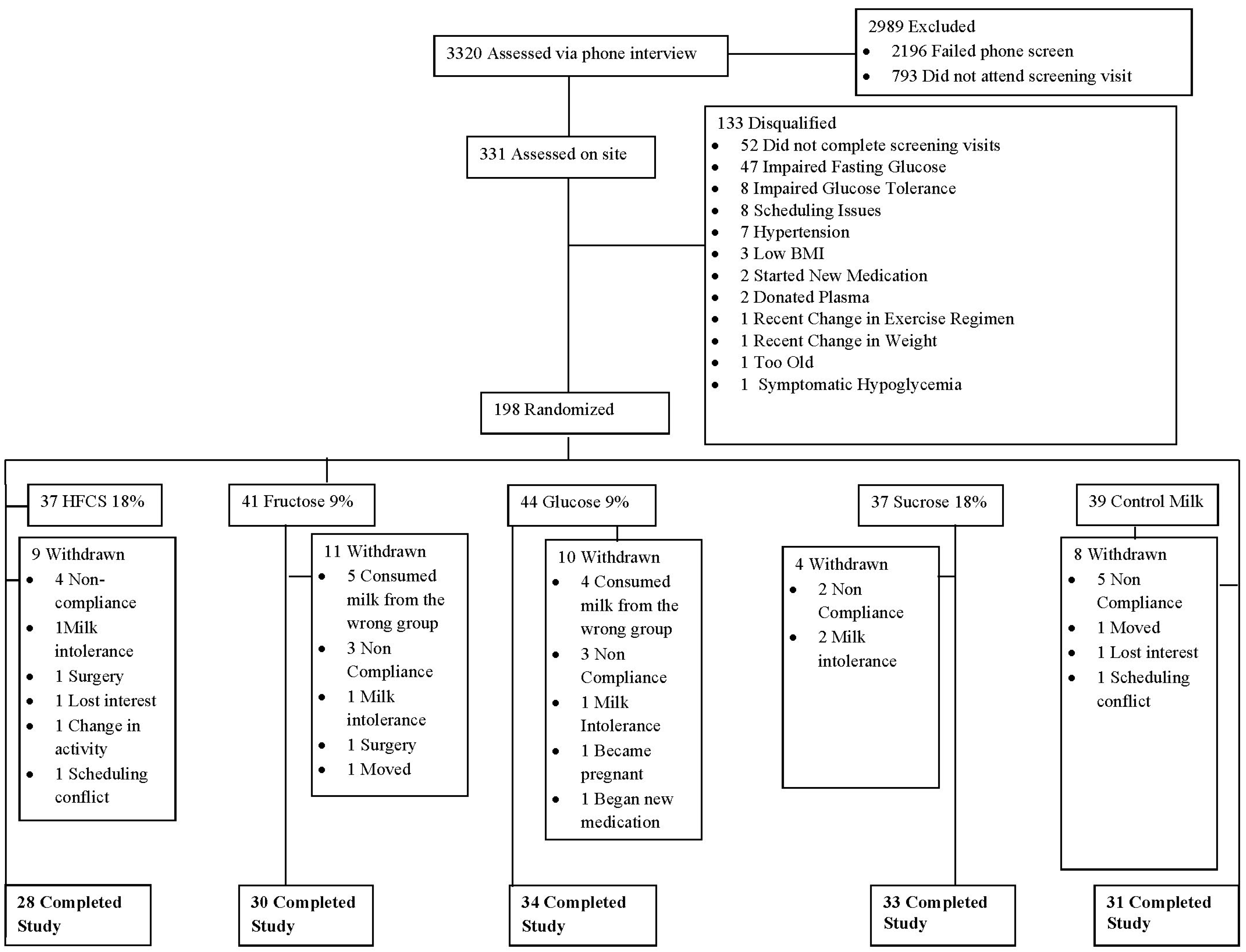
No Effect of Added Sugar Consumed at Median American Intake Level on Glucose Tolerance or Insulin Resistance
Abstract
:1. Introduction
2. Methods
2.1. Overview
| Group | Nutritional Interventions |
|---|---|
| Group 1 | HFCS sweetened milk with HFCS contributing 18% of estimated weight-maintenance energy intake |
| Group 2 | Fructose sweetened milk with fructose contributing 9% of estimated weight-maintenance energy intake |
| Group 3 | Glucose sweetened milk with glucose contributing 9% of estimated weight-maintenance energy intake |
| Group 4 | Sucrose sweetened milk with sucrose contributing 18% of estimated weight-maintenance energy intake |
| Group 5 | Total calories in unsweetened milk contributes 9% of estimated weight-maintenance energy intake |
2.2. Study Population
2.3. Intervention
2.4. Oral Glucose Tolerance Test and Blood Plasma Measurements
2.5. Derived Measurements
2.6. Statistical Analyses
3. Results
3.1. Demographic Information
3.2. Dietary Intake
| HFCS 18% n = 28 | Fructose 9% n = 30 | Glucose 9% n = 34 | Sucrose 18% n = 33 | Control n = 31 | All | p | |
|---|---|---|---|---|---|---|---|
| Age (years) | 36.5 ± 11.3 | 35.6 ± 10.4 | 37.0 ± 11.7 | 34.1 ± 11.0 | 35.3 ± 12.5 | 35.7 ± 11.4 | 0.864 |
| Gender | M = 11, F = 17 | M = 16, F = 14 | M = 17, F = 17 | M = 15, F = 18 | M = 10, F = 21 | M = 69, F = 87 | |
| Weight (kg) | 73.3 ± 13.2 | 74.3 ± 13.1 | 76.2 = 12.2 | 72.1 ±14.1 | 72.3 ± 12.2 | 73.7 ± 12.9 | 0.696 |
| BMI | 26.5 ± 3.5 | 26.0 ± 3.8 | 26.5 ± 3.3 | 25.6 ± 3.5 | 25.6 ± 3.6 | 26.0 ± 3.5 | 0.740 |
| Waist Circumference (cm) | 81.8 ± 10.7 | 81.3 ± 10.6 | 82.4 ± 10.1 | 80.8 ± 10.1 | 79.1 ± 8.8 | 81.1 ± 10.0 | 0.757 |
| Systolic Blood Pressure (mmHg) | 107.3 ± 11.1 | 105.8 ± 10.6 | 105.8 ± 8.7 | 107.2 ± 9.5 | 107.3 ± 9.0 | 106.7 ± 9.7 | 0.931 |
| Diastolic Blood Pressure (mmHg) | 68.5 ± 8.5 | 68.7 ± 7.2 | 68.1 ± 8.2 | 68.8 ± 7.7 | 69.6 ± 8.1 | 68.7 ± 7.9 | 0.957 |
| Cholesterol (mmol/L) | 4.9 ± 0.7 | 4.4 ± 0.8 | 5.0 ± 1.0 | 4.5 ± 0.9 | 4.4 ± 0.9 | 4.6 ± 0.9 | 0.022 |
| Triglycerides (mmol/L) | 1.3 ± 0.8 | 1.1 ± 0.7 | 1.4 ± 1.4 | 1.1 ± 0.6 | 0.8 ± 0.4 | 1.1 ± 0.9 | 0.121 |
| HDL (mmol/L) | 1.4 ± 0.4 | 1.3 ± 0.4 | 1.4 ± 0.4 | 1.4 ± 0.3 | 1.4 ± 0.5 | 1.4 ± 0.4 | 0.452 |
| LDL (mmol/L) | 2.9 ± 0.7 | 2.6 ± 0.6 | 2.9 ± 0.9 | 2.6 ± 0.8 | 2.6 ± 0.7 | 2.7 ± 0.8 | 0.160 |
| Glucose (mmol/L) | 5.1 ± 0.4 | 4.9 ± 0.4 | 5.0 ± 0.4 | 4.8 ± 0.4 | 5.0 ± 0.4 | 4.9 ± 0.4 | 0.058 |
