Hypolipidemic Effects and Safety of Lactobacillus Reuteri 263 in a Hamster Model of Hyperlipidemia
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials, Animals, and Experiment Design

2.2. HCD Composition
2.3. Liver and Fecal Lipid Analysis
2.4. Clinical Biochemical Profiles
2.5. Histological Staining of Tissues
2.6. Statistical Analysis
3. Results and Discussion
3.1. Hamster BW and Daily Intake
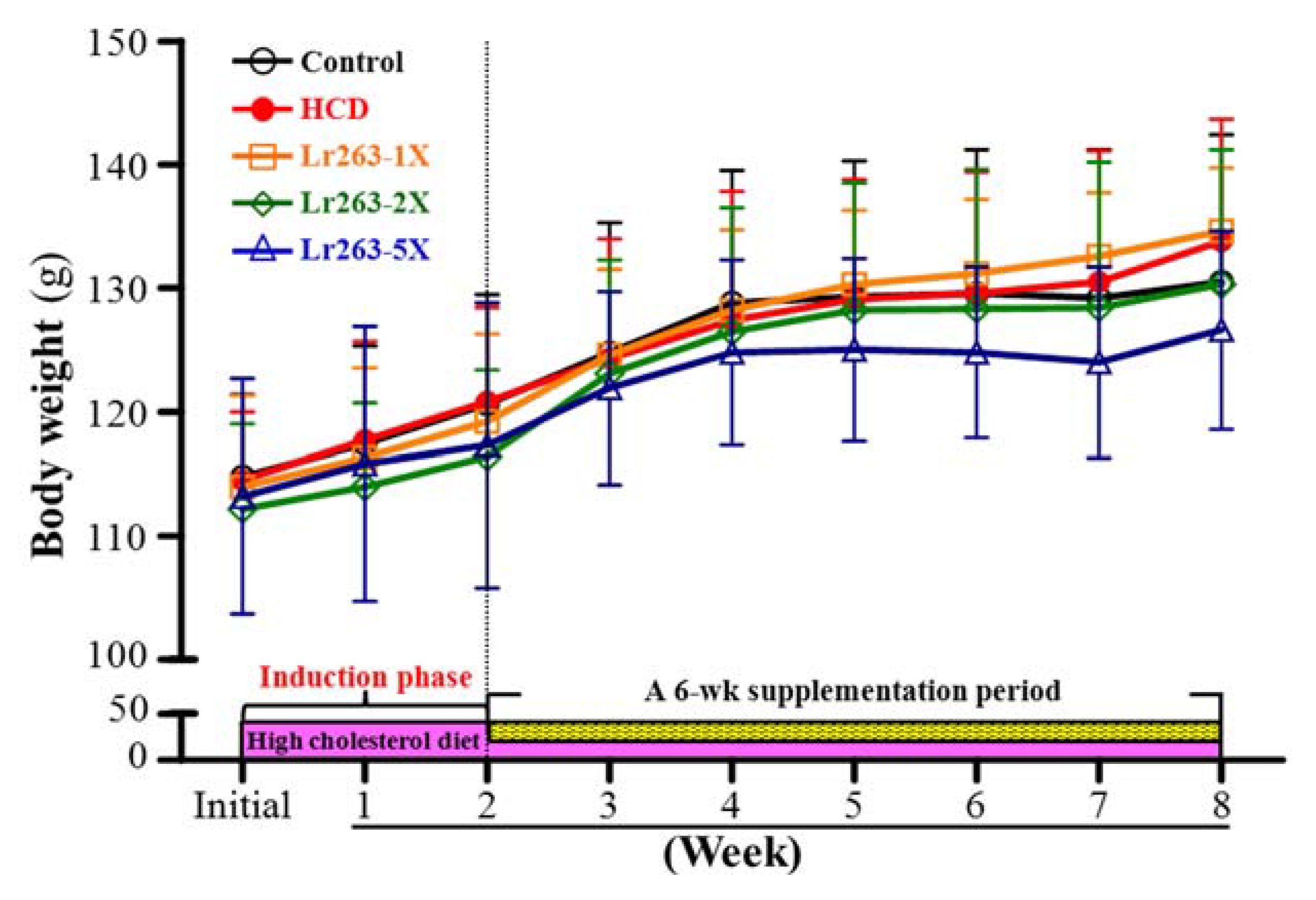
| Characteristics | Control | HCD | Lr263-1X | Lr263-2X | Lr263-5X | Trend Analysis |
|---|---|---|---|---|---|---|
| Initial BW (g) | 115 ± 7 | 114 ± 6 | 114 ± 7 | 112 ± 7 | 113 ± 9 | 0.7344 |
| Final BW (g) | 130 ± 12 | 134 ± 10 | 135 ± 5 | 130 ± 11 | 127 ± 8 | 0.0195 |
| Adaption phase Deit intake (g/hamster/day) | 8.02 ± 0.75 | 7.95 ± 1.04 | 8.00 ± 0.73 | 7.99 ± 0.69 | 8.05 ± 0.52 | 0.8111 |
| Induction phase Deit intake (g/hamster/day) | 9.08 ± 0.19 | 9.04 ± 0.45 | 9.09 ± 0.35 | 9.09 ± 0.32 | 9.01 ± 0.33 | 0.8223 |
| Lr263 Supplementation Deit intake (g/hamster/day) | 8.06 ± 0.68 | 8.23 ± 0.88 | 8.06 ± 0.94 | 8.13 ± 0.75 | 8.22 ± 0.68 | 0.8348 |
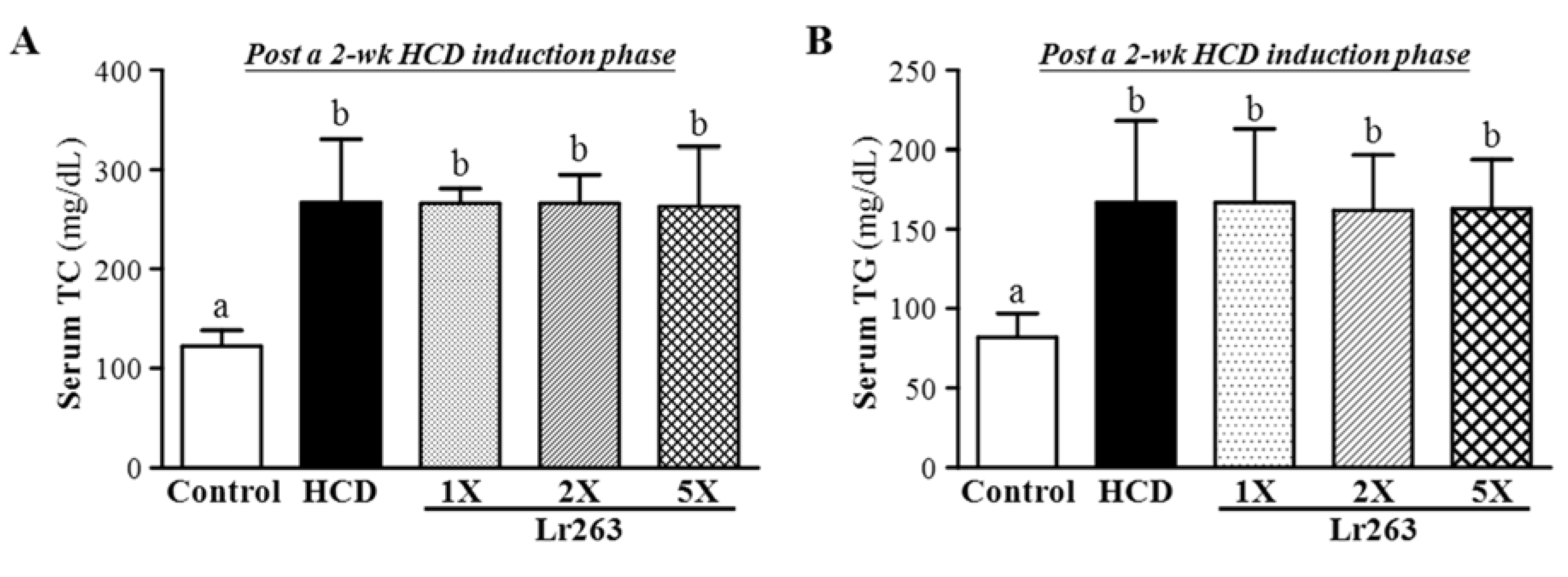
3.2. Effect of Two-Week HCD Induction on Serum TC and TG Levels
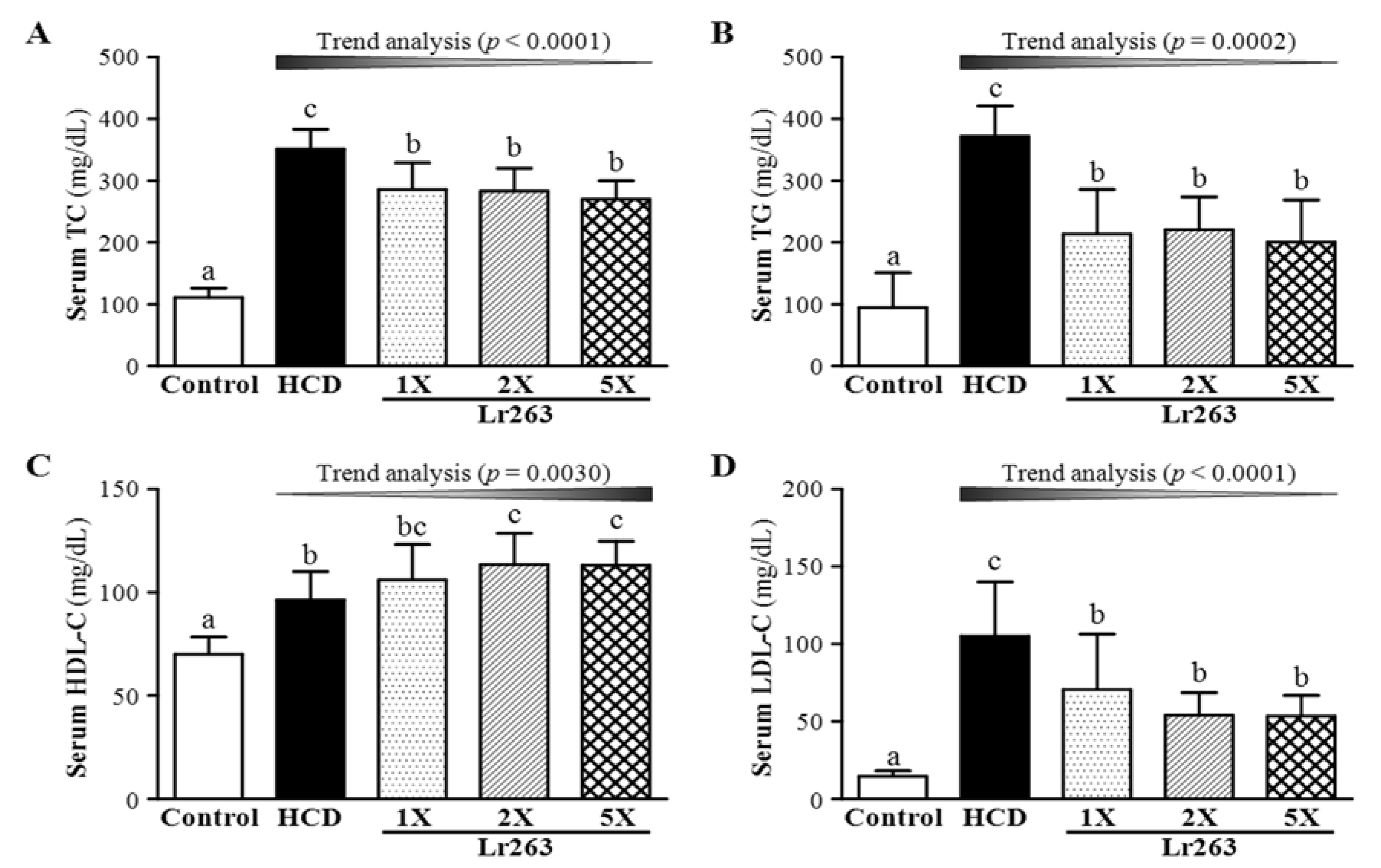
3.3. Effect of Six-Week Supplementation with Lr263 on Serum Lipid Levels and LDL-C/HDL-C Ratio in Hyperlipidemic Hamsters
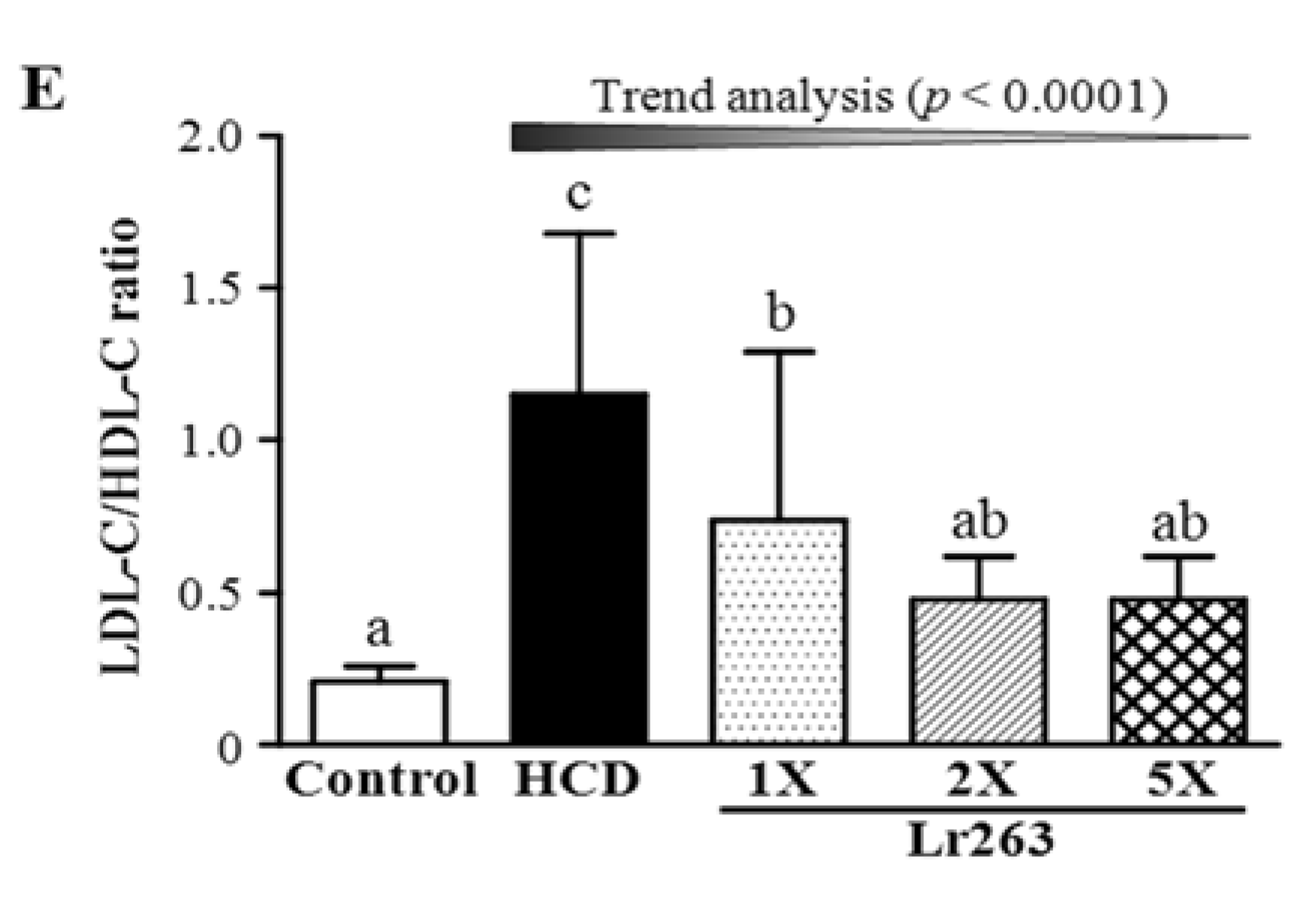
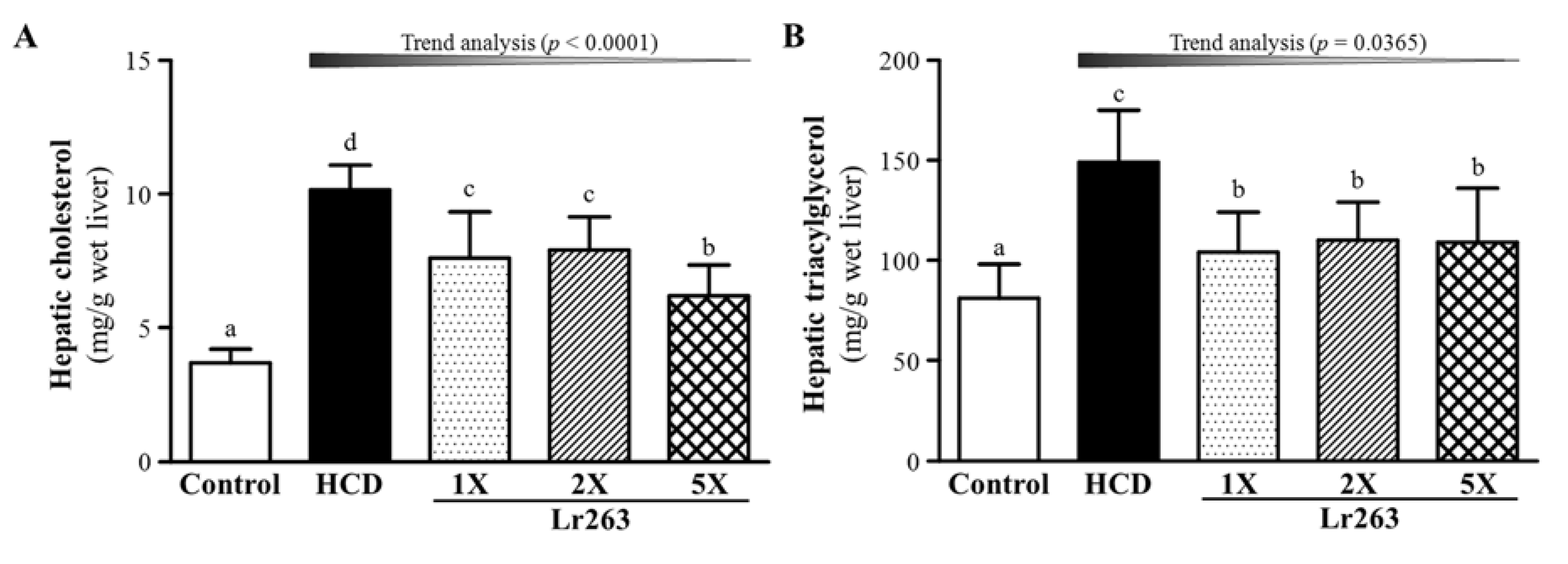
3.4. Effect of Six-Week Supplementation with Lr263 on Hepatic TC and TG Levels in Hyperlipidemic Hamsters
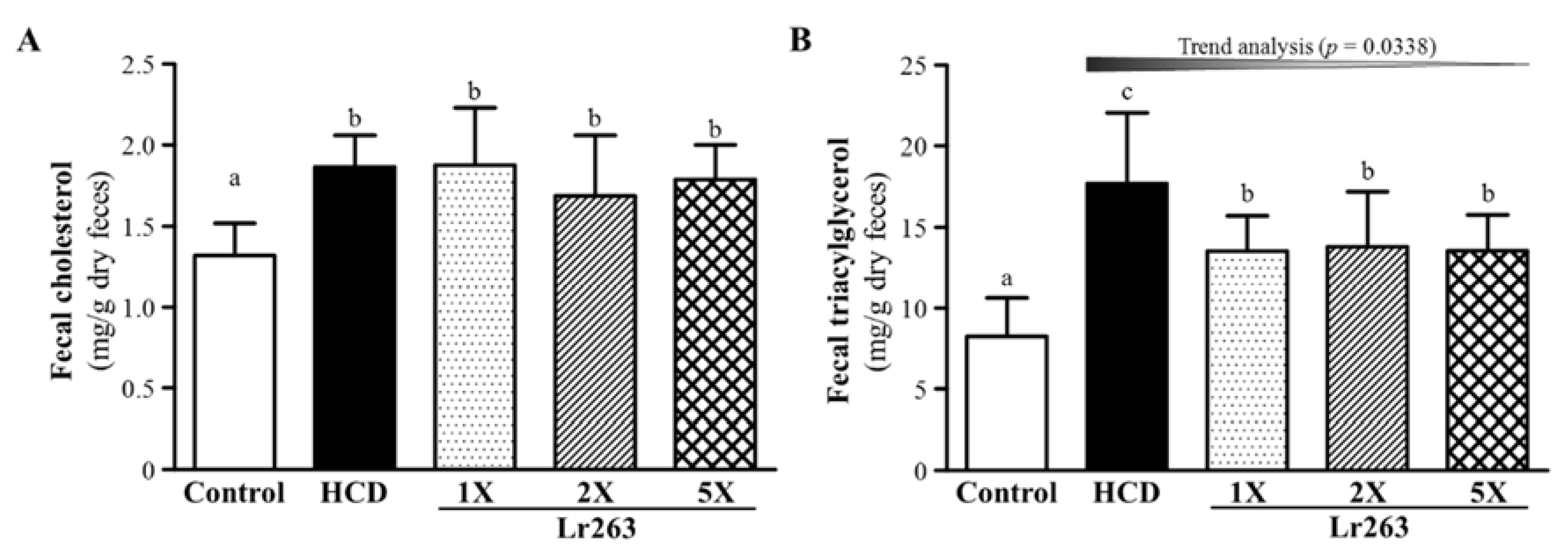
3.5. Effect of Six-Week Supplementation with Lr263 on Fecal TC and TG Levels in Hyperlipidemic Hamsters
3.6. Effect of Lr263 Supplementation on Tissue Weight at the End of the Experiment
