Oral Fat Sensing and CD36 Gene Polymorphism in Algerian Lean and Obese Teenagers
Abstract
:1. Introduction
2. Experimental Section
2.1. Subjects
| Parameters | Control Participants (n = 82) | Obese Participants (n = 83) |
|---|---|---|
| Age (years) | 13.92 ± 0.23 | 14.01 ± 0.19 |
| BMI z-score | 0.03 ± 0.00 | 2.67 ± 0.29 ** |
| Glycemia (mmol/L) | 4.41 ± 0.06 | 4.76 ± 0.05 * |
| TC (mmol/L) | 3.04 ± 0.08 | 3.39 ± 0.07 * |
| LDL-C (mmol/L) | 1.64 ± 0.07 | 2.00 ± 0.06 ** |
| HDL-C (mmol/L) | 1.08 ± 0.03 | 0.91 ± 0.02 ** |
| TG (mmol/L) | 0.74 ± 0.04 | 1.04 ± 0.05 ** |
| Insulin (pmol/L) | 45.98 ± 0.69 | 54.38 ± 2.22 ** |
| HOMA index | 1.29 ± 0.03 | 1.70 ± 0.12 ** |
2.1.1. Ethics
2.2. BMI z-Score
2.3. Determination of Fasting Blood Glucose and Lipids Parameters
2.4. Oleic Acid Sensitivity Analysis
2.5. Genotyping Analysis
2.6. Statistical Analysis
3. Results
3.1. Characteristics of the Participants
3.2. Blood Parameters
3.3. Oleic Acid Sensitivity
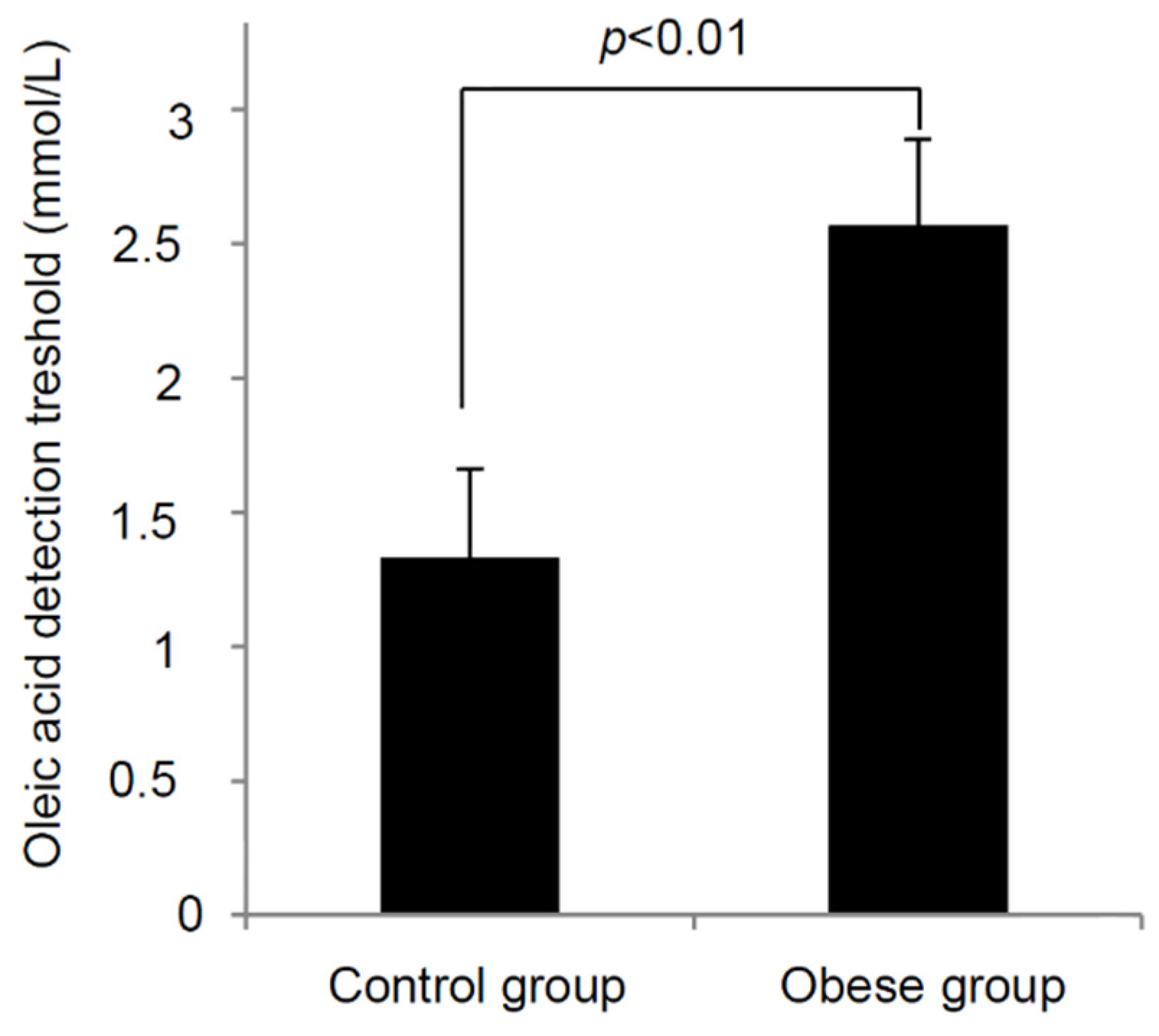
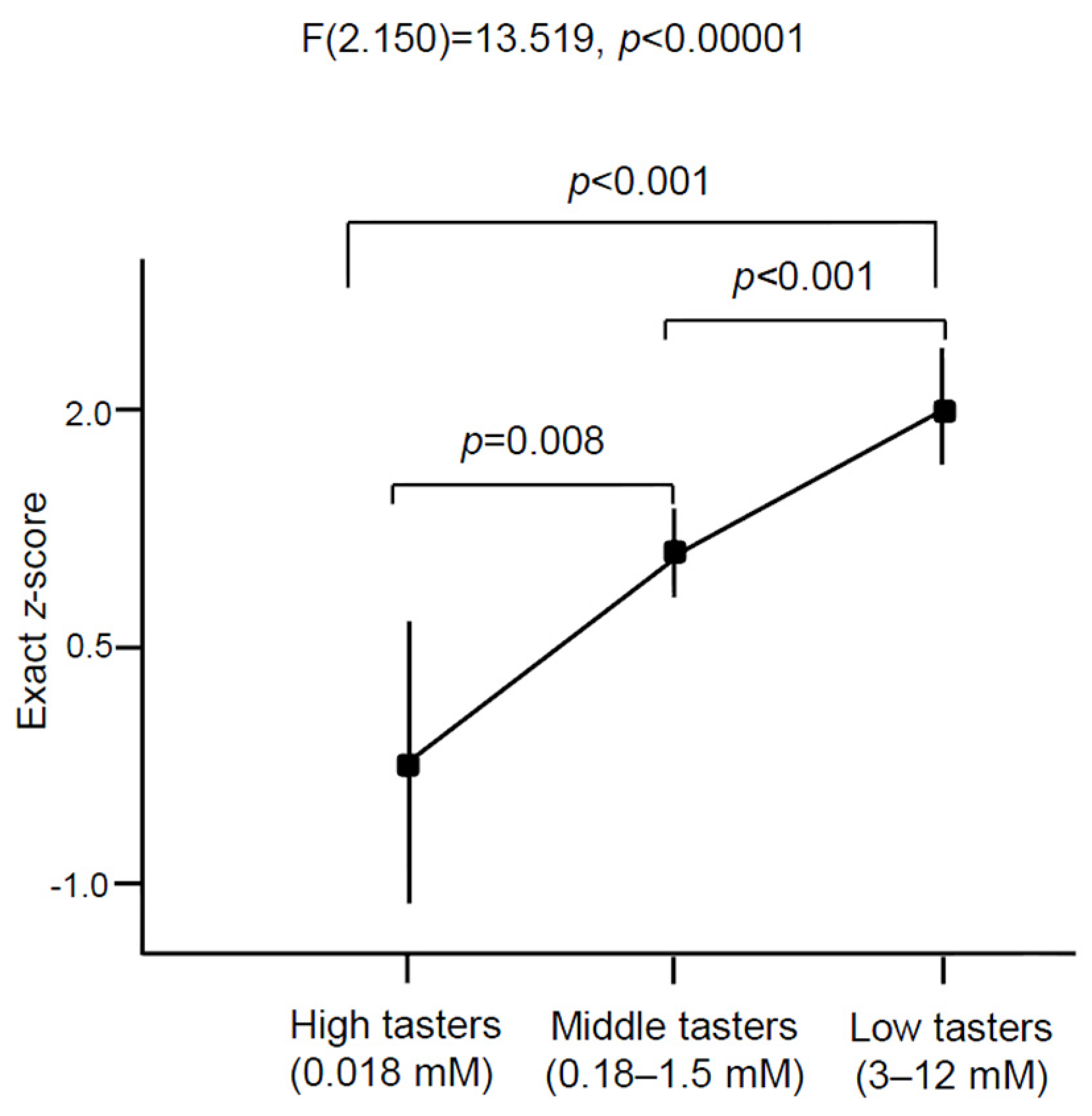
3.4. CD36 Genotyping
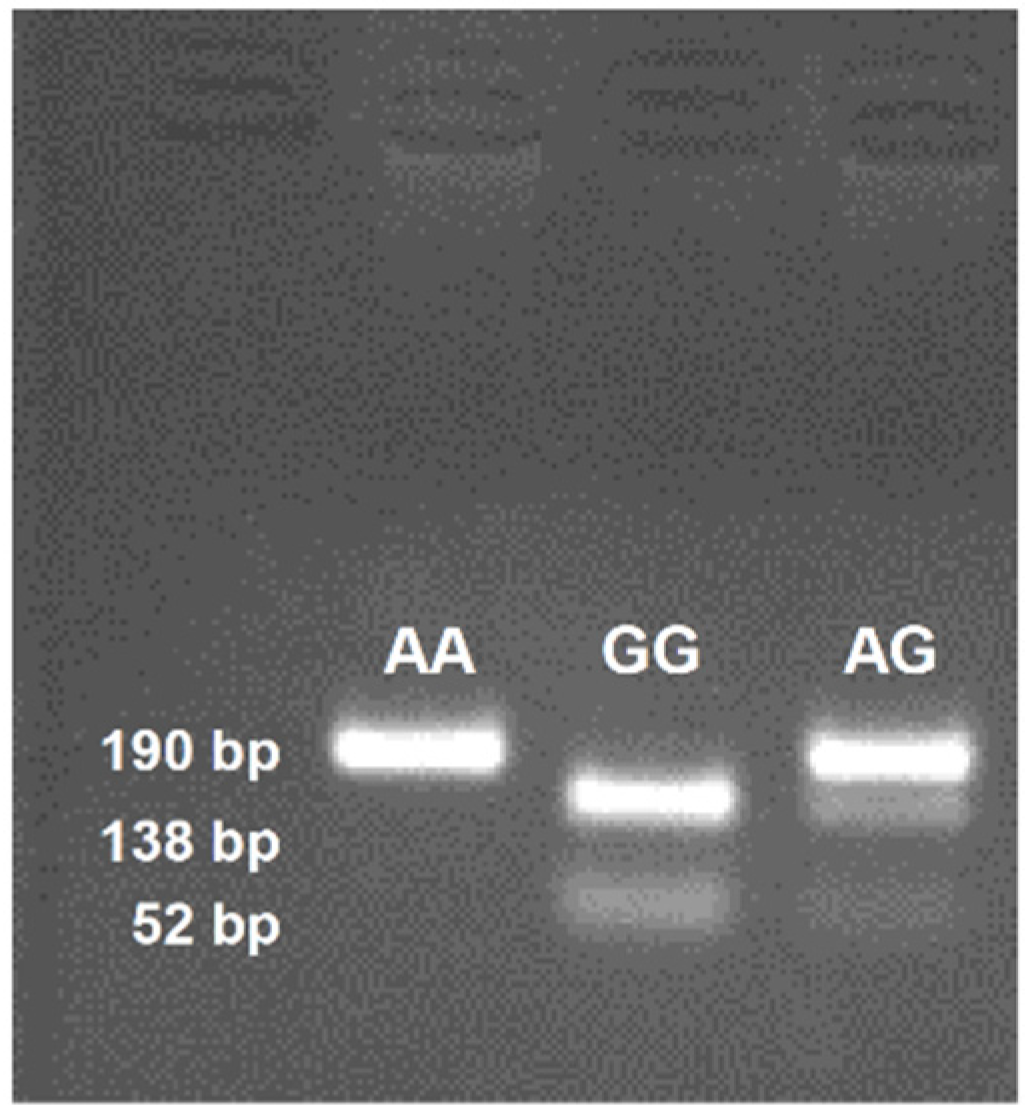
| Parameters | Control Participants (n = 82) | Obese Participants (n = 83) | Statistical Calculations |
|---|---|---|---|
| HWE χ2 | 2.67 | 3.05 | NS |
| Alleles (%) | p = 0.041 | ||
| A | 93 (56.7) | 113 (68.1) | OR = 1.63; 95% CI |
| G | 71 (43.3) | 53 (31.9) | RR = 1.28; 95% CI |
| Genotypes (%) | |||
| AA | 30 (36.6) | 35 (42.2) | p = 0.008 |
| AG | 33 (40.2) | 43 (51.8) | p = 0.002 |
| GG | 19 (23.2) | 5 (6.0) | - |
4. Discussion
Author Contributions
Conflicts of Interest
References
- George, A.M.; Jacob, A.G.; Fogelfeld, L. Lean diabetes mellitus: An emerging entity in the era of obesity. World J. Diabetes 2015, 6, 613–620. [Google Scholar] [PubMed]
- Ang, Y.N.; Wee, B.S.; Poh, B.K.; Ismail, M.N. Multifactorial Influences of Childhood Obesity. Curr. Obes. Rep. 2012, 2, 10–22. [Google Scholar] [CrossRef]
