Pomegranate Peel Extract Prevents Bone Loss in a Preclinical Model of Osteoporosis and Stimulates Osteoblastic Differentiation in Vitro
Abstract
:1. Introduction
2. Materials and Methods
2.1. Pomegranate Peel Extract
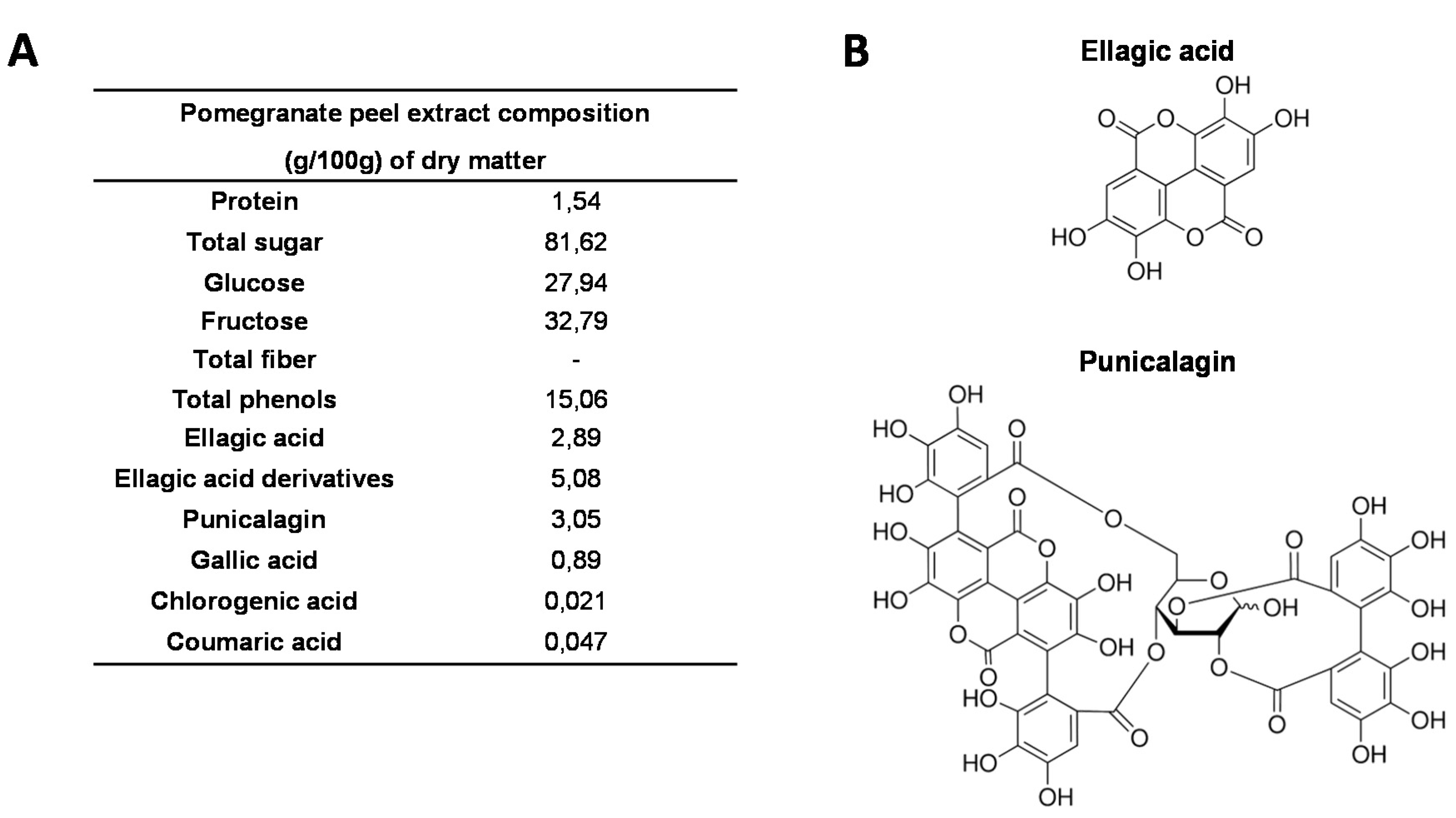
2.2. Animals Ethics
2.3. In Vitro Study Design: Serum Production
2.3.1. Cell Lines and Culture Conditions
2.3.2. Cell Proliferation
2.3.3. Alkaline Phosphatase (ALP) Activity Measurement
2.3.4. Tartrate-Resistant Acid Phosphatase (TRAP) Activity Measurement
2.3.5. Mineralization
2.4. In Vivo Study Design
2.4.1. Bone Mineral Density (BMD) Analysis
2.4.2. Bone Micro-Architecture Analysis
2.5. Taqman Low Density Arrays (TLDA)
2.6. Statistical Analysis
3. Results
3.1. nPGPE Promoted Osteoblast Differentiation
3.1.1. Cell Viability
3.1.2. Effect of PGPE on ALP Activity and Mineralization of MC3T3-E1 Cells
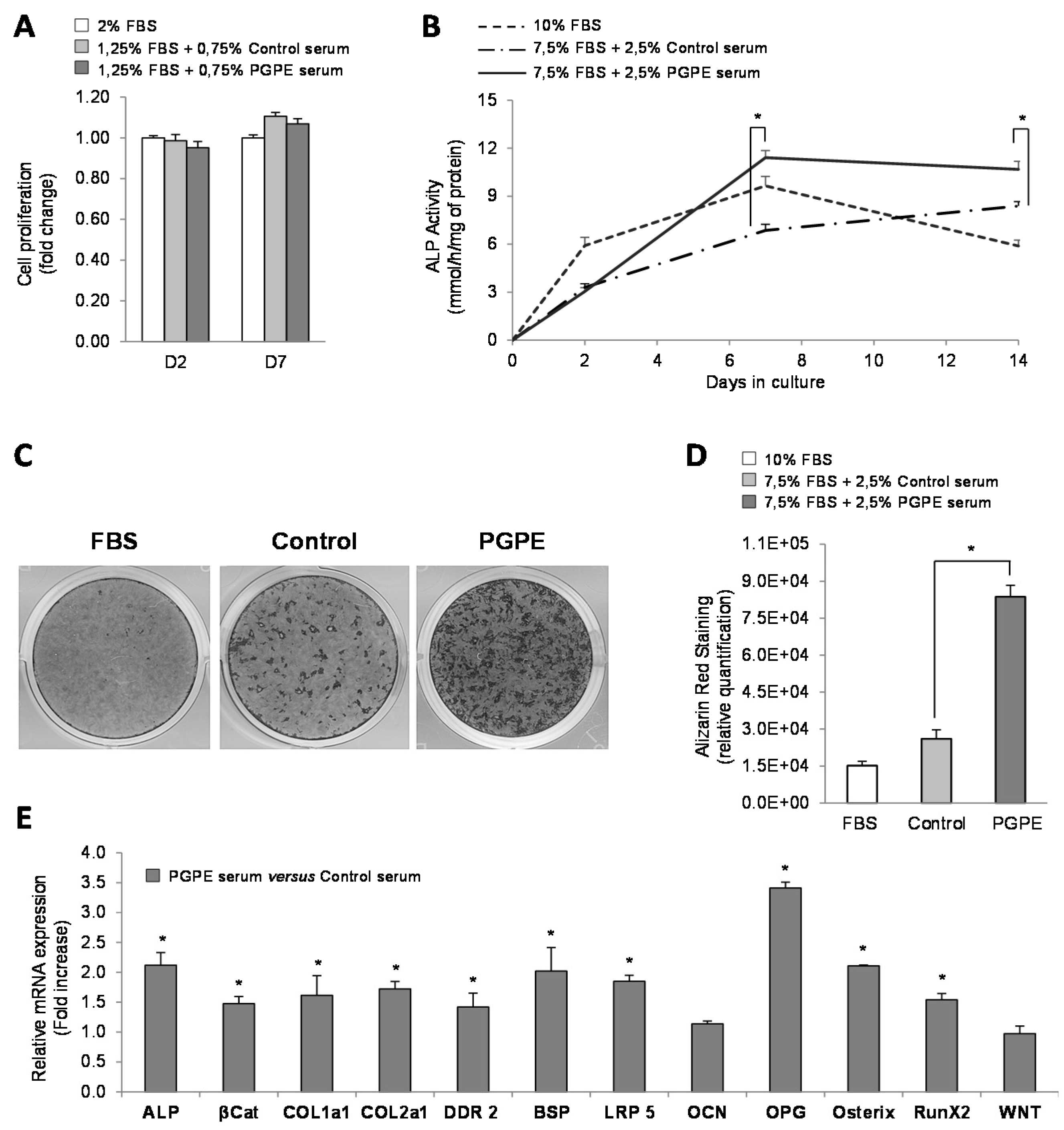
3.1.3. Effect of PGPE on transcriptional activity of MC3T3-E1 cells
3.2. PGPE Did Not Affect Osteoclast Activity but Impaired Transcription
3.2.1. Cell Viability
3.2.2. Effect of PGPE on TRAP Activity of RAW264.7 Cells
3.2.3. Effect of PGPE on Transcriptional Activity of RAW264.7 Cells
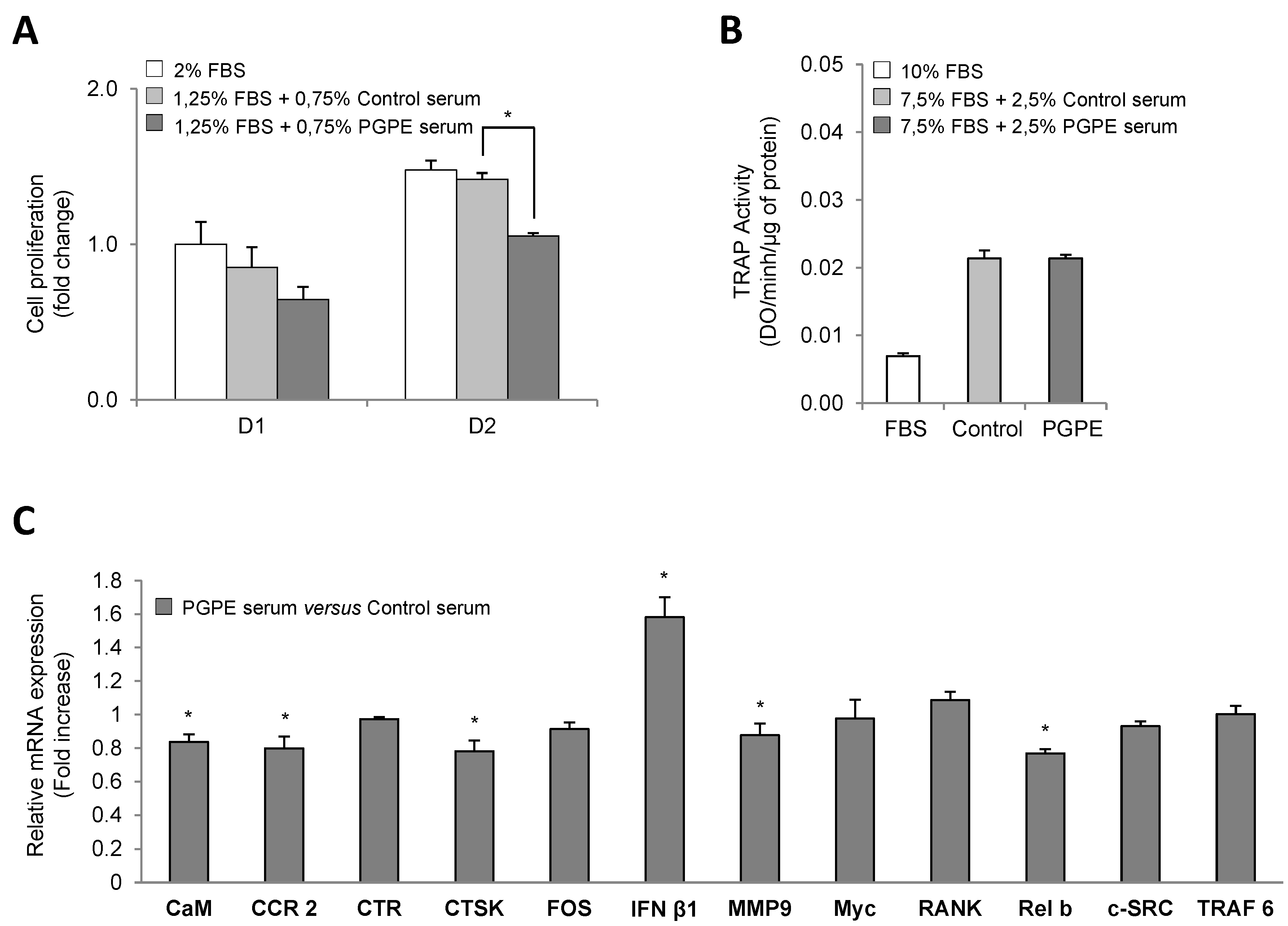
3.3. Confirmation of Cell Culture Results Using a Physiological in Vivo Approach
3.3.1. Validation of the Animal Model
| Sham | Ovariectomized | ||
|---|---|---|---|
| Normal Diet | Normal Diet | PGPE Diet | |
| (SH) | (OVX) | (OVX+PGPE) | |
| Initial body weight (g) | 18.9 ± 0.6 | 19.4 ± 0.6 | 19.0 ± 0.5 |
| Initial lean mass (% body weight) | 87.6 ± 3.8 | 87.1 ± 1.2 | 87.1 ± 0.9 |
| Initial fat mass (% body weight) | 6.6 ± 2.1 | 7.9 ± 1.3 | 8.0 ± 1.2 |
| Final body weight (g) | 19.3 ± 0.8 | 20.3 ± 1.2 | 19.9 ± 0.9 |
| Final lean mass (% body weight) | 88.9 ± 7.3 | 92.3 ± 3.0 | 91.6 ± 2.7 |
| Final fat mass (% body weight) | 9.8 ± 2.2 | 9.3 ± 1.6 | 9.0 ± 2.0 |
| Uterine weight (mg) | 84.4 ± 3.2 | 23.1 ± 2.8 # | 23.7 ± 2.8 |
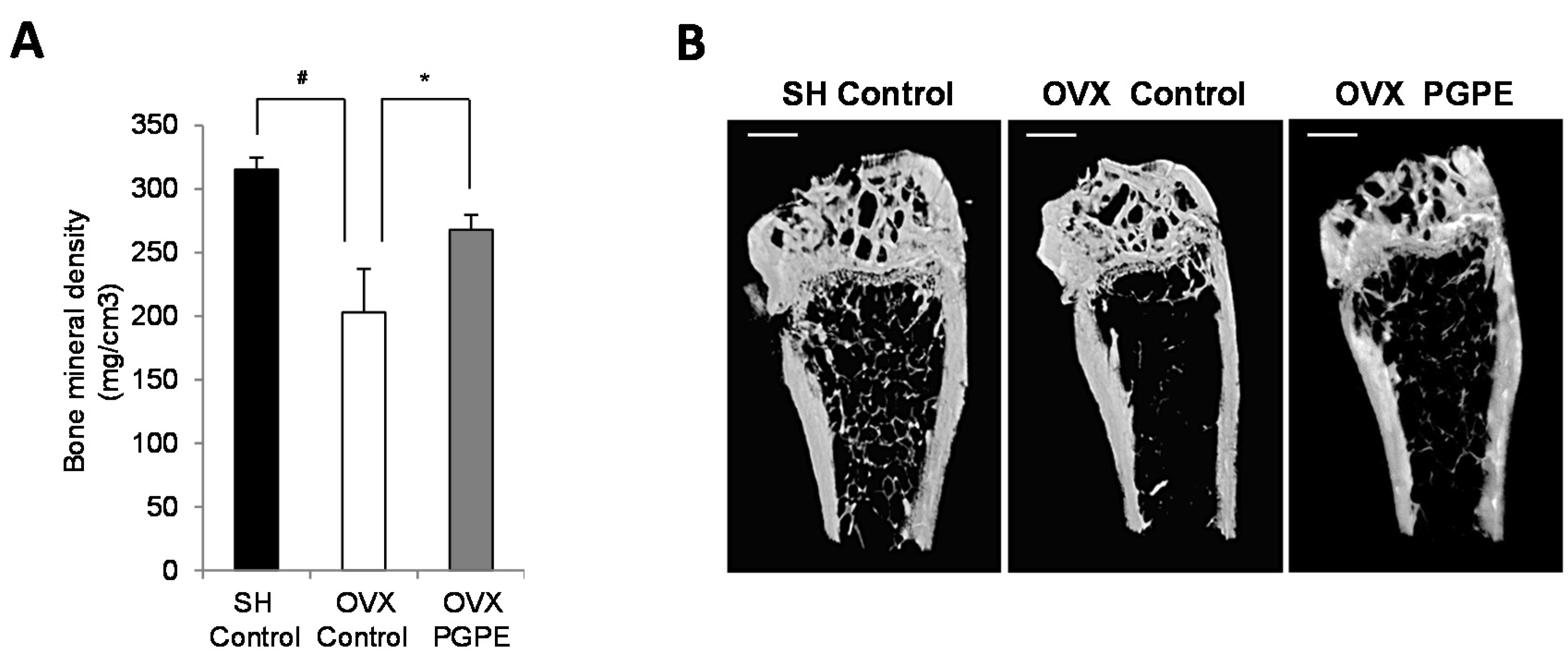
3.3.2. PGPE Consumption Improved Bone Mineral Density in Ovariectomized Mice
3.3.3. PGPE Preserved Bone Microarchitecture in Ovariectomized Mice
| Sham | Ovariectomized | ||
|---|---|---|---|
| Normal Diet | Normal Diet | PGPE Diet | |
| (SH) | (OVX) | (OVX+PGPE) | |
| BV/TV (%) | 12.7 ± 0.9 | 9.7 ± 0.6 # | 11.9 ± 0.76 * |
| TbTh (mm) | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| TbN (mm−1) | 2.0 ± 0.1 | 1.5 ± 0.1 # | 1.8 ± 0.07 * |
| TbSP (mm) | 0.2 ± 0.0 | 0.3 ± 9.97E-03 # | 0.3 ± 0.0 |
| SMI | 2.5 ± 0.1 | 2.6 ± 0.1 | 2.4 ± 0.04 * |
| Conn Dn (mm−3) | 278.4 ± 26.5 | 223.5 ± 8.8 | 224.3 ± 12.7 |
| Tb Pf (mm−1) | 25.5 ± 1.1 | 27.7 ± 1.0 | 23.2 ± 1.2 |
| Po tot (%) | 87.3 ± 1.0 | 90.3 ± 0.61 # | 88.1 ± 0.76 * |