| Insulin (pmol/L) | 67.4 ± 38.9 | 55.6 ± 31.9 | 55.6 ± 52.1 | 54.9 ± 33.3 | 54.2 ± 35.4 | 57.6 ± 38.9 | 0.687 |
| All | Time | HFCS 18% | Fructose 9% | Glucose 9% | Sucrose 18% | Control Milk | Interaction | ||
|---|---|---|---|---|---|---|---|---|---|
| Energy Intake (kcal) | Pre | 1994.5 ± 692.6 | <0.001 | 1897.4 ± 689.2 | 1988.1 ± 659.9 | 2031.7 ± 644.1 | 2050.6 ± 819.5 | 1979.9 ± 669.1 | 0.136 |
| Post | 2296.0 ± 669.0 | 2384.0 ± 598.4 | 2198.6 ± 691.7 | 2189.0 ± 524.1 | 2492.0 ± 835.3 | 2229.1 ± 637.3 | |||
| Fat (g) | Pre | 74.5 ± 30.5 | 0.837 | 70.4 ± 28.1 | 74.3 ± 23.4 | 75.2 ± 31.6 | 78.1 ± 39.2 | 73.4 ± 28.1 | 0.482 |
| Post | 73.8 ± 27.1 | 69.4 ± 21.2 | 71.8 ± 26.3 | 67.7 ± 21.4 | 80.8 ± 37.3 | 78.9 ± 24.2 | |||
| Carbohydrate (g) | Pre | 247.3 ± 92.6 | <0.001 | 232.0 ± 99.3 | 250.7 ± 100.8 | 256.3 ± 79.8 | 249.1 ± 93.6 | 244.6 ± 95.9 | 0.002 |
| Post | 305.2 ± 92.7 | 337.4 ± 93.1 *** | 286.9 ± 81.7 ¥ | 297.3 ± 74.8 *,¥ | 333.4 ± 106.2 *** | 275.0 ± 95.0 ¥ | |||
| Protein (g) | Pre | 88.7 ± 41.1 | <0.001 | 83.6 ± 35.3 | 86.0 ± 41.3 | 88.4 ± 33.5 | 93.9 ± 50.4 | 90.4 ± 44.3 | 0.834 |
| Post | 109.2 ± 43.0 | 108.4 ± 36.8 | 108.7 ± 59.9 | 103.8 ± 30.9 | 114.4 ± 47.8 | 110.9 ± 36.2 | |||
| Total Sugar (g) | Pre | 103.0 ± 51.3 | <0.001 | 96.0 ± 48.7 | 102.8 ± 57.8 | 107.7 ± 44.2 | 100.6 ± 45.7 | 106.3 ± 61.7 | <0.001 |
| Post | 174.4 ± 58.1 | 207.0 ± 58.9 *** | 155.7 ± 38.6 ***,† | 170.2 ± 42.8 ***,† | 204.5 ± 56.4 *** | 137.9 ± 62.1 **,† | |||
| Added Sugar (g) | Pre | 56.5 ± 38.5 | <0.001 | 49.3 ± 38.1 | 56.7 ± 35.5 | 59.5 ± 38.4 | 55.1 ± 35.6 | 60.3 ± 45.6 | <0.001 |
| Post | 98.2 ± 46.5 | 131.2 ± 32.8 *** | 82.3 ± 23.8 **,† | 93.8 ± 33.7 ***,† | 132.0 ± 44.4 *** | 54.8 ± 44.4 †,Δ |
3.3. Body Weight and Fasting Measures
3.4. Oral Glucose Tolerance Test
| All | Time | HFCS 18% | Fructose 9% | Glucose 9% | Sucrose 18% | Control | Interaction | ||
|---|---|---|---|---|---|---|---|---|---|
| Body Weight (kg) | Pre | 73.6 ± 13.0 | <0.001 | 73.8 ± 13.2 | 74.1 ± 13.3 | 76.2 ± 12.2 | 72.1 ± 14.1 | 72.3 ± 12.2 | 0.191 |
| Post | 74.5 ± 13.3 | 74.5 ± 13.8 | 74.7 ± 14.0 | 76.6 ± 12.1 | 73.4 ± 14.7 | 73.0 ± 12.4 | |||
| Glucose (mmol/L) | Pre | 4.9 ± 0.4 | 0.056 | 5.1 ± 0.4 | 4.8 ± 0.4 | 5.0 ± 0.4 | 4.8 ± 0.4 | 5.0 ± 0.4 | 0.738 |
| Post | 5.0 ± 0.5 | 5.2 ± 0.3 | 4.9 ± 0.7 | 5.0 ± 0.7 | 5.0 ± 0.4 | 5.0 ± 0.4 | |||
| Insulin (pmol/L) | Pre | 56.9 ± 38.9 | 0.169 | 68.1 ± 36.8 | 55.6 ± 32.6 | 55.6 ± 32.6 | 54.2 ± 33.3 | 54.2 ± 35.4 | 0.133 |
| Post | 61.8 ± 50.0 | 72.9 ± 51.4 | 79.2 ± 73.6 | 56.9 ± 52.1 | 55.6 ± 31.9 | 47.9 ± 28.5 | |||