| Organ Weight | Control | HCD | Lr263-1X | Lr263-2X | Lr263-5X | Trend Analysis |
|---|---|---|---|---|---|---|
| Liver (g) | 3.50 ± 0.37 a | 5.97 ± 0.28 c | 5.47 ± 0.32 b | 5.47 ± 0.37 b | 5.35 ± 0.31 b | 0.0003 |
| Kidney (g) | 1.17 ± 0.05 | 1.14 ± 0.05 | 1.15 ± 0.06 | 1.17 ± 0.08 | 1.13 ± 0.07 | 0.8553 |
| Heart (g) | 0.53 ± 0.04 | 0.50 ± 0.05 | 0.52 ± 0.06 | 0.53 ± 0.02 | 0.50 ± 0.03 | 0.8219 |
| EFP (g) | 2.12 ± 0.34 a | 2.98 ± 0.59 b | 2.56 ± 0.27 ab | 2.40 ± 0.49 a | 2.41 ± 0.40 a | 0.0095 |
| Relative liver (%) | 2.84 ± 0.19 a | 4.76 ± 0.52 c | 4.25 ± 0.39 b | 4.43 ± 0.25 bc | 4.48 ± 0.43 bc | 0.6720 |
| Relative kidney (%) | 0.96 ± 0.08 | 0.91 ± 0.07 | 0.89 ± 0.03 | 0.95 ± 0.07 | 0.95 ± 0.11 | 0.3636 |
| Relative heart (%) | 0.43 ± 0.04 | 0.40 ± 0.05 | 0.40 ± 0.04 | 0.43 ± 0.02 | 0.42 ± 0.03 | 0.0321 |
| Relative EFP (%) | 1.71 ± 0.16 a | 2.38 ± 0.56 b | 1.98 ± 0.19 a | 1.92 ± 0.25 a | 2.01 ± 0.28 a | 0.1194 |
3.7. Effect of Lr263 Supplementation on Biochemical Analyses at the End of the Experiment
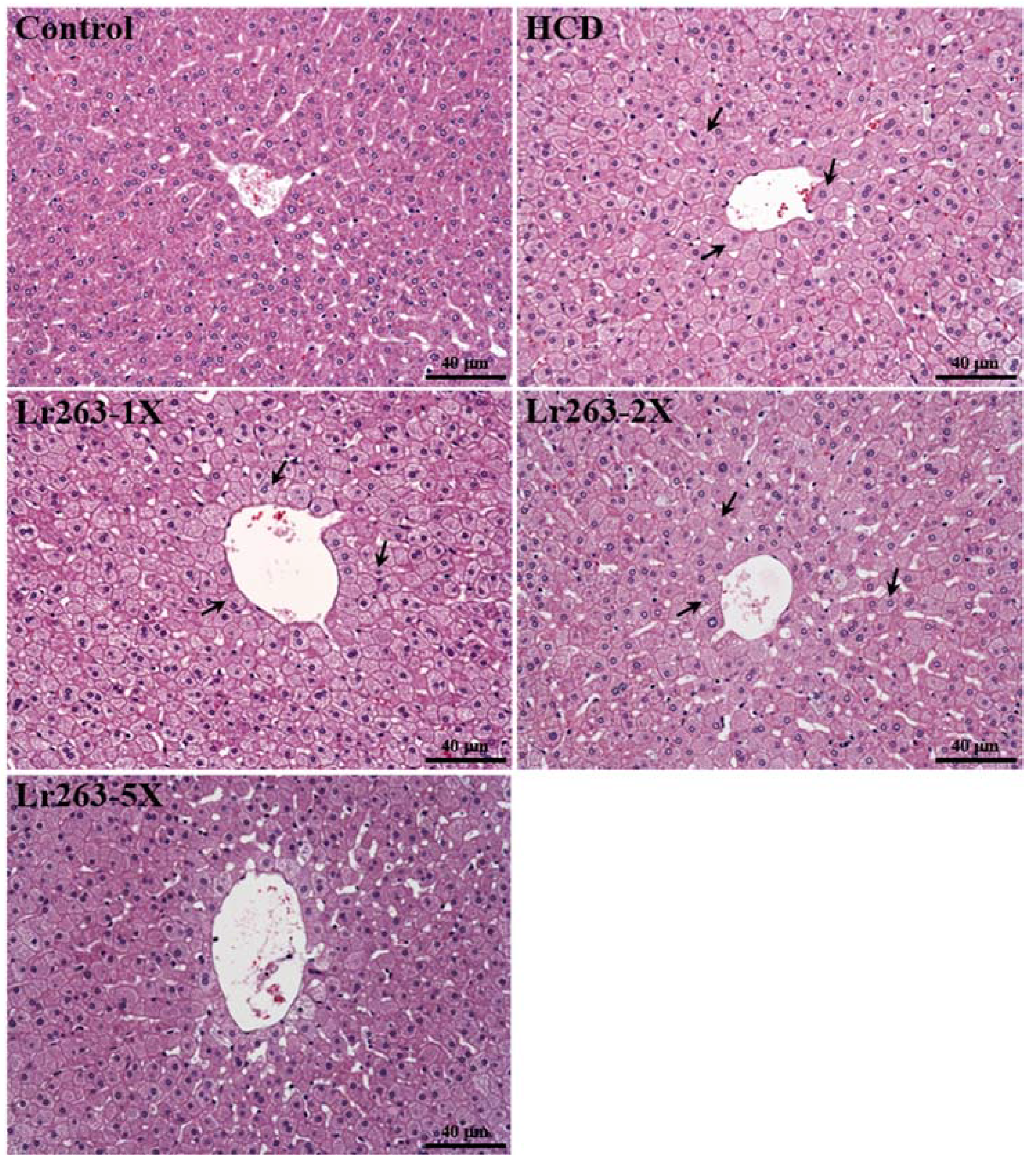
3.8. Effect of Lr263 Supplementation on Histology at the End of the Experiment
| Parameters | Control | HCD | Lr263-1X | Lr263-2X | Lr263-5X | Trend Analysis |
|---|---|---|---|---|---|---|
| AST (U/L) | 48 ± 10 | 46 ± 11 | 45 ± 8 | 43 ± 4 | 44 ± 4 | 0.8777 |
| ALT (U/L) | 72 ± 9 a | 101 ± 15 c | 90 ± 11 bc | 89 ± 12 b | 87 ± 5 b | 0.0143 |
| LDH (U/L) | 139 ± 25 | 142 ± 16 | 137 ± 16 | 138 ± 16 | 134 ± 13 | 0.3844 |
| Albumin (g/dL) | 3.6 ± 0.2 | 3.5 ± 0.2 | 3.6 ± 0.2 | 3.6 ± 0.2 | 3.6 ± 0.1 | 0.8501 |
| TP (g/dL) | 5.6 ± 0.3 | 5.5 ± 0.2 | 5.8 ± 0.2 | 5.7 ± 0.2 | 5.6 ± 0.2 | 0.8501 |
| BUN (mg/dL) | 17.9 ± 2.3 | 17.8 ± 1.8 | 17.9 ± 3.2 | 17.6 ± 0.8 | 17.7 ± 1.8 | 0.7215 |
