A Systematic Review of the Efficacy of Bioactive Compounds in Cardiovascular Disease: Phenolic Compounds
Abstract
:1. Introduction
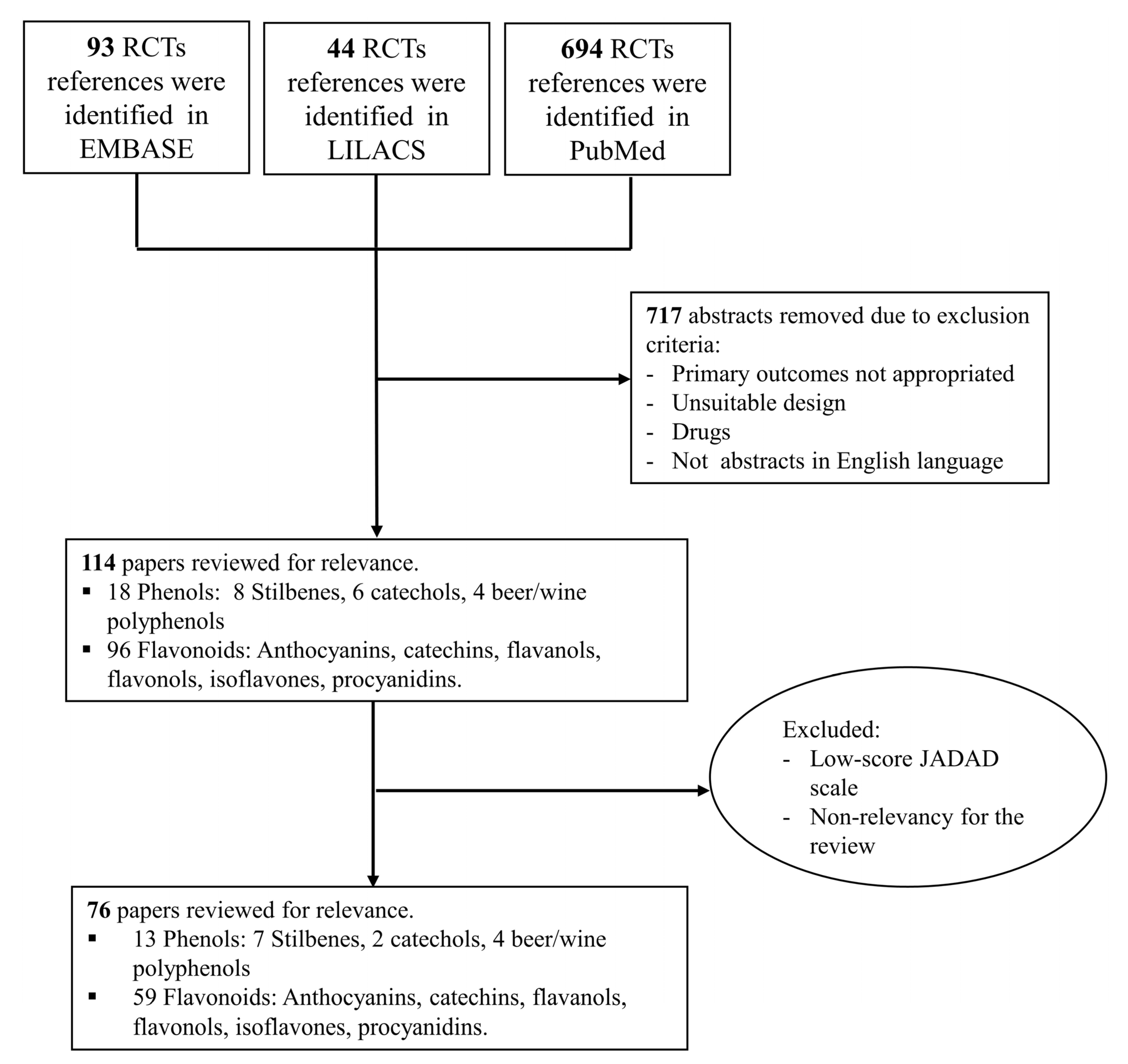
2. Methodology
2.1. Search Equation
2.2. Selection and Evaluation
3. Results and Discussion
| Group (Class) | Author/Date | Jadad Score | Design (Follow up) | (n) Population | Intervention | Outcomes | Significant Results |
|---|---|---|---|---|---|---|---|
| Phenols (Stilbenes) | Wong et al. (2011) [19] | 5 | 2B, X (1 h) | (19) Overweight/obese + ↑ BP men or post-menopausal women | 30 mg, 90 mg, 270 mg RSV vs. PCB | EF | ↑ EF, more with highest dose |
| Phenols (Stilbenes) | Wong et al. (2013) [20] * | 5 | 2B, X (1 h) | (28) obese subjects | Acute intervention: 75 mg/trans-resveratrol (Resvida) vs. PCB after chronic intervention | FMD | ↑ FMD |
| Phenols (Stilbenes) | Wong et al. (2013) [20] * | 5 | 2B, X (6 weeks) | (28) obese subjects | 75 mg/day trans-resveratrol (Resvida) vs. PCB | BP, AR, BMI, FMD | ↑ FMD |
| Phenols (Stilbenes) | Bo et al. (2013) [21] | 5 | 2B, X (60 days(wash-out 30 days)) | (50) Healthy smokers | 500 mg RSV/d vs. PCB | BP, Anthropometry, lipids profile, CHO metabolism, TAS, hsCRP, | ↓ hsCRP, TAG, ↑ TAS |
| Phenols (Stilbenes) | Militaru et al. (2013) [22] | 3 | 2B, Ctrl, PA (60 days) | (166) BMI 24–27 kg/m2, stable angina pectoris | 20 mg/day RSV, 20 mg/day RSV + 112 mg/day CF, 112 mg/day CF | Lipids profile, hsCRP, left ventricular function markers | ↓ TC, TAG greater in RSV, hsCRP greater in CF, NT-proBNP more effective RSV+CF |
| Phenols (Stilbenes) | Tomé-Carneiro et al. (2013) [23] | 4 | 3B, PCB (1 year) | (75) Stable CAD patients | 350 mg/day GE, 350 mg/day GE-RES vs. PCB (6 months); double dose next 6 months | PBMCs, inflammatory and fibrinolytic biomarkers | ↑ adiponectin, ↓ PAI-1, significantly activated or inhibited 6 key inflammation-related transcription factors in PBMCs |
| Phenols (Stilbenes) | Tomé-Carneiro et al. (2012) [24] | 4 | 3B, PCB (6 months) | (75) Primary prevention of CVD | 350 mg/day GE, 350 mg/day GE-RES vs. PCB | Lipids profile, oxidized LDL | ↓ LDLc, ApoB, LDLox and LDLox/ApoB ratio, ↑ nonHDLc/ApoB ratio in GE-RES |
| Phenols (Stilbenes) | Tomé-Carneiro et al. (2013) [25] | 5 | 3B, PCB, dose–response (1 year) | (35) T2D, HT with CAD | 350 mg/day GE, 350 mg/day GE-RES vs. PCB (6 months); double dose next 6 months | PBMCs, inflammatory, fibrinolytic biomarkers | ↓ CCL3, IL-1β, TNF-α expression, ↑ transcriptional repressor LRRFIP-1 in PBMCs with GE-RES |
| Phenols (Stilbenes) | Tomé-Carneiro et al. (2012) [26] | 4 | 3B, PCB (1 year) | (75) Primary prevention of CVD | 350 mg/day GE, 350 mg/day GE-RES vs. PCB (6 months); double dose next 6 months | Inflammatory and fibrinolytic biomarkers | ↓ CRP, TNF-α, PAI-1, IL-6/IL-10 ratio, sICAM ↑ IL-10, adiponectin in GE-RES |
| Phenols (Catechols) | Alwi et al. (2008) [27] | 4 | 2B, PCB (2 months) | (75) ACS patients | 45 mg/day, 90 mg/day or 180 mg/day curcumin vs. PCB | Lipids profile | Not significant effect |
| Phenols (Catechols) | Chuengsamarn et al. (2014) [28] | 5 | 2B, PCB (6 months) | (240) T2D patients | 750 mg/day curcumin vs. PCB | BP, anthropometry, lipids profile, adiponectin, leptin, CHO metabolism, PWV, uric acid | ↓ PWV, HOMA, TAG, uric acid, abdominal obesity and leptin, ↑ adiponectin. |
| Polyphenols (Wine/beer) | Botden et al. (2012) [29] | 4 | 2B, PCB, three-period X (4 weeks) | (61) HT subjects | 280 mg/day red wine polyphenols or 560 mg/day red wine polyphenols vs. PCB | BP | No significant effect |
| Polyphenols (Wine/beer) | Chiva-Blanch et al. (2014) [30] | 4 | 2B, PCB, X (4 weeks) | (36) High risk of CVD males | Beer (30 g alcohol/day), the equivalent amount of polyphenols in the form of non-alcoholic beer, or gin (30 g alcohol/day) | Circulating endothelial progenitor cells and EPC-mobilizing factors | Beer and non-alcoholic beer interventions, ↑-circulating EPC. No significant differences were observed after the gin period |
| Polyphenols (Wine/beer) | Chiva-Blanch et al. (2012) [31] | 3 | X (4 weeks) | (67) High risk of CVD males | Red wine (30 g alcohol/day), the equivalent amount of dealcoholized red wine, or gin (30 g alcohol/day) | BP and plasma nitric oxide | Dealcoholized red wine ↓ DBP and SBP |
| Polyphenols (Wine/beer) | Chiva-Blanch et al. (2012) [32] | 3 | X (4 weeks) | (67) High risk of CVD males | Red wine (30 g alcohol/day), the equivalent amount of dealcoholized red wine, or gin (30 g alcohol/day) | Inflammatory biomarkers | Alcohol ↑ IL-10 and ↓ macrophage-derived chemokine concentrations. Phenolic compounds of Red wine ↓ serum concentrations of ICAM-1, E-selectin, and IL-6 |
| Group (Class) | Author/ Date | Jadad Score | Design (Follow up) | (n) Population | Intervention | Outcomes | Significant Results |
|---|---|---|---|---|---|---|---|
| Flavonoids (Anthocyanins) | Kuntz (2014) [33] | 4 | 2B, PCB, X (14 days) | (30) Healthy females | 330 mL/day beverages (PCB, juice or smoothie with 8.9, 983.7 and 840.9 mg/L ACN, respectively) | Inflammatory and oxidative stress biomarkers | ↑ SOD and CAT after ACN. ↓ MDA after ACN ingestion. |
| Flavonoids (Anthocyanins) | Curtis et al. (2009) [34] | 5 | PCB, PA (12 weeks) | (57) Postmenopausal women | 500 mg/day ACN vs. PCB | BP, CHO metabolism, lipids profile, inflammatory biomarkers, platelet reactivity | No significant effect |
| Flavonoids (Anthocyanins) | Hassellund et al. (2013) [35] | 5 | 2B, PCB, X (4 weeks) | (31) Pre-hypertensive males | 640 mg/day ACN vs. PCB | Lipids profile, CHO metabolism, inflammatory and oxidative stress biomarkers | ↑ HDLc and glucose after anthocyanin versus PCB treatment. No effects were observed on inflammation or oxidative stress in vivo, except for vWf |
| Flavonoids (Anthocyanins) | Dohadwala et al. (2011) [36] | 4 | Open-label, (2 and 4 hour acute study) | (15) CAD subjects | 835 mg total polyphenols, 94 mg anthocyanins vs. PCB | Vascular function | No significant effect |
| Flavonoids (Anthocyanins) | Dohadwala et al. (2011) [36] | 4 | X, 2B, PCB (4 weeks, 2 week washout) | (44) CAD subjects | 835 mg total polyphenols, 94 mg anthocyanins vs. PCB | Vascular function | ↓ Carotid femoral pulse wave activity |
| Flavonoids (Catechins) | Miyazaki et al. (2013) [37] | 4 | 2B, PCB (14 weeks) | (52) Healthy subjects | 630.9 mg/day Green Tea Catechins vs. Ctrl | CVD risk markers | No significant effect |
