The Ratio of Dietary Branched-Chain Amino Acids is Associated with a Lower Prevalence of Obesity in Young Northern Chinese Adults: An Internet-Based Cross-Sectional Study
Abstract
:1. Introduction
2. Methods
2.1. Development of the IDQC
2.2. Reliability and Validity of the IDQC
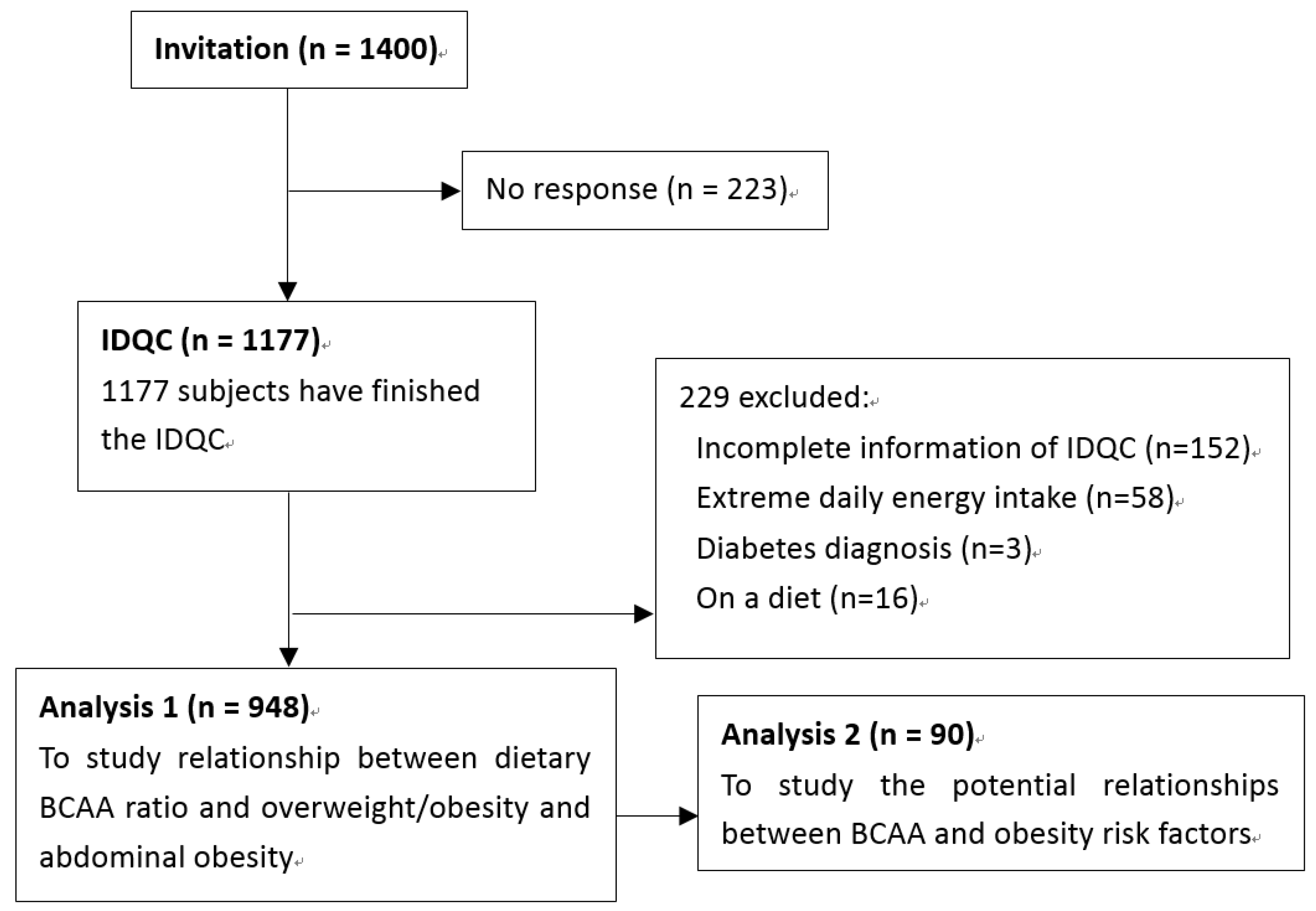
2.3. Participants and Exclusive Criteria
2.4. Dietary Nutrient Intake Calculation
2.5. Anthropometric Measurements
2.6. Definition of Overweight/Obesity and Abdominal Obesity
2.7. Laboratory Measurements
2.8. Statistical Analysis
3. Results
3.1. Characteristics and Dietary Intakes in the Study Population
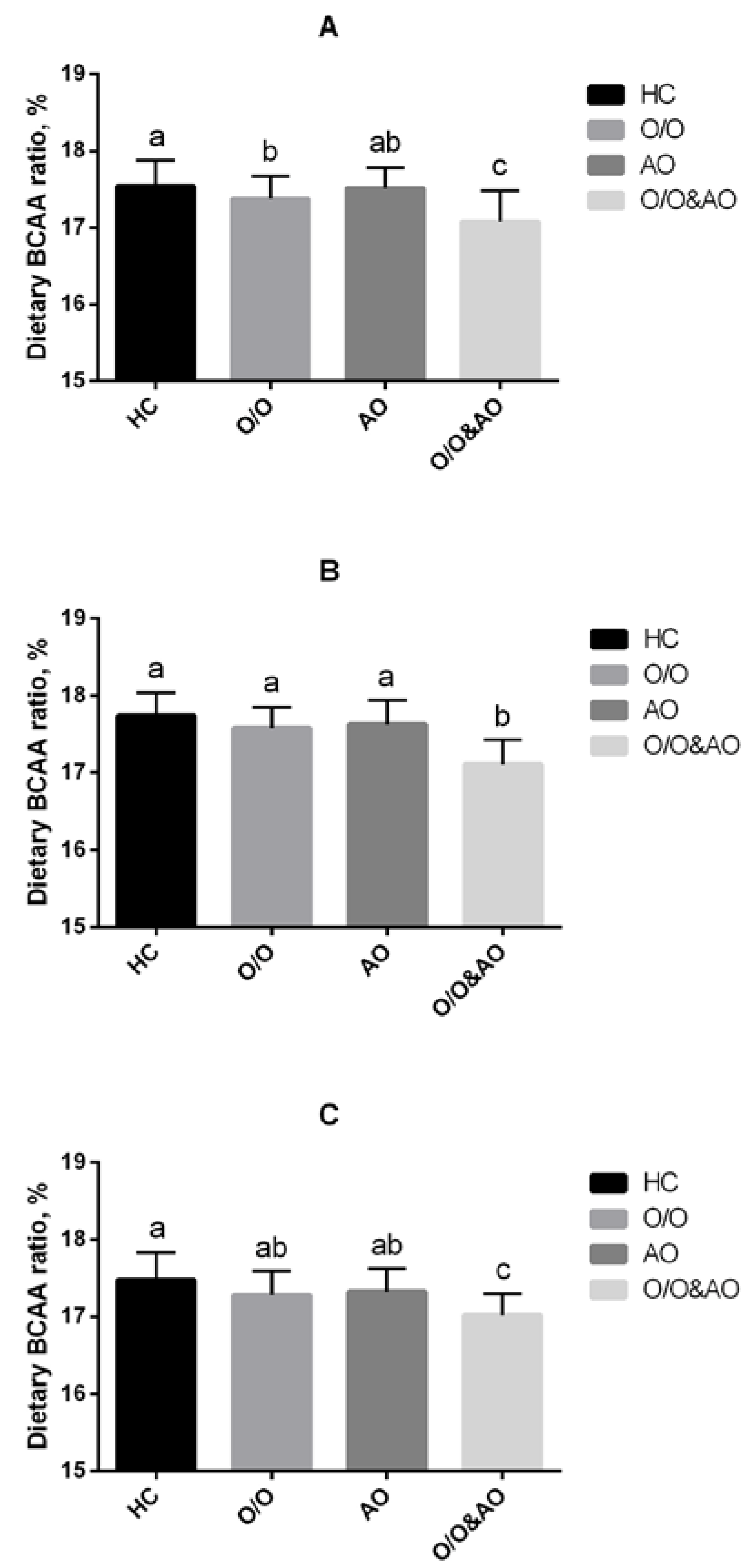
3.2. Dietary BCAA Ratios of Overweight/Obesity and Central Obese Participants Were Lower than Healthy Controls
| Characteristics | Quartiles of BCAAs (% Total Amino Acids Intake) | p | |||
|---|---|---|---|---|---|
| 1st (n = 237) | 2nd (n = 237) | 3rd (n = 237) | 4th (n = 237) | ||
| (≤17.07) | (>17.08 and ≤17.54) | (>17.54 and ≤17.94) | (>17.94) | ||
| Age, year | 22.5 ± 5.7 | 23.0 ± 6.3 | 22.2 ± 5.5 | 21.4 ± 4.1 | <0.01 |
| Gender | <0.001 | ||||
| Male, n (%) | 43 (18.1) | 70 (29.5) | 59 (24.9) | 87 (36.7) | |
| Female, n (%) | 194 (81.9) | 167 (70.5) | 178 (75.1) | 150 (63.3) | |
| Income per month | NS | ||||
| <2000 yuan, n (%) | 10 (4.2) | 8 (3.4) | 6 (2.5) | 6 (2.5) | |
| 2000–5000 yuan, n (%) | 220 (92.8) | 217 (91.6) | 221 (93.2) | 216 (91.1) | |
| ≥5000 yuan, n (%) | 7 (3.0) | 12 (5.1) | 10 (4.2) | 15 (6.3) | |
| Education | <0.05 | ||||
| Under college, n (%) | 7 (3.0) | 6 (2.5) | 12 (5.1) | 15 (6.3) | |
| Bachelor, n (%) | 210 (88.6) | 192 (81.0) | 199 (84.0) | 193 (81.4) | |
| Master or doctor, n (%) | 20 (8.4) | 39 (16.5) | 26 (11.0) | 29 (12.2) | |
| Labor | NS | ||||
| Light, n (%) | 23 (9.7) | 31 (13.1) | 26 (11.0) | 14 (5.9) | |
| Medium, n (%) | 214 (90.3) | 206 (86.9) | 211 (89.0) | 223 (94.1) | |
| Exercise | NS | ||||
| <10 h/week, n (%) | 103 (43.5) | 98 (41.4) | 100 (42.2) | 96 (40.5) | |
| 10–20 h/week, n (%) | 107 (45.1) | 111 (46.8) | 110 (46.4) | 103 (43.5) | |
| ≥20 h/week, n (%) | 27 (11.4) | 28 (11.8) | 27 (11.4) | 38 (16.0) | |
| Smoking and drinking * | |||||
| Current smoker, n (%) | 1 (0.4) | 11 (4.6) | 6 (2.5) | 7 (3.0) | <0.001 |
| Current drinker, n (%) | 15 (6.3) | 35 (14.8) | 31 (13.1) | 26 (11.0) | <0.05 |
| Weight, kg | 57.8 ± 10.2 | 59.2 ± 10.8 | 57.5 ± 9.3 | 57.5 ± 9.3 | NS |
| BMI, kg/m2 | 21.5 ± 2.7 | 21.5 ± 2.9 | 20.7 ± 2.4 | 20.5 ± 2.2 | <0.001 |
| <24 (normal), n (%) | 199 (84.0) | 199 (84.0) | 215 (90.7) | 219 (92.4) | <0.05 |
| Male, n (%) | 26 (11.0) | 55 (23.2) | 51 (21.5) | 80 (33.8) | <0.01 |
| Female, n (%) | 173 (73.0) | 144 (60.8) | 164 (69.2) | 139 (58.6) | <0.05 |
| ≥24 (overweight/obesity), n (%) | 38 (16.0) | 38 (16.0) | 22 (9.3) | 18 (7.6) | <0.05 |
| Male, n (%) | 17 (7.2) | 15 (6.3) | 8 (3.4) | 7 (3.0) | <0.05 |
| Female, n (%) | 21 (8.9) | 23 (9.7) | 14 (5.9) | 11 (4.6) | <0.05 |
