NO-Rich Diet for Lifestyle-Related Diseases
Abstract
:1. Introduction
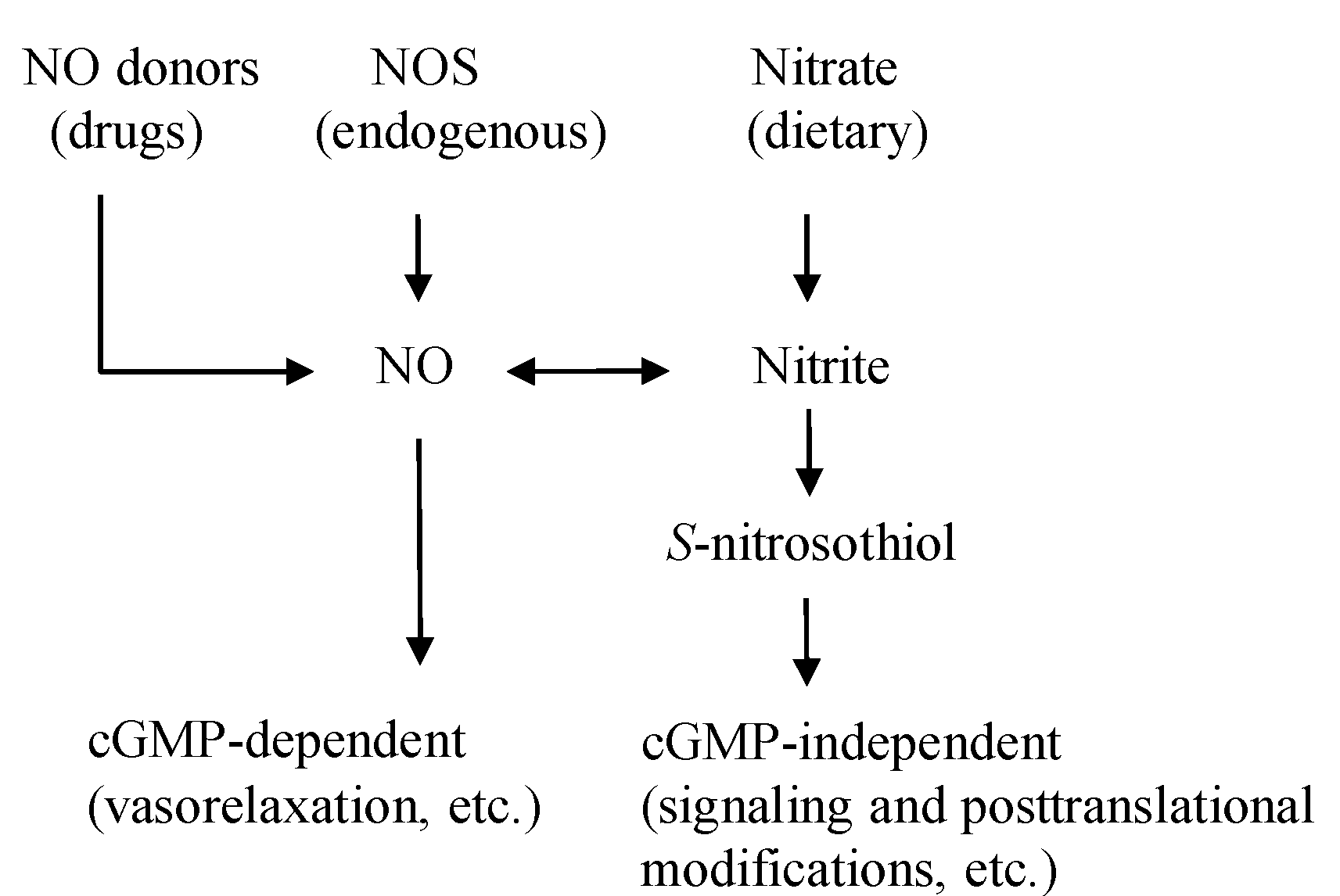
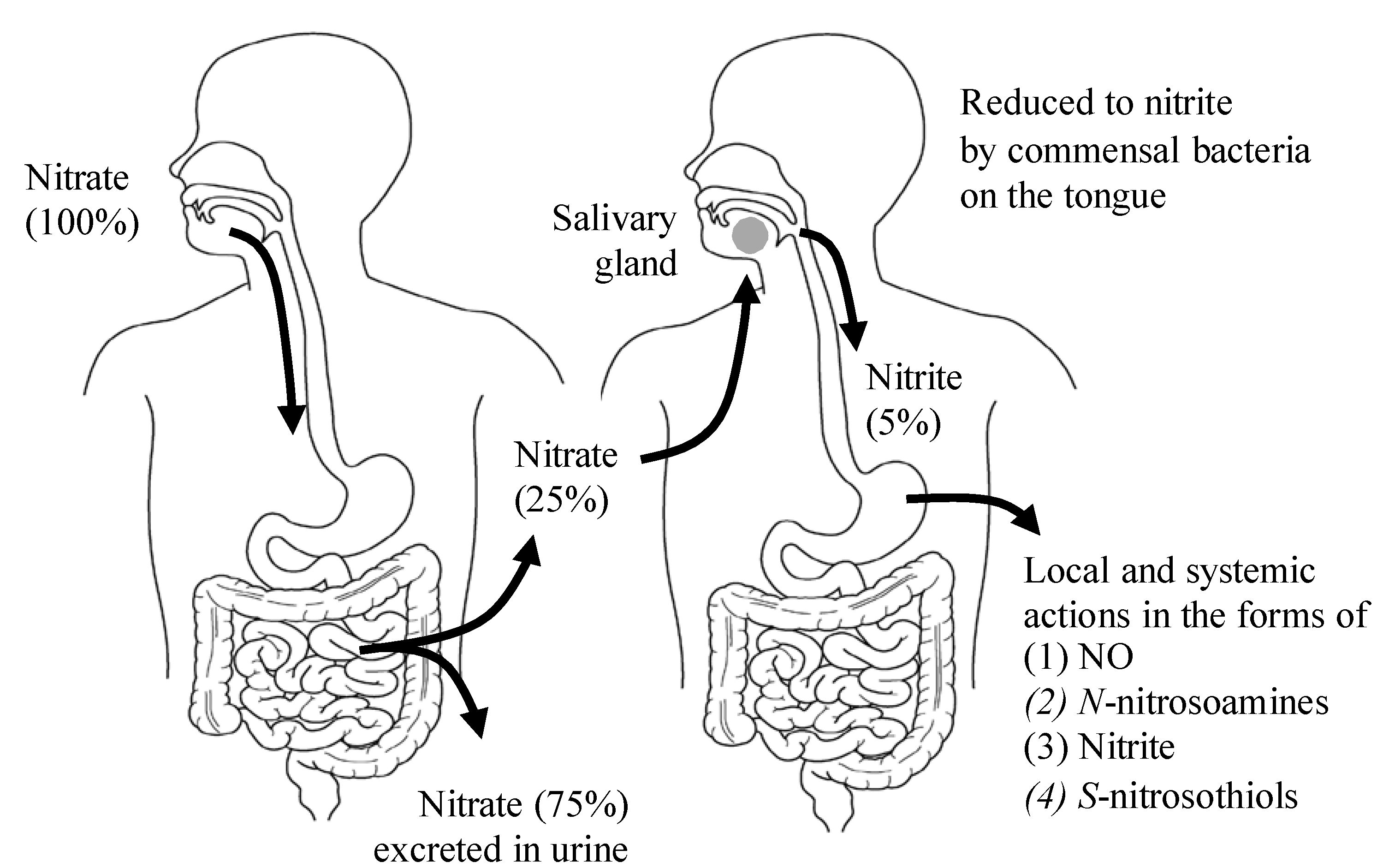
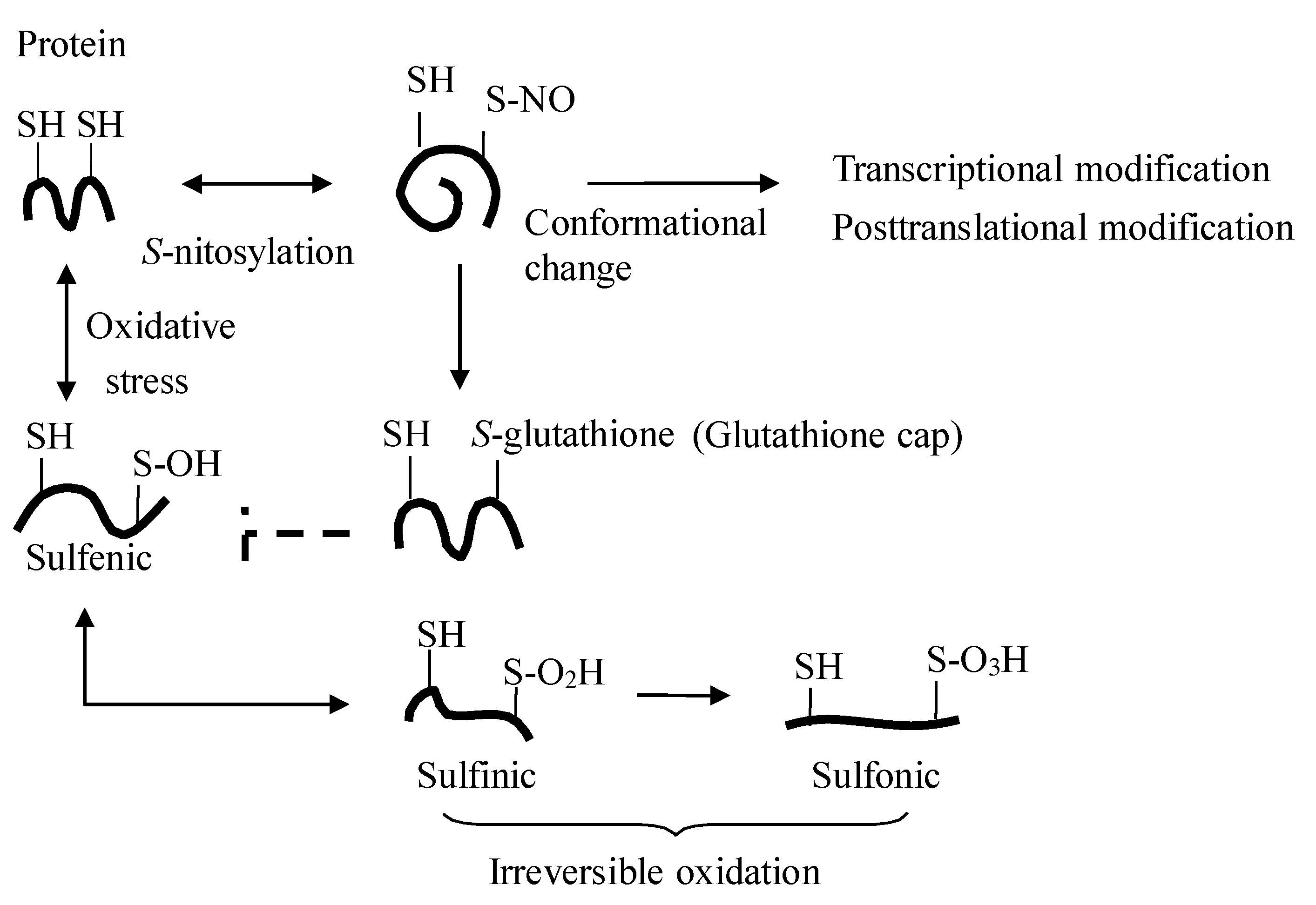
2. The Dietary Nitrate-nitrite-NO Pathway and Its Physiological Aspect
| Food product | Nitrate concentration (mg/100 g) | Nitrite concentration (mg/100 g) | Reference | ||
|---|---|---|---|---|---|
| Mean | Range | Mean | Range | ||
| Beets | 275.6 | 168–359 | 1.00 | 0.21–2.98 | [16] |
| Spinach | 233.3 | 53.5–366 | 0.70 | 0.0–1.29 | |
| Radishes | 168.0 | 76.4–250 | 0.01 | 0.0–0.1 | |
| Celery | 154.4 | 31.6–332 | 0.16 | 0.0–0.52 | |
| Lettuce | 85.0 | 7.9–217.1 | 0.06 | 0.001–0.97 | |
| Iceberg lettuce | 78.6 | 34.7–108 | 0.02 | 0.0–0.17 | |
| Mushroom | 59.0 | 1.9–8.5 | 0.80 | 0.0–3.8 | |
| Cabbage | 57.3 | 19.3–97.6 | 0.24 | 0.0–1.26 | |
| Broccoli | 39.4 | 2.9–114 | 0.06 | 0.001–0.95 | |
| Green beans | 38.6 | 16.5–61.1 | 0.05 | 0.0–0.25 | |
| Strawberries | 17.3 | 10.5–29.3 | 0.20 | 0.0–0.71 | |
| Banana | 13.7 | 8.8–21.4 | 0.21 | 0.0–0.95 | |
| Green pepper | 3.3 | 0.8–5.5 | 0.04 | 0.0–0.3 | |
| Spinach | 741.0 | - | 0.02 | - | [28] |
| Mustard greens | 116.0 | - | 0.003 | - | |
| Salad mix | 82.1 | - | 0.13 | - | |
| Cole slaw | 55.9 | - | 0.07 | - | |
| Broccoli | 39.5 | - | 0.07 | - | |
| Tomato | 39.2 | - | 0.03 | - | |
| Vegetable soup | 20.9 | - | 0.001 | - | |
| Hot dog | 9.0 | - | 0.05 | - | |
| Bacon | 5.5 | - | 0.38 | - | |
| Banana | 4.5 | - | 0.009 | - | |
| Pork tenderloin | 3.3 | - | 0.0 | - | |
| Bacon nitrite-free | 3.0 | - | 0.68 | - | |
| French fries | 2.0 | - | 0.17 | - | |
| Ham | 0.9 | - | 0.89 | - | |
| Fruit mix | 0.9 | - | 0.08 | - | |
| Orange | 0.8 | - | 0.02 | - | |
| Apple sauce | 0.3 | - | 0.008 | - | |
| Ketchup | 0.1 | - | 0.13 | - | |
| Carrots | 0.1 | - | 0.006 | - | |
| Nitrate concentration (mg/L) | Nitrite concentration (mg/L) | ||||
| Carrot juice | 27.55 | - | 0.036 | - | |
| Vegetable juice * | 26.17 | - | 0.092 | - | |
| Pomegranate juice | 12.93 | - | 0.069 | - | |
| Cranberry juice | 9.12 | - | 0.145 | - | |
| Acai juice | 0.56 | - | 0.013 | - | |
| Green tea | 0.23 | - | 0.007 | - | |
| 0 Min | 30 Min | ||
|---|---|---|---|
| Saliva | Nitrite (μM) | 104 ± 21 | 713 ± 150 |
| Nitrate (mM) | 0.19 ± 0.03 | 8.2 ± 1 | |
| S-NO (nM) | 25 ± 9.8 | 297 | |
| Plasma | Nitrite (μM) | 123 ± 19 | 229 ± 46 |
| Nitrate (mM) | 30 ± 4 | 432 ± 44 | |
| S-NO (nM) | 6.3 ± 1.4 | No significant change |
| Animal Model | Dietary Nitrate | Tissues | Effects of Dietary Nitrate | References |
|---|---|---|---|---|
| Uninephrectomized hypertension rat with high-salt diet. | Diets with 0.1 mM and 1 mM nitrate/kg/day for 8–11 weeks. | Kidney Heart Liver | Increase in plasma and tissue levels of nitrate and tissue levels of nitrosylation products. Reduction of oxidative stress and attenuation of renal injury, hypertension, cardiac hypertrophy and fibrosis. | [37] |
| C57BLK6 male mice with hypoxia-induced pulmonary hypertension. | 0.6 mM, 15 mM, and 45 mM nitrate/L in drinking water for 3 weeks. | Lung | Increase in plasma and lung levels of nitrite and cGMP. Reduction of right ventricular pressure and hypertrophy, and pulmonary vascular remodeling. | [38] |
| Male Wistar rat with hypoxic heart damage. | 0.7 mM/L nitrate in drinking water for 2 weeks. | Heart | Increase in plasma levels of nitrate and tissue levels of nitrite. Alleviation of metabolic abnormalities in the hypoxic heart. Improvement of myocardial energetics. | [39] |
3. Safety and Efficacy of Dietary Nitrate
4. Protective Effects of Dietary Nitrate/Nitrite on Lifestyle-Related Diseases
4.1. Insulin Resistance

4.2. Hypertension
4.3. Cardiac Ischemia/Reperfusion Injury
4.4. Chronic Obstructive Pulmonary Disease (COPD)
4.5. Cancer
4.6. Osteoporosis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- O’Donoghue, G.; Cunninggham, C.; Murphy, F.; Woods, C.; Aagaard-Hansen, J. Assessment and management of risk factors for the prevention of lifestyle-related disease: A cross-sectional survey of current activities, barriers and perceived training needs of primary care physiotherapists in the Republic of Ireland. Physiotharapy 2014, 100, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Bergmann, M.M.; Boeing, H.; Capewell, S. Healthy lifestyle behaviors and all-cause mortality among adults in the United States. Prev. Med. 2012, 55, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Manson, J.E.; Lee, I.M.; Cole, S.R.; Hennekens, C.H.; Willett, W.C.; Buring, J. Fruit and vegetable intake and risk of cardiovascular disease: The women’s health study. Am. J. Cin. Nutr. 2000, 72, 922–928. [Google Scholar]
- Joshipura, K.J.; Hu, F.B.; Manson, J.E.; Stampfer, M.J.; Rimm, E.B.; Speizer, F.E.; Colditz, G.; Ascherio, A.; Rosner, B.; Spiegelman, D.; et al. The effect of fruit and vegetable intake on risk for coronary heart disease. Ann. Intern. Med. 2001, 134, 1106–1114. [Google Scholar] [CrossRef] [PubMed]
- Bazzano, L.A.; He, J.; Ogden, L.G.; Loria, C.M.; Vupputuri, S.; Myers, L.; Whelton, P.K. Fruit and vegerable intake and risk of cardiovascular disease in US adults: The first National Health and Nutrition Examination Survey Epidemiologic Follow-up Study. Am. J. Clin. Nutr. 2002, 76, 93–99. [Google Scholar] [PubMed]
- Daucher, L.; Amouyel, P.; Hercberg, S.; Dallongeville, J. Fruit and vegetable consumption and risk of coronary heart disease: A meta-analysis of cohort studies. J. Nutr. 2006, 136, 2588–2593. [Google Scholar]