| Creatinine (mg/dL) | 0.12 ± 0.03 | 0.12 ± 0.02 | 0.13 ± 0.06 | 0.12 ± 0.04 | 0.12 ± 0.02 | 0.6189 |
| Glucose (mg/dL) | 99 ± 12 a | 151 ± 25 b | 110 ± 17 a | 103 ± 21 a | 98 ± 14 a | <0.0001 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tsai, T.Y.; Chu, L.H.; Lee, C.L.; Pan, T.M. Atherosclerosis-preventing activity of lactic acid bacteria-fermented milk soymilk supplemented with Momordica charantia. J. Agric. Food Chem. 2009, 57, 2065–2071. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.L.; Martoni, C.J.; Parent, M.; Prakash, S. Cholesterol-lowering efficacy of a microencapsulated bile salt hydrolase-active Lactobacillus reuteri NCIMB 30242 yoghurt formulation in hypercholesterolaemic adults. Br. J. Nutr. 2012, 107, 1505–1513. [Google Scholar] [CrossRef] [PubMed]
- Ljungh, A.; Wadström, T. Lactobacillus Molecular Biology: From Genomics to Probiotics. Caister Academic Press: Norfolk, England, 2009. [Google Scholar]
- Chen, Y.M.; Shih, T.W.; Chiu, C.P.; Pan, T.M.; Tsai, T.Y. Effects of lactic acid bacteria-fermented soy milk on melanogenesis in B16F0 melanocytes. J. Funct. Foods 2013, 5, 395–405. [Google Scholar] [CrossRef]
- Noohi, N.; Ebrahimipour, G.; Rohani, M.; Talebi, M.; Pourshafie, M.R. Phenotypic characteristics and probiotic potentials of Lactobacillus spp. isolated from poultry. Jundishapur J. Microbiol. 2014, 7, e17824. [Google Scholar] [CrossRef] [PubMed]
- Talarico, T.L.; Casas, I.A.; Chung, T.C.; Dobrogosz, W.J. Production and isolation of reuterin, a growth inhibitor produced by Lactobacillus reuteri. Antimicrob Agents Chemother. 1988, 32, 1854–1858. [Google Scholar] [CrossRef] [PubMed]
- Ojetti, V.; Ianiro, G.; Tortora, A.; D’Angelo, G.; di Rienzo, T.A.; Bibbò, S.; Gasbarrini, A. The effect of Lactobacillus reuteri supplementation in adults with chronic functional constipation: A randomized, double-blind, placebo-controlled trial. J. Gastrointestin. Liver Dis. 2014, 23, 387–391. [Google Scholar] [PubMed]