- Barnett, T.A.; Maximova, K.; Sabiston, C.M.; van Hulst, A.; Brunet, J.; Castonguay, A.L.; Bélanger, M.; O’Loughlin, J. Physical activity growth curves relate to adiposity in adolescents. Ann. Epidemiol. 2013, 23, 529–533. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Paradis, A.M.; Godin, G.; Pérusse, L.; Vohl, M.C. Associations between dietary patterns and obesity phenotypes. Int. J. Obes. 2009, 33, 1419–1426. [Google Scholar] [CrossRef] [PubMed]
- Rolls, E.T. Taste, olfactory, and food reward value processing in the brain. Prog. Neurobiol. 2015, 127–128, 64–90. [Google Scholar] [CrossRef] [PubMed]
- Gilbertson, T.A.; Khan, N.A. Cell signaling mechanisms of oro-gustatory detection of dietary fat: Advances and challenges. Prog. Lipid. Res. 2014, 53, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Silverstein, R.L.; Febbraio, M. CD36, a scavenger receptor involved in immunity, metabolism, angiogenesis, and behavior. Sci. Signal. 2010, 2, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Cartoni, C.; Yasumatsu, K.; Ohkuri, T.; Shigemura, N.; Yoshida, R.; Godinot, N.; le Coutre, J.; Ninomiya, Y.; Damak, S. Taste preference for fatty acids is mediated by GPR40 and GPR120. J. Neurosci. 2010, 30, 8376–8382. [Google Scholar] [CrossRef] [PubMed]
- Ancel, D.; Bernard, A.; Subramaniam, S.; Hirasawa, A.; Tsujimoto, G.; Hashimoto, T.; Passilly-Degrace, P.; Khan, N.A.; Besnard, P. The oral lipid sensor GPR120 is not indispensable for the orosensory detection of dietary lipids in the mouse. J. Lipid Res. 2015, 56, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Bacci, S.; Mlynarski, W.; Gottardo, L.; Soccio, T.; Menzaghi, C.; Iori, E.; Lager, R.A.; Shroff, A.R.; Gervino, E.V.; et al. A common haplotype at the CD36 locus is associated with high free fatty acid levels and increased cardiovascular risk in Caucasians. Hum. Mol. Genet. 2004, 13, 2197–2205. [Google Scholar] [CrossRef] [PubMed]