| BS/TV (mm−1) | 8.9 ± 0.5 | 6.7 ± 0.32 # | 7.9 ± 0.31 * |
3.3.4. Pomegranate Intake Was Associated with an Improved Expression Profile of Specific Bone Markers
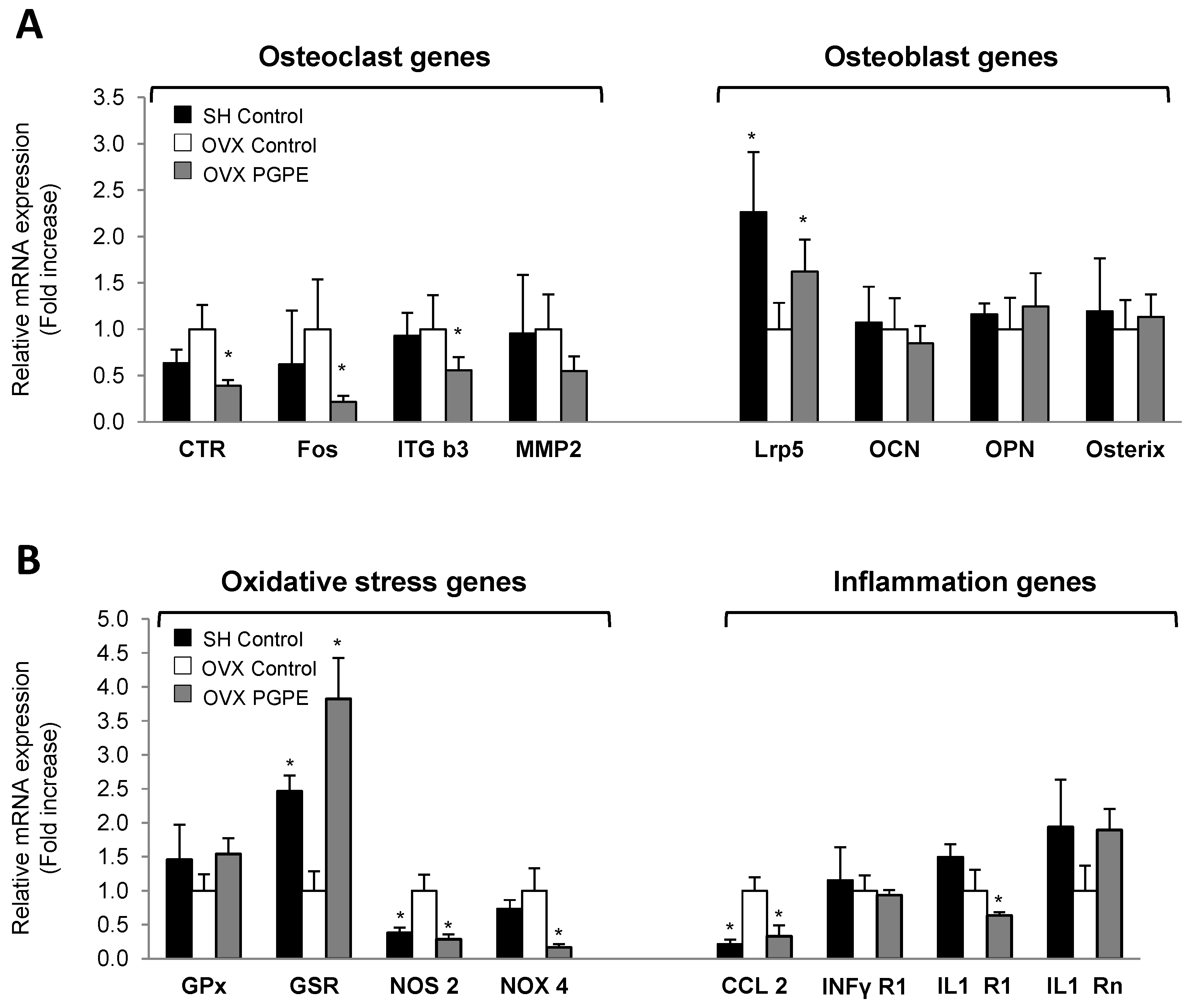
3.3.5. PGPE Enhanced Bone Inflammatory and Oxidative Status Marker Expression in Ovariectomized Mice
4. Discussion
Acknowledgments
Author Contributions
Conflict of Interest
References
- Rachner, T.D.; Khosla, S.; Hofbauer, L.C. Osteoporosis: Now and the future. Lancet 2011, 377, 1276–1287. [Google Scholar] [CrossRef]
- Sandhu, S.K.; Hampson, G. The pathogenesis, diagnosis, investigation and management of osteoporosis. J. Clin. Pathol. 2011, 64, 1042–1050. [Google Scholar] [CrossRef] [PubMed]
- Schulman, R.C.; Weiss, A.J.; Mechanick, J.I. Nutrition, bone, and aging: An integrative physiology approach. Curr. Osteoporos. Rep. 2011, 9, 184–195. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A.; Manach, C.; Morand, C.; Remesy, C.; Jimenez, L. Dietary polyphenols and the prevention of diseases. Crit. Rev. Food Sci. Nutr. 2005, 45, 287–306. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, D.; Ward, W.E. Detection of isoflavones in mouse tibia after feeding daidzein. J. Med. Food 2006, 9, 436–439. [Google Scholar] [CrossRef] [PubMed]
- Hagiwara, K.; Goto, T.; Araki, M.; Miyazaki, H.; Hagiwara, H. Olive polyphenol hydroxytyrosol prevents bone loss. Eur. J. Pharmacol. 2011, 662, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Ishimi, Y.; Yoshida, M.; Wakimoto, S.; Wu, J.; Chiba, H.; Wang, X.; Takeda, K.; Miyaura, C. Genistein, a soybean isoflavone, affects bone marrow lymphopoiesis and prevents bone loss in castrated male mice. Bone 2002, 31, 180–185. [Google Scholar] [CrossRef]
- Puel, C.; Mathey, J.; Agalias, A.; Kati-Coulibaly, S.; Mardon, J.; Obled, C.; Davicco, M.J.; Lebecque, P.; Horcajada, M.N.; Skaltsounis, A.L.; et al. Dose-response study of effect of oleuropein, an olive oil polyphenol, in an ovariectomy/inflammation experimental model of bone loss in the rat. Clin. Nutr. 2006, 25, 859–868. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Wang, X.X.; Chiba, H.; Higuchi, M.; Takasaki, M.; Ohta, A.; Ishimi, Y. Combined intervention of exercise and genistein prevented androgen deficiency-induced bone loss in mice. J. Appl. Physiol. 2003, 94, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Horcajada, M.N.; Offord, E. Naturally plant-derived compounds: Role in bone anabolism. Curr. Mol. Pharmacol. 2012, 5, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Yu, S. Genistein prevents bone resorption diseases by inhibiting bone resorption and stimulating bone formation. Biol. Pharm. Bull. 2003, 26, 780–786. [Google Scholar] [CrossRef] [PubMed]