- Joshipura, K.J.; Ascherio, A.; Manson, J.E.; Stampfer, M.J.; Rimm, E.B.; Speizer, F.E.; Hennekens, C.H.; Spiegelman, D.; Willett, W.C. Fruit and vegetable intake in relation to risk of ischemic stroke. JAMA 1999, 282, 1233–1239. [Google Scholar] [CrossRef] [PubMed]
- Davignon, J.; Ganz, P. Role of endothelial dysfunction in atherosclerosis. Circulation 2004, 109, III-27–III-32. [Google Scholar] [CrossRef] [PubMed]
- Hess, D.T.; Matsumoto, A.; Kim, S.O.; Marshall, H.E.; Stamler, J.S. Protein S-nitrosylation: Purview and parameters. Nat. Rev. Mol. Cell Biol. 2005, 6, 150–165. [Google Scholar] [CrossRef] [PubMed]
- Bryan, N.S.; Fernandez, B.O.; Bauer, S.M.; Garcia-Saura, M.F.; Milsom, A.B.; Rassaf, T.; Maloney, R.E.; Bharti, A.; Rodriguez, J.; Feelisch, M. Nitrite is a signaling molecule and regulator of gene expression in mammalian tissues. Nat. Chem. Biol. 2005, 1, 290–297. [Google Scholar] [CrossRef] [PubMed]
- West, M.B.; Hill, B.G.; Xuan, Y.T.; Bhatnagar, A. Protein glutathiolation by nitric oxide: An intracellular mechanism regulating redox protein modification. FASEB J. 2006, 20, E1049–E1060. [Google Scholar] [CrossRef] [PubMed]
- Larsen, F.J.; Schiffer, T.A.; Borniquel, S.; Sahlin, K.; Ekblom, B.; Lundberg, J.O.; Weitzberg, E. Dietary inorganic nitrate improves mitochondrial efficiency in humans. Cell Metab. 2011, 13, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Nair, K.S.; Irving, B.A.; Lanza, I.R. Can dietary nitrates enhance the efficiency of mitochondria? Cell Metab. 2011, 13, 117–118. [Google Scholar] [CrossRef] [PubMed]
- Melino, G.; Bernassola, F.; Knight, R.A.; Corasaniti, M.T.; Nistico, G.; Finazzi-Agro, A. S-nitrosylation regulates apoptosis. Nature 1997, 388, 432–433. [Google Scholar] [CrossRef] [PubMed]
- Weitzberg, E.; Lundberg, J.O. Nonenzymatic nitric oxide production in humans. Nitric Oxide 1998, 2, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Sindelar, J.J.; Milkowski, A.L. Human safety controversies surrounding nitrate and nitrite in the diet. Nitric Oxide 2012, 26, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Ysart, G.; Miller, P.; Barrett, G.; Farrington, D.; Lawrance, P.; Harrison, M. Dietary expoures to nitrate in the UK. Food Addit. Contamin. 1999, 16, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O.; Weitzberg, E.; Gladwin, M.T. The nitrate-nitrite-nitric oxide pathway in physiology and therapeutics. Nat. Rev. Drug Discov. 2008, 7, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O.; Gladwin, M.T.; Ahluwalia, A.; Benjamin, N.; Bryan, N.S.; Butler, A.; Cabrales, P.; Fago, A.; Feelisch, M.; Ford, P.C.; et al. Nitrate and nitrite in biology, nutrition and therapeutics. Nat. Chem. Biol. 2009, 5, 865–869. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O.; Govoni, M. Inorganic nitrate is a possible source for systemic generation of nitric oxide. Free Radic. Biol. Med. 2004, 37, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Spiegelhalder, B.; Eisenbrand, G.; Preussmann, R. Influence of dietary nitrate on nitrite content of human saliva: Possible relevance to in vivo formation of N-nitroso compounds. Food Cosmet. Toxicol. 1976, 14, 545–548. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Hellstrom, P.M.; Lundberg, J.M.; Alving, K. Greatly increased luminal nitric oxide in ulcerative colitis. Lancet 1994, 344, 1673–1674. [Google Scholar] [CrossRef]
- McKnight, G.M.; Smith, L.M.; Drummond, R.S.; Duncan, C.W.; Golden, M.; Benjamin, N. Chemical synthesis of nitric oxide in the stomach from dietary nitrate in humans. Gut 1997, 40, 211–214. [Google Scholar] [CrossRef] [PubMed]
- Takahama, U.; Oniki, T.; Hirota, S. Oxidation of quercetin by salivary components. Quercetin-dependent reduction of salivary nitrite under acidic conditions producing nitric oxide. J. Agric. Food Chem. 2002, 50, 4317–4322. [Google Scholar] [CrossRef] [PubMed]
- Björne, H.; Peterson, J.; Phillipson, M.; Weitzberg, E.; Holm, L.; Lundberg, J.O. Nitrite in saliva increases gastric mucosal blood flow and mucus thickness. J. Clin. Investig. 2004, 113, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Petersson, J.; Phillipson, M.; Jansson, E.A.; Patzak, A.; Lundberg, J.O.; Holm, L. Dietary nitrate increases gastric mucosal blood flow and mucosal defence. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 292, G718–G724. [Google Scholar] [CrossRef] [PubMed]
- Govoni, M.; Jansson, E.A.; Weitzberg, E.; Lundberg, J.O. The increase in plasma nitrite after a dietary nitrate load is markedly attenuated by an antibacterial mouthwash. Nitric Oxide 2008, 19, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Hord, N.G.; Tang, Y.; Bryan, N.S. Food sources of nitrates and nitrites: The physiologic context for potential health benefits. Am. J. Clin. Nutr. 2009, 90, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bryan, N.S.; Calvert, J.W.; Elrod, J.W.; Gundewar, S.; Ji, S.Y.; Lefer, D.J. Dietary nitrite supplementation protects against ischemia-reperfusion injury. Proc. Natl. Acad. Sci. USA 2007, 104, 19144–19149. [Google Scholar] [CrossRef] [PubMed]
- Raat, N.J.H.; Noguchi, A.C.; Liu, V.B.; Raghavachari, N.; Liu, D.; Xu, X.; Shiva, S.; Munson, P.J.; Gladwin, M.T. Dietary nitrate and nitrite modulate blood and organ nitrite and the cellular ischemic stress response. Free Radic. Biol. Med. 2009, 47, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Cosby, K.; Partovi, K.S.; Crawford, J.H.; Patel, R.P.; Reiter, C.D.; Martyr, S.; Yang, B.K.; Waclawiw, M.A.; Zalos, G.; Xu, X.; et al. Nitrite reduction to nitric oxide by deoxyhemoglobin vasodilates the human circulation. Nat. Med. 2003, 9, 1498–1505. [Google Scholar] [CrossRef] [PubMed]