| HOMA IR | Pre | 1.8 ± 1.3 | 0.101 | 2.2 ± 1.2 | 1.7 ± 1.0 | 1.8 ± 1.7 | 1.7 ± 1.1 | 1.7 ± 1.2 | 0.112 |
| Post | 2.0 ± 1.5 | 2.4 ± 1.8 | 2.4 ± 2.0 | 1.8 ± 1.3 | 1.8 ± 1.2 | 1.6 ± 1.0 |
| All | Time | HFCS 18% | Fructose 9% | Glucose 9% | Sucrose 18% | Control | Interaction | ||
|---|---|---|---|---|---|---|---|---|---|
| Glucose AUC (min·mmol/L) | Pre | 760.4 ± 166.5 | 0.271 | 793.7 ± 194.3 | 760.4 ± 172.1 | 799.2 ± 155.4 | 749.3 ± 183.2 | 710.4 ± 138.8 | 0.969 |
| Post | 777.0 ± 165.5 | 810.3 ± 194.3 | 782.6 ± 172.1 | 793.7 ± 144.3 | 765.9 ± 199.8 | 738.2 ± 127.7 | |||
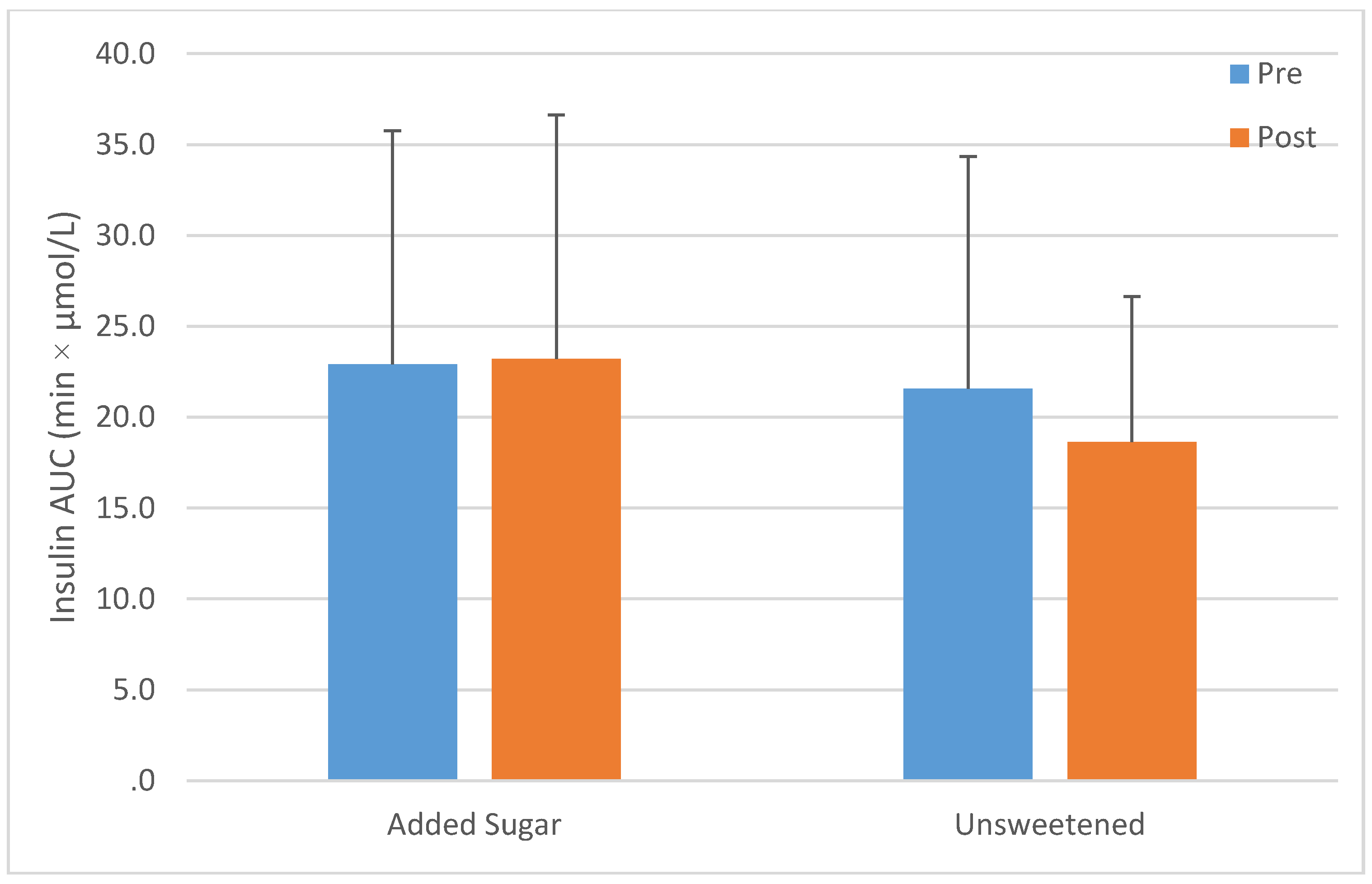
| Insulin AUC (min·µmol/L) | Pre | 22.9 ± 12.5 | 0.695 | 26.4 ± 18.8 | 19.4 ± 10.4 | 19.4 ± 8.3 | 26.4 ± 12.5 | 21.5 ± 12.5 | 0.005 |
| Post | 22.2 ± 12.5 | 22.9 ± 13.9 | 25.7 ± 11.8 ** | 20.8 ± 16.0 | 23.6 ± 11.8 | 18.8 ± 8.3 | |||
| Matsuda Insulin | Pre | 16.0 ± 24.3 | 0.681 | 21.3 ± 32.8 | 22.1 ± 45.8 | 12.9 ± 6.1 | 11.3 ± 6.8 | 14.7 ± 10.7 | 0.532 |
| Sensitivity Index | Post | 14.5 ± 26.6 | 28.0 ± 64.1 | 9.0 ± 4.2 | 12.4 ± 7.4 | 12.2 ± 10.0 | 13.8 ± 6.2 | ||
| Hepatic Insulin | Pre | 2.0 ± 1.6 | 0.941 | 2.7 ± 2.6 | 1.5 ± 0.5 | 1.6 ± 0.7 | 2.4 ± 1.6 | 2.0 ± 1.5 | 0.032 |
| Resistance | Post | 2.0 ± 1.2 | 2.2 ± 1.3 | 2.1 ± 0.8 * | 2.0 ± 1.5 | 2.4 ± 1.6 | 1.6 ± 0.6 |

4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hu, F.B. Globalization of diabetes: The role of diet, lifestyle, and genes. Diabetes Care 2011, 34, 1249–1257. [Google Scholar] [CrossRef] [PubMed]
- Shaw, J.E.; Sicree, R.A.; Zimmet, P.Z. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res. Clin. Pract. 2010, 87, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Danaei, G.; Finucane, M.M.; Lu, Y.; Singh, G.M.; Cowan, M.J.; Paciorek, C.J.; Lin, J.K.; Farzadfar, F.; Khang, Y.H.; Stevens, G.A.; et al. National, regional, and global trends in fasting plasma glucose and diabetes prevalence since 1980: Systematic analysis of health examination surveys and epidemiological studies with 370 country-years and 2.7 million participants. Lancet 2011, 378, 31–40. [Google Scholar] [CrossRef]
- International Diabetes Federation. Epidemiology and Morbidity. IDF Diabetes Atlas. Available online: https://www.idf.org/sites/default/files/IDF-Diabetes-Atlas-4th-edition.pdf (accessed on 8 April 2015).
- De Koning, L.; Malik, V.S.; Kellogg, M.D.; Rimm, E.B.; Willett, W.C.; Hu, F.B. Sweetened beverage consumption, incident coronary heart disease and biomarkers of risk in men. Circulation 2012, 125, 1735–1741. [Google Scholar] [CrossRef] [PubMed]