| Flavonoids (Catechins) | de Maat et al. (2000) [38] | 3 | 1B, PCB, PA (4 weeks) | (64) Healthy subjects | Black tea (3 g/day), green tea (3 g/day), green tea polyphenol isolate capsules (3.6 mg/day) and mineral water. | Inflammatory and endothelial markers | Negative correlation between the levels of the antioxidant β-carotene and the inflammation markers IL6 and fibrinogen |
| Flavonoids (Catechins) | Widmer et al. (2013) [39] | 3 | 2B, Ctrl (4 months) | (52) Early atherosclerosis | 30 mL/day simple Olive Oil vs. 30 mL/day of EGCG-supplemented Olive Oil | EF, inflammation and oxidative stress | Only significant when merging data of both groups the EF was improved. |
| Flavonoids (Catechins) | Nagao et al. (2007) [40] | 4 | 2B, PA (12 weeks) | (240) Visceral fat-type obesity | Green tea containing 583 mg/day catechins (catechin group) vs. 96 mg/day catechins (Ctrl group) | Anthropometric measurements, body fat composition and CVD risk | ↓ body weight, BMI, body fat ratio, body fat mass, waist circumference, hip circumference, visceral fat area, and subcutaneous fat area, SBP, LDLc |
| Flavonoids (Flavanols) | Farouque et al. (2006) [41] | 5 | 2B, PCB (6 weeks) | (40) Healthy males | Flavanol-rich chocolate bar and cocoa beverage (total flavanols, 444 mg/day) vs. matching isocaloric PCBs (total flavanols, 19.6 mg/day) | EF and adhesion molecules | No significant effect |
| Flavonoids (Flavanols) | Berry et al. (2010) [42] * | 4 | 2B, X (2 h, 3–7 days washout) | (21) overweight/obese subjects | HF, 701 mg or LF, 22 mg cocoa | BP, HR, FMD | ↑ DBP after exercise were attenuate by HF, improvement of FMD with HF |
| Flavonoids (Flavanols) | Davison et al. 2008 [43] * | 4 | 2B, PCB, PA (12 weeks) | (98) overweight/obese subjects | 902 mg cocoa flavanols/day vs. 36 mg cocoa flavanols/day With/without exercise protocol | BP, HDLc, LDLc, TG, HOMA, FMD | ↑ FMD at 6 and 12 weeks with HF vs. LF, ↑ DBP, BP mean, improvement in HOMA (independent of exercise) |
| Flavonoids (Flavanols) | West et al. (2014) [44] | 3 | 2B, PCB, X (4 weeks, 2 weeks washout) | (30) Middle-aged overweight | 37 g/day of dark chocolate and a sugar-free cocoa beverage (total flavanols = 814 mg/day) vs. low-flavonol chocolate and cocoa free beverage (total flavanols = 3 mg/day) | EF, BP | ↑ Basal and peak diameter of the brachial artery and basal blood flow volume. |
| Flavonoids (Flavanols) | Faridi et al. (2008) [45] | 4 | X, Ctrl, 1B (1 days, 7 days washout) | (45) Overweight subjects | Solid dark chocolate bar (821 mg flavanols) vs. cocoa-free PCB bar (0 mg flavanols) | EF, BP | Solid dark chocolate improved EF; also ↓ BP |
| Flavonoids (Flavanols) | Faridi et al. (2008) [45] | 4 | X, Ctrl, 1B (1 days, 7 days washout) | (44) Overweight subjects | Sugar-free cocoa (805.2 mg flavanols), sugared cocoa (805.2 mg flavanols), vs. PCB (0 mg flavanols). | EF, BP | Liquid cocoa ingestion improved EF; sugar-free cocoa ↓ BP |
| Flavonoids (Flavanols) | Davison et al. (2010) [46] | 3 | 2B, PA (6 weeks) | (52) Men and postmenopausal women with untreated mild HT | 33, 372, 712 or 1052 mg/day of cocoa flavanols | 24-h BP | No significant effect |
| Flavonoids (Flavanols) | Grassi et al. (2008) [47] | 3 | X, Ctrl, 1B (15 days) | (19) HT with Impaired glucose tolerance | Flavonol-rich dark chocolate (110.9 mg epicatechin, 36.12 mg catechin, 2.5 mg quercetin, 0.03 mg kaempferol, and 0.2 mg isorhamnetin)/d or flavonol-free white chocolate (0.04 mg/day catechins) | EF, IR, β-cell function, BP, CRP, TC | ↓ IR, BP, TC, LDLc. ↑ insulin sensitivity, EF |
| Flavonoids (Flavanols) | Flammer et al. (2012) [48] | 3 | 2B, PCB (2 hours) | (20) CHF patients | 40 g Flavonol rich chocolate (624 mg total flavanols) vs. 28.4 g Ctrl chocolate (0 mg flavanols | EF and platelet function in the short term | Improvement of vascular function in patients with CHF |
| Flavonoids (Flavanols) | Flammer et al. (2012) [48] | 3 | 2B, PCB (2 and 4 weeks) | (20) CHF patients | 40 g/day Flavonol rich chocolate (624 mg total flavanols) vs. 28.4 g/day Ctrl chocolate (0 mg flavanols) | EF and platelet function in long term by FMD | Improvement of vascular function in patients with CHF |
| Flavonoids (Flavanols) | Heiss et al. (2010) [49] | 3 | Ctrl, 2B, X (30 days) | (16) CAD patients | High-flavanol intervention (375 mg/day) and a macronutrient- and micronutrient-matched low-flavanol intervention (9 mg/day) twice daily | EF and enhancement and function of circulating angiogenic cells | ↑ EF, CD34+/KDR+-Circulating angiogenic cells. ↓ SBP |
| Flavonoids (Flavanols) | Horn et al. (2013) [50] | 3 | 2B, X (30 days) | (16) CAD patients | High-flavanol intervention (375 mg/day) and a macronutrient- and micronutrient-matched low-flavanol intervention (9 mg/day) twice daily | Circulating endothelial micro particles, markers of endothelial integrity, EF | ↑ Endothelial micro-particles and EF. Improvement of endothelial integrity |
| Flavonoids (Flavanols) | Balzer et al. (2008) [51] | 5 | 2B, PCB, three-period X (2 h) | (10) Diabetic subjects | Single-dose ingestion of cocoa, containing increasing concentrations of flavanols (75, 371, and 963 mg) | EF | Single ingestion of flavanol-containing cocoa was dose-dependently acute increases in circulating flavanols and EF |
| Flavonoids (Flavanols) | Balzer et al. (2008) [51] | 5 | 2B, PCB, PA (30 days) | (41) Diabetic subjects | 963 mg/day Flavanol-rich cocoa vs. nutrient-matched Ctrl (75 mg/day flavanols) | EF | Flavanol-containing cocoa ↑ baseline EF |
| Flavonoids (Flavonols) | Larson et al. (2012) [52] | 3 | 2B, PCB, X (1 days, 7 days washout) | (5) Healthy males | 1095 mg quercetin aglycone vs. PCB | Angiotensin-converting enzyme, endothelin-1, BP | No significant effect |
| Flavonoids (Flavonols) | Conquer et al. (1998) [53] | 3 | 2B (28 days) | (27) Healthy subjects | 4 capsules (1.0 g quercetin/day) vs. rice flour PCB | BP, lipids profile, thrombogenic risk factors | No significant effect |
| Flavonoids (Flavonols) | Suomela et al. (2006) [54] | 3 | 2B, PCB, X (4 weeks, 4 weeks washout) | (14) Healthy males | Oat meal with 78 mg/day flavonol aglycones (sea buckthorn) vs. Ctrl | CVD risk markers | No significant effect |