- Ormerod, J.O.M.; Ashrafian, H.; Maher, A.R.; Arif, S.; Steeples, V.; Born, G.V.R.; Egginton, S.; Feelisch, M.; Watkins, H.; Frenneaux, M.P. The role of vascular myoglobin in nitrite-mediated blood vessel relaxation. Cardiovasc. Res. 2011, 89, 560–565. [Google Scholar] [CrossRef] [PubMed]
- Shiva, S.; Sack, M.N.; Greer, J.J.; Duranski, M.; Ringwood, L.A.; Burwell, L.; Wang, X.; MacArthur, P.H.; Shoja, A.; Raghavachari, N.; et al. Nitrite augments tolerance to ischemia/reperfusion injury via the modulation of mitochondrial electron transfer. J. Exp. Med. 2007, 204, 2089–2102. [Google Scholar] [CrossRef] [PubMed]
- Sonoda, K.; Ohtake, K.; Kubo, Y.; Uchida, H.; Uchida, M.; Natsume, H.; Kobayashi, M.; Kobayashi, J. Aldehyde dehydrogenase 2 partly mediates hypotensive effect of nitrite on L-NAME-induced hypertension in normoxic rat. Clin. Exp. Hypertens. 2014, 36, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Richardson, R.S.; Noyszewski, E.A.; Kendrick, K.F.; Leigh, J.S.; Wagner, P.D. Myoglobin O2 desaturation during exercise. Evidence of limited O2 transport. J. Clin. Investig. 1996, 96, 1916–1926. [Google Scholar] [CrossRef] [PubMed]
- Larsen, F.J.; Weitzberg, E.; Lundberg, J.O.; Ekblom, B. Dietary nitrate reduces maximal oxygen consumption while maintaining work performance in maximal exercise. Free Radic. Biol. Med. 2010, 48, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Carlström, M.; Persson, A.E.G.; Larsson, E.; Hezel, M.; Scheffer, P.G.; Teerlink, T.; Weitzberg, E.; Lundberg, J.O. Dietary nitrate attenuates oxidative stress, prevents cardiac and renal injuries, and reduces blood pressure in salt-induced hypertension. Cardiovasc. Res. 2011, 89, 574–585. [Google Scholar] [CrossRef] [PubMed]
- Baliga, R.S.; Milsom, A.B.; Ghosh, S.M.; Trinder, S.L.; MacAllister, R.J.; Ahluwalia, A.; Hobbs, A.J. Dietary nitrate ameliorates pulmonary hypertension cytoprotective role for endothelial nitric oxide synthase and xanthine oxidoreductase. Circulation 2012, 125, 2922–2932. [Google Scholar] [CrossRef] [PubMed]
- Ashmore, T.; Fernandez, B.O.; Branco-Price, C.; West, J.A.; Cowburn, A.S.; Heather, L.C.; Griffin, J.L.; Johnson, R.S.; Feelisch, M.; Murray, A.J. Dietary nitrate increases arginine availability and protects mitochondrial complex I and energetics in the hypoxic rat heart. J. Physiol. 2014, 592, 4715–4731. [Google Scholar] [CrossRef] [PubMed]
- Stamler, J.S.; Lamas, S.; Fang, F.C. Nitrosylation: The prototypic redox-based signaling mechanism. Cell 2001, 106, 675–683. [Google Scholar] [CrossRef]
- Knobeloch, L.; Salna, B.; Hogan, A.; Postle, J.; Anderson, H. Blue babies and nitrate-contaminated well water. Environ. Health Perspect. 2000, 108, 675–678. [Google Scholar] [CrossRef] [PubMed]
- Comly, H.H. Cyanosis in infants caused by nitrates in well water. JAMA 1945, 129, 112–116. [Google Scholar] [CrossRef]
- Avery, A.A. Infantile methemoglobinemia: Reexamining the role of drinking water nitrates. Environ. Health Perspect. 1999, 107, 583–586. [Google Scholar] [CrossRef] [PubMed]
- Mensinga, T.T.; Speijers, G.J.; Meulenbelt, J. Health implications of exposure to environmental nitrogenous compounds. Toxicol. Rev. 2003, 22, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Gangolli, S.D.; van den Brandt, P.A.; Feron, V.J.; Janzowskyd, C.; Koemane, J.H.; Speijersf, G.J.A.; Spiegelhalderg, B.; Walkerh, R.; Wishnoki, J.S. Nitrate, nitrite and N-nitroso compounds. Eur. J. Pharmacol. 1994, 292, 1–38. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.H.; Aickin, M.; Champagne, C.; Craddick, S.; Sacks, F.M.; McCarron, P.; Most-Windhauser, M.M.; Rukenbrod, F.; Haworth, L.; Dash-Sodium Collaborative Research Group. Food group sources of nutrients in the dietary pattern s of the DASH-Sodium trial. J. Am. Diet. Assoc. 2003, 103, 488–496. [Google Scholar] [PubMed]
- Kobayashi, J. Nitric oxide and insulin resistance. Immunoendocrinology 2015, 2, 1. [Google Scholar]
- Das, U.N. Insulin: An endogenous cardioprotector. Curr. Opin. Crit. Care 2003, 9, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Abel, E.D. Insulin signaling in heart muscle: Lessons from genetically engineered mouse models. Curr. Hypertens. Rep. 2004, 6, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Montagnani, M.; Koh, K.K.; Quon, M.J. Reciprocal relationships between insulin resistance and endothelial dysfunction: Molecular and pathphysiological mechanismns. Circulation 2006, 113, 1888–1904. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Gao, F.; Ma, X.L. Insulin says NO to cardiovascular disease. Cardiovasc. Res. 2011, 89, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, A.X.; Aylor, K.; Barrett, E.J. Nitric oxide directly promotes vascular endothelial insulin transport. Diabetes 2013, 62, 4030–4042. [Google Scholar] [CrossRef] [PubMed]
- Kim, F.; Pham, M.; Rizzo, N.O.; Morton, G.J.; Wisse, B.E.; Kirk, E.A.; Chait, A.; Schwartz, M.W. Vascular inflammation, insulin resistance and reduced nitric oxide production precede the onset of peripheral insulin resistance. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1982–1988. [Google Scholar] [CrossRef] [PubMed]