- Montonen, J.; Jarvinen, R.; Knekt, P.; Heliovaara, M.; Reunanen, A. Consumption of sweetened beverages and intakes of fructose and glucose predict type 2 diabetes occurrence. J. Nutr. 2007, 137, 1447–1454. [Google Scholar] [PubMed]
- Le, K.A.; Ith, M.; Kreis, R.; Faeh, D.; Bortolotti, M.; Tran, C.; Boesch, C.; Tappy, L. Fructose overconsumption causes dyslipidemia and ectopic lipid deposition in healthy subjects with and without a family history of type 2 diabetes. Am. J. Clin. Nutr. 2009, 89, 1760–1765. [Google Scholar] [CrossRef] [PubMed]
- Schulze, M.B.; Hoffmann, K.; Manson, J.E.; Willett, W.C.; Meigs, J.B.; Weikert, C.; Heidemann, C.; Colditz, G.A.; Hu, F.B. Dietary pattern, inflammation, and incidence of type 2 diabetes in women. Am. J. Clin. Nutr. 2005, 82, 675–684. [Google Scholar] [PubMed]
- Goran, M.I.; Ulijaszek, S.J.; Ventura, E.E. High fructose corn syrup and diabetes prevalence: A global perspective. Global Public Health 2013, 8, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Yoffe, P.; Hills, N.; Lustig, R.H. The relationship of sugar to population-level diabetes prevalence: An econometric analysis of repeated cross-sectional data. PLoS ONE 2013, 8, e57873. [Google Scholar] [CrossRef] [PubMed]
- Bray, G.A. Fructose and risk of cardiometabolic disease. Curr. Atheroscler. Rep. 2012, 14, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.; Segal, M.; Sautin, Y.; Nakagawa, T.; Feig, D.I.; Kang, D.H.; Gersch, M.S.; Benner, S.; Sanchez-Lozada, L.G. Potential role of sugar (fructose) in the epidemic of hypertension, obesity and the metabolic syndrome, diabetes, kidney disease, and cardiovascular disease. Am. J. Clin. Nutr. 2007, 86, 899–906. [Google Scholar] [PubMed]
- Sanchez-Lozada, L.G.; Le, M.; Segal, M.; Johnson, R.J. How safe is fructose for persons with or without diabetes? Am. J. Clin. Nutr. 2008, 88, 1189–1190. [Google Scholar] [PubMed]
- The InterAct Consortium. Consumption of sweet beverages and type 2 diabetes incidence in European adults: Results from EPIC-InterAct. Diabetologia 2013, 56, 1520–1530. [Google Scholar]
- Sluijs, I.; Beulens, J.W.J.; van der Schouw, Y.T.; van der A, D.L.; Buckland, G.; Kuijsten, A.; Schulze, M.B.; Amiano, P.; Ardanaz, E.; Balkau, B. Dietary glycemic index, glycemic load, and digestible carbohydrate intake are not associated with risk of type 2 diabetes in eight European countries. J. Nutr. 2013, 143, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Pooranaperundevi, M.; Sumiyabanu, M.S.; Viswanathan, P.; Sundarapandiyan, R.; Anuradha, C.V. Insulin resistance induced by high-fructose diet potentiates carbon tetrachloride hepatotoxicity. Toxicol. Ind. Health 2010, 26, 89–104. [Google Scholar] [CrossRef] [PubMed]
