Long-Term Effect of Docosahexaenoic Acid Feeding on Lipid Composition and Brain Fatty Acid-Binding Protein Expression in Rats
Abstract
:1. Introduction
2. Experimental Section
2.1. Animals
| Fatty Acid | Control Diet (Cnt) | DHA Diet (ω-3) |
|---|---|---|
| g/100 g of total fatty acids | ||
| C14:0 | 0.1 ± 0.0 | 0.4 ± 0.0 |
| C16:0 | 6.7 ± 0.3 | 6.2 ± 0.1 |
| C16:1ω-7 | 0.2 ± 0.0 | 0.2 ± 0.1 |
| C18:0 | 38.8 ± 1.2 | 40.6 ± 0.2 |
| C18:1ω-9 | 29.0 ± 1.7 | 24.8 ± 0.3 |
| C18:2ω-6 (LA) | 21.2 ± 0.5 | 21.6 ± 0.0 |
| C20:0 | 0.9 ± 0.0 | 0.9 ± 0.0 |
| C18:3ω-3 (ALA) | 1.7 ± 0.1 | 3.3 ± 0.1 |
| C20:3ω-6 | 0.4 ± 0.1 | 0.4 ± 0.1 |
| C20:4ω-6 (AA) | 0.4 ± 0.0 | 0.4 ± 0.0 |
| C22:6ω-3 (DHA) | 0 | 0.9 ± 0.1 |
2.2. Brain Phospholipids Fatty Acid Composition
2.3. Western Blot Analysis
2.4. Statistical Analysis
3. Results
3.1. Effect of a DHA-Rich Diet on Dam and Pup Brain Fatty Acid Composition
3.1.1. Dam Brain Fatty Acid Composition: DHA-Rich Diet Does Not Alter AA and DHA Levels
3.1.2. Three-Week Old Pup Brain Fatty Acid Composition: DHA-Rich Diet Increases DHA and Decreases AA Content
| Fatty Acids | Cnt (n = 9) | ω-3 (n = 4) | p-Value * |
|---|---|---|---|
| % of total fatty acids | |||
| 14:0 | 0.22 ± 0.07 | 0.29 ± 0.16 | 0.63 |
| 15:0 | 0.45 ± 0.15 | 0.52 ± 0.29 | 0.82 |
| 16:0 | 22.2 ± 0.15 | 22.9 ± 0.17 * | 0.03 |
| 16:1ω-9 | 0.49 ± 0.03 | 0.51 ± 0.03 | 0.63 |
| 18:0 | 25.0 ± 0.22 | 25.1 ± 0.68 | 0.94 |
| 18:1ω-9 | 17.7 ± 0.25 | 17.5 ± 0.65 | 0.81 |
| 18:1c11 | 4.5 ± 0.38 | 4.4 ± 0.59 | 0.88 |
| 18:2ω-6 | 0.70 ± 0.01 | 0.77 ± 0.02 * | 0.004 |
| 20:0 | 0.37 ± 0.02 | 0.37 ± 0.04 | 0.94 |
| 18:3ω-3 | 1.1 ± 0.05 | 1.1 ± 0.07 | 0.74 |
| 20:2ω-6 | 0.09 ± 0.008 | 0.09 ± 0.003 | 0.64 |
| 20:3ω-6 | 0.81 ± 0.05 | 0.77 ± 0.06 | 0.62 |
| 20:4ω-6 | 8.9 ± 0.25 | 8.3 ± 0.11 | 0.12 |
| 24:0 | 1.2 ± 0.07 | 1.2 ± 0.15 | 0.86 |
| 24:1ω-9 | 2.7 ± 0.12 | 2.5 ± 0.06 | 0.14 |
| 22:4ω-6 | 0.53 ± 0.04 | 0.24 ± 0.09 * | 0.004 |
| 22:5ω-6 | 0.05 ± 0.008 | 0.06 ± 0.01 | 0.32 |
| 20:5ω-3 | 0.73 ± 0.05 | 0.73 ± 0.1 | 0.99 |
| 22:5ω-3 | 0.12 ± 0.01 | 0.16 ± 0.01 | 0.07 |
| 22:6ω-3 | 12.0 ± 0.28 | 12.6 ± 0.30 | 0.19 |
| Total MUFA | 25.3 ± 0.32 | 24.8 ± 0.07 | 0.16 |
| Total SFA | 49.5 ± 0.33 | 50.3 ± 0.44 | 0.18 |
| ω-3 PUFA | 13.9 ± 0.27 | 14.6 ± 0.36 | 0.19 |
| ω-6 PUFA | 11.1 ± 0.27 | 10.2 ± 0.13 | 0.05 |
| Total PUFA | 25.1 ± 0.34 | 24.8 ± 0.35 | 0.65 |
| 18:2ω-6:20:4ω-6 | 0.08 ± 0.003 | 0.09 ± 0.003 * | 0.02 |
| ω-6:ω-3 PUFA | 0.8 ± 0.03 | 0.7 ± 0.02 * | 0.046 |
| Fatty Acids | Cnt (n = 15) | ω-3 (n = 9) | * p-Value |
|---|---|---|---|
| % of total fatty acids | |||
| 14:0 | 0.42 ± 0.01 | 0.37 ± 0.02 | 0.05 |
| 15:0 | 0.77 ± 0.09 | 0.61 ± 0.12 | 0.29 |
| 16:0 | 27.5 ± 0.23 | 26.1 ± 0.69 | 0.08 |
| 16:1ω-9 | 1.1 ± 0.03 | 1.0 ± 0.04 | 0.20 |