- Rassi, C.M.; Lieberherr, M.; Chaumaz, G.; Pointillart, A.; Cournot, G. Down-regulation of osteoclast differentiation by daidzein via caspase 3. J. Bone Miner. Res. 2002, 17, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Trzeciakiewicz, A.; Habauzit, V.; Horcajada, M.N. When nutrition interacts with osteoblast function: Molecular mechanisms of polyphenols. Nutr. Res. Rev. 2009, 22, 68–81. [Google Scholar] [CrossRef] [PubMed]
- De Wilde, A.; Lieberherr, M.; Colin, C.; Pointillart, A. A low dose of daidzein acts as an erbeta-selective agonist in trabecular osteoblasts of young female piglets. J. Cell Physiol. 2004, 200, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Santiago-Mora, R.; Casado-Diaz, A.; de Castro, M.D.; Quesada-Gomez, J.M. Oleuropein enhances osteoblastogenesis and inhibits adipogenesis: The effect on differentiation in stem cells derived from bone marrow. Osteoporos. Int. 2011, 22, 675–684. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, E.; Yamaguchi, M. Anabolic effect of genistein in osteoblastic MC3T3-E1 cells. Int. J. Mol. Med. 2000, 5, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Lansky, E.P.; Newman, R.A. Punica granatum (pomegranate) and its potential for prevention and treatment of inflammation and cancer. J. Ethnopharmacol. 2007, 109, 177–206. [Google Scholar] [CrossRef] [PubMed]
- Viuda-Martos, M.; Fernández-López, J.; Pérez-Álvarez, J.A. Pomegranate and its many functional components as related to human health: A review. Compr. Rev. Food Sci. Food Saf. 2010, 9, 635–654. [Google Scholar] [CrossRef]
- Rahimi, H.R.; Arastoo, M.; Ostad, S.N. A comprehensive review of punica granatum (pomegranate) properties in toxicological, pharmacological, cellular and molecular biology researches. Iran J. Pharm. Res. 2012, 11, 385–400. [Google Scholar] [PubMed]
- Kim, Y.H.; Choi, E.M. Stimulation of osteoblastic differentiation and inhibition of interleukin-6 and nitric oxide in Mc3t3-E1 cells by pomegranate ethanol extract. Phytother. Res. 2009, 23, 737–739. [Google Scholar] [CrossRef] [PubMed]
- Rosenblat, M.; Volkova, N.; Aviram, M. Pomegranate juice (PJ) consumption antioxidative properties on mouse macrophages, but not pj beneficial effects on macrophage cholesterol and triglyceride metabolism, are mediated via pj-induced stimulation of macrophage pon2. Atherosclerosis 2010, 212, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Hollebeeck, S.; Winand, J.; Herent, M.F.; During, A.; Leclercq, J.; Larondelle, Y.; Schneider, Y.J. Anti-inflammatory effects of pomegranate (Punica granatum L.) husk ellagitannins in Caco-2 cells, an in vitro model of human intestine. Food Funct. 2012, 3, 875–885. [Google Scholar] [CrossRef] [PubMed]
