation of the futile cycling of ethanol in chronic alcoholic rats. Alcohol. Clin. Exp. Res. 1994, 18, 1377–1385. [Google Scholar] [CrossRef] [PubMed]
- Pan, A.; Hu, F.B. Effects of carbohydrates on satiety: Differences between liquid and solid food. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Almiron-Roig, E.; Flores, S.Y.; Drewnowski, A. No difference in satiety or in subsequent energy intakes between a beverage and a solid food. Physiol. Behav. 2004, 82, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, K.A.; Shikany, J.M.; Keating, K.D.; Allison, D.B. Will reducing sugar-sweetened beverage consumption reduce obesity? Evidence supporting conjecture is strong, but evidence when testing effect is weak. Obes. Rev. 2013, 14, 620–633. [Google Scholar] [CrossRef] [PubMed]
- Hone-Blanchet, A.; Fecteau, S. Overlap of food addiction and substance use disorders definitions: Analysis of animal and human studies. Neuropharmacology 2014, 85, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Malik, V.S.; Hu, F.B. Sweeteners and risk of obesity and type 2 diabetes: The role of sugar-sweetened beverages. Curr. Diab. Rep. 2012, 12, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.Z.; Empie, M.W. Lack of findings for the association between obesity risk and usual sugar-sweetened beverage consumption in adults--a primary analysis of databases of CSFII-1989–1991, CSFII-1994–1998, NHANES III, and combined NHANES 1999–2002. Food Chem. Toxicol. 2007, 45, 1523–1536. [Google Scholar] [CrossRef] [PubMed]
- Forshee, R.A.; Anderson, P.A.; Storey, M.L. Sugar-sweetened beverages and body mass index in children and adolescents: A meta-analysis. Am. J. Clin. Nutr. 2008, 87, 1662–1671. [Google Scholar] [PubMed]
- Malik, V.S.; Schulze, M.B.; Hu, F.B. Intake of sugar-sweetened beverages and weight gain: A systematic review. Am. J. Clin. Nutr. 2006, 84, 274–288. [Google Scholar] [PubMed]
- Trumbo, P.R.; Rivers, C.R. Systematic review of the evidence for an association between sugar-sweetened beverage consumption and risk of obesity. Nutr. Rev. 2014, 72, 566–574. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute: Table 1B. Mean intake of energy and mean contribution (kcal) of various foods among US population, by age, NHANES 2005–2006. Available online: http://appliedresearch.cancer.gov/diet/foodsources/energy/table1b.html (accessed on 7 March 2014).
- De Castro, J.M. Accommodation of particular foods or beverages into spontaneously ingested evening meals. Appetite 1994, 23, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Mattes, R.D.; Shikany, J.M.; Kaiser, K.A.; Allison, D.B. Nutritively sweetened beverage consumption and body weight: A systematic review and meta-analysis of randomized experiments. Obes. Rev. 2011, 12, 346–365. [Google Scholar] [CrossRef] [PubMed]
- Tucker, R.M.; Mattes, R.D. Satiation, satiety: The puzzle of solids and liquids. In Satiation, Satiety and the Control of Food Intake: Theory and Practice; Blundell, J., Bellisle, F., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2014; pp. 182–201. [Google Scholar]
- Hu, F.B. Resolved: There is sufficient scientific evidence that decreasing sugar-sweetened beverage consumption will reduce the prevalence of obesity and obesity-related diseases. Obes. Rev. 2013, 14, 606–619. [Google Scholar] [CrossRef] [PubMed]
- Briefel, R.R.; Wilson, A.; Cabili, C.; Hedley-Dodd, A. Reducing calories and added sugars by improving children’s beverage choices. J. Acad. Nutr. Diet. 2013, 113, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Lasater, G.; Piernas, C.; Popkin, B.M. Beverage patterns and trends among school-aged children in the US, 1989–2008. Nutr. J. 2011, 10, 103. [Google Scholar] [CrossRef] [PubMed]
- James, J.; Kerr, D. Prevention of childhood obesity by reducing soft drinks. Int. J. Obes. 2005, 29 (Suppl. 2), S54–S57. [Google Scholar] [CrossRef]
- Blume, H. LAUSD to remove chocolate, strawberry milk from schools, superintendent says. Los Angeles Times. Available online: http://articles.latimes.com/so11/apr/28/local/la-me-lausd-milk-20110428 (accessed on 4 February 2015).