| 18:0 | 23.7 ± 0.16 | 23.7 ± 0.3 | 0.99 |
| 18:1ω-9 | 13.3 ± 0.17 | 14.4 ± 0.79 | 0.22 |
| 18:1c11 | 2.7 ± 0.09 | 2.9 ± 0.20 | 0.16 |
| 18:2ω-6 | 1.1 ± 0.03 | 1.2 ± 0.04 | 0.07 |
| 20:0 | 0.25 ± 0.02 | 0.36 ± 0.08 | 0.23 |
| 18:3ω-3 | 0.38 ± 0.03 | 0.58 ± 0.15 | 0.21 |
| 20:2ω-6 | 0.18 ± 0.008 | 0.20 ± 0.02 | 0.19 |
| 20:3ω-6 | 0.70 ± 0.03 | 0.99 ± 0.14 | 0.07 |
| 20:4ω-6 | 11.6 ± 0.16 | 10.6 ± 0.43 * | 0.04 |
| 24:0 | 0.31 ± 0.04 | 0.53 ± 0.16 | 0.19 |
| 24:1ω-9 | 3.2 ± 0.04 | 2.8 ± 0.04 * | <0.0001 |
| 22:4ω-6 | 1.4 ± 0.03 | 0.49 ± 0.02 * | <0.0001 |
| 22:5ω-6 | 0.03 ± 0.003 | 0.05 ± 0.01 | 0.14 |
| 20:5ω-3 | 0.39 ± 0.04 | 0.52 ± 0.14 | 0.42 |
| 22:5ω-3 | 0.14 ± 0.006 | 0.14 ± 0.01 | 0.88 |
| 22:6ω-3 | 10.8 ± 0.23 | 12.3 ± 0.43 * | 0.003 |
| Total MUFA | 20.3 ± 0.23 | 21.2 ± 0.98 | 0.40 |
| Total SFA | 52.9 ± 0.31 | 51.6 ± 0.78 | 0.14 |
| ω-3 PUFA | 11.7 ± 0.21 | 13.5 ± 0.27 * | <0.0001 |
| ω-6 PUFA | 14.9 ± 0.13 | 13.5 ± 0.28 * | <0.0001 |
| Total PUFA | 26.7 ± 0.31 | 27.0 ± 0.44 | 0.56 |
| ω-6 PUFA:ω-3 PUFA | 1.3 ± 0.02 | 0.99 ± 0.02 * | <0.0001 |
| 20:4ω-6:22:6ω-3 | 1.1 ± 0.02 | 0.86 ± 0.02 * | <0.0001 |
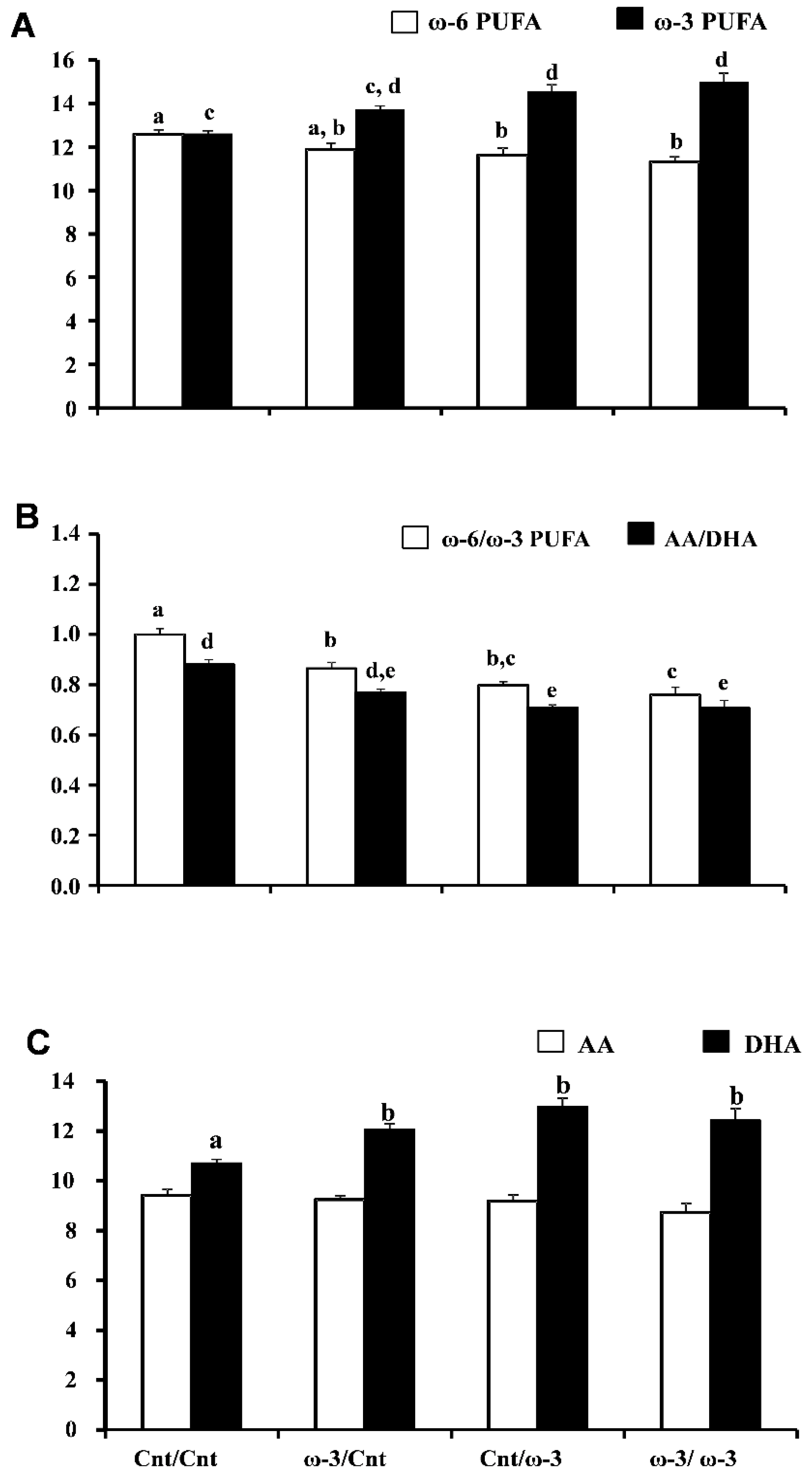
3.1.3. Six-Week Old Pup Brain Fatty Acid Composition: DHA-Rich Diet Maintains Elevated DHA Content after Pups Are Transferred to a Cnt Diet
| Fatty Acids | Treatment Groups (Dam diet/Pup Diet) | |||
|---|---|---|---|---|
| Cnt/Cnt (n = 10) | ω-3/Cnt (n = 7) | Cnt/ω-3 (n = 8) | ω-3/ω-3 (n = 8) | |
| % of Total Fatty Acids | ||||
| 14:0 | 0.17 ± 0.006 | 0.17 ± 0.004 | 0.17 ± 0.004 | 0.16 ± 0.005 |
| 15:0 | 0.61 ± 0.21 | 0.77 ± 0.27 | 0.74 ± 0.22 | 1.1 ± 0.17 |
| 16:0 | 22.9 ± 0.28 | 24.5 ± 1.1 | 23.6 ± 0.34 | 21.8 ± 0.72 |
| 16:1ω-9 | 0.48 ± 0.01 | 0.55 ± 0.03 | 0.50 ± 0.02 | 0.48 ± 0.02 |
| 18:0 | 25.2 ± 0.46 | 24.5 ± 0.89 | 25.4 ± 0.49 | 23.7 ± 0.62 |
| 18:1ω-9 | 16.5 ± 0.39 | 16.4 ± 0.74 | 16.0 ± 0.21 | 18.3 ± 0.86 |
| 18:1c11 | 4.0 ± 0.31 | 3.2 ± 0.32 | 3.3 ± 0.34 | 3.1 ± 0.21 |
| 18:2ω-6 | 0.78 ± 0.04 a | 0.80 ± 0.03 a | 0.82 ± 0.03 a,b | 0.94 ± 0.06 b |
| 20:0 | 0.48 ± 0.05 a,b | 0.45 ± 0.06 a,b | 0.38 ± 0.01 a | 0.58 ± 0.09 b |
| 18:3ω-3 | 0.99 ± 0.12 | 0.78 ± 0.11 | 0.78 ± 0.05 | 1.4 ± 0.28 |
| 20:2ω-6 | 0.16 ± 0.02 | 0.13 ± 0.008 | 0.15 ± 0.009 | 0.19 ± 0.03 |
| 20:3ω-6 | 0.87 ± 0.07 a,b | 0.83 ± 0.11 a,b | 0.77 ± 0.02 a | 1.1 ± 0.09 b |
| 20:4ω-6 | 9.4 ± 0.23 | 9.3 ± 0.15 | 9.2 ± 0.25 | 8.7 ± 0.35 |
| 24:0 | 1.1 ± 0.11 | 0.95 ± 0.19 | 0.79 ± 0.03 | 1.3 ± 0.27 |
| 24:1ω-9 | 3.1 ± 0.06 a | 2.7 ± 0.04 b | 2.7 ± 0.1 b | 2.7 ± 0.08 b |
| 22:4ω-6 | 1.3 ± 0.08 a | 0.81 ± 0.32 a,b | 0.68 ± 0.08 a,b | 0.28 ± 0.03 b |
| 22:5ω-6 | 0.03 ± 0.003 | 0.04 ± 0.005 | 0.03 ± 0.001 | 0.05 ± 0.01 |
| 22:5ω-3 | 0.11 ± 0.007 a | 0.13 ± 0.009 a,b | 0.15 ± 0.02 a,b | 0.16 ± 0.009 b |