- Larrosa, M.; Gonzalez-Sarrias, A.; Yanez-Gascon, M.J.; Selma, M.V.; Azorin-Ortuno, M.; Toti, S.; Tomas-Barberan, F.; Dolara, P.; Espin, J.C. Anti-inflammatory properties of a pomegranate extract and its metabolite urolithin-A in a colitis rat model and the effect of colon inflammation on phenolic metabolism. J. Nutr. Biochem. 2010, 21, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Kasimsetty, S.G.; Bialonska, D.; Reddy, M.K.; Ma, G.; Khan, S.I.; Ferreira, D. Colon cancer chemopreventive activities of pomegranate ellagitannins and urolithins. J. Agric. Food Chem. 2010, 58, 2180–2187. [Google Scholar] [CrossRef] [PubMed]
- Larrosa, M.; Tomas-Barberan, F.A.; Espin, J.C. The dietary hydrolysable tannin punicalagin releases ellagic acid that induces apoptosis in human colon adenocarcinoma Caco-2 cells by using the mitochondrial pathway. J. Nutr. Biochem. 2006, 17, 611–625. [Google Scholar] [CrossRef] [PubMed]
- Syed, D.N.; Chamcheu, J.C.; Mukhtar, V.M. Pomegranate extracts and cancer prevention: Molecular and cellular activities. Anticancer Agents Med. Chem. 2012, 13, 1149–1161. [Google Scholar] [CrossRef]
- Aviram, M.; Volkova, N.; Coleman, R.; Dreher, M.; Reddy, M.K.; Ferreira, D.; Rosenblat, M. Pomegranate phenolics from the peels, arils, and flowers are antiatherogenic: Studies in vivo in atherosclerotic apolipoprotein e-deficient (e 0) mice and in vitro in cultured macrophages and lipoproteins. J. Agric. Food Chem. 2008, 56, 1148–1157. [Google Scholar] [CrossRef] [PubMed]
- Rosenblat, M.; Volkova, N.; Coleman, R.; Aviram, M. Pomegranate byproduct administration to apolipoprotein e-deficient mice attenuates atherosclerosis development as a result of decreased macrophage oxidative stress and reduced cellular uptake of oxidized low-density lipoprotein. J. Agric. Food Chem. 2006, 54, 1928–1935. [Google Scholar] [CrossRef] [PubMed]
- Fuhrman, B.; Volkova, N.; Aviram, M. Pomegranate juice inhibits oxidized LDL uptake and cholesterol biosynthesis in macrophages. J. Nutr. Biochem. 2005, 16, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Esmaillzadeh, A.; Tahbaz, F.; Gaieni, I.; Alavi-Majd, H.; Azadbakht, L. Concentrated pomegranate juice improves lipid profiles in diabetic patients with hyperlipidemia. J. Med. Food 2004, 7, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Ortiz, M.; Martinez-Abundis, E.; Espinel-Bermudez, M.C.; Perez-Rubio, K.G. Effect of pomegranate juice on insulin secretion and sensitivity in patients with obesity. Ann. Nutr. Metab. 2011, 58, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Bagri, P.; Ali, M.; Aeri, V.; Bhowmik, M.; Sultana, S. Antidiabetic effect of punica granatum flowers: Effect on hyperlipidemia, pancreatic cells lipid peroxidation and antioxidant enzymes in experimental diabetes. Food Chem. Toxicol. 2009, 47, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Fawole, O.A.; Makunga, N.P.; Opara, U.L. Antibacterial, antioxidant and tyrosinase-inhibition activities of pomegranate fruit peel methanolic extract. BMC Complement Altern. Med. 2012, 12, 200. [Google Scholar] [CrossRef] [PubMed]