- Lewis, G.F.; Uffelman, K.; Naples, M.; Szeto, L.; Haidari, M.; Adeli, K. Intestinal lipoprotein overproduction, a nearly recognized component of insulin resistance, is ameliorated by the insulin sensitizer rosiglitazone: Studies in the fructose-fed Syrian golden hamster. Endocrinology 2005, 146, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Thorburn, A.W.; Storlien, L.H.; Jenkins, A.B.; Khouri, S.; Kraegen, E.W. Fructose-induced in vivo insulin resistance and elevated plasma triglyceride levels in rats. Am. J. Clin. Nutr. 1989, 49, 1155–1163. [Google Scholar] [PubMed]
- Rizkalla, S.W.; Boillot, J.; Tricottet, V.; Fontvieille, A.M.; Luo, J.; Salzman, J.L.; Camilleri, J.P.; Slama, G. Effects of chronic dietary fructose with and without copper supplementation on glycaemic control, adiposity, insulin binding to adipocytes and glomerular basement membrane thickness in normal rats. Br. J. Nutr. 1993, 70, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Maiztegui, B.; Borelli, M.I.; Raschia, M.A.; Del Zotto, H.; Gagliardino, J.J. Islet adaptive changes to fructose-induced insulin resistance: Beta-cell mass, glucokinase, glucose metabolism, and insulin secretion. J. Endocrinol. 2009, 200, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Mielke, J.G.; Taghibiglou, C.; Liu, L.; Zhang, Y.; Jia, Z.; Adeli, K.; Wang, Y.T. A biochemical and functional characterization of diet-induced brain insulin resistance. J. Neurochem. 2005, 93, 1568–1578. [Google Scholar] [CrossRef] [PubMed]
- Stanhope, K.L.; Schwarz, J.M.; Keim, N.L.; Griffen, S.C.; Bremer, A.A.; Graham, J.L.; Hatcher, B.; Cox, C.L.; Dyachenko, A.; Zhang, W. Consuming fructose-sweetened, not glucose-sweetened, beverages increases visceral adiposity and lipids and decreases insulin sensitivity in overweight/obese humans. J. Clin. Investig. 2009, 119, 1322–1334. [Google Scholar] [CrossRef] [PubMed]
- Cozma, A.I.; Ha, V.; de Souza, R.J.; Sievenpiper, J. Sweeteners and Diabetes. In Fructose, High Fructose Corn Syrup, Sucrose and Health; Rippe, J.M., Ed.; Springer Publishing: New York, NY, USA, 2014. [Google Scholar]