| Flavonoids (Flavonols) | Edwards et al. (2007) [55] | 3 | 2B, PCB, X (28 days) | (41) Prehypertension and hypertension | 730 mg quercetin/day vs. PCB | BP, oxidative stress | ↓ BP in hypertensive group |
| Flavonoids (Flavonols) | Larson et al. (2012) [52] | 3 | 2B, PCB, X (1 days, 2 days washout) | (12) HT stage 1 males | 1095 mg quercetin aglycone vs. PCB | Angiotensin-converting enzyme, endothelin-1, BP | ↓ BP in Hypertensive men |
| Group (Class) | Author/Date | Jadad Score | Design (Follow up) | (n) Population | Intervention | Outcomes | Significant Results |
|---|---|---|---|---|---|---|---|
| Flavonoids (Isoflavones) | McVeigh et al. (2006) [56] | 3 | 1B, X (57 days, 4 weeks washout) | (35) Healthy males | Milk protein isolate (MPI), low-isoflavone soy protein isolate (low-iso SPI; 1.64 ± 0.19 mg aglycone isoflavones/day), and high-isoflavone SPI (high-iso SPI; 61.7 ± 7.4 mg aglycone isoflavones/day) | Lipids profile | ↓ TC/HDLc, LDLc/HDLc, and Apo B/Apo A-I with both SPI treatments than with MPI treatment |
| Flavonoids (Isoflavones) | Sanders et al. (2002) [57] | 3 | X (17 days, 25 days washout) | (22) Healthy subjects | 56 vs. 2 mg isoflavones/day | Lipids profile, fibrinogen, and active TGF-β, factor VII coagulant and PAI-1 | ↑ HDL and Apo A1 in high-isoflavone |
| Flavonoids (Isoflavones) | Thorp et al. (2008) [58] * | 5 | 2B, PCB, X (6 weeks) | (91) Hypercholesterolemia | 24 g SP+70–80 mg ISOs (diet S) vs. 12 g SP + 12 g dairy protein (DP) + 70–80 mg ISOs (diet SD) vs. 24 g DP without ISOs (diet D) | HDLc, LDLc, TC | No significant effect |
| Flavonoids (Isoflavones) | Atkinson et al. (2004) [59] | 5 | 2B, PCB (12 months) | (205) Female | 43.5 mg red clover-derived isoflavones/day vs. PCB | Lipids profile, BP, fibrinogen and PAI-1 | No significant effect |
| Flavonoids (Isoflavones) | Marini et al. (2010) [60] | 5 | 2B, PCB (24 months) | (138) Females with low bone mass | 54 mg/day genistein aglycone vs. PCB | Lipids profile, CHO metabolism, HOMA, fibrinogen, osteoprotegerin and homocysteine | ↓ fasting glucose and insulin, HOMA, fibrinogen and homocysteine |
| Flavonoids (Isoflavones) | Hodis et al. (2011) [61] | 5 | 2B, PCB (2 years) | (350) Postmenopausal women | 25 g/day soy protein (91 mg/day aglycone isoflavone equivalents) vs. PCB | Atherosclerosis progression | No significant effect |
| Flavonoids (Isoflavones) | Atteritano et al. (2007) [62] | 5 | 2B, PCB (24 months) | (191) Postmenopausal women | 54 mg/day genistein vs. PCB | Lipids profile, CHO metabolism, HOMA, fibrinogen, sVCAM-1, sICAM-1, 8-iso-PGF2α, and osteoprotegerin | ↓ Fasting glucose and insulin as well as HOMA, fibrinogen, 8-iso-PGF2α, sICAM-1, and sVCAM-1. ↑ Serum osteoprotegerin |
| Flavonoids (Isoflavones) | Garrido et al. (2006) [63] | 3 | PCB (12 weeks) | (29) Postmenopausal women | 100 mg/day isoflavones vs. PCB | Lipids profile, CHO metabolism and platelet thromboxane A2 receptor density. BP, BMI, subcutaneous fat | ↓ Thromboxane A2 after the experimental treatment. |
| Flavonoids (Isoflavones) | Hall et al. (2005) [64] | 4 | 2B, PCB, X (8 weeks, 8 weeks washout) | (117) Postmenopausal women | Isoflavone-enriched (genistein-to-daidzein ratio of 2:1; 50 mg/day) vs. PCB cereal | Inflammatory and vascular homeostasis biomarkers | ↓ CRP |
| Flavonoids (Isoflavones) | Rios et al. (2008) [65] | 3 | 2B, PCB (6 months) | (47) Postmenopausal women | 40 mg/day isoflavone vs. casein PCB | Lipids profile | No significant effect |
| Flavonoids (Isoflavones) | Villa et al. (2009) [66] | 3 | PCB (24 weeks) | (50) Postmenopausal women | 54 mg/day genistein vs. PCB | Anthropometric measures, lipid profile, CHO metabolism and C-peptide evaluation, IR and EF | HOMA and fasting glucose levels significantly improved |
| Flavonoids (Isoflavones) | Liu et al. (2012) [67] | 4 | 2B, PCB (6 months) | (180) Postmenopausal women | 15 g/day soy protein and 100 mg/day isoflavone (Soy group), vs. 15 g/day milk protein and 100 mg/day isoflavone (Iso group) vs. 15 g/day milk protein (PCB) | Lipids profile, inflammatory markers and composite cardiovascular | No significant effect |
| Flavonoids (Isoflavones) | Yang et al. (2012) [68] | 3 | Open-labelled, prospective (24 week) | (130) Healthy Taiwanese postmenopausal women | 35 mg/day vs. 70 mg/day soy extractª | Lipids profile | ↓ TC, LDLc in patients with TC >200 mg/dL |
| Flavonoids (Isoflavones) | Liu et al. (2013) [67] | 5 | 2B, PCB (6 months) | (270) Pre-hypertensive women | 40 g/day soy flour (whole soy group), 40 g/day low-fat milk powder + 63 mg/day daidzein (daidzein group), vs. 40 g/day low-fat milk powder (PCB) | Anthropometric indicators and body composition | No significant effect |
| Flavonoids (Isoflavones) | Aubertin-Leheudre et al. (2008) [69] | 3 | 2B, PCB (6 months) | (50) Obese postmenopausal women | 70 mg/day isoflavones vs. PCB | Body composition (DXA), and Lipid profile and CHO metabolism | No significant effect |
| Flavonoids (Isoflavones) | Choquette et al. (2011) [70] | 4 | 2B, PCB (6 months) | (100) Overweight to obese postmenopausal women | PCB or isoflavones (70 mg/day) or exercise + PCB or exercise + isoflavones (70 mg/day). Exercise consisted of three weekly sessions of resistance training and aerobics | Body composition, lipids profile, CHO metabolism and HOMA. | No significant effect |
| Flavonoids (Isoflavones) | Aubertin-Leheudre M et al. (2007) [71] | 3 | 2B, PCB (12 months) | (56) Obese postmenopausal women | 70 mg/day isoflavonesb (+weight loss exercise program from the 6 months) vs. PCB | Anthropometry, lipids profile, CHO metabolism, CRP | ↓ body weight, BMI, total and abdominal FM (kg and %), ↑ FFM/FM ratio with exercise program |
| Flavonoids (Isoflavones) | Hodgson et al. (1999) [72] | 3 | 2B, PCB, PA (8 weeks) | (59) High-normal BP | 55 mg/day isoflavonoid vs. PCB | 8-iso-PGF2α | No significant effect |
| Flavonoids (Isoflavones) | Sagara et al. (2004) [73] | 3 | PCB, 2B, PA (5 weeks) | (61) Men with relatively higher BP or TC | Diets containing at least 20 g/day soy protein + 80 mg/day isoflavones vs. PCB diets | BP and Lipid profile | ↓ BP, TC and non-HDLc and ↑ HDLc. |
| Flavonoids (Isoflavones) | Clerici et al. (2007) [74] | 4 | Ctrl, PA (8 weeks) | (62) Hypercholesterolemia | 80 g serving/d (33 mg/day isoflavones + negligible soy protein + led to a serum isoflavone concentration of 222 +/- 21 nmol/L) vs. Ctrl group | Lipids profile, hsCRP, urinary 8-iso-PGF2α, and EF | ↓ LDLc, TC |