- Laffranchi, R.; Gogvadze, V.; Richter, C.; Spinas, G.A. Nitric oxide (nitrogen monoxide, NO) stimulates insulin secretion by inducing calcium release from mitochondria. Biochem. Biophys. Res. Commun. 1995, 217, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Nystrom, T.; Ortsater, H.; Huang, Z.; Zhang, F.; Larsen, F.J.; Weitzberg, E.; Lundbergb, J.O.; Sjöholma, A. Inorganic nitrite stimulates pancreatic islet blood flow and insulin secretion. Free Radic. Biol. Med. 2012, 53, 1017–1023. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.J.; Kim, H.S.; Park, H.S.; Kim, M.O.; Kim, M.; Yun, J.Y.; Kim, E.H.; Lee, S.A.; Lee, S.H.; Koh, E.H.; et al. Nitric oxide increases Insulin sensitivity in skeletal muscle by improving mitochondrial function and insulin signaling. Korean Diabetes J. 2009, 33, 198–205. [Google Scholar] [CrossRef]
- Rizzo, N.O.; Maloney, E.; Pham, M.; Luttrell, I.; Wessells, H.; Tateya, S.; Daum, G.; Handa, P.; Schwartz, M.W.; Kim, F. Reduced NO-cGMP signaling contributes to vascular inflammation and insulin resistance induced by high-fat feeding. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Richey, J.M. The vascular endothelium, a benign restrictive barrier? No! Role of nitric oxide in regulating insulin action. Diabetes 2013, 62, 4006–4008. [Google Scholar] [CrossRef] [PubMed]
- Khoo, N.K.H.; Mo, L.; Zharikov, S.; Kamga, C.; Quesnelle, K.; Golin-Bisello, F.; Li, L.; Wang, Y.; Shiva, S. Nitrite augments glucose uptake in adipocytes through the protein kinase A-dependent stimulation of mitochondrial fusion. Free Radic. Biol. Med. 2014, 70, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Draznin, B. Molecular mechanisms of insulin resistance: Serine phosphorylation of insulin receptor substrate-1 and increased expression of p85α. The two sides of a coin. Diabetes 2006, 55, 2392–2397. [Google Scholar] [CrossRef] [PubMed]
- Carvalho-Filho, M.A.; Ueno, M.; Hirabara, S.M.; Seabra, A.B.; Carvalheria, J.B.C.; Oliveira, M.G.; Velloso, L.A.; Curi, R.; Saad, M.J.A. S-nitrosation of the insulin receptor, insulin receptor substrate 1, and protein kinase B/Akt: A novel mechanism of insulin resistance. Diabetes 2005, 54, 959–967. [Google Scholar] [CrossRef] [PubMed]
- Fisher-Wellman, K.H.; Neufer, P.D. Linking mitochondrial bioenergetics to insulin resistance via redox biology. Trends Endocrinol. Metab. 2012, 23, 142–152. [Google Scholar] [CrossRef] [PubMed]
- De Luca, C.; Olefsky, J.M. Inflammation and insulin resistance. FEBS Lett. 2008, 582, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Hsu, M.F.; Meng, T.C. Enhancement of insulin responsiveness by nitric oxide-mediated inactivation of protein-tyrosine phosphatases. J. Biol. Chem. 2010, 285, 7919–7928. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Torregrossa, A.C.; Potts, A.; Pierini, D.; Aranke, M.; Garg, H.K.; Bryan, N.S. Dietary nitrite improves insulin signaling through GLUT4 translocation. Free Rad. Biol. Med. 2014, 67, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Carlström, M.; Larsen, F.J.; Nystrom, T.; Hazel, M.; Borniquel, S.; Weitzberg, E.; Lundberg, J.O. Dietary inorganic nitrate reverses features of metabolic syndrome in endothelial nitric oxide synthase-deficient mice. Proc. Natl. Acad. Sci. USA 2010, 107, 17716–17720. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, K.; Nakano, G.; Ehara, N.; Sonoda, K.; Ito, J.; Uchida, H.; Kobayashi, J. Dietary nitrite supplementation improves insulin resistance in type 2 diabetic KKA(y) mice. Nitric Oxide 2015, 44, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Khalifi, S.; Rahimipour, A.; Jeddi, S.; Ghanbari, M.; Kazerouni, F.; Ghasemi, A. Dietary nitrate improves glucose tolerance and lipid profile in an animal model of hyperglycemia. Nitric oxide 2015, 44, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Biasucci, L.M.; Graziani, F.; Rizzello, V.; Liuzzo, G.; Guidone, C.; Caterina, A.R.D.; Brugaletta, S.; Mingrone, G.; Crea, F. Paradoxical preservation of vascular function in severe obesity. Am. J. Med. 2010, 123, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Assar, M.E.I.; Adana, J.C.R.D.; Angulo, J.; Martinez, M.L.P.; Matias, A.H.; Rodriguez-Manas, L. Preserved endothelial function in human obesity in the absence of insulin resistance. J. Transl. Med. 2013, 11, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Sansbury, B.E.; Cummins, T.D.; Tang, Y.; Hellmann, J.; Holden, C.R.; Harbeson, H.M.A.; Chen, Y.; Patel, R.P.; Spite, M.; Bhatnagar, A.; et al. Overexpression of endothelial nitric oxide synthase prevents diet-induced obesity and regulates adipocyte phenotype. Circ. Res. 2012, 111, 1176–1189. [Google Scholar] [CrossRef] [PubMed]
- Ness, A.R.; Powles, J.W. Fruit and vegetables, and cardiovascular disease: A review. Int. J. Epidemiol. 1997, 26, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Van’t Veer, P.; Jansen, M.C.; Klerk, M.; Kok, F.J. Fruits and vegetables in the prevention of cancer and cardiovascular disease. Public Health Nutr. 2000, 3, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Bazzano, L.A.; Serdula, M.K.; Liu, S. Dietary intake of fruits and vegetables and risk of cardiovascular disease. Curr. Atheroscler. Rep. 2003, 5, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R., 3rd; Simons-Morton, D.G.; et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A clinical trial of the effects of dietary patterns on blood pressure. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, A.; Mitchell, K.; Blackwell, J.; Vanhatalo, A.; Jones, A.M. High-nitrate vegetable diet increases nitrate and nitrite concentrations and reduces blood pressure in healthy women. Public Health Nutr. 2015. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, F.M.; Shiva, S. Nitrite anion provides potent cytoprotective and antiapoptotic effects as adjunctive therapy to reperfusion for acute myocardial infarction. Circulation 2008, 117, 2986–2994. [Google Scholar] [CrossRef] [PubMed]