- Saris, W.H.; Astrup, A.; Prentice, A.M.; Zunft, H.J.; Formiguera, X.; Verboeket-van de Venne, W.P.; Raben, A.; Poppitt, S.D.; Seppelt, B.; Johnston, S.; et al. Randomized controlled trial of changes in dietary carbohydrate/fat ratio and simple vs. complex carbohydrates on body weight and blood lipids: The CARMEN study. The Carbohydrate Ratio Management in European National diets. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 1310–1318. [Google Scholar] [CrossRef] [PubMed]
- Cozma, A.I.; Sievenpiper, J.L.; de Souza, R.J.; Chiavaroli, L.; Ha, V.; Wang, D.D.; Mirrahimi, A.; Yu, M.E.; Carleton, A.J.; Di Buono, M.; et al. Effect of fructose on glycemic control in diabetes: A systematic review and meta-analysis of controlled feeding trials. Diabetes Care 2012, 35, 1611–1620. [Google Scholar] [CrossRef] [PubMed]
- Colditz, G.A.; Manson, J.E.; Stampfer, M.J.; Rosner, B.; Willett, W.C.; Speizer, F.E. Diet and risk of clinical diabetes in women. Am. J. Clin. Nutr. 1992, 55, 1018–1023. [Google Scholar] [PubMed]
- Meyer, K.A.; Kushi, L.H.; Jacobs, D.R., Jr.; Slavin, J.; Sellers, T.A.; Folsom, A.R. Carbohydrates, dietary fiber, and incident type 2 diabetes in older women. Am. J. Clin. Nutr. 2000, 71, 921–930. [Google Scholar] [PubMed]
- Janket, S.J.; Manson, J.E.; Sesso, H.; Buring, J.E.; Liu, S. A prospective study of sugar intake and risk of type 2 diabetes in women. Diabetes Care 2003, 26, 1008–1105. [Google Scholar] [CrossRef] [PubMed]
- Hodge, A.M.; English, D.R.; O’Dea, K.; Giles, G.G. Glycemic index and dietary fiber and the risk of type 2 diabetes. Diabetes Care 2004, 27, 2701–2706. [Google Scholar] [CrossRef] [PubMed]
- Morino, K.; Petersen, K.F.; Shulman, G.I. Molecular mechanisms of insulin resistance in humans and their potential links with mitochondrial dysfunction. Diabetes 2006, 55, S9–S15. [Google Scholar] [CrossRef] [PubMed]
- Seppala-Lindroos, A.; Vehkavaara, S.; Hakkinen, A.M.; Goto, T.; Westerbacka, J.; Sovijarvi, A.; Halavaara, J.; Yki-Jarvinen, H. Fat accumulation in the liver is associated with defects in insulin suppression of glucose production and serum free fatty acids independent of obesity in normal men. J. Clin. Endocrinol. Metab. 2002, 87, 3023–3028. [Google Scholar] [CrossRef] [PubMed]
- Hellerstein, M.K.; Schwarz, J.M.; Neese, R.A. Regulation of hepatic de novo lipogenesis in humans. Annu. Rev. Nutr. 1996, 16, 523–557. [Google Scholar] [CrossRef] [PubMed]
- Hellerstein, M.K. No common energy currency: De novo lipogenesis as the road less traveled. Am. J. Clin. Nutr. 2001, 74, 707–708. [Google Scholar] [PubMed]