| 22:6ω-3 | 10.7 ± 0.15 a | 12.1 ± 0.25 b | 13.0 ± 0.35 b | 12.4 ± 0.45 b |
| Total MUFA | 24.1 ± 0.37 a,c | 22.9 ± 0.56 a,b | 22.4 ± 0.44 b | 24.7 ± 0.75 c |
| Total SFA | 50.4 ± 0.36 | 51.4 ± 0.42 | 51.1 ± 0.58 | 48.7 ± 0.90 |
| ω-3 PUFA | 12.6 ± 0.13 a | 13.7 ± 0.14 a,b | 14.6 ± 0.29 b | 15.0 ± 0.40 b |
| ω-6 PUFA | 12.6 ± 0.21 a | 11.9 ± 0.31 a,b | 11.6 ± 0.32 b | 11.3 ± 0.23 b |
| Total PUFA | 25.2 ± 0.19 | 25.6 ± 0.34 | 26.2 ± 0.58 | 26.3 ± 0.34 |
| 18:2ω-6:20:4ω-6 | 0.08 ± 0.006 a | 0.09 ± 0.005 a,b | 0.09 ± 0.004 a,b | 0.11 ± 0.01 b |
| ω-6 PUFA:ω-3 PUFA | 0.99 ± 0.02 a | 0.87 ± 0.02 b | 0.79 ± 0.01 b,c | 0.76 ± 0.03 c |
| 20:4ω-6:22:6ω-3 | 0.88 ± 0.02 a | 0.77 ± 0.01 a,b | 0.71 ± 0.01 b | 0.71 ± 0.03 b |
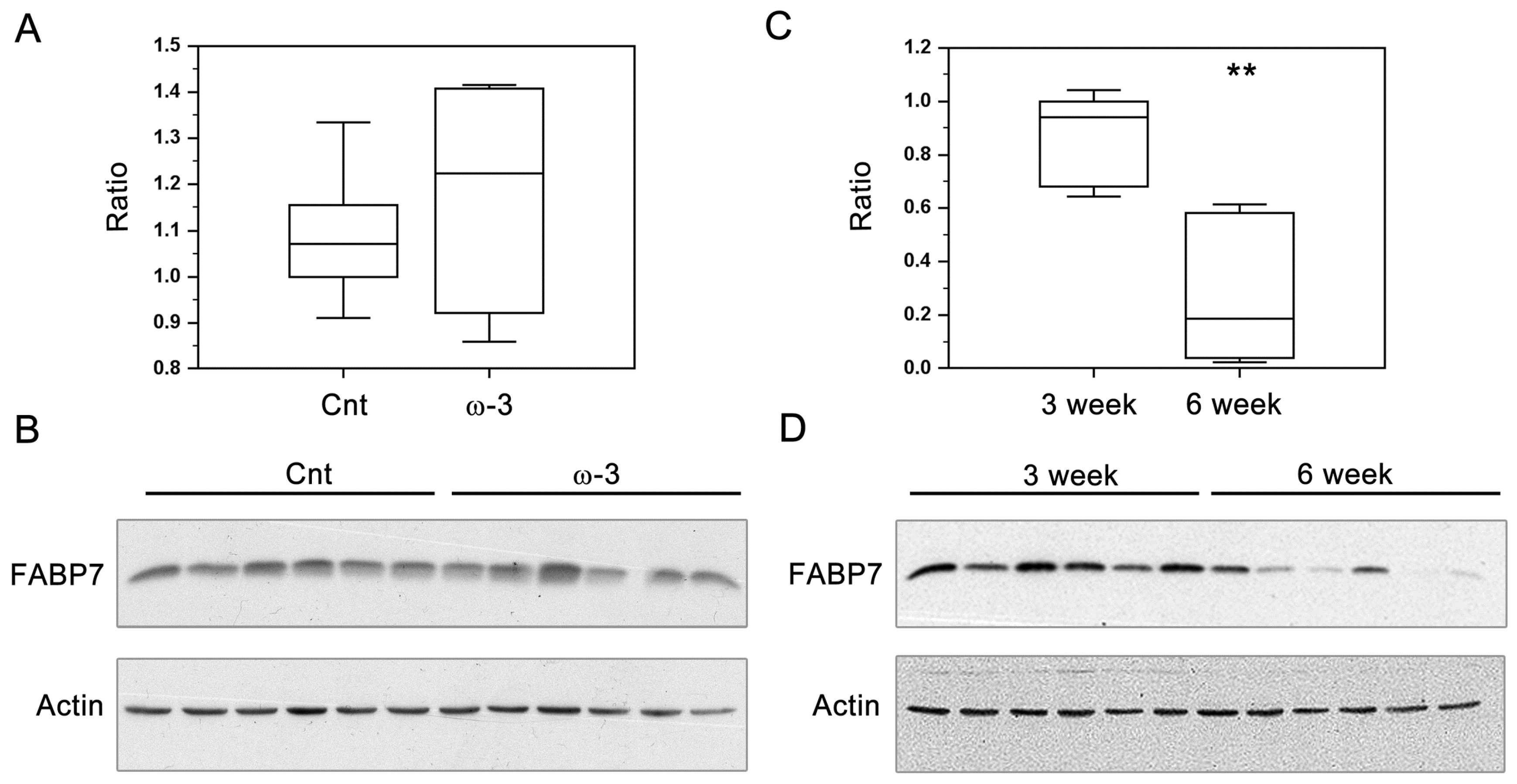
3.2. FABP7 Expression in Three-Week and Six-Week Pups

4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- De Graaf-Peters, V.B.; Hadders-Algra, M. Ontogeny of the human central nervous system: What is happening when? Early Hum. Dev. 2006, 82, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Tau, G.Z.; Peterson, B.S. Normal development of brain circuits. Neuropsychopharmacology 2010, 35, 147–168. [Google Scholar] [CrossRef] [PubMed]
- Dekaban, A.S. Changes in brain weights during the span of human life: Relation of brain weights to body heights and body weights. Ann. Neurol. 1978, 4, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Clandinin, M.T. Brain development and assessing the supply of polyunsaturated fatty acid. Lipids 1999, 34, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Clandinin, M.T.; Chappell, J.E.; Leong, S.; Heim, T.; Swyer, P.R.; Chance, G.W. Intrauterine fatty acid accretion rates in human brain: Implications for fatty acid requirements. Early Hum. Dev. 1980, 4, 121–129. [Google Scholar] [CrossRef]
- Martinez, M.; Mougan, I. Fatty acid composition of human brain phospholipids during normal development. J. Neurochem. 1998, 71, 2528–2533. [Google Scholar] [CrossRef] [PubMed]
- Semple, B.D.; Blomgren, K.; Gimlin, K.; Ferriero, D.M.; Noble-Haeusslein, L.J. Brain development in rodents and humans: Identifying benchmarks of maturation and vulnerability to injury across species. Prog. Neurobiol. 2013, 106–107, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Green, P.; Yavin, E. Fatty acid composition of late embryonic and early postnatal rat brain. Lipids 1996, 31, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Purvis, J.M.; Clandinin, M.T.; Hacker, R.R. Fatty acid accretion during perinatal brain growth in the pig. A model for fatty acid accretion in human brain. Comp. Biochem. Physiol. B 1982, 72, 195–199. [Google Scholar] [CrossRef]