| Flavonoids (Isoflavones) | Meyer et al. (2004) [75] | 3 | PCB, X (5 weeks, without washout) | (23) Mildly hypercholesterolemic and/or hypertensive | Soy-based milk (30 g/day soy protein + 80 mg/day isoflavones) + yoghurt (treatment) vs. equivalent dairy products (Ctrl) | BP, arterial compliance, lipid profile, fatty acids | No significant effect |
| Flavonoids (Isoflavones) | Jenkins et al. (2002) [76] | 3 | 1B (1 month, 2 weeks washout) | (41) Postmenopausal women with hypercholesterolemia | A low-fat dairy food Ctrl diet, high- (50 g soy protein and 73 mg isoflavones/day), low- (52 g soy protein and 10 mg isoflavones/day) isoflavone soy food diets | BP, lipids profile, oxidized LDL, calculated CAD risk | Soy diets ↓ TC estimated CAD risk, TC/HDLc, LDLc/HDLc, ApoB/A-I. Blood lipid and BP changes, the calculated CAD risk ↓ with the soy diets |
| Flavonoids (Isoflavones) | Blum et al. (2003) [77] | 4 | 2B, PCB, X (6 weeks, 1 month washout) | (24) Postmenopausal women with hypercholesterolemia | 25 g/day soy protein vs. PCB | Vascular inflammation biomarkers | No significant effect |
| Flavonoids (Isoflavones) | Teede et al. (2006) [78] | 3 | Ctrl, X (3 months) | (41) Hypertensive postmenopausal | Soy cereal (40 g/day soy protein + 118 mg/day isoflavones) vs. gluten PCB cereal | BP, arterial function | ↑ 24 hour HR, area under curve of 24 h SBP |
| Flavonoids (Isoflavones) | Cicero et al. (2013) [79] | 4 | Ctrl, 1B prospective study with PA (12 weeks) | (40) Mildly dyslipidemic postmenopausal women | 60 mg/day soy isoflavones + 500 mg/day berberine vs. PCB (1 tablet/d) | BP, HOMA, lipids profile, metalloproteinase | Isoflavones-berberine experienced a significant improvement in plasma lipid and metalloproteinase serum levels. |
| Flavonoids (Isoflavones) | Curtis et al. (2013) [80] | 5 | 2B, PCB, PA (1 year) | (180) Postmenopausal women with T2D | 27 g/day flavonoid-enriched chocolate (containing 850 mg flavan-3-ols [90 mg epicatechin] + 100 mg isoflavones [aglycone equivalents)] /d) vs. PCB. | Intima-media thickness of the common carotid artery, pulse wave velocity, augmentation index, BP, and vascular biomarkers | Only pulse pressure variability improved |
| Flavonoids (Isoflavones) | Curtis et al. (2013) [80] | 5 | PA, PCB (1 year) | (93) Postmenopausal women with T2D | 27 g/day flavonoid-enriched chocolate (containing 850 mg flavan-3-ols [90 mg epicatechin] + 100 mg isoflavones [aglycone equivalents)] /d) vs. PCB. | HOMA and QUICKI, lipid profile, BP | Estimated 10-year total coronary heart disease risk (derived from UK Prospective Diabetes Study algorithm) was attenuated after flavonoid intervention |
| Flavonoids (Isoflavones) | Chan et al. (2008) [81] | 5 | 2B, PCB (12 weeks) | (102) Prior ischemic stroke | 80 mg/day isoflavone supplement vs. PCB | EF, nitro-glycerine-mediated dilatation, BP, HR, CHO metabolism, haemoglobin A1c, and oxidative stress biomarkers | ↓ serum hsCRP and improved brachial EF in patients with clinically manifest atherosclerosis |
| Flavonoids (Isoflavones) | Webb et al. (2008) [82] | 4 | 2B, PA (5 days) | (71) Subjects with CAD | Isoflavone-intact soy protein (75 mg/day of isoflavones) vs. isoflavone-free PCB | Stimulated coronary blood flow, Basal and stimulated coronary artery luminal diameters | No significant effect |
| Flavonoids (Isoflavones) | Fanti et al. (2006) [83] | 3 | 2B, Crtl, prospective, pilot study (8 weeks) | (32) ESRD patients with systemic inflammation | Nutritional supplements (soy groups) containing 26–54 mg isoflavones aglycones vs. isoflavone-free milk-based supplements (Ctrl group) | Inflammatory biomarkers | Inverse correlation between blood isoflavones levels and CRP, positive correlation between blood isoflavones levels and IGF-1 |
| Flavonoids (Procyanidins) | Ras et al. (2013) [84] | 5 | 2B, PCB, PA (8 weeks) | (70) Healthy subjects | 300 mg/day Grape Seed Extract vs. PCB | BP | No significant effect |
| Flavonoids (Procyanidins) | Yubero et al. (2013) [85] | 3 | 2B, PCB, X (56 days) | (60) Healthy subjects | 700 mg/day the Grape Extract (Eminol®) vs. PCB | CVD risk and oxidative stress markers | ↓TC, LDLc and ↑ TAC and vitamin E. |
| Flavonoids (Procyanidins) | Asher et al. (2012) [86] | 5 | 2B, PCB, four-period X (3.5 days, 4 days washout) | (21) Pre-hypertensive or mildly hypertensive adults | Hawthorn Extract (1000, 1500, and 2500 mg/day) vs. PCB | EF and nitric oxide release | No significant effect |
| Flavonoids (Procyanidins) | Liu et al. (2004) [87] | 3 | PCB, 2B, PA (12 weeks) | (58) HT subjects | 100 mg/day Pycnogenol vs. PCB | Endothelin | ↓ Calcium antagonist nifedipine. ↓ endothelin-1 concentration and ↑ of 6-keto prostaglandin F1a. |
| Flavonoids (Procyanidins) | Enseleit et al. (2012) [88] | 5 | 2B, PCB, X (8 weeks, 2 weeks washout) | (23) Patients with stable CAD | 200 mg/day Pycnogenol vs. PCB | EF, oxidation and inflammatory markers, platelet adhesion and 24 h BP | EF improvement. ↓ 8-iso-PGF2α |
| Flavonoids (Procyanidins) | Mellen et al. (2010) [89] | 3 | 2B, PCB, X (4 weeks, 4 weeks washout) | (50) Patients with CAD | 1300 mg/day muscadine grape seed vs. PCB | EF, oxidation and inflammatory markers, antioxidant status | No significant effect |
| Flavonoids (Procyanidins) | Tauchert et al. (2002) [90] | 3 | 2B, PCB (16 weeks) | (209) Chronic stable heart failure patients | 1800 mg/day crataegus extract WS 1442 or 900 mg/day crataegus extract WS 1442 vs. PCB | Typical heart failure symptoms | Typical heart failure symptoms as rated by the patients were ↓ to a greater extent |
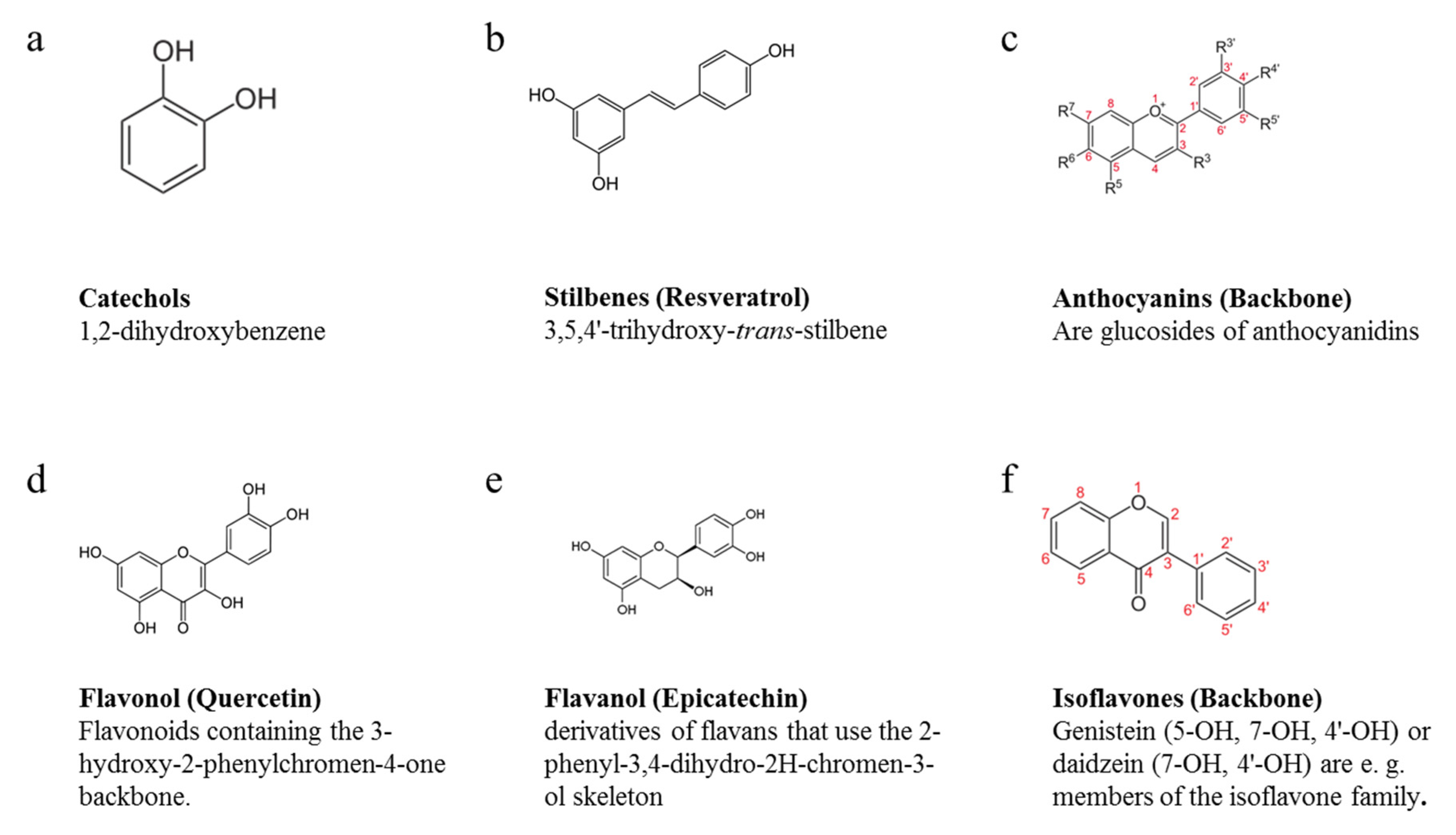
3.1. Simple Phenols
3.1.1. Stilbenes
3.1.2. Catechols
3.1.3. Beer or Wine Polyphenols
3.2. Polyphenols
3.2.1. Anthocyanins
3.2.2. Catechins
3.2.3. Flavanols
3.2.4. Flavonols
3.2.5. Isoflavones
3.2.6. Procyanidins
4. Limitations and Future Perspectives
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Martínez-Augustin, O.; Aguilera, C.M.; Gil-campos, M.; Sánchez de Medina, F.; Gil, A. Bioactive anti-obesity food components. Int. J. Vitam. Nutr. Res. 2012, 82, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Organization, W.H. Obesity and overweight. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/ (accessed on 14 October 2014).