- Peterson, K.F.; Shulman, G.I. Etiology of insulin resistance. Am. J. Med. 2007, 119, S10–S16. [Google Scholar] [CrossRef] [PubMed]
- Shulman, G.I. Ectopic fat in insulin resistance, dyslipidemia, and cardiometabolic disease. N. Engl. J. Med. 2014, 371, 1131–1141. [Google Scholar] [CrossRef] [PubMed]
- Lowndes, J.; Kawiecki, D.; Yu, Z.; Rippe, J.M. No Dose Response Relationship in the Effects of Commonly Consumed Sugars on Risk Factors for Diabetes across a Range of Typical Human Consumption Levels. Food Nutr. Sci. 2015, 6, 101–111. [Google Scholar] [CrossRef]
- Mifflin, M.D.; St. Jeor, S.T.; Hill, L.A.; Scott, B.J.; Daugherty, S.A.; Koh, Y.O. A new predictive equation for resting energy expenditure in healthy individuals. Am. J. Clin. Nutr. 1990, 51, 241–247. [Google Scholar] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Ghani, M.A.; Matsuda, M.; Balas, B.; DeFronzo, R.A. Muscle and liver insulin resistance indexes derived from the oral glucose tolerance test. Diabetes Care 2007, 30, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, M.; DeFronzo, R.A. Insulin sensitivity indices obtained from oral glucose tolerance testing: Comparison with the euglycemic insulin clamp. Diabetes Care 1999, 22, 1462–1470. [Google Scholar] [CrossRef] [PubMed]
- Johnston, R.D.; Stephenson, M.C.; Crossland, H.; Cordon, S.M.; Palcidi, E.; Cox, E.F.; Taylor, M.A.; Aithal, G.P.; MacDonald, I.A. No difference between high fructose and high glucose diets on liver tricylglycerol or biochemistry in healthy overweight men. Gastroenterology 2013, 145, 1016–1025. [Google Scholar] [CrossRef] [PubMed]
- Lê, K.-A.; Faeh, D.; Stettler, R.; Ith, M.; Kreis, R.; Vermathen, P.; Boesch, C.; Ravussin, E.; Tappy, L. A 4-wk high-fructose diet alters lipid metabolism without affecting insulin sensitivity or ectopic lipids in healthy humans. Am. J. Clin. Nutr. 2006, 84, 1374–1379. [Google Scholar] [PubMed]
- Latulippe, M.E.; Skoog, S.M. Fructose malabsorption and intolerance: Effects of fructose with and without simultaneous glucose ingestion. Crit. Rev. Food Sci. Nutr. 2011, 51, 583–592. [Google Scholar] [CrossRef] [PubMed]
- Tappy, L.; Egli, L.; Tran, C. Metabolism of Nutritive Sweeteners in Humans. In Fructose, High Fructose Corn Syrup, Sucrose and Health; Rippe, J.M., Ed.; Springer Publishing: New York, NY, USA, 2014. [Google Scholar]