- Xiang, M.; Alfven, G.; Blennow, M.; Trygg, M.; Zetterstrom, R. Long-chain polyunsaturated fatty acids in human milk and brain growth during early infancy. Acta Paediatr. 2000, 89, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Ruan, C.; Liu, X.; Man, H.; Ma, X.; Lu, G.; Duan, G.; DeFrancesco, C.A.; Connor, W.E. Milk composition in women from five different regions of China: The great diversity of milk fatty acids. J. Nutr. 1995, 125, 2993–2998. [Google Scholar] [PubMed]
- Pedersen, L.; Lauritzen, L.; Brasholt, M.; Buhl, T.; Bisgaard, H. Polyunsaturated fatty acid content of mother's milk is associated with childhood body composition. Pediatr. Res. 2012, 72, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Sherry, C.L.; Oliver, J.S.; Marriage, B.J. Docosahexaenoic acid supplementation in lactating women increases breast milk and plasma docosahexaenoic acid concentrations and alters infant omega 6:3 fatty acid ratio. Prostaglandins Leukot. Essent. Fatty Acids 2015, 95, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Brenna, J.T.; Varamini, B.; Jensen, R.G.; Diersen-Schade, D.A.; Boettcher, J.A.; Arterburn, L.M. Docosahexaenoic and arachidonic acid concentrations in human breast milk worldwide. Am. J. Clin. Nutr. 2007, 85, 1457–1464. [Google Scholar] [PubMed]
- Neuringer, M.; Connor, W.E.; Lin, D.S.; Barstad, L.; Luck, S. Biochemical and functional effects of prenatal and postnatal omega 3 fatty acid deficiency on retina and brain in rhesus monkeys. Proc. Natl. Acad. Sci. USA 1986, 83, 4021–4025. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.Z.; Mita, R.; Beaulieu, M.; Gao, Z.; Godbout, R. Fatty acid binding proteins in brain development and disease. Int. J. Dev. Biol. 2010, 54, 1229–1239. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.Z.; Li, X.; Godbout, R. A novel fatty acid-binding protein (FABP) gene resulting from tandem gene duplication in mammals: Transcription in rat retina and testis. Genomics 2008, 92, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Balendiran, G.K.; Schnutgen, F.; Scapin, G.; Borchers, T.; Xhong, N.; Lim, K.; Godbout, R.; Spener, F.; Sacchettini, J.C. Crystal structure and thermodynamic analysis of human brain fatty acid-binding protein. J. Biol. Chem. 2000, 275, 27045–27054. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Hatten, M.E.; Heintz, N. Brain lipid-binding protein (BLBP): A novel signaling system in the developing mammalian CNS. Neuron 1994, 12, 895–908. [Google Scholar] [CrossRef]
- Mita, R.; Beaulieu, M.J.; Field, C.; Godbout, R. Brain fatty acid-binding protein and omega-3/omega-6 fatty acids: Mechanistic insight into malignant glioma cell migration. J. Biol. Chem. 2010, 285, 37005–37015. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Bollen, A.W.; Aldape, K.D.; Gupta, N. Nuclear FABP7 immunoreactivity is preferentially expressed in infiltrative glioma and is associated with poor prognosis in EGFR-overexpressing glioblastoma. BMC Cancer 2006. [Google Scholar] [CrossRef] [PubMed]