| WC, cm | 73.6 ± 8.2 | 72.8 ± 8.4 | 72.1 ± 7.0 | 71.2 ± 7.3 | <0.01 |
| Abdominal obesity, n (%) | 25 (10.5) | 22 (9.3) | 13 (5.5) | 13 (5.5) | <0.05 |
| Male, n (%) | 14 (5.9) | 10 (4.2) | 9 (3.8) | 7 (3.0) | <0.05 |
| Female, n (%) | 11 (4.6) | 12 (5.1) | 4 (1.7) | 6 (2.5) | <0.05 |
| Overweight/obesity & abdominal obesity, n (%) | 18 (7.6) | 17 (7.2) | 7 (3.0) | 6 (2.5) | <0.05 |
| Male, n (%) | 11 (4.6) | 11 (4.6) | 5 (2.1) | 3 (1.3) | <0.05 |
| Female, n (%) | 7 (3.0) | 6 (2.5) | 2 (0.9) | 3 (1.3) | <0.05 |
| SBP, mmHg | 112.2 ± 12.5 | 114.2 ± 10.0 | 111.8 ± 12.0 | 113.6 ± 11.4 | NS |
| DBP, mmHg | 77.1 ± 10.3 | 78.4 ± 8.9 | 76.3 ± 10.3 | 77.4 ± 8.8 | NS |
| Dietary intakes # | |||||
| Energy, kcal/day | 2250.3 ± 54.8 | 2460.5 ± 52.5 | 2442.6 ± 57.2 | 2392.9 ± 61.1 | <0.01 |
| Total carbohydrate, g/day | 373.9 ± 9.5 | 376.1 ± 8.5 | 368.0 ± 9.3 | 353.6 ± 10.0 | NS |
| % energy | 64.1 ± 0.7 | 58.8 ± 0.6 | 57.7 ± 0.6 | 57.4 ± 0.7 | <0.001 |
| Total fat, g/day | 58.9 ± 2.4 | 75.3 ± 2.5 | 75.9 ± 2.3 | 76.0 ± 2.6 | <0.001 |
| % energy | 22.7 ± 0.6 | 26.9 ± 0.6 | 27.7 ± 0.5 | 28.3 ± 0.6 | <0.001 |
| Cholesterol, mg/day | 257.5 ± 15.2 | 405.3 ± 14.9 | 421.1 ± 15.5 | 433.5 ± 17.2 | <0.001 |
| Fiber, g/day | 18.5 ± 0.8 | 19.6 ± 0.6 | 17.5 ± 0.6 | 14.1 ± 0.6 | <0.001 |
| Total protein, g/day | 75.0 ± 2.1 | 88.0 ± 2.1 | 88.9 ± 2.4 | 85.3 ± 2.5 | <0.001 |
| % energy | 13.2 ± 0.1 | 14.3 ± 0.1 | 14.6 ± 0.1 | 14.3 ± 0.2 | <0.001 |
| Total amino acids, g/day | 48.6 ± 1.6 | 53.6 ± 1.5 | 52.8 ± 1.8 | 48.8 ± 1.9 | NS |
| BCAAs | 8.1 ± 0.3 | 9.3 ± 0.3 | 9.4 ± 0.3 | 8.9 ± 0.3 | <0.01 |
| Isoleucine | 2.1 ± 0.1 | 2.4 ± 0.1 | 2.4 ± 0.1 | 2.3 ± 0.1 | <0.05 |
| Leucine | 3.7 ± 0.1 | 4.2 ± 0.1 | 4.3 ± 0.1 | 4.1 ± 0.2 | <0.05 |
| Valine | 2.4 ± 0.1 | 2.7 ± 0.1 | 2.7 ± 0.1 | 2.5 ± 0.1 | <0.05 |
3.3. Dietary BCAA Ratio Was Inversely Associated with BMI and Waist Circumference in the Study Population
| Parameters | BMI | Waist | ||
|---|---|---|---|---|
| r | p | r | p | |
| Overall | ||||
| Isoleucine | −0.160 | <0.001 | −0.113 | <0.001 |
| Leucine | −0.172 | <0.001 | −0.177 | <0.001 |
| Valine | −0.109 | <0.01 | −0.133 | <0.01 |
| BCAAs | −0.197 | <0.001 | −0.187 | <0.001 |
| Male | ||||
| Isoleucine | −0.279 | <0.001 | −0.233 | <0.001 |
| Leucine | −0.224 | <0.001 | −0.209 | <0.001 |
| Valine | −0.187 | <0.05 | −0.115 | <0.05 |
| BCAAs | −0.278 | <0.001 | −0.256 | <0.001 |
| Female | ||||
| Isoleucine | −0.124 | <0.001 | −0.113 | <0.001 |
| Leucine | −0.182 | <0.001 | −0.257 | <0.001 |
| Valine | −0.110 | <0.05 | −0.102 | <0.05 |
| BCAAs | −0.196 | <0.001 | −0.254 | <0.001 |
3.4. Associations between BCAA Ratio and Prevalence of Overweight/Obesity, and Abdominal Obesity
| BCAA Quartiles | Quartile 1 | Quartile 2 | Quartile 3 | Quartile 4 |
|---|---|---|---|---|
| Overweight/obesity | ||||
| All | ||||
| Crude | 1 | 1.000 (0.612, 1.633) | 0.536 (0.306, 0.938) * | 0.430 (0.238, 0.779) ** |