- Abdollahzadeh, S.; Mashouf, R.; Mortazavi, H.; Moghaddam, M.; Roozbahani, N.; Vahedi, M. Antibacterial and antifungal activities of Punica granatum peel extracts against oral pathogens. J. Dent. 2011, 8, 1–6. [Google Scholar]
- Haidari, M.; Ali, M.; Ward Casscells, S., 3rd; Madjid, M. Pomegranate (punica granatum) purified polyphenol extract inhibits influenza virus and has a synergistic effect with oseltamivir. Phytomedicine 2009, 16, 1127–1136. [Google Scholar] [CrossRef] [PubMed]
- Johanningsmeier, S.D.; Harris, G.K. Pomegranate as a functional food and nutraceutical source. Annu. Rev. Food Sci. Technol. 2011, 2, 181–201. [Google Scholar] [CrossRef] [PubMed]
- Haber, S.L.; Joy, J.K.; Largent, R. Antioxidant and antiatherogenic effects of pomegranate. Am. J. Health Syst. Pharm. 2011, 68, 1302–1305. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Yang, J.; Wei, J.; Li, Y.; Xu, J.; Jiang, Y. Antioxidant activities of peel, pulp and seed fractions of common fruits as determined by frap assay. Nutr. Res. 2003, 23, 1719–1726. [Google Scholar] [CrossRef]
- Noda, Y.; Kaneyuki, T.; Mori, A.; Packer, L. Antioxidant activities of pomegranate fruit extract and its anthocyanidins: Delphinidin, cyanidin, and pelargonidin. J. Agric. Food Chem. 2002, 50, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Krause, K.H. Aging: A revisited theory based on free radicals generated by NOX family NADPH oxidases. Exp. Gerontol. 2007, 42, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Probst-Hensch, N.M. Chronic age-related diseases share risk factors: Do they share pathophysiological mechanisms and why does that matter? Swiss Med. Wkly. 2010. [Google Scholar] [CrossRef] [PubMed]
- Syslova, K.; Bohmova, A.; Mikoska, M.; Kuzma, M.; Pelclova, D.; Kacer, P. Multimarker screening of oxidative stress in aging. Oxid. Med Cell. Longev. 2014, 2014, 562–860. [Google Scholar] [CrossRef] [PubMed]
- Balcerczyk, A.; Gajewska, A.; Macierzynska-Piotrowska, E.; Pawelczyk, T.; Bartosz, G.; Szemraj, J. Enhanced antioxidant capacity and anti-ageing biomarkers after diet micronutrient supplementation. Molecules 2014, 19, 14794–14808. [Google Scholar] [CrossRef] [PubMed]
- Howcroft, T.K.; Campisi, J.; Louis, G.B.; Smith, M.T.; Wise, B.; Wyss-Coray, T.; Augustine, A.D.; McElhaney, J.E.; Kohanski, R.; Sierra, F. The role of inflammation in age-related disease. Aging (Albany NY) 2013, 5, 84–93. [Google Scholar] [PubMed]