- Perk, J.; de Backer, G.; Gohlke, H.; Graham, I.; Reiner, Ž.; Verschuren, M.; Albus, C.; Benlian, P.; Boysen, G.; Cifkova, R.; et al. European Guidelines on cardiovascular disease prevention in clinical practice (version 2012). Eur. Heart J. 2012, 33, 1635–1701. [Google Scholar] [PubMed]
- Stone, N.J.; Robinson, J.G.; Lichtenstein, A.H.; Bairey Merz, C.N.; Blum, C.B.; Eckel, R.H.; Goldberg, A.C.; Gordon, D.; Levy, D.; Lloyd-Jones, D.M.; et al. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: A report of the American college of cardiology/American heart association task force on practice guidelines. J. Am. Coll. Cardiol. 2014, 63, 2889–2934. [Google Scholar] [CrossRef] [PubMed]
- Kitts, D.D. Bioactive substances in food: Identification and potential uses. Can. J. Physiol. Pharmacol. 1994, 72, 423–434. [Google Scholar] [CrossRef] [PubMed]
- Kris-Etherton, P.M.; Hecker, K.D.; Bonanome, A.; Coval, S.M.; Binkoski, A.E.; Hilpert, K.F.; Griel, A.E.; Etherton, T.D. Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am. J. Med. 2002, 113, 71S–88S. [Google Scholar] [CrossRef]
- Sarriá, B.; Martínez-López, S.; Sierra-Cinos, J.L.; Garcia-Diz, L.; Goya, L.; Mateos, R.; Bravo, L. Effects of bioactive constituents in functional cocoa products on cardiovascular health in humans. Food Chem. 2015, 174, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Huxley, R.R.; Neil, H.A.W. The relation between dietary flavonol intake and coronary heart disease mortality: A meta-analysis of prospective cohort studies. Eur. J. Clin. Nutr. 2003, 57, 904–908. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Liu, X.; Bai, Y.; Wang, X.; Sun, K.; Chen, J.; Hui, R. Effect of oral isoflavone supplementation on vascular endothelial function in postmenopausal women: A meta-analysis of randomized placebo-controlled trials. Am. J. Clin. Nutr. 2009, 91, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-M.; Nie, Z.-L.; Zhou, B.; Lian, X.-Q.; Zhao, H.; Gao, W.; Wang, Y.-S.; Jia, E.-Z.; Wang, L.-S.; Yang, Z.-J. Flavonols intake and the risk of coronary heart disease: A meta-analysis of cohort studies. Atherosclerosis 2012, 222, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Hooper, L.; Kroon, P.A.; Rimm, E.B.; Cohn, J.S.; Harvey, I.; Le Cornu, K.A.; Ryder, J.J.; Hall, W.L.; Cassidy, A. Flavonoids, flavonoid-rich foods, and cardiovascular risk: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2008, 88, 38–50. [Google Scholar] [PubMed]
- FDA. FDA Basics—Dietary Supplements. Available online: http://www.fda.gov/AboutFDA/Transparency/Basics/ucm193949.htm (accessed on 13 Feburbary 2015).
- Lekakis, J.; Rallidis, L.S.; Andreadou, I.; Vamvakou, G.; Kazantzoglou, G.; Magiatis, P.; Skaltsounis, A.-L.; Kremastinos, D.T. Polyphenolic compounds from red grapes acutely improve endothelial function in patients with coronary heart disease. Eur. J. Cardiovasc. Prev. Rehabil. 2005, 12, 596–600. [Google Scholar] [CrossRef] [PubMed]
- Quiles, J.L.; Mesa, M.D.; Ramírez-Tortosa, C.L.; Aguilera, C.M.; Battino, M.; Gil, Á.; Ramírez-Tortosa, M.C. Curcuma longa extract supplementation reduces oxidative stress and attenuates aortic fatty streak development in rabbits. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 1225–1231. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, M.P.; McKay, D.L.; Blumberg, J.B. Flavonoid Basics: Chemistry, Sources, Mechanisms of Action, and Safety. J. Nutr. Gerontol. Geriatr. 2012, 31, 176–189. [Google Scholar] [CrossRef] [PubMed]
- Scottish Intercollegiate Guidelines Network. Risk Estimation and the Prevention of Cardiovascular Disease (Guideline 97); Scottish Intercollegiate Guidelines Network: Edinburgh, UK, 2007. [Google Scholar]
- Jadad, A.R.; Moore, R.A.; Carroll, D.; Jenkinson, C.; Reynolds, D.J.; Gavaghan, D.J.; McQuay, H.J. Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef]
- Uauy, R.E.A. Fats and fatty acids in human nutrition, Report of an expert consultation. FAO Food Nutr. Pap. 2008, 550, 189. [Google Scholar]
- Wong, R.H.X.; Howe, P.R.C.; Buckley, J.D.; Coates, A.M.; Kunz, I.; Berry, N.M. Acute resveratrol supplementation improves flow-mediated dilatation in overweight/obese individuals with mildly elevated blood pressure. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.H.X.; Berry, N.M.; Coates, A.M.; Buckley, J.D.; Bryan, J.; Kunz, I.; Howe, P.R.C. Chronic resveratrol consumption improves brachial flow-mediated dilatation in healthy obese adults. J. Hypertens. 2013, 31, 1819–1827. [Google Scholar] [CrossRef] [PubMed]
- Bo, S.; Ciccone, G.; Castiglione, A.; Gambino, R.; de Michieli, F.; Villois, P.; Durazzo, M.; Cavallo-Perin, P.; Cassader, M. Anti-inflammatory and antioxidant effects of resveratrol in healthy smokers a randomized, double-blind, placebo-controlled, cross-over trial. Curr. Med. Chem. 2013, 20, 1323–1331. [Google Scholar] [CrossRef] [PubMed]
- Militaru, C.; Donoiu, I.; Craciun, A.; Scorei, I.D.; Bulearca, A.M.; Scorei, R.I. Oral resveratrol and calcium fructoborate supplementation in subjects with stable angina pectoris: Effects on lipid profiles, inflammation markers, and quality of life. Nutrition 2013, 29, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Tomé-Carneiro, J.; Gonzálvez, M.; Larrosa, M.; Yáñez-Gascón, M.J.; García-Almagro, F.J.; Ruiz-Ros, J.A.; Tomás-Barberán, F.A.; García-Conesa, M.T.; Espín, J.C. Grape resveratrol increases serum adiponectin and downregulates inflammatory genes in peripheral blood mononuclear cells: A triple-blind, placebo-controlled, one-year clinical trial in patients with stable coronary artery disease. Cardiovasc. Drugs Ther. 2013, 27, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Tomé-Carneiro, J.; Gonzálvez, M.; Larrosa, M.; García-Almagro, F.J.; Avilés-Plaza, F.; Parra, S.; Yáñez-Gascón, M.J.; Ruiz-Ros, J.A.; García-Conesa, M.T.; Tomás-Barberán, F.A.; et al. Consumption of a grape extract supplement containing resveratrol decreases oxidized LDL and ApoB in patients undergoing primary prevention of cardiovascular disease: A triple-blind, 6-month follow-up, placebo-controlled, randomized trial. Mol. Nutr. Food Res. 2012, 56, 810–821. [Google Scholar] [CrossRef] [PubMed]
- Tomé-Carneiro, J.; Larrosa, M.; Yáñez-Gascón, M.J.; Dávalos, A.; Gil-Zamorano, J.; Gonzálvez, M.; García-Almagro, F.J.; Ruiz Ros, J.A.; Tomás-Barberán, F.A.; Espín, J.C.; et al. One-year supplementation with a grape extract containing resveratrol modulates inflammatory-related microRNAs and cytokines expression in peripheral blood mononuclear cells of type 2 diabetes and hypertensive patients with coronary artery disease. Pharmacol. Res. 2013, 72, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Tomé-Carneiro, J.; Gonzálvez, M.; Larrosa, M.; Yáñez-Gascón, M.J.; García-Almagro, F.J.; Ruiz-Ros, J.A.; García-Conesa, M.T.; Tomás-Barberán, F.A.; Espín, J.C. One-year consumption of a grape nutraceutical containing resveratrol improves the inflammatory and fibrinolytic status of patients in primary prevention of cardiovascular disease. Am. J. Cardiol. 2012, 110, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Alwi, I.; Santoso, T.; Suyono, S.; Sutrisna, B.; Suyatna, F.D.; Kresno, S.B.; Ernie, S. The effect of curcumin on lipid level in patients with acute coronary syndrome. Acta Med. Indones. 2008, 40, 201–210. [Google Scholar] [PubMed]
- Chuengsamarn, S.; Rattanamongkolgul, S.; Phonrat, B.; Tungtrongchitr, R.; Jirawatnotai, S. Reduction of atherogenic risk in patients with type 2 diabetes by curcuminoid extract: A randomized controlled trial. J. Nutr. Biochem. 2014, 25, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Botden, I.P.G.; Draijer, R.; Westerhof, B.E.; Rutten, J.H.W.; Langendonk, J.G.; Sijbrands, E.J.G.; Danser, A.H.J.; Zock, P.L.; van den Meiracker, A.H. Red wine polyphenols do not lower peripheral or central blood pressure in high normal blood pressure and hypertension. Am. J. Hypertens. 2012, 25, 718–723. [Google Scholar] [CrossRef] [PubMed]
- Chiva-Blanch, G.; Condines, X.; Magraner, E.; Roth, I.; Valderas-Martínez, P.; Arranz, S.; Casas, R.; Martínez-Huélamo, M.; Vallverdú-Queralt, A.; Quifer-Rada, P.; et al. The non-alcoholic fraction of beer increases stromal cell derived factor 1 and the number of circulating endothelial progenitor cells in high cardiovascular risk subjects: A randomized clinical trial. Atherosclerosis 2014, 233, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Chiva-Blanch, G.; Urpi-Sarda, M.; Ros, E.; Arranz, S.; Valderas-Martínez, P.; Casas, R.; Sacanella, E.; Llorach, R.; Lamuela-Raventos, R.M.; Andres-Lacueva, C.; et al. Dealcoholized red wine decreases systolic and diastolic blood pressure and increases plasma nitric oxide: Short communication. Circ. Res. 2012, 111, 1065–1068. [Google Scholar] [CrossRef] [PubMed]