| Model 1 | 1 | 0.978 (0.564, 1.697) | 0.501 (0.268, 0.937) * | 0.403 (0.208, 0.780) ** |
| Model 2 | 1 | 0.958 (0.543, 1.688) | 0.509 (0.265, 0.976) * | 0.392 (0.195, 0.789) ** |
| Model 3 | 1 | 0.963 (0.546, 1.700) | 0.508 (0.265, 0.972) * | 0.389 (0.193, 0.783) ** |
| Male | ||||
| Crude | 1 | 0.468 (0.218, 1.006) | 0.185 (0.073, 0.469) ** | 0.161 (0.061, 0.430) ** |
| Model 1 | 1 | 0.542 (0.229, 1.280) | 0.255 (0.096, 0.678) ** | 0.186 (0.063, 0.551) ** |
| Model 2 | 1 | 0.594 (0.242, 1.454) | 0.303 (0.107, 0.853) * | 0.206 (0.064, 0.659) ** |
| Model 3 | 1 | 0.525 (0.210, 1.312) | 0.264 (0.093, 0.752) * | 0.170 (0.053, 0.550) ** |
| Female | ||||
| Crude | 1 | 0.883 (0.449, 1.733) | 0.726 (0.359, 1.470) | 0.469 (0.213, 1.034) |
| Model 1 | 1 | 0.696 (0.344, 1.453) | 0.635 (0.298, 1.356) | 0.404 (0.174, 0.941) * |
| Model 2 | 1 | 0.644 (0.306, 1.354) | 0.529 (0.242, 1.157) | 0.310 (0.125, 0.733) * |
| Model 3 | 1 | 0.628 (0.298, 1.325) | 0.533 (0.243, 1.165) | 0.316 (0.127, 0.789) * |
| Abdominal obesity | ||||
| All | ||||
| Crude | 1 | 0.872 (0.477, 1.594) | 0.496 (0.247, 0.996) * | 0.494 (0.246, 0.991) * |
| Model 1 | 1 | 0.704 (0.350, 1.414) | 0.343 (0.147, 0.801) * | 0.368 (0.166, 0.818) * |
| Model 2 | 1 | 0.710 (0.348, 1.450) | 0.349 (0.147, 0.832) * | 0.379 (0.165, 0.868) * |
| Model 3 | 1 | 0.671 (0.325,1.385) | 0.351 (0.145, 0.845) * | 0.376 (0.161, 0.876) * |
| Male | ||||
| Crude | 1 | 0.464 (0.205, 1.048) | 0.417 (0.181, 0.959) * | 0.101 (0.028, 0.359) ** |
| Model 1 | 1 | 0.441 (0.169, 1.150) | 0.426 (0.164, 1.106) | 0.084 (0.018, 0.397) ** |
| Model 2 | 1 | 0.433 (0.164, 1.144) | 0.417 (0.153, 1.136) | 0.079 (0.016, 0.380) ** |
| Model 3 | 1 | 0.351 (0.127, 0.969) * | 0.383 (0.138, 1.067) | 0.066 (0.013, 0.333) ** |
| Female | ||||
| Crude | 1 | 1.455 (0.541, 3.916) | 0.565 (0.162, 1.965) | 0.710 (0.221, 2.282) |
| Model 1 | 1 | 1.256 (0.443, 3.556) | 0.538 (0.150, 1.930) | 0.685 (0.204, 2.302) |
| Model 2 | 1 | 1.331 (0.456, 3.883) | 0.582 (0.155, 2.182) | 0.730 (0.202, 2.643) |
| Model 3 | 1 | 1.299 (0.445, 3.797) | 0.578 (0.154, 2.171) | 0.748 (0.206, 2.713) |
3.5. Dietary BCAA Ratio was Negatively Associated with 2 h-PG in Overall and Male Young Adults
| Parameters | FBG | 2 h-PG | FBI | HOMA-IR | TG | TC | HDL | LDL | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| r | p | r | p | r | p | r | p | r | p | r | p | r | p | r | p | |
| Overall | ||||||||||||||||
| Isoleucine | −0.089 | 0.216 | −0.102 | 0.109 | −0.096 | 0.671 | −0.131 | 0.213 | −0.082 | 0.356 | −0.136 | 0.712 | 0.146 | 0.181 | −0.079 | 0.471 |
| Leucine | −0.128 | 0.125 | −0.157 | 0.129 | −0.135 | 0.289 | −0.194 | 0.289 | −0.121 | 0.331 | −0.077 | 0.893 | 0.067 | 0.540 | −0.193 | 0.077 |
| Valine | −0.119 | 0.365 | −0.107 | 0.168 | −0.097 | 0.531 | −0.102 | 0.547 | −0.078 | 0.677 | −0.010 | 0.972 | 0.040 | 0.715 | 0.010 | 0.926 |
| BCAA | −0.205 | 0.066 | −0.316 | 0.037 | −0.182 | 0.139 | −0.221 | 0.095 | −0.098 | 0.543 | 0.061 | 0.698 | 0.026 | 0.810 | 0.031 | 0.781 |