- Holz, C.; Busjahn, A.; Mehling, H.; Arya, S.; Boettner, M.; Habibi, H.; Lang, C. Significant Reduction in Helicobacter pylori Load in Humans with Non-viable Lactobacillus reuteri DSM17648: A Pilot Study. Probiotics Antimicrob. Proteins 2014. [Google Scholar] [CrossRef]
- Sima, A.; Bulla, A.; Simionescu, N. Experimental obstructive coronary atherosclerosis in the hyperlipidemic hamster. J. Submicrosc. Cytol. Pathol. 1990, 22, 1–16. [Google Scholar] [PubMed]
- Sullivan, M.P.; Cerda, J.J.; Robbins, F.L.; Burgin, C.W. The gerbil, hamster, and guinea pig as rodent models for hyperlipidemia. Lab. Anim. Sci. 1993, 43, 575–578. [Google Scholar] [PubMed]
- Stancu, C.S.; Sanda, G.M.; Deleanu, M.; Sima, A.V. Probiotics determine hypolipidemic and antioxidant effects in hyperlipidemic hamsters. Mol. Nutr. Food. Res. 2014, 58, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.; Sun, J.; Xia, S.; Li, L.; Li, Y.; Wang, P.; Shi, Y.; Le, G. Effects of different Lactobacillus reuteri on inflammatory and fat storage in high-fat diet-induced obesity mice model. J. Funct. Foods 2015, 14, 424–434. [Google Scholar] [CrossRef]
- Singh, T.P.; Malik, R.K.; Katkamwar, S.G.; Kaur, G. Hypocholesterolemic effects of Lactobacillus reuteri LR6 in rats fed on high-cholesterol diet. Int. J. Food Sci. Nutr. 2015, 66, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.C.; Huang, W.C.; Chiu, C.C.; Chang, Y.K.; Huang, C.C. Whey protein improves exercise performance and biochemical profiles in trained mice. Med. Sci. Sports Exerc. 2014, 46, 1517–1524. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Leung, K.Y.; Cao, Y.; Huang, Y.; Ratnayake, W.M.; Chen, Z.Y. Alpha-linolenic acid but not conjugated linolenic acid is hypocholesterolaemic in hamsters. Br. J. Nutr. 2005, 93, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.M.; Wu, F.H. Comparison of animal models of hyperlipidemia. Zhong Xi Yi Jie He Xue Bao 2004, 2, 132–134. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wang, H.; Jiao, R.; Peng, C.; Wong, Y.M.; Yeung, V.S.; Huang, Y.; Chen, Z.Y. Choosing hamsters but not rats as a model for studying plasma cholesterol-lowering activity of functional foods. Mol. Nutr. Food Res. 2009, 53, 921–930. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Li, K.; Tang, S.; Xiao, Y. Study on animal models for hyperlipidemia. Wei Sheng Yan Jiu 2002, 31, 97–99. [Google Scholar] [PubMed]
- Lee, C.L.; Tsai, T.Y.; Wang, J.J.; Pan, T.M. In vivo hypolipidemic effects and safety of low dosage Monascus powder in a hamster model of hyperlipidemia. Appl. Microbiol. Biotechnol. 2006, 70, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.L.; Hung, H.K.; Wang, J.J.; Pan, T.M. Red mold dioscorea has greater hypolipidemic and antiatherosclerotic effect than traditional red mold rice and unfermented dioscorea in hamsters. J. Agric. Food Chem. 2007, 55, 7162–7169. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Fan, S.; Xie, X.; Xue, N.; Jin, X.; Wang, L. Novel PPAR pan agonist, ZBH ameliorates hyperlipidemia and insulin resistance in high fat diet induced hyperlipidemic hamster. PLoS ONE. 2014, 9, e96056. [Google Scholar] [CrossRef] [PubMed]
- Yonejima, Y.; Ushida, K.; Mori, Y. Effect of lactic acid bacteria on lipid metabolism and fat synthesis in mice fed a high-fat diet. Biosci. Microbiota Food Health 2013, 32, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.I.; Gibson, G.R. Effects of consumption of probiotics and prebiotics on serum lipid levels in humans. Crit. Rev. Biochem. Mol. Biol. 2002, 37, 259–281. [Google Scholar] [CrossRef] [PubMed]