- Mita, R.; Coles, J.E.; Glubrecht, D.D.; Sung, R.; Sun, X.; Godbout, R. B-FABP-expressing radial glial cells: The malignant glioma cell of origin? Neoplasia 2007, 9, 734–744. [Google Scholar] [CrossRef] [PubMed]
- Elsherbiny, M.E.; Emara, M.; Godbout, R. Interaction of brain fatty acid-binding protein with the polyunsaturated fatty acid environment as a potential determinant of poor prognosis in malignant glioma. Prog. Lipid Res. 2013, 52, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Maximin, E.; Langelier, B.; Aioun, J.; Al-Gubory, K.H.; Bordat, C.; Lavialle, M.; Heberden, C. Fatty acid binding protein 7 and n-3 poly unsaturated fatty acid supply in early rat brain development. Dev. Neurobiol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Ozias, M.K.; Carlson, S.E.; Levant, B. Maternal parity and diet (n-3) polyunsaturated fatty acid concentration influence accretion of brain phospholipid docosahexaenoic acid in developing rats. J. Nutr. 2007, 137, 125–129. [Google Scholar] [PubMed]
- Anderson, G.J. Developmental sensitivity of the brain to dietary n-3 fatty acids. J. Lipid Res. 1994, 35, 105–111. [Google Scholar] [PubMed]
- Robinson, L.E.; Field, C.J. Dietary long-chain (n-3) fatty acids facilitate immune cell activation in sedentary, but not exercise-trained rats. J. Nutr. 1998, 128, 498–504. [Google Scholar] [PubMed]
- Lien, E.L.; Boyle, F.G.; Yuhas, R.J.; Kuhlman, C.F. Effect of maternal dietary arachidonic or linoleic acid on rat pup fatty acid profiles. Lipids 1994, 29, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Layne, K.S.; Goh, Y.K.; Jumpsen, J.A.; Ryan, E.A.; Chow, P.; Clandinin, M.T. Normal subjects consuming physiological levels of 18:3 (n-3) and 20:5 (n-3) from flaxseed or fish oils have characteristic differences in plasma lipid and lipoprotein fatty acid levels. J. Nutr. 1996, 126, 2130–2140. [Google Scholar] [PubMed]
- Godbout, R.; Bisgrove, D.A.; Shkolny, D.; Day, R.S., 3rd. Correlation of B-FABP and GFAP expression in malignant glioma. Oncogene 1998, 16, 1955–1962. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, V.K.; Huang, B.X.; Kim, H.Y. Effects of docosahexaenoic acid on mouse brain synaptic plasma membrane proteome analyzed by mass spectrometry and (16)O/(18)O labeling. J. Proteome Res. 2011, 10, 5472–5480. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, P. The laboratory rat: Relating its age with human's. Int. J. Prev. Med. 2013, 4, 624–630. [Google Scholar] [PubMed]