| Male | ||||||||||||||||
| Isoleucine | −0.017 | 0.618 | −0.132 | 0.124 | −0.101 | 0.457 | −0.221 | 0.210 | −0.079 | 0.652 | −0.128 | 0.589 | 0.279 | 0.081 | 0.100 | 0.539 |
| Leucine | −0.375 | 0.017 | −0.202 | 0.055 | −0.199 | 0.382 | −0.139 | 0.472 | −0.211 | 0.219 | −0.022 | 0.940 | −0.099 | 0.544 | −0.122 | 0.400 |
| Valine | −0.132 | 0.203 | −0.112 | 0.106 | −0.163 | 0.291 | −0.178 | 0.313 | −0.035 | 0.864 | −0.058 | 0.921 | 0.243 | 0.130 | −0.012 | 0.944 |
| BCAA | −0.252 | 0.051 | −0.373 | 0.001 | −0.178 | 0.117 | −0.213 | 0.117 | −0.119 | 0.421 | −0.079 | 0.689 | 0.161 | 0.322 | −0.067 | 0.682 |
| Female | ||||||||||||||||
| Isoleucine | −0.277 | 0.083 | −0.068 | 0.279 | −0.033 | 0.981 | −0.067 | 0.668 | −0.098 | 0.443 | −0.071 | 0.783 | 0.101 | 0.535 | −0.150 | 0.357 |
| Leucine | −0.118 | 0.468 | −0.129 | 0.158 | −0.181 | 0.421 | −0.126 | 0.479 | −0.112 | 0.312 | −0.099 | 0.769 | −0.002 | 0.990 | 0.158 | 0.332 |
| Valine | −0.258 | 0.108 | −0.107 | 0.235 | −0.102 | 0.765 | −0.132 | 0.348 | −0.084 | 0.547 | −0.035 | 0.891 | −0.072 | 0.660 | 0.197 | 0.222 |
| BCAA | −0.246 | 0.073 | −0.211 | 0.045 | −0.168 | 0.235 | −0.186 | 0.138 | −0.093 | 0.455 | −0.067 | 0.621 | −0.046 | 0.780 | −0.061 | 0.707 |
3.6. Dietary BCAA Ratio Was Positively Associated with Serum LEP and ADPN in Overall and Male Adults
| Parameters | LEP | ADPN | TNF-α | IL-6 | CRP | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| r | p | r | p | r | p | r | p | r | p | |
| Overall | ||||||||||
| Isoleucine | 0.113 | 0.331 | 0.108 | 0.365 | −0.123 | 0.222 | −0.117 | 0.421 | −0.137 | 0.192 |
| Leucine | 0.192 | 0.109 | 0.212 | 0.099 | −0.162 | 0.169 | −0.124 | 0.265 | −0.256 | 0.043 |
| Valine | 0.098 | 0.421 | 0.088 | 0.643 | −0.102 | 0.289 | −0.101 | 0.348 | −0.127 | 0.231 |
| BCAA | 0.211 | 0.045 | 0.231 | 0.042 | −0.191 | 0.055 | −0.115 | 0.281 | −0.229 | 0.035 |
| Male | ||||||||||
| Isoleucine | 0.147 | 0.264 | 0.131 | 0.273 | −0.131 | 0.231 | −0.122 | 0.487 | −0.161 | 0.181 |
| Leucine | 0.201 | 0.052 | 0.233 | 0.047 | −0.139 | 0.372 | −0.098 | 0.544 | −0.281 | 0.023 |
| Valine | 0.111 | 0.389 | 0.079 | 0.586 | −0.104 | 0.313 | −0.137 | 0.286 | −0.116 | 0.312 |
| BCAA | 0.235 | 0.03 | 0.236 | 0.021 | −0.193 | 0.107 | −0.117 | 0.331 | −0.275 | 0.031 |
| Female | ||||||||||
| Isoleucine | 0.102 | 0.412 | 0.117 | 0.452 | −0.087 | 0.628 | −0.111 | 0.367 | −0.145 | 0.211 |
| Leucine | 0.189 | 0.131 | 0.178 | 0.142 | −0.116 | 0.437 | −0.145 | 0.287 | −0.236 | 0.030 |
| Valine | 0.102 | 0.423 | 0.099 | 0.398 | −0.141 | 0.333 | −0.094 | 0.621 | −0.133 | 0.233 |
| BCAA | 0.198 | 0.071 | 0.218 | 0.044 | −0.171 | 0.102 | −0.136 | 0.221 | −0.219 | 0.040 |
3.7. Dietary BCAA Ratio Was Positively Associated with Serum LEP and ADPN in Overall and Male Adults
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Blomstrand, E.; Eliasson, J.; Karlsson, H.K.; Kohnke, R. Branched-chain amino acids activate key enzymes in protein synthesis after physical exercise. J. Nutr. 2006, 136, 269S–273S. [Google Scholar] [PubMed]