- Taranto, M.P.; Medici, M.; Perdigon, G.; Ruiz Holgado, A.P.; Valdez, G.F. Effect of Lactobacillus reuteri on the prevention of hypercholesterolemia in mice. J. Dairy Sci. 2000, 83, 401–403. [Google Scholar] [CrossRef] [PubMed]
- Barona, J.; Fernandez, M.L. Dietary cholesterol affects plasma lipid levels, the intravascular processing of lipoproteins and reverse cholesterol transport without increasing the risk for heart disease. Nutrients 2012, 4, 1015–1025. [Google Scholar] [CrossRef] [PubMed]
- Taranto, M.P.; Fernandez Murga, M.L.; Lorca, G.; de Valdez, G.F. Bile salts and cholesterol induce changes in the lipid cell membrane of Lactobacillus reuteri. J. Appl. Microbiol. 2003, 95, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Begley, M.; Hill, C.; Gahan, C.G. Bile salt hydrolase activity in probiotics. Appl. Environ. Microbiol. 2006, 72, 1729–1738. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.C.; Lin, P.P.; Hsieh, Y.M.; Zhang, Z.Y.; Wu, H.C.; Huang, C.C. Cholesterol-lowering potentials of lactic acid bacteria based on bile-salt hydrolase activity and effect of potent strains on cholesterol metabolism in vitro and in vivo. Sci. World J. 2014. [Google Scholar] [CrossRef]
- Friedman, M.; Fitch, T.E.; Levin, C.E.; Yokoyama, W.H. Feeding tomatoes to hamsters reduces their plasma low-density lipoprotein cholesterol and triglycerides. J. Food Sci. 2000, 65, 897–900. [Google Scholar] [CrossRef]