- Wauquier, F.; Barquissau, V.; Leotoing, L.; Davicco, M.J.; Lebecque, P.; Mercier, S.; Philippe, C.; Miot-Noirault, E.; Chardigny, J.M.; Morio, B.; et al. Borage and fish oils lifelong supplementation decreases inflammation and improves bone health in a murine model of senile osteoporosis. Bone 2012, 50, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Lacativa, P.G.; Farias, M.L. Osteoporosis and inflammation. Arq Bras Endocrinol. Metabol. 2010, 54, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Wauquier, F.; Leotoing, L.; Coxam, V.; Guicheux, J.; Wittrant, Y. Oxidative stress in bone remodelling and disease. Trends Mol. Med. 2009, 15, 468–477. [Google Scholar] [CrossRef] [PubMed]
- Manolagas, S.C.; Parfitt, A.M. What old means to bone. Trends Endocrinol. Metab. 2010, 21, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Koh, J.M.; Lee, Y.S.; Kim, Y.S.; Kim, D.J.; Kim, H.H.; Park, J.Y.; Lee, K.U.; Kim, G.S. Homocysteine enhances bone resorption by stimulation of osteoclast formation and activity through increased intracellular ros generation. J. Bone Miner. Res. 2006, 21, 1003–1011. [Google Scholar] [CrossRef] [PubMed]
- Ozgocmen, S.; Kaya, H.; Fadillioglu, E.; Aydogan, R.; Yilmaz, Z. Role of antioxidant systems, lipid peroxidation, and nitric oxide in postmenopausal osteoporosis. Mol. Cell. Biochem. 2007, 295, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Zahin, M.; Aqil, F.; Ahmad, I. Broad spectrum antimutagenic activity of antioxidant active fraction of Punica granatum L. Peel extracts. Mutat. Res. 2010, 703, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Tzulker, R.; Glazer, I.; Bar-Ilan, I.; Holland, D.; Aviram, M.; Amir, R. Antioxidant activity, polyphenol content, and related compounds in different fruit juices and homogenates prepared from 29 different pomegranate accessions. J. Agric. Food Chem. 2007, 55, 9559–9570. [Google Scholar] [CrossRef] [PubMed]
- Ismail, T.; Sestili, P.; Akhtar, S. Pomegranate peel and fruit extracts: A review of potential anti-inflammatory and anti-infective effects. J. Ethnopharmacol. 2012, 143, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Mousavinejad, G.; Emam-Djomeh, Z.; Rezaei, K.; Khodaparast, M.H.H. Identification and quantification of phenolic compounds and their effects on antioxidant activity in pomegranate juices of eight iranian cultivars. Food Chem. 2009, 115, 1274–1278. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [PubMed]
- Installation expérimentale de nutrition. Available online: https://www6.clermont.inra.fr/unh/PlateauTechniques/Installation-Experimentale-de-Nutrition (accessed on 26 October 2015).