- Arai, Y.; Funatsu, N.; Numayama-Tsuruta, K.; Nomura, T.; Nakamura, S.; Osumi, N. Role of FABP7, a downstream gene of PAX6, in the maintenance of neuroepithelial cells during early embryonic development of the rat cortex. J. Neurosci. 2005, 25, 9752–9761. [Google Scholar] [CrossRef] [PubMed]
- Matsumata, M.; Sakayori, N.; Maekawa, M.; Owada, Y.; Yoshikawa, T.; Osumi, N. The effects of FABP7 and FABP5 on postnatal hippocampal neurogenesis in the mouse. Stem Cells 2012, 30, 1532–1543. [Google Scholar] [CrossRef] [PubMed]
- Priego, T.; Sanchez, J.; Garcia, A.P.; Palou, A.; Pico, C. Maternal dietary fat affects milk fatty acid profile and impacts on weight gain and thermogenic capacity of suckling rats. Lipids 2013, 48, 481–495. [Google Scholar] [CrossRef] [PubMed]
- Bourre, J.M.; Dumont, O.S.; Piciotti, M.J.; Pascal, G.A.; Durand, G.A. Dietary alpha-linolenic acid deficiency in adult rats for 7 months does not alter brain docosahexaenoic acid content, in contrast to liver, heart and testes. Biochim. Biophys. Acta 1992, 1124, 119–122. [Google Scholar] [CrossRef]
- Lin, Y.H.; Shah, S.; Salem, N., Jr. Altered essential fatty acid metabolism and composition in rat liver, plasma, heart and brain after microalgal DHA addition to the diet. J. Nutr. Biochem. 2010, 22, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.A.; Muhlhausler, B.; Makrides, M. Conversion of linoleic acid and alpha-linolenic acid to long-chain polyunsaturated fatty acids (LCPUFAs), with a focus on pregnancy, lactation and the first 2 years of life. Matern. Child Nutr. 2011, 7, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Wainwright, P.E.; Huang, Y.S.; Simmons, V.; Mills, D.E.; Ward, R.P.; Ward, G.R.; Winfield, D.; McCutcheon, D. Effects of prenatal ethanol and long-chain n-3 fatty acid supplementation on development in mice. 2. Fatty acid composition of brain membrane phospholipids. Alcohol. Clin. Exp. Res. 1990, 14, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Wijendran, V.; Lawrence, P.; Diau, G.Y.; Boehm, G.; Nathanielsz, P.W.; Brenna, J.T. Significant utilization of dietary arachidonic acid is for brain adrenic acid in baboon neonates. J. Lipid Res. 2002, 43, 762–767. [Google Scholar] [PubMed]
- Anderson, G.J.; Connor, W.E.; Corliss, J.D. Docosahexaenoic acid is the preferred dietary n-3 fatty acid for the development of the brain and retina. Pediatr. Res. 1990, 27, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Cunnane, S.C.; Francescutti, V.; Brenna, J.T.; Crawford, M.A. Breast-fed infants achieve a higher rate of brain and whole body docosahexaenoate accumulation than formula-fed infants not consuming dietary docosahexaenoate. Lipids 2000, 35, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Garg, M.L.; Sebokova, E.; Thomson, A.B.; Clandinin, M.T. Delta 6-desaturase activity in liver microsomes of rats fed diets enriched with cholesterol and/or omega 3 fatty acids. Biochem. J. 1988, 249, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Garg, M.L.; Thomson, A.B.; Clandinin, M.T. Effect of dietary cholesterol and/or omega 3 fatty acids on lipid composition and delta 5-desaturase activity of rat liver microsomes. J. Nutr. 1988, 118, 661–668. [Google Scholar] [PubMed]