- Mencarelli, A.; Cipriani, S.; Renga, B.; Bruno, A.; D’Amore, C.; Distrutti, E.; Fiorucci, S. VSL#3 resets insulin signaling and protects against NASH and atherosclerosis in a model of genetic dyslipidemia and intestinal inflammation. PLoS One 2012, 7, e45425. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Egusa, S.; Fukuda, M. Isoflavone and protein constituents of lactic acid-fermented soy milk combine to prevent dyslipidemia in rats fed a high cholesterol diet. Nutrients 2014, 6, 5704–5723. [Google Scholar] [CrossRef] [PubMed]
- Jo, S.Y.; Choi, E.A.; Lee, J.J.; Chang, H.C. Characterization of starter kimchi fermented with Leuconostoc kimchii GJ2 and its cholesterol-lowering effects in rats fed a high-fat and high-cholesterol diet. J. Sci. Food Agric. 2014. [Google Scholar] [CrossRef]
- Chawda, H.M.; Mandavia, D.R.; Parmar, P.H.; Baxi, S.N.; Tripathi, C.R. Hypolipidemic activity of a hydroalcoholic extract of Cyperus scariosus Linn. root in guinea pigs fed with a high cholesterol diet. Chin. J. Nat. Med. 2014, 12, 819–826. [Google Scholar]
- Gilat, T.; Leikin-Frenkel, A.; Goldiner, I.; Juhel, C.; Lafont, H.; Gobbi, D.; Konikoff, F.M. Prevention of diet-induced fatty liver in experimental animals by the oral administration of a fatty acid bile acid conjugate (FABAC). Hepatology 2003, 38, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Hathout, A.S.; Mohamed, S.R.; El-Nekeety, A.A.; Hassan, N.S.; Aly, S.E.; Abdel-Wahhab, M.A. Ability of Lactobacillus casei and Lactobacillus reuteri to protect against oxidative stress in rats fed aflatoxins-contaminated diet. Toxicon 2011, 58, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Forsberg, A.; Abrahamsson, T.R.; Björkstén, B.; Jenmalm, M.C. Pre-and post-natal Lactobacillus reuteri supplementation decreases allergen responsiveness in infancy. Clin. Exp. Allergy 2013, 43, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Martoni, C.; Bhathena, J.; Jones, M.L. Investigation of microencapsulated BSH active Lactobacillus in the simulated human GI tract. J. Biomed. Biotechnol. 2007, 2007, 1–9. [Google Scholar] [CrossRef]