- Chiva-Blanch, G.; Urpi-Sarda, M.; Llorach, R.; Rotches-Ribalta, M.; Guillen, M.; Casas, R.; Arranz, S.; Valderas-Martinez, P.; Portoles, O.; Corella, D. Differential effects of polyphenols and alcohol of red wine on the expression of adhesion molecules and inflammatory cytokines related to atherosclerosis: A randomized clinical trial (vol 95, pg 326, 2012). Am. J. Clin. Nutr. 2012, 95, 1506. [Google Scholar] [CrossRef] [PubMed]
- Kuntz, S.; Kunz, C.; Herrmann, J.; Borsch, C.H.; Abel, G.; Dietrich, H.; Rudloff, S.; Fröhling, B.; Dietrich, H.; Rudloff, S. Anthocyanins from fruit juices improve the antioxidant status of healthy young female volunteers without affecting anti-inflammatory parameters: Results from the randomised, double-blind, placebo-controlled, cross-over ANTHONIA (ANTHOcyanins in Nutrition Investigation Alliance) study. Br. J. Nutr. 2014, 112, 925–936. [Google Scholar] [PubMed]
- Curtis, P.J.; Kroon, P.A.; Hollands, W.J.; Walls, R.; Jenkins, G.; Kay, C.D.; Cassidy, A. Cardiovascular disease risk biomarkers and liver and kidney function are not altered in postmenopausal women after ingesting an elderberry extract rich in anthocyanins for 12 weeks. J. Nutr. 2009, 139, 2266–2271. [Google Scholar] [CrossRef] [PubMed]
- Hassellund, S.S.; Flaa, A.; Kjeldsen, S.E.; Seljeflot, I.; Karlsen, A.; Erlund, I.; Rostrup, M. Effects of anthocyanins on cardiovascular risk factors and inflammation in pre-hypertensive men: A double-blind randomized placebo-controlled crossover study. J. Hum. Hypertens. 2012, 27, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Dohadwala, M.M.; Holbrook, M.; Hamburg, N.M.; Shenouda, S.M.; Chung, W.B.; Titas, M.; Kluge, M.A.; Wang, N.; Palmisano, J.; Milbury, P.E.; et al. A Effects of cranberry juice consumption on vascular function in patients with coronary artery disease 1–3. Am. J. Clin. Nutr. 2011, 93, 934–940. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, R.; Kotani, K.; Ayabe, M.; Tsuzaki, K.; Shimada, J.; Sakane, N.; Takase, H.; Ichikawa, H.; Yonei, Y.; Ishii, K. Minor effects of green tea catechin supplementation on cardiovascular risk markers in active older people: A randomized controlled trial. Geriatr. Gerontol. Int. 2013, 13, 622–629. [Google Scholar] [CrossRef] [PubMed]
- De Maat, M.P.; Pijl, H.; Kluft, C.; Princen, H.M. Consumption of black and green tea had no effect on inflammation, haemostasis and endothelial markers in smoking healthy individuals. Eur. J. Clin. Nutr. 2000, 54, 757–763. [Google Scholar] [CrossRef] [PubMed]
- Widmer, R.J.; Freund, M.A.; Flammer, A.J.; Sexton, J.; Lennon, R.; Romani, A.; Mulinacci, N.; Vinceri, F.F.; Lerman, L.O.; Lerman, A. Beneficial effects of polyphenol-rich olive oil in patients with early atherosclerosis. Eur. J. Nutr. 2012, 52, 1223–1231. [Google Scholar] [CrossRef] [PubMed]
- Nagao, T.; Hase, T.; Tokimitsu, I. A green tea extract high in catechins reduces body fat and cardiovascular risks in humans. Obesity (Silver Spring). 2007, 15, 1473–1483. [Google Scholar] [CrossRef] [PubMed]
- Farouque, H.M.O.; Leung, M.; Hope, S.A.; Baldi, M.; Schechter, C.; Cameron, J.D.; Meredith, I.T. Acute and chronic effects of flavanol-rich cocoa on vascular function in subjects with coronary artery disease: A randomized double-blind placebo-controlled study. Clin. Sci. (Lond). 2006, 111, 71–80. [Google Scholar] [PubMed]
- Berry, N.M.; Davison, K.; Coates, A.M.; Buckley, J.D.; Howe, P.R.C. Impact of cocoa flavanol consumption on blood pressure responsiveness to exercise. Br. J. Nutr. 2010, 103, 1480–1484. [Google Scholar] [CrossRef] [PubMed]
- Davison, K.; Coates, A.M.; Buckley, J.D.; Howe, P.R.C. Effect of cocoa flavanols and exercise on cardiometabolic risk factors in overweight and obese subjects. Int. J. Obes. (Lond). 2008, 32, 1289–1296. [Google Scholar] [CrossRef] [PubMed]
- West, S.G.; McIntyre, M.D.; Piotrowski, M.J.; Poupin, N.; Miller, D.L.; Preston, A.G.; Wagner, P.; Groves, L.F.; Skulas-Ray, A.C. Effects of dark chocolate and cocoa consumption on endothelial function and arterial stiffness in overweight adults. Br. J. Nutr. 2014, 111, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Faridi, Z.; Njike, V.Y.; Dutta, S.; Ali, A.; Katz, D.L. Acute dark chocolate and cocoa ingestion and endothelial function: A randomized controlled crossover trial. Am. J. Clin. Nutr. 2008, 88, 58–63. [Google Scholar] [PubMed]
- Davison, K.; Berry, N.M.; Misan, G.; Coates, A.M.; Buckley, J.D.; Howe, P.R.C. Dose-related effects of flavanol-rich cocoa on blood pressure. J. Hum. Hypertens. 2010, 24, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Desideri, G.; Necozione, S.; Lippi, C.; Casale, R.; Properzi, G.; Blumberg, J.B.; Ferri, C. Blood pressure is reduced and insulin sensitivity increased in glucose-intolerant, hypertensive subjects after 15 days of consuming high-polyphenol dark chocolate. J. Nutr. 2008, 138, 1671–1676. [Google Scholar] [PubMed]
- Flammer, A.J.; Sudano, I.; Wolfrum, M.; Thomas, R.; Enseleit, F.; Périat, D.; Kaiser, P.; Hirt, A.; Hermann, M.; Serafini, M.; et al. Cardiovascular effects of flavanol-rich chocolate in patients with heart failure. Eur. Heart J. 2012, 33, 2172–2180. [Google Scholar] [CrossRef] [PubMed]
- Heiss, C.; Jahn, S.; Taylor, M.; Real, W.M.; Angeli, F.S.; Wong, M.L.; Amabile, N.; Prasad, M.; Rassaf, T.; Ottaviani, J.I.; et al. Improvement of endothelial function with dietary flavanols is associated with mobilization of circulating angiogenic cells in patients with coronary artery disease. J. Am. Coll. Cardiol. 2010, 56, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Horn, P.; Amabile, N.; Angeli, F.S.; Sansone, R.; Stegemann, B.; Kelm, M.; Springer, M.L.; Yeghiazarians, Y.; Schroeter, H.; Heiss, C. Dietary flavanol intervention lowers the levels of endothelial microparticles in coronary artery disease patients. Br. J. Nutr. 2013, 111, 1245–1252. [Google Scholar] [CrossRef] [PubMed]
- Balzer, J.; Rassaf, T.; Heiss, C.; Kleinbongard, P.; Lauer, T.; Merx, M.; Heussen, N.; Gross, H.B.; Keen, C.L.; Schroeter, H.; et al. Sustained Benefits in Vascular Function Through Flavanol-Containing Cocoa in Medicated Diabetic Patients. A Double-Masked, Randomized, Controlled Trial. J. Am. Coll. Cardiol. 2008, 51, 2141–2149. [Google Scholar] [CrossRef] [PubMed]
- Larson, A.; Witman, M.A.H.; Guo, Y.; Ives, S.; Richardson, R.S.; Bruno, R.S.; Jalili, T.; Symons, J.D. Acute, quercetin-induced reductions in blood pressure in hypertensive individuals are not secondary to lower plasma angiotensin-converting enzyme activity or endothelin-1: Nitric oxide. Nutr. Res. 2012, 32, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Conquer, J.A.; Maiani, G.; Azzini, E.; Raguzzini, A.; Holub, B.J. Supplementation with quercetin markedly increases plasma quercetin concentration without effect on selected risk factors for heart disease in healthy subjects. J. Nutr. 1998, 128, 593–597. [Google Scholar] [PubMed]
- Suomela, J.P.; Ahotupa, M.; Yang, B.; Vasankari, T.; Kallio, H. Absorption of flavonols derived from sea buckthorn (Hippopha?? rhamnoides L.) and their effect on emerging risk factors for cardiovascular disease in humans. J. Agric. Food Chem. 2006, 54, 7364–7369. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.L.; Lyon, T.; Litwin, S.E.; Rabovsky, A.; Symons, J.D.; Jalili, T. Quercetin reduces blood pressure in hypertensive subjects. J. Nutr. 2007, 137, 2405–2411. [Google Scholar] [PubMed]
- McVeigh, B.L.; Dillingham, B.L.; Lampe, J.W.; Duncan, A.M. Effect of soy protein varying in isoflavone content on serum lipids in healthy young men. Am. J. Clin. Nutr. 2006, 83, 244–251. [Google Scholar] [PubMed]
- Sanders, T.A.B.; Dean, T.S.; Grainger, D.; Miller, G.J.; Wiseman, H. Moderate intakes of intact soy protein rich in isoflavones compared with ethanol-extracted soy protein increase HDL but do not influence transforming growth factor β1 concentrations and hemostatic risk factors for coronary heart disease in healthy subjets. Am. J. Clin. Nutr. 2002, 76, 373–377. [Google Scholar] [PubMed]
- Thorp, A.A.; Howe, P.R.C.; Mori, T.A.; Coates, A.M.; Buckley, J.D.; Hodgson, J.; Mansour, J.; Meyer, B.J. Soy food consumption does not lower LDL cholesterol in either equol or nonequol producers. Am. J. Clin. Nutr. 2008, 88, 298–304. [Google Scholar] [PubMed]