- Taber, D.R.; Chriqui, J.F.; Powell, L.M.; Chaloupka, F.J. Banning all sugar-sweetened beverages in middle schools: Reduction of in-school access and purchasing but not overall consumption. Arch. Pediatr. Adolesc. Med. 2012, 166, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Chriqui, J.F.; Pickel, M.; Story, M. Influence of school competitive food and beverage policies on obesity, consumption, and availability: A systematic review. JAMA Pediatr. 2014, 168, 279–286. [Google Scholar] [CrossRef] [PubMed]
- OECD. Obesity update. Available online: http://www.oecd.org/els/health-systems/Obesity-Update-2014.pdf (accessed on 17 February 2015).
- Stern, D.; Piernas, C.; Barquera, S.; Rivera, J.A.; Popkin, B.M. Caloric beverages were major sources of energy among children and adults in Mexico, 1999–2012. J. Nutr. 2014, 144, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Action on sugar. Childhood Obesity Action Plan. Prepared for Jeremy Hunt—UK Secretary of State for Health. Available online: http://www.actiononsalt.org.uk/actiononsugar/Press%20Release%20/133979.pdf (accessed on 17 February 2015).
- Daily Express. Jeremy Hunt’s Plans to Ban Fizzy Drinks from All Schools. Available online: http://www.express.co.uk/life-style/health/406898/Jeremy-Hunt-s-plans-to-ban-fizzy-drinks-from-all-schools (accessed on 17 February 2015).
- Gómez-Martínez, S.; Martín, A.; Romeo, J.; Castillo, M.; Mesena, M.; Baraza, J.C.; Jiménez-Pavón, D.; Redondo, C.; Zamora, S.; Marcos, A. Is soft drink consumption associated with body composition? A cross-sectional study in Spanish adolescents. Nutr. Hosp. 2009, 24, 97–102. [Google Scholar] [PubMed]
- Valente, H.; Teixeira, V.; Padrão, P.; Bessa, M.; Cordeiro, T.; Moreira, A.; Mitchell, V.; Lopes, C.; Mota, J.; Moreira, P. Sugar-sweetened beverage intake and overweight in children from a Mediterranean country. Public Health Nutr. 2011, 14, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Isacco, L.; Lazaar, N.; Ratel, S.; Thivel, D.; Aucouturier, J.; Doré, E.; Meyer, M.; Duché, P. The impact of eating habits on anthropometric characteristics in French primary school children. Child. Care Health Dev. 2010, 36, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Welsh, J.A.; Cogswell, M.E.; Rogers, S.; Rockett, H.; Mei, Z.; Grummer-Strawn, L.M. Overweight among low-income preschool children associated with the consumption of sweet drinks: Missouri, 1999–2002. Pediatrics 2005, 115, e223–e229. [Google Scholar] [CrossRef] [PubMed]
- Mathias, K.C.; Slining, M.M.; Popkin, B.M. Foods and beverages associated with higher intake of sugar-sweetened beverages. Am. J. Prev. Med. 2013, 44, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Energy Drinks “Should be Banned” for Under-16s. Available online: http://www.bbc.com/news/health-31623771 (accessed on 17 February 2015).
- De Boer, M.; Scharf, R.J.; Demmer, R.T. Sugar-Sweetened Beverages and Weight Gain in 2- to 5-Year-Old Children. Pediatrics 2013, 132, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Ariza, A.J.; Chen, E.H.; Binns, H.J.; Christoffel, K.K. Risk factors for overweight in five- to six year- old Hispanic-American children: A pilot study. J. Urban Health 2004, 81, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Kral, T.V.E.; Stunkard, A.J.; Berkowitz, R.I.; Stallings, V.A.; Moore, R.H.; Faith, M.S. Beverage consumption patterns of children born at different risk of obesity. Obesity 2008, 16, 1802–1808. [Google Scholar] [CrossRef] [PubMed]
- Kosova, E.C.; Auinger, P.; Bremer, A.A. The relationships between sugar-sweetened beverage intake and cardiometabolic markers in young children. J. Acad. Nutr. Diet. 2013, 113, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Troiano, R.P.; Briefel, R.R.; Carroll, M.D.; Bialostosky, K. Energy and fat intakes of children and adolescents in the United States: Data from the national health and nutrition examination surveys. Am. J. Clin. Nutr. 2000, 72 (Suppl. 5), 1343S–1353S. [Google Scholar] [PubMed]
- Dubois, L.; Farmer, A.; Girard, M.; Peterson, K. Regular sugar-sweetened beverage consumption between meals increases risk of overweight among preschool-aged children. J. Am. Diet. Assoc. 2007, 107, 929–934. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, T.M.; Yang, S.J.; Nicklas, T.A. Beverage intake among preschool children and its effect on weight status. Pediatrics 2006, 118, e1010–e1018. [Google Scholar] [CrossRef] [PubMed]