- Ozdener, M.H.; Subramaniam, S.; Sundaresan, S.; Sery, O.; Hashimoto, T.; Asakawa, Y.; Besnard, P.; Abumrad, N.A.; Khan, N.A. CD36- and GPR120-mediated Ca2+ signaling in human taste bud cells mediates differential responses to fatty acids and is altered in obese mice. Gastroenterology 2014, 146, 995–1005. [Google Scholar] [CrossRef] [PubMed]
- Love-Gregory, L.; Sherva, R.; Schappe, T.; Qi, J.S.; McCrea, J.; Klein, S.; Connelly, M.A.; Abumrad, N.A. Common CD36 SNPs reduce protein expression and may contribute to a protective atherogenic profile. Hum. Mol. Genet. 2011, 20, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ling, Z.Y.; Deng, S.B.; Du, H.A.; Yin, Y.H.; Yuan, J.; She, Q.; Chen, Y.Q. Associations between CD36 gene polymorphisms and susceptibility to coronary artery heart disease. Braz. J. Med. Biol. Res. 2014, 47, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, M.; Gautam, S.; Saxena, M.; Bidb, H.K.; Agrawalc, C.G. Association of CD36 gene variants rs1761667 (G > A) and rs1527483 (C > T) with Type 2 diabetes in North Indian population. Int. J. Diabetes. Mellit. 2010, 2, 179–183. [Google Scholar] [CrossRef]
- Pepino, M.Y.; Love-Gregory, L.; Klein, S.; Abumrad, N.A. The fatty acid translocase gene CD36 and lingual lipase influence oral sensitivity to fat in obese subjects. J. Lipid Res. 2012, 53, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Mrizak, I.; Šerý, O.; Plesnik, J.; Arfa, A.; Fekih, M.; Bouslema, A.; Zaouali, M.; Tabka, Z.; Khan, N.A. The A allele of cluster of differentiation 36 (CD36) SNP 1761667 associates with decreased lipid taste perception in obese Tunisian women. Br. J. Nutr. 2015, 113, 1330–1337. [Google Scholar] [CrossRef] [PubMed]
- Sayed, A.; Šerý, O.; Plesnik, J.; Daoudi, H.; Rouabah, A.; Rouabah, L.; Khan, N.A. CD36 AA genotype is associated with decreased lipid taste perception in young obese, but not lean, children. Int. J. Obes. 2015, 6, 920–924. [Google Scholar] [CrossRef] [PubMed]