- Stipanuk, M.H. Leucine and protein synthesis: Mtor and beyond. Nutr. Rev. 2007, 65, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Mourier, A.; Bigard, A.X.; de Kerviler, E.; Roger, B.; Legrand, H.; Guezennec, C.Y. Combined effects of caloric restriction and branched-chain amino acid supplementation on body composition and exercise performance in elite wrestlers. Int. J. Sports Med. 1997, 18, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Layman, D.K. The role of leucine in weight loss diets and glucose homeostasis. J. Nutr. 2003, 133, 261S–267S. [Google Scholar] [PubMed]
- Layman, D.K.; Shiue, H.; Sather, C.; Erickson, D.J.; Baum, J. Increased dietary protein modifies glucose and insulin homeostasis in adult women during weight loss. J. Nutr. 2003, 133, 405–410. [Google Scholar] [PubMed]
- Ikehara, O.; Kawasaki, N.; Maezono, K.; Komatsu, M.; Konishi, A. Acute and chronic treatment of l-isoleucine ameliorates glucose metabolism in glucose-intolerant and diabetic mice. Biol. Pharm. Bull. 2008, 31, 469–472. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Kobayashi, H.; Mawatari, K.; Sato, J.; Bajotto, G.; Kitaura, Y.; Shimomura, Y. Effects of branched-chain amino acid supplementation on plasma concentrations of free amino acids, insulin, and energy substrates in young men. J. Nutr. Sci. Vitaminol. 2011, 57, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Donato, J., Jr.; Pedrosa, R.G.; Cruzat, V.F.; Pires, I.S.; Tirapegui, J. Effects of leucine supplementation on the body composition and protein status of rats submitted to food restriction. Nutrition 2006, 22, 520–527. [Google Scholar] [CrossRef] [PubMed]
- Mourier, A.; Gautier, J.F.; de Kerviler, E.; Bigard, A.X.; Villette, J.M.; Garnier, J.P.; Duvallet, A.; Guezennec, C.Y.; Cathelineau, G. Mobilization of visceral adipose tissue related to the improvement in insulin sensitivity in response to physical training in NIDDM. Effects of branched-chain amino acid supplements. Diabetes Care 1997, 20, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.Q.; Xun, P.; Bujnowski, D.; Daviglus, M.L.; van Horn, L.; Stamler, J.; He, K.; Group, I.C.R. Higher branched-chain amino acid intake is associated with a lower prevalence of being overweight or obese in middle-aged East Asian and Western adults. J. Nutr. 2011, 141, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.F. Effect of body mass index on all-cause mortality and incidence of cardiovascular diseases—Report for meta-analysis of prospective studies open optimal cut-off points of body mass index in Chinese adults. Biomed. Environ. Sci. 2002, 15, 245–252. [Google Scholar] [PubMed]
- Logue, J. Obesity is an independent risk factor for death from CHD. Practitioner 2011, 255, 5. [Google Scholar] [PubMed]