- Duranski, M.R.; Greer, J.J.; Dejam, A. Cytoprotective effects of nitrite during in vivo ischemia-reperfusion of the heart and liver. J. Clin. Investig. 2005, 115, 1232–1240. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.; Bond, R.; McLean, P.; Uppal, R.; Benjamin, N.; Ahluwalia, A. Reduction of nitrite to nitric oxide during ischemia protects against myocardial ischemia-reperfusion damage. Proc. Natl. Acad. Sci. USA 2004, 101, 13683–13688. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.E.; Su, J.; Fu, X.; Hsu, A.; Gross, G.J.; Tweddell, J.S.; Hogg, N. Nitrite confers protection against myocardial infarction: Role of xanthine oxidoreductase, NADPH oxidase and K(ATP) channels. J. Mol. Cell Cardiol. 2007, 43, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Bryan, N.S.; Calvert, J.W.; Gundewar, S.; Lefer, D.J. Dietary nitrite restores NO homeostasis and is cardioprotective in endothelial nitric oxide synthase-deficient mice. Free Radic. Biol. Med. 2008, 45, 468–474. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G., III; Tsao, P.S.; Mulloy, D.; Lefer, A.M. Cardioprotective effects of acidified sodium nitrite in myocardial ischemia with reperfusion. J. Pharmacol. Exp. Ther. 1990, 252, 35–41. [Google Scholar] [PubMed]
- Larsen, F.J.; Ekblom, B.; Sahlin, K.; Lundberg, J.O.; Weitzberg, E. Effects of dietary nitrate on blood pressure in healthy volunteers. N. Engl. J. Med. 2006, 355, 2792–2793. [Google Scholar] [CrossRef] [PubMed]
- Kapil, V.; Milsom, A.B.; Okorie, M.; Maleki-Toyserkani, S.; Akram, F.; Rehman, F.; Arghandawi, S.; Pearl, V.; Benjamin, N.; Loukogeorgakis, S.; et al. Inorganic nitrate supplementation lowers blood pressure in humans: Role for nitrite-derived NO. Hypertension 2010, 56, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.J.; Patel, N.; Loukogeorgakis, S.; Okorie, M.; Aboud, Z.; Misra, S.; Rashid, R.; Miall, P.; Deanfield, J.; Benjamin, N.; et al. Acute blood pressure lowering, vasoprotective, and antiplatelet properties of dietary nitrate via bioconversion to nitrite. Hypertension 2008, 51, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Vanhatalo, A.; Bailey, S.J.; Blackwell, J.R.; DiMenna, F.J.; Pavey, T.G.; Wilkerson, D.P.; Benjamin, N.; Winyard, P.G.; Jones, A.M. Acute and chronic effects of dietary nitrate supplementation on blood pressure and the physiological responses to moderate-intensity and incremental exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 68, R1121–R1131. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, D.A.; George, T.W.; Lovegrove, J.A. The effects of dietary nitrate on blood pressure and endothelial function: A review of human intervention studies. Nutr. Res. Rev. 2013, 26, 210–222. [Google Scholar] [CrossRef] [PubMed]
- Siervo, M.; Lala, J.; Ogbonmwan, I.; Mathers, J.C. Inorganic nitrate and beetroot juice supplementation reduces blood pressure in adults: A systematic review and meta-analysis. J. Nutr. 2013, 143, 818–826. [Google Scholar] [CrossRef] [PubMed]
- Kapil, V.; Khambata, R.S.; Robertson, A.; Caulfield, M.J.; Ahluwalia, A. Dietary nitrate provides sustained blood pressure lowering in hypertensive patients. Hypertension 2015, 65, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Sindler, A.L.; DeVan, A.E.; Fleenor, B.S.; Seals, D.R. Inorganic nitrite supplementation for healthy arterial aging. J. Appl. Physiol. 2014, 116, 463–477. [Google Scholar] [CrossRef] [PubMed]
- Bhushan, S.; Kondo, K.; Polhemus, D.J.; Otsuka, H.; Nicholson, C.K.; Tao, Y.X.; Huang, H.; Georgiopoulou, V.V.; Murohara, T.; Calvert, J.W.; et al. Nitrite therapy improves left ventricular function during heart failure via restoration of nitric oxide-mediated cytoprotective signaling. Circ. Res. 2014, 114, 1281–1291. [Google Scholar] [CrossRef] [PubMed]
- Leist, M.; Single, B.; Castoldi, A.F.; Kuhnle, S.; Nicotera, P. Intracellular adenosine triphosphate (ATP) concentration: A switch in the decision between apoptosis and necrosis. J. Exp. Med. 1997, 185, 1481–1486. [Google Scholar] [CrossRef] [PubMed]
- Eguchi, Y.; Shimizu, S.; Tsujimoto, Y. Intracellular ATP levels determine cell death fate by apoptosis or necrosis. Cancer Res. 1997, 57, 1835–1840. [Google Scholar] [PubMed]
- Murata, I.; Nozaki, R.; Ooi, K.; Ohtake, K.; Kimura, S.; Ueda, H.; Nakano, G.; Sonoda, K.; Inoue, Y.; Uchida, H.; et al. Nitrite reduces ischemia/reperfusion-induced muscle damage and improves survival rates in rat crush injury model. J. Trauma Acute Care Surg. 2012, 72, 1548–1554. [Google Scholar] [CrossRef] [PubMed]
- Shiva, S.; Wang, X.; Ringwood, L.A.; Xu, X.; Yuditskaya, S.; Annavajjhala, V.; Miyajima, H.; Hogg, N.; Harris, Z.L.; Gladwin, M.T. Ceruloplasmin is a NO oxidase and nitrite synthase that determines endocrine NO homeostasis. Nat. Chem. Biol. 2006, 9, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Bryan, N.S.; Rassaf, T.; Maloney, R.E.; Rodriguez, C.M.; Saijo, F.; Rodriguez, J.R.; Feelisch, M. Cellular targets and mechanisms of nitros(yl)ation: An insight into their nature and kinetics in vivo. Proc. Natl. Acad. Sci. USA 2004, 101, 4308–4313. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.P.; Greer, J.J.M.; Kakkar, A.K.; Ware, P.D.; Turnage, R.H.; Hicks, M.; van Haeren, R.; de Crom, R.; Kawashima, S.; Yokoyama, M.; et al. Endothelial nitric oxide synthase overexpression attenuates myocardial reperfusion injury. Am. J. Physiol. Heart Circ. Physiol. 2004, 286, H276–H282. [Google Scholar] [CrossRef] [PubMed]