- Melis, M.; Sollai, G.; Muroni, P.; Crnjar, R.; Barbarossa, I.T. Associations between orosensory perception of oleic acid, the common single nucleotide polymorphisms (rs1761667 and rs1527483) in the CD36 gene, and 6-n-propylthiouracil (PROP) tasting. Nutrients 2015, 7, 2068–2084. [Google Scholar] [CrossRef] [PubMed]
- Dietz, H. Critical periods in childhood for the development of obesity. Am. J. Clin. Nutr. 1994, 59, 955–999. [Google Scholar] [PubMed]
- Parsons, T.J.; Power, C.; Logan, S.; Summerbell, C.D. Childhood predictors of adult obesity: A systematic review. Int. J. Obes. Relat. Metab. Disord. 1999, 23, S1–S107. [Google Scholar] [PubMed]
- Skilton, M.R.; Marks, G.B.; Ayer, J.G.; Garden, F.L.; Garnett, S.P.; Harmer, J.A.; Leeder, S.R.; Toelle, B.G.; Webb, K.; Baur, L.A.; et al. Weight gain in infancy and vascular risk factors in later childhood. Pediatrics 2013, 131, e1821–e1828. [Google Scholar] [CrossRef] [PubMed]
- Janssen, I.; Katzmarzyk, P.T.; Srinivasan, S.R.; Chen, W.; Malina, R.M.; Bouchard, C.; Berenson, G.S. Utility of childhood BMI in the prediction of adulthood disease: Comparison of national and international references. Obes. Res. 2005, 13, 1106–1115. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, L.; Rinaldi, C.M. The prediction of preschool children’s weight from family environment factors: Gender-linked differences. Eat. Behav. 2010, 11, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Must, A.; Anderson, S.E. Body mass index in children and adolescents: considerations for population-based applications. Int. J. Obes. 2006, 30, 590–594. [Google Scholar] [CrossRef] [PubMed]
- Hughes, A.R.; Sherriff, A.; Lawlor, D.A.; Ness, A.R.; Reilly, J.J. Incidence of obesity during childhood and adolescence in a large contemporary cohort. Prev. Med. 2011, 52, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Natalie, S.; Suchindran, C.; North, K.E.; Popkin, B.M.; Gordon-Larsen, P. Association of adolescent obesity with risk of severe obesity in adulthood. JAMA 2010, 304, 2042–2047. [Google Scholar] [PubMed]