- Sabokbar, A.; Millett, P.J.; Myer, B.; Rushton, N. A rapid, quantitative assay for measuring alkaline phosphatase activity in osteoblastic cells in vitro. Bone Miner. 1994, 27, 57–67. [Google Scholar] [CrossRef]
- Trzeciakiewicz, A.; Habauzit, V.; Mercier, S.; Barron, D.; Urpi-Sarda, M.; Manach, C.; Offord, E.; Horcajada, M.N. Molecular mechanism of hesperetin-7-O-glucuronide, the main circulating metabolite of hesperidin, involved in osteoblast differentiation. J. Agric. Food Chem. 2010, 58, 668–675. [Google Scholar] [CrossRef] [PubMed]
- Wittrant, Y.; Gorin, Y.; Woodruff, K.; Horn, D.; Abboud, H.E.; Mohan, S.; Abboud-Werner, S.L. High D(+)glucose concentration inhibits rankl-induced osteoclastogenesis. Bone 2008, 42, 1122–1130. [Google Scholar] [CrossRef] [PubMed]
- Kalu, D.N. The ovariectomized rat model of postmenopausal bone loss. Bone Miner. 1991, 15, 175–191. [Google Scholar] [CrossRef]
- Lei, Z.; Xiaoying, Z.; Xingguo, L. Ovariectomy-associated changes in bone mineral density and bone marrow haematopoiesis in rats. Int. J. Exp. Pathol. 2009, 90, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Bruker. Available online: http://bruker-microct.com/products/downloads.htm (accessed on 26 October 2015).
- XLSTAT. Available online: https://www.xlstat.com/fr/ (accessed on 26 October 2015).
- Espin, J.C.; Gonzalez-Barrio, R.; Cerda, B.; Lopez-Bote, C.; Rey, A.I.; Tomas-Barberan, F.A. Iberian pig as a model to clarify obscure points in the bioavailability and metabolism of ellagitannins in humans. J. Agric. Food Chem. 2007, 55, 10476–10485. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Sarrias, A.; Gimenez-Bastida, J.A.; Garcia-Conesa, M.T.; Gomez-Sanchez, M.B.; Garcia-Talavera, N.V.; Gil-Izquierdo, A.; Sanchez-Alvarez, C.; Fontana-Compiano, L.O.; Morga-Egea, J.P.; Pastor-Quirante, F.A.; et al. Occurrence of urolithins, gut microbiota ellagic acid metabolites and proliferation markers expression response in the human prostate gland upon consumption of walnuts and pomegranate juice. Mol. Nutr. Food Res. 2010, 54, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Patel, C.; Dadhaniya, P.; Hingorani, L.; Soni, M.G. Safety assessment of pomegranate fruit extract: Acute and subchronic toxicity studies. Food Chem. Toxicol. 2008, 46, 2728–2735. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C. Molecular mechanisms of osteoblast-specific transcription factor osterix effect on bone formation. Beijing Da Xue Xue Bao 2012, 44, 659–665. [Google Scholar] [PubMed]
- Zhang, Y.; Su, J.; Yu, J.; Bu, X.; Ren, T.; Liu, X.; Yao, L. An essential role of discoidin domain receptor 2 (DDR2) in osteoblast differentiation and chondrocyte maturation via modulation of Runx2 activation. J. Bone Miner. Res. 2011, 26, 604–617. [Google Scholar] [CrossRef] [PubMed]
- Sreekumar, S.; Sithul, H.; Muraleedharan, P.; Azeez, J.M.; Sreeharshan, S. Pomegranate fruit as a rich source of biologically active compounds. BioMed. Res. Int. 2014, 2014, 686921. [Google Scholar] [CrossRef] [PubMed]
- Boyle, W.J.; Simonet, W.S.; Lacey, D.L. Osteoclast differentiation and activation. Nature 2003, 423, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Vaira, S.; Johnson, T.; Hirbe, A.C.; Alhawagri, M.; Anwisye, I.; Sammut, B.; O'Neal, J.; Zou, W.; Weilbaecher, K.N.; Faccio, R.; et al. RelB is the NF-kB subunit downstream of nik responsible for osteoclast differentiation. Proc. Natl. Acad. Sci. USA 2008, 105, 3897–3902. [Google Scholar] [CrossRef] [PubMed]
- Coelho, L.F.; Magno de Freitas Almeida, G.; Mennechet, F.J.; Blangy, A.; Uze, G. Interferon-alpha and -beta differentially regulate osteoclastogenesis: Role of differential induction of chemokine cxcl11 expression. Proc. Natl. Acad. Sci. USA 2005, 102, 11917–11922. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Murray, M.A.; Garrett, I.R.; Gutierrez, G.E.; Nyman, J.S.; Mundy, G.; Fast, D.; Gellenbeck, K.W.; Chandra, A.; Ramakrishnan, S. A targeted approach for evaluating preclinical activity of botanical extracts for support of bone health. J. Nutr. Sci. 2014, 3, e13. [Google Scholar] [CrossRef] [PubMed]
- Park, E.K.; Kim, M.S.; Lee, S.H.; Kim, K.H.; Park, J.Y.; Kim, T.H.; Lee, I.S.; Woo, J.T.; Jung, J.C.; Shin, H.I.; et al. Furosin, an ellagitannin, suppresses rankl-induced osteoclast differentiation and function through inhibition of map kinase activation and actin ring formation. Biochem. Biophys. Res. Commun. 2004, 325, 1472–1480. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Summanen, P.H.; Komoriya, T.; Henning, S.M.; Lee, R.P.; Carlson, E.; Heber, D.; Finegold, S.M. Pomegranate ellagitannins stimulate growth of gut bacteria in vitro: Implications for prebiotic and metabolic effects. Anaerobe 2015, 34, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, S.; Ismail, T.; Fraternale, D.; Sestili, P. Pomegranate peel and peel extracts: Chemistry and food features. Food Chem. 2015, 174, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Bialonska, D.; Ramnani, P.; Kasimsetty, S.G.; Muntha, K.R.; Gibson, G.R.; Ferreira, D. The influence of pomegranate by-product and punicalagins on selected groups of human intestinal microbiota. Int. J. Food Microbiol. 2010, 140, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Lutz, T.; Scharrer, E. Effect of short-chain fatty acids on calcium absorption by the rat colon. Exp. Physiol. 1991, 76, 615–618. [Google Scholar] [CrossRef] [PubMed]
- Roberfroid, M.; Gibson, G.R.; Hoyles, L.; McCartney, A.L.; Rastall, R.; Rowland, I.; Wolvers, D.; Watzl, B.; Szajewska, H.; Stahl, B.; et al. Prebiotic effects: Metabolic and health benefits. Br. J. Nutr. 2010, 104, S1–S63. [Google Scholar] [CrossRef] [PubMed]