- Rassaf, T.; Flögel, U.; Drexhage, C.; Hendgen-Cotta, U.; Kelm, M.; Schrader, J. Nitrite reductase function of deoxymyoglobin: Oxygen sensor and regulator of cardiac energetics and function. Circ. Res. 2007, 100, 1749–1754. [Google Scholar] [CrossRef] [PubMed]
- Hendgen-Cotta, U.B.; Merx, M.W.; Shiva, S.; Schmitz, J.; Becher, S.; Klare, J.P.; Steinhoff, H.J.; Goedecke, A.; Schrader, J.; et al. Nitrite reductase activity of myoglobin regulates respiration and cellular viability in myocardial ischemia-reperfusion injury. Proc. Natl. Acad. Sci. USA 2008, 105, 10256–10261. [Google Scholar] [CrossRef] [PubMed]
- Shiva, S.; Gladwin, M.T. Nitrite mediates cytoprotection after ischemia-reperfusion by modulating mitochondrial function. Basic Res. Cardiol. 2009, 104, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Calvert, J.W.; Lefer, D.J. Myocardial protection by nitrite. Cardiovasc. Res. 2009, 83, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Vivekananthan, D.; Penn, M.S.; Sapp, S.K.; Hsu, A.; Topol, E.J. Use of antioxidant vitamins for the prevention of cardiovascular disease: Meta-analysis of randomized trials. Lancet 2003, 361, 2017–2023. [Google Scholar] [CrossRef]
- Bjelakovic, G.; Nikolova, D.; Gluud, L.L.; Simonetti, R.G.; Gluud, C. Mortality in randomized trials of antioxidant supplements for primary and secondary prevention: Systemic review and meta-analysis. JAMA 2007, 297, 842–857. [Google Scholar] [CrossRef] [PubMed]
- Sesso, H.D.; Buring, J.E.; Christen, W.G.; Kurth, T.; Belanger, C.; MacFadyen, J.; Bubes, V.; Manson, J.E.; Glynn, R.J.; Gaziano, J.M. Vitamins E and C in the prevention of cardiovascular disease in men. JAMA 2008, 300, 2123–2133. [Google Scholar] [CrossRef] [PubMed]
- Hung, H.C.; Joshipura, K.J.; Jiang, R.; Hu, F.B.; Hunter, D.; Smith-Warner, S.A.; Colditz, G.A.; Rosner, B.; Spiegelman, D.; Willett, W.C. Fruit and vegetable intake and risk of major chronic disease. J. Natl. Cancer Inst. 2004, 96, 1577–1584. [Google Scholar] [CrossRef] [PubMed]
- Wedzicha, J.A.; Seemungal, T.A.R. COPD exacerbations: Defining their cause and prevention. Lancet 2007, 370, 786–796. [Google Scholar] [CrossRef]
- Varraso, R.; Chiuve, S.E.; Fung, T.T.; Barr, R.G.; Hu, F.B.; Willett, W.C.; Camargo, C.A. Alternate healthy eating index 2010 and risk of chronic obstructive pulmonary disease among US women and men: Prospective study. Brit. Med. J. 2015, 350, h286. [Google Scholar] [CrossRef] [PubMed]
- Jiang, R.; Paik, D.C.; Hankinson, J.L.; Barr, R.G. Cured meat consumption, lung function, and chronic obstructive pulmonary disease among United States adult. Am. J. Respir. Crit. Care Med. 2007, 175, 798–804. [Google Scholar] [CrossRef] [PubMed]
- De Batlle, J.; Mendez, M.; Romieu, I.; Balcells, E.; Benet, M.; Donaire-Gonzalez, D.; Ferrer, J.J.; Orozco-Levi, M.; Anto, J.M.; Garcia-Aymerich, J. Cured meat consumption increases risk of readmission in COPD patients. Eur. Respir. J. 2012, 40, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Varraso, R.; Jiang, R.; Barr, R.G.; Willett, W.C.; Carlos, A. Prospective study of cured meats consumption and risk of chronic obstructive pulmonary disease in men. Am. J. Epidemiol. 2007, 166, 1438–1445. [Google Scholar] [CrossRef] [PubMed]
- Shuval, H.I.; Gruener, N. Epidemiological and toxicological aspects of nitrates and nitrites in the environment. Am. J. Public Health 1972, 62, 1045–1052. [Google Scholar] [CrossRef] [PubMed]
- Hsu, J.; Arcot, J.; Lee, N.A. Nitrate and nitrite quantification from cured meat and vegetables and their estimated dietary intake in Australians. Food Chem. 2009, 115, 334–339. [Google Scholar] [CrossRef]
- Archer, D.L. Evidence that ingested nitrate and nitrite are beneficial to health. J. Food Prot. 2002, 65, 872–875. [Google Scholar] [PubMed]
- Cassens, R.G. Residual nitrite in cured meat. Food Technol. 1997, 51, 53–55. [Google Scholar]
- Romieu, I.; Trenga, C. Diet and obstructive lung diseases. Epidemiol. Rev. 2001, 23, 268–287. [Google Scholar] [CrossRef] [PubMed]
- Romieu, I. Nutrition and lung health. Int. J. Tuberc. Lung Dis. 2005, 9, 362–374. [Google Scholar] [PubMed]
- Denny, S.I.; Thompson, R.L.; Margetts, B.M. Dietary factors in the pathogenesis of asthma and chronic obstructive pulmonary disease. Curr. Allergy Asthma Rep. 2003, 3, 130–136. [Google Scholar] [CrossRef] [PubMed]
- McKeever, T.M.; Scrivener, S.; Broadfield, E.; Jones, Z.; Britton, J.; Lewis, S.A. Prospective study of diet and decline in lung function in a general population. Am. J. Respir. Crit. Care Med. 2002, 165, 1299–1303. [Google Scholar] [CrossRef] [PubMed]
- Butland, B.K.; Fehily, A.M.; Elwood, P.C. Diet, lung function, and lung function decline in a cohort of 2512 middle aged men. Thorax 2000, 55, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Smit, H.A.; Grievink, L.; Tabak, C. Dietary influences on chronic obstructive lung disease and asthma: A review of the epidemiological evidence. Proc. Nutr. Soc. 1999, 58, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Carey, I.M.; Strachan, D.P.; Cook, D.G. Effects of changes in fresh fruit consumption on ventilator function in healthy British adults. Am. J. Respir. Crit. Care Med. 1998, 158, 728–733. [Google Scholar] [CrossRef] [PubMed]