- Blum, J.W.; Jacobsen, D.J.; Donnelly, J.E. Beverage consumption patterns in elementary school aged children across a two-year period. J. Am. Coll. Nutr. 2005, 24, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Newby, P.K.; Peterson, K.E.; Berkey, C.S.; Leppert, J.; Willett, W.C.; Colditz, G.A. Beverage consumption is not associated with changes in weight and body mass index among low income preschool children in North Dakota. J. Am. Diet. Assoc. 2004, 104, 1086–1094. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Artalejo, F.; García, E.L.; Gorgojo, L.; Garcés, C.; Royo, M.A.; Martín Moreno, J.M.; Benavente, M.; Macías, A.; de Oya, M. Investigators of the Four Provinces Study. Consumption of bakery products, sweetened soft drinks and yogurt among children aged 6–7 years: Association with nutrient intake and overall diet quality. Br. J. Nutr. 2003, 89, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Grimes, C.A.; Riddell, L.J.; Campbell, K.J.; Nowson, C.A. Dietary salt intake, sugar-sweetened beverage consumption, and obesity risk. Pediatrics 2013, 131, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Han, E.; Powell, L.M. Consumption patterns of sugar-sweetened beverages in the United States. J. Acad. Nutr. Diet. 2013, 113, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Louie, J.C.; Flood, V.M.; Hector, D.J.; Rangan, A.M.; Gill, T.P. Dairy consumption and overweight and obesity: A systematic review of prospective cohort studies. Obes. Rev. 2011, 12, e582–e592. [Google Scholar] [CrossRef] [PubMed]
- Huh, S.Y.; Rifas-Shiman, S.L.; Rich-Edwards, J.W.; Taveras, E.M.; Gillman, M.W. Prospective association between milk intake and adiposity in preschool-aged children. J. Am. Diet. Assoc. 2010, 110, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Saelens, B.E.; Couch, S.C.; Wosje, K.S.; Stark, L.J.; Daniels, S.R. Relations among milk and non-milk beverage consumption, calcium, and relative weight in high-weight status children. J. Clin. Psychol. Med. Settings 2006, 13, 117–125. [Google Scholar] [CrossRef]
- Fulgoni, V.L.; Quann, E.E. National trends in beverage consumption in children from birth to 5 years: Analysis of NHANES across three decades. Nutr. J. 2012, 11, 92. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Early Childhood Obesity Prevention Policies; The National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Dove, E.R.; Hodgson, J.M.; Puddey, I.B.; Beilin, L.J.; Lee, Y.P.; Mori, T.A. Skim milk compared with a fruit drink acutely reduces appetite and energy intake in overweight men and women. Am. J. Clin. Nutr. 2009, 90, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Mehrabani, S.; Salehi-Abargouei, A.; Asemi, M.; Mehrabani, S.; Feizi, A.; Safavi, S.M. Effect of low-fat milk consumption compared to apple Juice and water on the energy intake among 10–12-year-old obese boys: A three-way cross-over clinical trial. Int. J. Prev. Med. 2014, 5, 405–411. [Google Scholar]
- Rehm, C.D.; Drewnowski, A.; Monsivais, P. Potential population-level nutritional impact of replacing whole and reduced-fat milk with low-fat and skim milk among US children aged 2–19 years. J. Nutr. Educ. Behav. 2015, 47, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Poppitt, S.D.; Swann, D.L.; Black, A.E.; Prentice, A.M. Assessment of selective under-reporting of food intake by obese women in a metabolic facility. Int. J. Obes. 1998, 22, 303–311. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poppitt, S.D. Beverage Consumption: Are Alcoholic and Sugary Drinks Tipping the Balance towards Overweight and Obesity? Nutrients 2015, 7, 6700-6718. https://doi.org/10.3390/nu7085304
Poppitt SD. Beverage Consumption: Are Alcoholic and Sugary Drinks Tipping the Balance towards Overweight and Obesity? Nutrients. 2015; 7(8):6700-6718. https://doi.org/10.3390/nu7085304
Chicago/Turabian StylePoppitt, Sally D. 2015. "Beverage Consumption: Are Alcoholic and Sugary Drinks Tipping the Balance towards Overweight and Obesity?" Nutrients 7, no. 8: 6700-6718. https://doi.org/10.3390/nu7085304
APA StylePoppitt, S. D. (2015). Beverage Consumption: Are Alcoholic and Sugary Drinks Tipping the Balance towards Overweight and Obesity? Nutrients, 7(8), 6700-6718. https://doi.org/10.3390/nu7085304