- Atkinson, C.; Oosthuizen, W.; Scollen, S.; Loktionov, A.; Day, N.E.; Bingham, S. A Modest protective effects of isoflavones from a red clover-derived dietary supplement on cardiovascular disease risk factors in perimenopausal women, and evidence of an interaction with ApoE genotype in 49–65 year-old women. J. Nutr. 2004, 134, 1759–1764. [Google Scholar] [PubMed]
- Marini, H.; Bitto, A.; Altavilla, D.; Burnett, B.P.; Polito, F.; di Stefano, V.; Minutoli, L.; Atteritano, M.; Levy, R.M.; Frisina, N.; et al. Efficacy of genistein aglycone on some cardiovascular risk factors and homocysteine levels: A follow-up study. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Hodis, H.N.; Mack, W.J.; Kono, N.; Azen, S.P.; ShoupeJ, D.; Hwang-Levine, J.; Petitti, D.; Whitfield-Maxwell, L.; Yan, M.; Franke, A.A.; et al. Isoflavone Soy Protein Supplementation and Atherosclerosis in Healthy Postmenopausal Women: A Randomized Controlled Trial. Stroke 2012, 42, 3168–3175. [Google Scholar] [CrossRef] [PubMed]
- Atteritano, M.; Marini, H.; Minutoli, L.; Polito, F.; Bitto, A.; Altavilla, D.; Mazzaferro, S.; D’Anna, R.; Cannata, M.L.; Gaudio, A.; et al. Effects of the phytoestrogen genistein on some predictors of cardiovascular risk in osteopenic, postmenopausal women: A two-year randomized, double-blind, placebo-controlled study. J. Clin. Endocrinol. Metab. 2007, 92, 3068–3075. [Google Scholar] [CrossRef] [PubMed]
- Garrido, A.; de la Maza, M.P.; Hirsch, S.; Valladares, L. Soy isoflavones affect platelet thromboxane A2 receptor density but not plasma lipids in menopausal women. Maturitas 2006, 54, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Hall, W.L.; Vafeiadou, K.; Hallund, J.; Bügel, S.; Koebnick, C.; Reimann, M.; Ferrari, M.; Branca, F.; Talbot, D.; Dadd, T.; et al. Soy-isoflavone-enriched foods and inflammatory biomarkers of cardiovascular disease risk in postmenopausal women: Interactions with genotype and equol production. Am. J. Clin. Nutr. 2005, 82, 1260–1268. [Google Scholar] [PubMed]
- Rios, D.R.A.; Rodrigues, E.T.; Cardoso, A.P.Z.; Montes, M.B.A.; Franceschini, S.A.; Toloi, M.R.T. Effects of isoflavones on the coagulation and fibrinolytic system of postmenopausal women. Nutrition 2008, 24, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Villa, P.; Costantini, B.; Suriano, R.; Perri, C.; Macrì, F.; Ricciardi, L.; Panunzi, S.; Lanzone, A. The differential effect of the phytoestrogen genistein on cardiovascular risk factors in postmenopausal women: Relationship with the metabolic status. J. Clin. Endocrinol. Metab. 2009, 94, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.M.; Ho, S.C.; Chen, Y.M.; Ho, Y.P. The effects of isoflavones combined with soy protein on lipid profiles, C-reactive protein and cardiovascular risk among postmenopausal Chinese women. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.S.; Wang, S.Y.; Yang, Y.C.; Su, C.H.; Lee, F.K.; Chen, S.C.; Tseng, C.Y.; Jou, H.J.; Huang, J.P.; Huang, K.E. Effects of standardized phytoestrogen on Taiwanese menopausal women. Taiwan J. Obstet. Gynecol. 2012, 51, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Aubertin-Leheudre, M.; Lord, C.; Khalil, A.; Dionne, I.J. Isoflavones and clinical cardiovascular risk factors in obese postmenopausal women: A randomized double-blind placebo-controlled trial. J. Womens Health (Larchmt) 2008, 17, 1363–1369. [Google Scholar] [CrossRef] [PubMed]
- Choquette, S.; Riesco, É.; Cormier, É.; Dion, T.; Aubertin-Leheudre, M.; Dionne, I.J. Effects of soya isoflavones and exercise on body composition and clinical risk factors of cardiovascular diseases in overweight postmenopausal women: A 6-month double-blind controlled trial. Br. J. Nutr. 2011, 105, 1199–1209. [Google Scholar] [PubMed]
- Aubertin-Leheudre, M.; Lord, C.; Khalil, A.; Dionne, I.J. Effect of 6 months of exercise and isoflavone supplementation on clinical cardiovascular risk factors in obese postmenopausal women: A randomized, double-blind study. Menopause 2007, 14, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, J.M.; Puddey, I.B.; Croft, K.D.; Mori, T.A.; Rivera, J.; Beilin, L.J. Isoflavonoids do not inhibit in vivo lipid peroxidation in subjects with high-normal blood pressure. Atherosclerosis 1999, 145, 167–172. [Google Scholar] [CrossRef]
- Sagara, M.; Kanda, T.; NJelekera, M.; Teramoto, T.; Armitage, L.; Birt, N.; Birt, C.; Yamori, Y. Effects of dietary intake of soy protein and isoflavones on cardiovascular disease risk factors in high risk, middle-aged men in Scotland. J. Am. Coll. Nutr. 2004, 23, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Clerici, C.; Setchell, K.D.R.; Battezzati, P.M.; Pirro, M.; Giuliano, V.; Asciutti, S.; Castellani, D.; Nardi, E.; Sabatino, G.; Orlandi, S.; et al. Pasta naturally enriched with isoflavone aglycons from soy germ reduces serum lipids and improves markers of cardiovascular risk. J. Nutr. 2007, 137, 2270–2278. [Google Scholar] [PubMed]
- Meyer, B.J.; Larkin, T.A.; Owen, A.J.; Astheimer, L.B.; Tapsell, L.C.; Howe, P.R.C. Limited lipid-lowering effects of regular consumption of whole soybean foods. Ann. Nutr. Metab. 2004, 48, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.A.; Kendall, C.W.C.; Jackson, C.J.C.; Connelly, P.W.; Parker, T.; Faulkner, D.; Vidgen, E.; Cunnane, S.C.; Leiter, L.A.; Josse, R.G. Effects of high- and low-isoflavone soyfoods on blood lipids, oxidized LDL, homocysteine, and blood pressure in hyperlipidemic men and women. Am. J. Clin. Nutr. 2002, 76, 365–372. [Google Scholar] [PubMed]
- Blum, A.; Lang, N.; Peleg, A.; Vigder, F.; Israeli, P.; Gumanovsky, M.; Lupovitz, S.; Elgazi, A.; Ben-Ami, M. Effects of oral soy protein on markers of inflammation in postmenopausal women with mild hypercholesterolemia. Am. Heart J. 2003, 145, e7. [Google Scholar] [CrossRef] [PubMed]
- Teede, H.J.; Giannopoulos, D.; Dalais, F.S.; Hodgson, J.; McGrath, B.P. Randomised, controlled, cross-over trial of soy protein with isoflavones on blood pressure and arterial function in hypertensive subjects. J. Am. Coll. Nutr. 2006, 25, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.G.; Tartagni, E.; Ferroni, A.; de Sando, V.; Grandi, E.; Borghi, C. Combined nutraceutical approach to postmenopausal syndrome and vascular remodeling biomarkers. J. Altern. Complement. Med. 2013, 19, 582–587. [Google Scholar] [CrossRef] [PubMed]
- Curtis, P.J.; Potter, J.; Kroon, P.A.; Wilson, P.; Dhatariya, K.; Sampson, M.; Cassidy, A. Vascular function and atherosclerosis progression after 1 y of flavonoid intake in statin-treated postmenopausal women with type 2 diabetes: A double-blind randomized controlled trial. Am. J. Clin. Nutr. 2013, 97, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.H.; Lau, K.K.; Yiu, K.H.; Li, S.W.; Chan, H.T.; Fong, D.Y.T.; Tam, S.; Lau, C.P.; Tse, H.F. Reduction of C-reactive protein with isoflavone supplement reverses endothelial dysfunction in patients with ischaemic stroke. Eur. Heart J. 2008, 29, 2800–2807. [Google Scholar] [CrossRef] [PubMed]
- Webb, C.M.; Hayward, C.S.; Mason, M.J.; Ilsley, C.D.; Collins, P. Coronary vasomotor and blood flow responses to isoflavone-intact soya protein in subjects with coronary heart disease or risk factors for coronary heart disease. Clin. Sci. 2008, 115, 353. [Google Scholar] [CrossRef] [PubMed]
- Fanti, P.; Asmis, R.; Stephenson, T.J.; Sawaya, B.P.; Franke, A.A. Positive effect of dietary soy in ESRD patients with systemic inflammation—Correlation between blood levels of the soy isoflavones and the acute-phase reactants. Nephrol. Dial. Transplant. 2006, 21, 2239–2246. [Google Scholar] [CrossRef] [PubMed]
- Ras, R.T.; Zock, P.L.; Zebregs, Y.E.M.P.; Johnston, N.R.; Webb, D.J.; Draijer, R. Effect of polyphenol-rich grape seed extract on ambulatory blood pressure in subjects with pre- and stage I hypertension. Br. J. Nutr. 2013, 110, 2234–2241. [Google Scholar] [CrossRef] [PubMed]
- Yubero, N.; Sanz-Buenhombre, M.; Guadarrama, A.; Villanueva, S.; Carrión, J.M.; Larrarte, E.; Moro, C. LDL cholesterol-lowering effects of grape extract used as a dietary supplement on healthy volunteers. Int. J. Food Sci. Nutr. 2013, 64, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Asher, G.N.; Viera, A.J.; Weaver, M.A.; Dominik, R.; Caughey, M.; Hinderliter, A.L. Effect of hawthorn standardized extract on flow mediated dilation in prehypertensive and mildly hypertensive adults: A randomized, controlled cross-over trial. BMC Complement. Altern. Med. 2012, 12, 26. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wei, J.; Tan, F.; Zhou, S.; Würthwein, G.; Rohdewald, P. Antidiabetic effect of Pycnogenol® French maritime pine bark extract in patients with diabetes type II. Life Sci. 2004, 75, 2505–2513. [Google Scholar] [CrossRef] [PubMed]