- Lorenzo, C.; Okoloise, M.; Williams, K.; Stern, M.P.; Haffner, S.M. The metabolic syndrome as predictor of type 2 diabetes: The San Antonio Heart Study. Diabetes Care 2003, 26, 3153–3159. [Google Scholar] [CrossRef] [PubMed]
- De Simone, G.; Devereux, R.B.; Chinali, M.; Roman, M.J.; Best, L.G.; Welty, T.K.; Lee, E.T.; Howard, B.V.; the Strong Heart Study Investigators. Risk factors for arterial hypertension in adults with initial optimal blood pressure: The strong heart study. Hypertension 2006, 47, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Howard, B.V.; Ruotolo, G.; Robbins, D.C. Obesity and dyslipidemia. Endocrinol. Metab. Clin. North Am. 2003, 32, 855–867. [Google Scholar] [CrossRef]
- De Pergola, G.; Silvestris, F. Obesity as a major risk factor for cancer. J. Obes. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Xi, B.; Liang, Y.; He, T.; Reilly, K.H.; Hu, Y.; Wang, Q.; Yan, Y.; Mi, J. Secular trends in the prevalence of general and abdominal obesity among Chinese adults, 1993–2009. Obes. Rev. 2012, 13, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Gardner, C.D.; Kiazand, A.; Alhassan, S.; Kim, S.; Stafford, R.S.; Balise, R.R.; Kraemer, H.C.; King, A.C. Comparison of the atkins, zone, ornish, and learn diets for change in weight and related risk factors among overweight premenopausal women: The A to Z weight loss study: A randomized trial. JAMA 2007, 297, 969–977. [Google Scholar] [CrossRef] [PubMed]
- Li, G.Q.; Li, Y.C.; Feng, R.N.; Jiang, Y.S. Reliabiliry and validity of internet-based food frequency questionnaire. J. Harbin Med. Univ. 2014, 5, 376–380. [Google Scholar]
- Yang, Y.; Wang, G.; Guo, X. China Food Composition Tables; Peking University Medical Press: Beijing, China, 2009. [Google Scholar]
- Hu, J.; Wallace, D.C.; Jones, E.; Liu, H. Cardiometabolic health of Chinese older adults with diabetes living in Beijing, China. Public. Health. Nurs. 2009, 26, 500–511. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Belobrajdic, D.P.; McIntosh, G.H.; Owens, J.A. A high-whey-protein diet reduces body weight gain and alters insulin sensitivity relative to red meat in wistar rats. J. Nutr. 2004, 134, 1454–1458. [Google Scholar] [PubMed]
- Zhang, Y.; Guo, K.; LeBlanc, R.E.; Loh, D.; Schwartz, G.J.; Yu, Y.H. Increasing dietary leucine intake reduces diet-induced obesity and improves glucose and cholesterol metabolism in mice via multimechanisms. Diabetes 2007, 56, 1647–1654. [Google Scholar] [CrossRef] [PubMed]
- Larosa, J.C.; Fry, A.G.; Muesing, R.; Rosing, D.R. Effects of high-protein, low-carbohydrate dieting on plasma lipoproteins and body weight. J. Am. Diet. Assoc. 1980, 77, 264–270. [Google Scholar] [PubMed]
- Sargrad, K.R.; Homko, C.; Mozzoli, M.; Boden, G. Effect of high protein vs. high carbohydrate intake on insulin sensitivity, body weight, hemoglobin A1c, and blood pressure in patients with type 2 diabetes mellitus. J. Am. Diet. Assoc. 2005, 105, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Weigle, D.S.; Breen, P.A.; Matthys, C.C.; Callahan, H.S.; Meeuws, K.E.; Burden, V.R.; Purnell, J.Q. A high-protein diet induces sustained reductions in appetite, ad libitum caloric intake, and body weight despite compensatory changes in diurnal plasma leptin and ghrelin concentrations. Am. J. Clin. Nutr. 2005, 82, 41–48. [Google Scholar] [PubMed]