- Njoroge, S.W.; Laposata, M.; Katrangi, W.; Seegmiller, A.C. DHA and EPA reverse cystic fibrosis-related FA abnormalities by suppressing FA desaturase expression and activity. J. Lipid Res. 2012, 53, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Wadhwani, N.S.; Manglekar, R.R.; Dangat, K.D.; Kulkarni, A.V.; Joshi, S.R. Effect of maternal micronutrients (folic acid, vitamin B12) and omega 3 fatty acids on liver fatty acid desaturases and transport proteins in wistar rats. Prostaglandins Leukot. Essent. Fatty Acids 2012, 86, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.K.; Holman, R.T.; Lubozynski, M.F.; Dyck, P.J. Changes in fatty acid composition of peripheral nerve myelin in essential fatty acid deficiency. Arch. Biochem. Biophys. 1980, 204, 175–180. [Google Scholar] [CrossRef]
- O'Brien, J.S.; Sampson, E.L. Fatty acid and fatty aldehyde composition of the major brain lipids in normal human gray matter, white matter, and myelin. J. Lipid Res. 1965, 6, 545–551. [Google Scholar] [PubMed]
- Sargent, J.R.; Coupland, K.; Wilson, R. Nervonic acid and demyelinating disease. Med. Hypotheses. 1994, 42, 237–242. [Google Scholar] [CrossRef]
- Haubner, L.; Sullivan, J.; Ashmeade, T.; Saste, M.; Wiener, D.; Carver, J. The effects of maternal dietary docosahexaenoic acid intake on rat pup myelin and the auditory startle response. Dev. Neurosci. 2007, 29, 460–467. [Google Scholar] [CrossRef] [PubMed]
- Delion, S.; Chalon, S.; Herault, J.; Guilloteau, D.; Besnard, J.C.; Durand, G. Chronic dietary alpha-linolenic acid deficiency alters dopaminergic and serotoninergic neurotransmission in rats. J. Nutr. 1994, 124, 2466–2476. [Google Scholar] [PubMed]
- Carrie, I.; Clement, M.; de Javel, D.; Frances, H.; Bourre, J.M. Specific phospholipid fatty acid composition of brain regions in mice. Effects of n-3 polyunsaturated fatty acid deficiency and phospholipid supplementation. J. Lipid Res. 2000, 41, 465–472. [Google Scholar] [PubMed]
- Jumpsen, J.; Lien, E.L.; Goh, Y.K.; Clandinin, M.T. Small changes of dietary (n-6) and (n-3)/fatty acid content ration alter phosphatidylethanolamine and phosphatidylcholine fatty acid composition during development of neuronal and glial cells in rats. J. Nutr. 1997, 127, 724–731. [Google Scholar] [PubMed]