- Enseleit, F.; Sudano, I.; Périat, D.; Winnik, S.; Wolfrum, M.; Flammer, A.J.; Fröhlich, G.M.; Kaiser, P.; Hirt, A.; Haile, S.R.; et al. Effects of Pycnogenol on endothelial function in patients with stable coronary artery disease: A double-blind, randomized, placebo-controlled, cross-over study. Eur. Heart J. 2012, 33, 1589–1597. [Google Scholar] [CrossRef] [PubMed]
- Mellen, P.B.; Daniel, K.R.; Brosnihan, K.B.; Hansen, K.J.; Herrington, D.M. Effect of muscadine grape seed supplementation on vascular function in subjects with or at risk for cardiovascular disease: A randomized crossover trial. J. Am. Coll. Nutr. 2010, 29, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Tauchert, M. Efficacy and safety of crataegus extract WS 1442 in comparison with placebo in patients with chronic stable New York Heart Association class-III heart failure. Am. Heart J. 2002, 143, 910–915. [Google Scholar] [CrossRef] [PubMed]
- Tang, P.C.-T.; Ng, Y.-F.; Ho, S.; Gyda, M.; Chan, S.-W. Resveratrol and cardiovascular health—Promising therapeutic or hopeless illusion? Pharmacol. Res. 2014, 90, 88–115. [Google Scholar] [CrossRef] [PubMed]
- Shechter, M.; Issachar, A.; Marai, I.; Koren-Morag, N.; Freinark, D.; Shahar, Y.; Shechter, A.; Feinberg, M.S. Long-term association of brachial artery flow-mediated vasodilation and cardiovascular events in middle-aged subjects with no apparent heart disease. Int. J. Cardiol. 2009, 134, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Yeboah, J.; Crouse, J.R.; Hsu, F.C.; Burke, G.L.; Herrington, D.M. Brachial flow-mediated dilation predicts incident cardiovascular events in older adults: The cardiovascular health study. Circulation 2007, 115, 2390–2397. [Google Scholar] [CrossRef] [PubMed]
- Rossi, R.; Nuzzo, A.; Origliani, G.; Modena, M.G. Prognostic Role of Flow-Mediated Dilation and Cardiac Risk Factors in Post-Menopausal Women. J. Am. Coll. Cardiol. 2008, 51, 997–1002. [Google Scholar] [CrossRef] [PubMed]
- Williams, I.L.; Chowienczyk, P.J.; Wheatcroft, S.B.; Patel, A.G.; Sherwood, R.A.; Momin, A.; Shah, A.M.; Kearney, M.T. Endothelial function and weight loss in obese humans. Obes. Surg. 2005, 15, 1055–1060. [Google Scholar] [CrossRef] [PubMed]
- Widlansky, M.E.; Duffy, S.J.; Hamburg, N.M.; Gokce, N.; Warden, B.A.; Wiseman, S.; Keaney, J.F.; Frei, B.; Vita, J.A. Effects of black tea consumption on plasma catechins and markers of oxidative stress and inflammation in patients with coronary artery disease. Free Radic. Biol. Med. 2005, 38, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Shenouda, S.M.; Widlansky, M.E.; Chen, K.; Xu, G.; Holbrook, M.; Tabit, C.E.; Hamburg, N.M.; Frame, A.A.; Caiano, T.L.; Kluge, M.A.; et al. Altered mitochondrial dynamics contributes to endothelial dysfunction in diabetes mellitus. Circulation 2011, 124, 444–453. [Google Scholar] [CrossRef] [PubMed]
- Hartman, M.-L.; Shirihai, O.S.; Holbrook, M.; Xu, G.; Kocherla, M.; Shah, A.; Fetterman, J.L.; Kluge, M.A.; Frame, A.A.; Hamburg, N.M.; et al. A Relation of mitochondrial oxygen consumption in peripheral blood mononuclear cells to vascular function in type 2 diabetes mellitus. Vasc. Med. 2014, 19, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Sahebkar, A. A systematic review and meta-analysis of randomized controlled trials investigating the effects of curcumin on blood lipid levels. Clin. Nutr. 2013, 33, 406–414. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Boscá, A.; Soler, A.; Carrión, M.A.; Díaz-Alperi, J.; Bernd, A.; Quintanilla, C.; Quintanilla Almagro, E.; Miquel, J. An hydroalcoholic extract of Curcuma longa lowers the apo B/apo A ratio. Implications for atherogenesis prevention. Mech. Ageing Dev. 2000, 119, 41–47. [Google Scholar] [CrossRef]
- Weisberg, S.P.; Leibel, R.; Tortoriello, D.V. Dietary curcumin significantly improves obesity-associated inflammation and diabetes in mouse models of diabesity. Endocrinology 2008, 149, 3549–3558. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Zheng, S.; Chen, A. Curcumin eliminates leptin’s effects on hepatic stellate cell activation via interrupting leptin signaling. Endocrinology 2009, 150, 3011–3020. [Google Scholar] [CrossRef] [PubMed]
- Rotondo, S.; di Castelnuovo, A.; de Gaetano, G. The relationship between wine consumption and cardiovascular risk: From epidemiological evidence to biological plausibility. Ital. Heart J. 2001, 2, 1–8. [Google Scholar] [PubMed]
- Wang, X.; Ouyang, Y.Y.; Liu, J.; Zhao, G. Flavonoid intake and risk of CVD: A systematic review and meta-analysis of prospective cohort studies. Br. J. Nutr. 2013, 111, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.; Bryant, S.; Huntley, A.L. Green tea and green tea catechin extracts: An overview of the clinical evidence. Maturitas 2012, 73, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Khawaja, O.; Gaziano, J.M.; Djoussé, L. Chocolate and coronary heart disease: A systematic review. Curr. Atheroscler. Rep. 2011, 13, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.M.; Zhao, D.; Nie, Z.L.; Zhao, H.; Zhou, B.; Gao, W.; Wang, L.S.; Yang, Z.J. Flavonol intake and stroke risk: A meta-analysis of cohort studies. Nutrition 2014, 30, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Hollman, P.C.H.; Geelen, A.; Kromhout, D. Dietary flavonol intake may lower stroke risk in men and women. J. Nutr. 2010, 140, 600–604. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.M.; Ho, S.C.; Chen, Y.M.; Woo, J. A six-month randomized controlled trial of whole soy and isoflavones daidzein on body composition in equol-producing postmenopausal women with prehypertension. J. Obes. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Curtis, P.J.; Sampson, M.; Potter, J.; Dhatariya, K.; Kroon, P.A.; Cassidy, A. Chronic Ingestion of Flavan-3-ols and and Lipoprotein Status and Attenuates With Type 2 Diabetes. Diabetes Care 2012, 35, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Azadbakht, L.; Atabak, S.; Esmaillzadeh, A. Soy protein intake, cardiorenal indices, and C-reactive protein in type 2 diabetes with nephropathy: A longitudinal randomized clinical trial. Diabetes Care 2008, 31, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Hong, K.; Saltsman, P.; DeShields, S.; Bellman, M.; Thames, G.; Liu, Y.; Wang, H.-J.; Elashoff, R.; Heber, D. Long-term efficacy of soy-based meal replacements vs an individualized diet plan in obese type II DM patients: Relative effects on weight loss, metabolic parameters, and C-reactive protein. Eur. J. Clin. Nutr. 2005, 59, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, Y.; Iso, H.; Ishihara, J.; Okada, K.; Inoue, M.; Tsugane, S. Association of dietary intake of soy, beans, and isoflavones with risk of cerebral and myocardial infarctions in Japanese populations: The Japan Public Health Center-based (JPHC) study cohort I. Circulation 2007, 116, 2553–2562. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.-Y.; Wang, P.; He, K.; Qin, L.-Q. Effect of soy isoflavones on circulating C-reactive protein in postmenopausal women: Meta-analysis of randomized controlled trials. Menopause 2011, 18, 1256–1262. [Google Scholar] [CrossRef] [PubMed]
- Vanhoutte, P.M.; Shimokawa, H.; Tang, E.H.C.; Feletou, M. Endothelial dysfunction and vascular disease. Acta Physiol. (Oxf). 2009, 196, 193–222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pase, M.P.; Grima, N.A.; Sarris, J. The effects of dietary and nutrient interventions on arterial stiffness: A systematic review. Am. J. Clin. Nutr. 2011, 93, 446–454. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rangel-Huerta, O.D.; Pastor-Villaescusa, B.; Aguilera, C.M.; Gil, A. A Systematic Review of the Efficacy of Bioactive Compounds in Cardiovascular Disease: Phenolic Compounds. Nutrients 2015, 7, 5177-5216. https://doi.org/10.3390/nu7075177
Rangel-Huerta OD, Pastor-Villaescusa B, Aguilera CM, Gil A. A Systematic Review of the Efficacy of Bioactive Compounds in Cardiovascular Disease: Phenolic Compounds. Nutrients. 2015; 7(7):5177-5216. https://doi.org/10.3390/nu7075177
Chicago/Turabian StyleRangel-Huerta, Oscar D., Belen Pastor-Villaescusa, Concepcion M. Aguilera, and Angel Gil. 2015. "A Systematic Review of the Efficacy of Bioactive Compounds in Cardiovascular Disease: Phenolic Compounds" Nutrients 7, no. 7: 5177-5216. https://doi.org/10.3390/nu7075177
APA StyleRangel-Huerta, O. D., Pastor-Villaescusa, B., Aguilera, C. M., & Gil, A. (2015). A Systematic Review of the Efficacy of Bioactive Compounds in Cardiovascular Disease: Phenolic Compounds. Nutrients, 7(7), 5177-5216. https://doi.org/10.3390/nu7075177