- Anthony, J.C.; Anthony, T.G.; Kimball, S.R.; Jefferson, L.S. Signaling pathways involved in translational control of protein synthesis in skeletal muscle by leucine. J. Nutr. 2001, 131, 856S–860S. [Google Scholar] [PubMed]
- Kimball, S.R.; Jefferson, L.S. Signaling pathways and molecular mechanisms through which branched-chain amino acids mediate translational control of protein synthesis. J. Nutr. 2006, 136, 227S–231S. [Google Scholar] [PubMed]
- Cota, D.; Proulx, K.; Smith, K.A.; Kozma, S.C.; Thomas, G.; Woods, S.C.; Seeley, R.J. Hypothalamic mtor signaling regulates food intake. Science 2006, 312, 927–930. [Google Scholar] [CrossRef] [PubMed]
- Magni, P.; Motta, M.; Martini, L. Leptin: A possible link between food intake, energy expenditure, and reproductive function. Regul. Pept. 2000, 92, 51–56. [Google Scholar] [CrossRef]
- Lynch, C.J.; Gern, B.; Lloyd, C.; Hutson, S.M.; Eicher, R.; Vary, T.C. Leucine in food mediates some of the postprandial rise in plasma leptin concentrations. Am. J. Physiol. Endocrinol. Metab. 2006, 291, E621–E630. [Google Scholar] [CrossRef] [PubMed]
- Leclercq-Meyer, V.; Marchand, J.; Woussen-Colle, M.C.; Giroix, M.H.; Malaisse, W.J. Multiple effects of leucine on glucagon, insulin, and somatostatin secretion from the perfused rat pancreas. Endocrinology 1985, 116, 1168–1174. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, A.E.; Carroll, P.B.; Atwater, I.J. Effects of leucine on insulin secretion and beta cell membrane potential in mouse islets of langerhans. Pancreas 1991, 6, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Najafi, H.; Daikhin, Y.; Nissim, I.B.; Collins, H.W.; Yudkoff, M.; Matschinsky, F.M.; Stanley, C.A. Regulation of leucine-stimulated insulin secretion and glutamine metabolism in isolated rat islets. J. Biol. Chem. 2003, 278, 2853–2858. [Google Scholar] [CrossRef] [PubMed]
- Doi, M.; Yamaoka, I.; Nakayama, M.; Mochizuki, S.; Sugahara, K.; Yoshizawa, F. Isoleucine, a blood glucose-lowering amino acid, increases glucose uptake in rat skeletal muscle in the absence of increases in AMP-activated protein kinase activity. J. Nutr. 2005, 135, 2103–2108. [Google Scholar] [PubMed]
- Doi, M.; Yamaoka, I.; Nakayama, M.; Sugahara, K.; Yoshizawa, F. Hypoglycemic effect of isoleucine involves increased muscle glucose uptake and whole body glucose oxidation and decreased hepatic gluconeogenesis. Am. J. Physiol. Endocrinol. Metab. 2007, 292, E1683–E1693. [Google Scholar] [CrossRef] [PubMed]
- Nagata, C.; Nakamura, K.; Wada, K.; Tsuji, M.; Tamai, Y.; Kawachi, T. Branched-chain amino acid intake and the risk of diabetes in a Japanese community: The Takayama study. Am. J. Epidemiol. 2013, 178, 1226–1232. [Google Scholar] [CrossRef] [PubMed]