- Lavrador, M.S.; Abbes, P.T.; Escrivão, M.A.; Taddei, J.A. Cardiovascular risks in adolescents with different degrees of obesity. Arq. Bras. Cardiol. 2011, 96, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Weiss, R.; Dziura, J.; Burgert, T.S.; Tamborlane, W.V.; Taksali, S.E.; Yeckel, C.W.; Allen, K.; Lopes, M.; Savoye, M.; Morrison, J.; et al. Obesity and the metabolic syndrome in children and adolescents. N. Engl. J. Med. 2004, 350, 2362–2374. [Google Scholar] [CrossRef] [PubMed]
- Ruel, I.L.; Gaudet, D.; Perron, P.; Bergeron, J.; Julien, P.; Lamarche, B.; Québec LipD Study. Effect of obesity on HDL and LDL particle sizes in carriers of the null P207L or defective D9N mutation in the lipoprotein lipase gene: The Québec LipD Study. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Srinivasan, S.; Webber, L.; Wattigney, W.A.; Berenson, G.S. Association of fasting insulin level with serum lipid and lipoprotein levels in children, adolescents, and young adults: The Bogalusa Heart Study. Arch. Intern. Med. 1995, 155, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Okumura, M.J.; Davis, M.M.; Herman, W.H.; Gurney, J.G. Prevalence and determinants of insulin resistance among U.S. adolescents: A population-based study. Diabetes Care. 2006, 29, 2427–2432. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.E.; Seimon, R.V.; Keast, R.S.; Clifton, P.M.; Feinle-Bisset, C. Marked differences in gustatory and gastrointestinal sensitivity to oleic acid between lean and obese men. Am. J. Clin. Nutr. 2011, 93, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Chevrot, M.; Bernard, A.; Ancel, D.; Buttet, M.; Martin, C.; Abdoul-Azize, S.; Merlin, J.F.; Poirier, H.; Niot, I.; Khan, N.A.; et al. Obesity alters the gustatory perception of lipids in the mouse: Plausible involvement of lingual CD36. J. Lipid Res. 2013, 54, 2485–2494. [Google Scholar] [CrossRef] [PubMed]