- Ooi, L.G.; Ahmad, R.; Yuen, K.H. Lactobacillus acidophilus CHO-220 and inulin reduced plasma total cholesterol and low-density lipoprotein cholesterol via alteration of lipid transporters. J. Dairy. Sci. 2010, 93, 5048–5058. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.L.; Martoni, C.J.; Prakash, S. Cholesterol lowering and inhibition of sterol absorption by Lactobacillus reuteri NCIMB 30242: A randomized controlled trial. Eur. J. Clin. Nutr. 2012, 66, 1234–1241. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, W.-C.; Chen, Y.-M.; Kan, N.-W.; Ho, C.-S.; Wei, L.; Chan, C.-H.; Huang, H.-Y.; Huang, C.-C. Hypolipidemic Effects and Safety of Lactobacillus Reuteri 263 in a Hamster Model of Hyperlipidemia. Nutrients 2015, 7, 3767-3782. https://doi.org/10.3390/nu7053767
Huang W-C, Chen Y-M, Kan N-W, Ho C-S, Wei L, Chan C-H, Huang H-Y, Huang C-C. Hypolipidemic Effects and Safety of Lactobacillus Reuteri 263 in a Hamster Model of Hyperlipidemia. Nutrients. 2015; 7(5):3767-3782. https://doi.org/10.3390/nu7053767
Chicago/Turabian StyleHuang, Wen-Ching, Yi-Ming Chen, Nai-Wen Kan, Chun-Sheng Ho, Li Wei, Ching-Hung Chan, Hui-Yu Huang, and Chi-Chang Huang. 2015. "Hypolipidemic Effects and Safety of Lactobacillus Reuteri 263 in a Hamster Model of Hyperlipidemia" Nutrients 7, no. 5: 3767-3782. https://doi.org/10.3390/nu7053767
APA StyleHuang, W.-C., Chen, Y.-M., Kan, N.-W., Ho, C.-S., Wei, L., Chan, C.-H., Huang, H.-Y., & Huang, C.-C. (2015). Hypolipidemic Effects and Safety of Lactobacillus Reuteri 263 in a Hamster Model of Hyperlipidemia. Nutrients, 7(5), 3767-3782. https://doi.org/10.3390/nu7053767