- Weitzberg, E.; Lundberg, J.O. Novel aspects of dietary nitrate and human health. Annu. Rev. Nutr. 2013, 33, 129–159. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, H.; Ohshima, H.; Pignatelli, B. Inhibitors of endogenous nitrosation. Mechanisms and implications in human cancer prevention. Mutat. Res. 1988, 202, 307–324. [Google Scholar] [CrossRef]
- Pannala, A.S.; Mani, A.R.; Spencer, J.P.E.; Skinner, V.; Bruckdorfer, K.R.; Moore, K.P.; Rice-Evans, C.A. The effect of dietary nitrate on salivery, plasma, and urinary nitrate metabolism in humans. Free Radic. Biol. Med. 2003, 34, 576–584. [Google Scholar] [CrossRef]
- Larsen, F.J.; Schiffer, T.A.; Ekblom, B.; Mattsson, M.P.; Checa, A.; Wheelock, C.E.; Nystrom, T.; Lundberg, J.O.; Weitzberg, E. Dietary nitrate reduces resting metabolic rate: A randomized, crossover study in humans. Am. J. Clin. Nutr. 2014, 99, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Ricciardolo, F.L.M.; Sterk, P.J.; Gaston, B.; Folkerts, G. Nitric oxide in health and disease of the respiratory system. Physiol. Rev. 2004, 84, 731–765. [Google Scholar] [CrossRef] [PubMed]
- Hansel, T.T.; Kharitonov, S.A.; Donnelly, L.E.; Erin, E.M.; Currie, M.G.; Moore, W.M.; Manning, P.T.; Recker, D.P.; Barnes, P.J. A selective inhibitor of inducible nitric oxide synthase inhibits exhaled breath nitric oxide in healthy volunteers and asthmatics. FASEB J. 2003, 17, 1298–1300. [Google Scholar] [CrossRef] [PubMed]
- Brindicci, C.; Ito, K.; Resta, O.; Pride, N.B.; Barnes, P.J.; Kharitonov, S.A. Exhaled nitric oxide from lung periphery is increased in COPD. Eur. Respir. J. 2005, 26, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Berry, M.J.; Justus, N.W.; Hauser, J.I.; Case, A.H.; Helms, C.C.; Basu, S.; Rogers, Z.; Lewis, M.T.; Miller, G.D. Dietary nitrate supplementation improves exercise performance and decreases blood pressure in COPD patients. Nitric Oxide 2014. [Google Scholar] [CrossRef] [PubMed]
- Kerley, C.P.; Cahill, K.; Bolger, K.; McGowan, A.; Burke, C.; Faul, J.; Cormican, L. Dietary nitrate supplementation in COPD: An acute, double-blind, randomized, placebo-controlled, crossover trial. Nitric Oxide 2015, 44, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Iijima, K.; Grant, J.; McElroy, K.; Fyfe, V.; Preston, T.; McColl, K.E. Novel mechanism of nitrosative stress from dietary nitrate with relevance to gastro-oesophageal junction cancers. Cartinogenesis 2003, 24, 1951–1960. [Google Scholar] [CrossRef] [PubMed]
- Magee, P.N.; Barnes, J.M. The production of malignant primary hepatic tumours in the rat by feeding dimethylnitrosamine. Br. J. Cancer 1956, 10, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Mirvish, S.S. N-nitroso compounds: Their chemical and in vivo formation and possible importance as environmental carcinogenesis. J. Toxicol. Environ. Health 1977, 2, 1267–1277. [Google Scholar] [CrossRef] [PubMed]
- Mirvish, S.S. Role of N-nitroso compounds (NOC) and N-nitrosation in etiology of gastric, esophageal, nasopharyngeal and bladder cancer and contribution to cancer of known exposures to NOC. Cancer Lett. 1995, 93, 17–48. [Google Scholar] [CrossRef]
- Bryan, N.S.; Alexander, D.D.; Coughlin, J.R.; Milkowski, A.L.; Boffetta, P. Ingested nitrate and nitrite and stomach cancer risk: An updated review. Food Chem. Toxicol. 2012, 50, 3646–3665. [Google Scholar] [CrossRef] [PubMed]
- Buiatti, E.; Palli, D.; Decarli, A.; Amadori, D.; Avellini, C.; Bianchi, S.; Biserni, R.; Cipriani, F.; Cocco, P.; Giacosa, A.; et al. A case-control study of gastric cancer and diet in Italy. Int. J. Cancer 1989, 44, 611–616. [Google Scholar] [CrossRef] [PubMed]
- Ward, M.H.; López-Carrillo, L. Dietary factors and the risk of gastric cancer in Mexico city. Am. J. Epidemiol. 1999, 149, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Van den Brandt, P.A.; Botterweck, A.A.M.; Goldbohm, A. Salt intake, cured meat consumption, refrigerator use and stomach cancer incidence: A prospective cohort study (Netherlands). Cancer Cause Control 2003, 14, 427–438. [Google Scholar] [CrossRef]
- Kuhnle, G.G.C.; Story, G.W.; Reda, T.; Mani, A.R.; Moore, K.P.; Lunn, J.C.; Bingham, S.A. Diet-induced endogenous formation of nitroso compounds in the GI tract. Free Radic. Biol. Med. 2007, 43, 1040–1047. [Google Scholar] [CrossRef] [PubMed]
- Hogg, N. Red meat and colon cancer: Heme proteins and nitrite in the gut. A commentary on “Diet-induced endogenous formation of nitroso compounds in the GI tract”. Free Radic. Biol. Med. 2007, 43, 1037–1039. [Google Scholar] [CrossRef] [PubMed]
- McEvoy, C.T.; Temple, N.; Woodside, J.V. Vegetarian diets, low-meat diets and health: A review. Public Health Nutr. 2012, 15, 2287–2294. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, M.; Winyard, P.G.; Benjamin, N. Dietary nitrate-good or bad? Nitric Oxide 2010, 22, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Milkowski, A.; Garg, H.K.; Coughlin, J.R.; Bryan, N.S. Nutritional epidemiology in the context of nitric oxide biology: A risk-benefit evaluation for dietary nitrite and nitrate. Nitric Oxide 2010, 15, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Bradbury, K.E.; Appleby, P.N.; Key, T.J. Fruit, vegetable, and fiber intake in relation to cancer risk: Findings from the European Prospective Investigation into Cancer and Nutrition (EPIC). Am. J. Clin. Nutr. 2014, 100, 394S–398S. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.W.; Kim, H.; Kim, KH. NF-κB, inducible nitric oxide synthase and apoptosis by Helicobacter pylori infection. Free Radic. Biol. Med. 2001, 31, 355–366. [Google Scholar] [CrossRef]