- Martin, D.D.; Robbins, M.E.; Spector, A.A.; Wen, B.C.; Hussey, D.H. The fatty acid composition of human gliomas differs from that found in nonmalignant brain tissue. Lipids 1996, 31, 1283–1288. [Google Scholar] [CrossRef] [PubMed]
- Kaloshi, G.; Mokhtari, K.; Carpentier, C.; Taillibert, S.; Lejeune, J.; Marie, Y.; Delattre, J.Y.; Godbout, R.; Sanson, M. FABP7 expression in glioblastomas: Relation to prognosis, invasion and EGFR status. J. Neurooncol. 2007, 84, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Anthony, T.E.; Klein, C.; Fishell, G.; Heintz, N. Radial glia serve as neuronal progenitors in all regions of the central nervous system. Neuron 2004, 41, 881–890. [Google Scholar] [PubMed]
- Kurtz, A.; Zimmer, A.; Schnutgen, F.; Bruning, G.; Spener, F.; Muller, T. The expression pattern of a novel gene encoding brain-fatty acid binding protein correlates with neuronal and glial cell development. Development 1994, 120, 2637–2649. [Google Scholar] [PubMed]
- Xu, L.Z.; Sanchez, R.; Sali, A.; Heintz, N. Ligand specificity of brain lipid-binding protein. J. Biol. Chem. 1996, 271, 24711–24719. [Google Scholar] [PubMed]
- Pelerin, H.; Jouin, M.; Lallemand, M.S.; Alessandri, J.M.; Cunnane, S.C.; Langelier, B.; Guesnet, P. Gene expression of fatty acid transport and binding proteins in the blood-brain barrier and the cerebral cortex of the rat: Differences across development and with different DHA brain status. Prostaglandins. Leukot. Essent. Fatty Acids 2014, 91, 213–220. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elsherbiny, M.E.; Goruk, S.; Monckton, E.A.; Richard, C.; Brun, M.; Emara, M.; Field, C.J.; Godbout, R. Long-Term Effect of Docosahexaenoic Acid Feeding on Lipid Composition and Brain Fatty Acid-Binding Protein Expression in Rats. Nutrients 2015, 7, 8802-8817. https://doi.org/10.3390/nu7105433
Elsherbiny ME, Goruk S, Monckton EA, Richard C, Brun M, Emara M, Field CJ, Godbout R. Long-Term Effect of Docosahexaenoic Acid Feeding on Lipid Composition and Brain Fatty Acid-Binding Protein Expression in Rats. Nutrients. 2015; 7(10):8802-8817. https://doi.org/10.3390/nu7105433
Chicago/Turabian StyleElsherbiny, Marwa E., Susan Goruk, Elizabeth A. Monckton, Caroline Richard, Miranda Brun, Marwan Emara, Catherine J. Field, and Roseline Godbout. 2015. "Long-Term Effect of Docosahexaenoic Acid Feeding on Lipid Composition and Brain Fatty Acid-Binding Protein Expression in Rats" Nutrients 7, no. 10: 8802-8817. https://doi.org/10.3390/nu7105433
APA StyleElsherbiny, M. E., Goruk, S., Monckton, E. A., Richard, C., Brun, M., Emara, M., Field, C. J., & Godbout, R. (2015). Long-Term Effect of Docosahexaenoic Acid Feeding on Lipid Composition and Brain Fatty Acid-Binding Protein Expression in Rats. Nutrients, 7(10), 8802-8817. https://doi.org/10.3390/nu7105433