- Temple, E.C.; Hutchinson, I.; Laing, D.G.; Jinks, A.L. Taste development: Differential growth rates of tongue regions in humans. Brain Res. Dev. Brain Res. 2002, 135, 65–70. [Google Scholar] [CrossRef]
- Laugerette, F.; Passilly-Degrace, P.; Patris, B.; Niot, I.; Febbraio, M.; Montmayeur, J.P.; Besnard, P. CD36 involvement in orosensory detection of dietary lipids, spontaneous fat preference, and digestive secretions. J. Clin. Invest. 2005, 115, 3177–3184. [Google Scholar] [CrossRef] [PubMed]
- Ichimura, A.; Hirasawa, A.; Poulain-Godefroy, O.; Bonnefond, A.; Hara, T.; Yengo, L.; Kimura, I.; Leloire, A.; Liu, N.; Iida, K.; et al. Dysfunction of lipid sensor GPR120 leads to obesity in both mouse and human. Nature 2012, 483, 350–354. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.E.; Keast, R.S. Recent fat intake modulates fat taste sensitivity in lean and overweight subjects. Int. J. Obes. 2012, 36, 834–842. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC‐BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daoudi, H.; Plesník, J.; Sayed, A.; Šerý, O.; Rouabah, A.; Rouabah, L.; Khan, N.A. Oral Fat Sensing and CD36 Gene Polymorphism in Algerian Lean and Obese Teenagers. Nutrients 2015, 7, 9096-9104. https://doi.org/10.3390/nu7115455
Daoudi H, Plesník J, Sayed A, Šerý O, Rouabah A, Rouabah L, Khan NA. Oral Fat Sensing and CD36 Gene Polymorphism in Algerian Lean and Obese Teenagers. Nutrients. 2015; 7(11):9096-9104. https://doi.org/10.3390/nu7115455
Chicago/Turabian StyleDaoudi, Hadjer, Jiří Plesník, Amira Sayed, Omar Šerý, Abdelkader Rouabah, Leila Rouabah, and Naim Akhtar Khan. 2015. "Oral Fat Sensing and CD36 Gene Polymorphism in Algerian Lean and Obese Teenagers" Nutrients 7, no. 11: 9096-9104. https://doi.org/10.3390/nu7115455
APA StyleDaoudi, H., Plesník, J., Sayed, A., Šerý, O., Rouabah, A., Rouabah, L., & Khan, N. A. (2015). Oral Fat Sensing and CD36 Gene Polymorphism in Algerian Lean and Obese Teenagers. Nutrients, 7(11), 9096-9104. https://doi.org/10.3390/nu7115455