- Sreeja, S.; Santhosh Kumar, T.R.; Lakshmi, B.S. Pomegranate extract demonstrate a selective estrogen receptor modulator profile in human tumor cell lines and in vivo models of estrogen deprivation. J. Nutr. Biochem. 2012, 23, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Al-Obaidi, M.M.; Al-Bayaty, F.H.; Al Batran, R.; Hassandarvish, P.; Rouhollahi, E. Protective effect of ellagic acid on healing alveolar bone after tooth extraction in rat—A histological and immunohistochemical study. Arch. Oral Biol. 2014, 59, 987–999. [Google Scholar] [CrossRef] [PubMed]
- Al-Obaidi, M.M.; Al-Bayaty, F.H.; Al Batran, R.; Hussaini, J.; Khor, G.H. Impact of ellagic acid in bone formation after tooth extraction: An experimental study on diabetic rats. Sci. World J. 2014, 2014, 908098. [Google Scholar] [CrossRef] [PubMed]
- Satpathy, S.; Patra, A.; Purohit, A.P. Estrogenic activity of Punica granatum L. Pell estract. Asian Pac. J. Reprod. 2013, 2, 19–24. [Google Scholar] [CrossRef]
- Bahtiar, A.; Arifin, S.; Razalifha, A.; Qomariah, N.; Wuyung, P.; Arsianti, A. Polar fraction of Punica granatum L. Peel extract increased osteoblast number on ovariectomized rat bone. Int. J. Herbal Med. 2014, 2, 65–70. [Google Scholar]
- Kang, S.J.; Choi, B.R.; Kim, S.H.; Yi, H.Y.; Park, H.R.; Kim, D.C.; Choi, S.H.; Han, C.H.; Park, S.J.; Song, C.H.; et al. Dried pomegranate potentiates anti-osteoporotic and anti-obesity activities of red clover dry extracts in ovariectomized rats. Nutrients 2015, 7, 2622–2647. [Google Scholar] [CrossRef] [PubMed]
- Papoutsi, Z.; Kassi, E.; Chinou, I.; Halabalaki, M.; Skaltsounis, L.A.; Moutsatsou, P. Walnut extract (juglans regia l.) and its component ellagic acid exhibit anti-inflammatory activity in human aorta endothelial cells and osteoblastic activity in the cell line ks483. Br. J. Nutr. 2008, 99, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Papoutsi, Z.; Kassi, E.; Tsiapara, A.; Fokialakis, N.; Chrousos, G.P.; Moutsatsou, P. Evaluation of estrogenic/antiestrogenic activity of ellagic acid via the estrogen receptor subtypes eralpha and erbeta. J. Agric. Food Chem. 2005, 53, 7715–7720. [Google Scholar] [CrossRef] [PubMed]
- Mundy, G.R. Osteoporosis and inflammation. Nutr. Rev. 2007, 65, S147–S151. [Google Scholar] [CrossRef] [PubMed]
- Middha, S.K.; Usha, T.; Pande, V. HPLC evaluation of phenolic profile, nutritive content, and antioxidant capacity of extracts obtained from punica granatum fruit peel. Adv. Pharmacol. Sci. 2013, 2013, 296236. [Google Scholar] [PubMed]
- Shukla, M.; Gupta, K.; Rasheed, Z.; Khan, K.A.; Haqqi, T.M. Consumption of hydrolyzable tannins-rich pomegranate extract suppresses inflammation and joint damage in rheumatoid arthritis. Nutrition 2008, 24, 733–743. [Google Scholar] [CrossRef] [PubMed]
- Balbir-Gurman, A.; Fuhrman, B.; Braun-Moscovici, Y.; Markovits, D.; Aviram, M. Consumption of pomegranate decreases serum oxidative stress and reduces disease activity in patients with active rheumatoid arthritis: A pilot study. Isr. Med. Assoc. J. 2011, 13, 474–479. [Google Scholar] [PubMed]
- Lee, C.J.; Chen, L.G.; Liang, W.L.; Wanga, C. Anti-inflammatory effects of punica granatum linne in vitro and in vivo. Food Chem. 2010, 118, 315–322. [Google Scholar] [CrossRef]
- Iwatake, M.; Okamoto, K.; Tanaka, T.; Tsukuba, T. Punicalagin attenuates osteoclast differentiation by impairing nfatc1 expression and blocking AKT- and JNK-dependent pathways. Mol. Cell. Biochem. 2015, 407, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Bialonska, D.; Kasimsetty, S.G.; Khan, S.I.; Ferreira, D. Urolithins, intestinal microbial metabolites of pomegranate ellagitannins, exhibit potent antioxidant activity in a cell-based assay. J. Agric. Food Chem. 2009, 57, 10181–10186. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Sarrias, A.; Larrosa, M.; Tomas-Barberan, F.A.; Dolara, P.; Espin, J.C. NF-κB-dependent anti-inflammatory activity of urolithins, gut microbiota ellagic acid-derived metabolites, in human colonic fibroblasts. Br. J. Nutr. 2010, 104, 503–512. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spilmont, M.; Léotoing, L.; Davicco, M.-J.; Lebecque, P.; Miot-Noirault, E.; Pilet, P.; Rios, L.; Wittrant, Y.; Coxam, V. Pomegranate Peel Extract Prevents Bone Loss in a Preclinical Model of Osteoporosis and Stimulates Osteoblastic Differentiation in Vitro. Nutrients 2015, 7, 9265-9284. https://doi.org/10.3390/nu7115465
Spilmont M, Léotoing L, Davicco M-J, Lebecque P, Miot-Noirault E, Pilet P, Rios L, Wittrant Y, Coxam V. Pomegranate Peel Extract Prevents Bone Loss in a Preclinical Model of Osteoporosis and Stimulates Osteoblastic Differentiation in Vitro. Nutrients. 2015; 7(11):9265-9284. https://doi.org/10.3390/nu7115465
Chicago/Turabian StyleSpilmont, Mélanie, Laurent Léotoing, Marie-Jeanne Davicco, Patrice Lebecque, Elisabeth Miot-Noirault, Paul Pilet, Laurent Rios, Yohann Wittrant, and Véronique Coxam. 2015. "Pomegranate Peel Extract Prevents Bone Loss in a Preclinical Model of Osteoporosis and Stimulates Osteoblastic Differentiation in Vitro" Nutrients 7, no. 11: 9265-9284. https://doi.org/10.3390/nu7115465
APA StyleSpilmont, M., Léotoing, L., Davicco, M.-J., Lebecque, P., Miot-Noirault, E., Pilet, P., Rios, L., Wittrant, Y., & Coxam, V. (2015). Pomegranate Peel Extract Prevents Bone Loss in a Preclinical Model of Osteoporosis and Stimulates Osteoblastic Differentiation in Vitro. Nutrients, 7(11), 9265-9284. https://doi.org/10.3390/nu7115465