- Wilson, K.T.; Ramanujam, K.S.; Mobley, H.L.; Musselman, R.F.; James, S.P.; Meltzer, S.J. Helicobacter pylori stimulates inducible nitric oxide synthase expression and activity in a murine macrophage cell line. Gastroenterology 1996, 111, 1524–1533. [Google Scholar] [CrossRef]
- Jaiswal, M.; LaRusso, N.F.; Gores, G.J. Nitric oxide in gastrointestinal epithelial cell cartinogenesis: Linking inflammation to oncogenesis. Am. J. Physiol. Gastrointest. Liver physiol. 2001, 281, G626–G634. [Google Scholar] [PubMed]
- Zhao, K.; Whiteman, M.; Spencer, J.P.; Halliwell, B. DNA damage by nitrite and peroxynitrite: Protection by dietary phenols. Methods Enzymol. 2001, 335, 296–307. [Google Scholar] [PubMed]
- Wink, D.A.; Vodovotz, Y.; Laval, J.; Laval, F.; Dewhirst, M.W.; Mitchell, J.B. The multifaceted roles of nitric oxide in cancer. Carcinogenesis 1998, 19, 711–721. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, J.R.; Xie, K. Tumors face NO problems? Cancer Res. 2006, 66, 6459–6462. [Google Scholar] [CrossRef] [PubMed]
- Brot, C.; Jorgensen, N.R.; Sorensen, O.H. The influence of smoking on vitamin D status and calcium metabolism. Eur. J. Clin. Nutr. 1999, 53, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Maurel, D.B.; Boisseau, N.; Benhamou, C.L.; Jaffre, C. Alcohol and bone: Review of dose effects and mechanisms. Osteoporos. Int. 2012, 23, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Holbrook, T.L.; Barrett-Connor, E.; Wingard, D.L. Dietary calcium and risk of hip fracture: 14-year prospective population study. Lancet 1988, 332, 1046–1049. [Google Scholar] [CrossRef]
- Feskanich, D.; Willett, W.C.; Colditz, G.A. Calcium, vitamin D, milk consumption, and hip fractures: A prospective study among postmenopausal women. Am. J. Clin. Nutr. 2003, 77, 504–511. [Google Scholar] [PubMed]
- Muraki, S.; Yamamoto, S.; Ishibashi, H.; Oka, H.; Yoshimura, N.; Kawaguchi, H.; Nakamura, K. Diet and lifestyle associated with increased bone mineral density: Cross-sectional study of Japanese elderly women at an osteoporosis outpatient clinic. J. Orthop. Sci. 2007, 12, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Van’t Hof, R.J.; Ralston, S.H. Nitric oxide and bone. Immunology 2001, 103, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Armour, K.E.; van’t Hof, R.J.; Grabowski, P.S.; Reid, D.M.; Ralston, S.H. Evidence for pathogenic role of nitric oxide in inflammation-induced osteoporosis. J. Bone Miner. Res. 1999, 14, 2137–2142. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Yan, H.; Hou, W.; Wu, P.; Tian, J.; Tian, L.; Zhu, B.; Ma, J.; Lu, S. Relationships between endothelial nitric oxide synthase gene polymorphisms and osteoporosis in postmenopausal women. J. Zhejiang. Univ. Sci. B 2009, 10, 609–618. [Google Scholar] [CrossRef] [PubMed]
- Wimalawansa, S.J. Nitric oxide: Novel therapy for osteoporosis. Expert Opin. Pharmacother. 2008, 9, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Wimalawansa, S.J.; de Marco, G.; Gangula, P.; Yallampalli, C. Nitric oxide donor alleviates ovariectomy-induced bone loss. Bone 1996, 18, 301–304. [Google Scholar] [CrossRef]
- Hao, Y.J.; Tang, Y.; Chen, F.B.; Pei, F.X. Different doses of nitric oxide donor prevent osteoporosis in ovariectomized rats. Clin. Orthop. Relat. Res. 2005, 435, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Jamal, S.A.; Reid, L.S.; Hamilton, C.J. The effects of organic nitrates on osteoporosis: A systematic review. Osteoporos. Int. 2013, 24, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Prynne, C.J.; Mishra, G.D.; O’Connell, M.A.; Muniz, G.; Laskey, M.A.; Yan, L.; Prentice, A.; Ginty, F. Fruit and vegetable intakes and bone mineral status: A cross sectional study in 5 age and sex cohorts. Am. J. Clin. Nutr. 2006, 83, 1420–1428. [Google Scholar] [PubMed]
- Tucker, K.L.; Hannan, M.T.; Chen, H.; Cupples, L.A.; Wilson, P.W.; Kiel, D.P. Potassium, magnesium, and fruit and vegetable intakes are associated with greater bone mineral density in elderly men and women. Am. J. Clin. Nutr. 1999, 69, 727–736. [Google Scholar] [PubMed]
- Macdonald, H.M.; New, S.A.; Golden, M.H.; Campbell, M.K.; Reid, D.M. Nutritional associations with bone loss during the menopausal transition: Evidence of a beneficial effect of calcium, alcohol, and fruit and vegetable nutrients and of a detrimental effect of fatty acids. Am. J. Clin. 2004, 79, 155–165. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kobayashi, J.; Ohtake, K.; Uchida, H. NO-Rich Diet for Lifestyle-Related Diseases. Nutrients 2015, 7, 4911-4937. https://doi.org/10.3390/nu7064911
Kobayashi J, Ohtake K, Uchida H. NO-Rich Diet for Lifestyle-Related Diseases. Nutrients. 2015; 7(6):4911-4937. https://doi.org/10.3390/nu7064911
Chicago/Turabian StyleKobayashi, Jun, Kazuo Ohtake, and Hiroyuki Uchida. 2015. "NO-Rich Diet for Lifestyle-Related Diseases" Nutrients 7, no. 6: 4911-4937. https://doi.org/10.3390/nu7064911
APA StyleKobayashi, J., Ohtake, K., & Uchida, H. (2015). NO-Rich Diet for Lifestyle-Related Diseases. Nutrients, 7(6), 4911-4937. https://doi.org/10.3390/nu7064911