- Ohno, T.; Tanaka, Y.; Sugauchi, F.; Orito, E.; Hasegawa, I.; Nukaya, H.; Kato, A.; Matunaga, S.; Endo, M.; Tanaka, Y.; et al. Suppressive effect of oral administration of branched-chain amino acid granules on oxidative stress and inflammation in HCV-positive patients with liver cirrhosis. Hepatol. Res. 2008, 38, 683–688. [Google Scholar] [CrossRef] [PubMed]
- Torres-Leal, F.L.; Fonseca-Alaniz, M.H.; Teodoro, G.F.; de Capitani, M.D.; Vianna, D.; Pantaleao, L.C.; Matos-Neto, E.M.; Rogero, M.M.; Donato, J., Jr.; Tirapegui, J. Leucine supplementation improves adiponectin and total cholesterol concentrations despite the lack of changes in adiposity or glucose homeostasis in rats previously exposed to a high-fat diet. Nutr. Metab. (Lond.) 2011, 8, 62. [Google Scholar] [CrossRef] [PubMed]
- Zemel, M.B.; Bruckbauer, A. Effects of a leucine and pyridoxine-containing nutraceutical on fat oxidation, and oxidative and inflammatory stress in overweight and obese subjects. Nutrients 2012, 4, 529–541. [Google Scholar] [CrossRef] [PubMed]
- Hsu, M.C.; Chien, K.Y.; Hsu, C.C.; Chung, C.J.; Chan, K.H.; Su, B. Effects of BCAA, arginine and carbohydrate combined drink on post-exercise biochemical response and psychological condition. Chin. J. Physiol. 2011, 54, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Herbst, K.L.; Bhasin, S. Testosterone action on skeletal muscle. Curr. Opin. Clin. Nutr. Metab. Care 2004, 7, 271–277. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.-C.; Li, Y.; Liu, L.-Y.; Chen, Y.; Zi, T.-Q.; Du, S.-S.; Jiang, Y.-S.; Feng, R.-N.; Sun, C.-H. The Ratio of Dietary Branched-Chain Amino Acids is Associated with a Lower Prevalence of Obesity in Young Northern Chinese Adults: An Internet-Based Cross-Sectional Study. Nutrients 2015, 7, 9573-9589. https://doi.org/10.3390/nu7115486
Li Y-C, Li Y, Liu L-Y, Chen Y, Zi T-Q, Du S-S, Jiang Y-S, Feng R-N, Sun C-H. The Ratio of Dietary Branched-Chain Amino Acids is Associated with a Lower Prevalence of Obesity in Young Northern Chinese Adults: An Internet-Based Cross-Sectional Study. Nutrients. 2015; 7(11):9573-9589. https://doi.org/10.3390/nu7115486
Chicago/Turabian StyleLi, Yan-Chuan, Ying Li, Li-Yan Liu, Yang Chen, Tian-Qi Zi, Shan-Shan Du, Yong-Shuai Jiang, Ren-Nan Feng, and Chang-Hao Sun. 2015. "The Ratio of Dietary Branched-Chain Amino Acids is Associated with a Lower Prevalence of Obesity in Young Northern Chinese Adults: An Internet-Based Cross-Sectional Study" Nutrients 7, no. 11: 9573-9589. https://doi.org/10.3390/nu7115486
APA StyleLi, Y.-C., Li, Y., Liu, L.-Y., Chen, Y., Zi, T.-Q., Du, S.-S., Jiang, Y.-S., Feng, R.-N., & Sun, C.-H. (2015). The Ratio of Dietary Branched-Chain Amino Acids is Associated with a Lower Prevalence of Obesity in Young Northern Chinese Adults: An Internet-Based Cross-Sectional Study. Nutrients, 7(11), 9573-9589. https://doi.org/10.3390/nu7115486