- Silbernagel, G.; Machann, J.; Unmuth, S.; Schick, F.; Stefan, N.; Hans, U.; Ring, H.; Fritsche, A. Effects of 4-week very-high-fructose/glucose diets on insulin sensitivity, visceral fat and intrahepatic lipids: An exploratory trial. Br. J. Nutr. 2011, 106, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Pan, A.; Sun, Q.; Bernstein, A.M.; Schulze, M.B.; Manson, J.E.; Willett, W.C.; Hu, F.B. Red meat consumption and risk of type 2 diabetes: 3 Cohorts of US adults and an updated meta-analysis. Am. J. Clin. Nutr. 2011, 94, 1088–1096. [Google Scholar] [CrossRef] [PubMed]
- Halton, T.L.; Willett, W.C.; Liu, S.; Manson, J.E.; Stampfer, M.J.; Hu, F.B. Potato and French fry consumption and risk of type 2 diabetes in women. Am. J. Clin. Nutr. 2006, 83, 284–290. [Google Scholar] [PubMed]
- Barclay, A.W.; Brand-Miller, J. The Australian paradox: A substantial decline in sugars intake over the same timeframe that overweight and obesity have increased. Nutrients 2012, 3, 491–504. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Total added sugars, have not resulted in a decrease of obesity or diabetes in the US. Available online: http://www.cdc.gov/diabetes/statistics/slides/maps_diabetesobesity_trends.pdf (accessed on 8 April 2015).
- Welsh, J.A.; Sharma, A.J.; Grellinger, L.; Vos, M.B. Consumption of added sugars is decreasing in the United States. Am. J. Clin. Nutr. 2011, 94, 726–734. [Google Scholar] [CrossRef] [PubMed]
- Marriott, B.; Cole, N.; Lee, E. National estimates of dietary fructose intake increased from 1977 to 2004 in the United States. J. Nutr. 2009, 139, 1228S–1235S. [Google Scholar] [CrossRef] [PubMed]
- Dabfinn, A.; Norat, T.; Romundstad, P.; Vatten, L.J. Dairy products and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis of cohort studies. Am. J. Clin. Nutr. 2013, 98, 1066–1083. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lowndes, J.; Sinnett, S.S.; Rippe, J.M. No Effect of Added Sugar Consumed at Median American Intake Level on Glucose Tolerance or Insulin Resistance. Nutrients 2015, 7, 8830-8845. https://doi.org/10.3390/nu7105430
Lowndes J, Sinnett SS, Rippe JM. No Effect of Added Sugar Consumed at Median American Intake Level on Glucose Tolerance or Insulin Resistance. Nutrients. 2015; 7(10):8830-8845. https://doi.org/10.3390/nu7105430
Chicago/Turabian StyleLowndes, Joshua, Stephanie S. Sinnett, and James M. Rippe. 2015. "No Effect of Added Sugar Consumed at Median American Intake Level on Glucose Tolerance or Insulin Resistance" Nutrients 7, no. 10: 8830-8845. https://doi.org/10.3390/nu7105430
APA StyleLowndes, J., Sinnett, S. S., & Rippe, J. M. (2015). No Effect of Added Sugar Consumed at Median American Intake Level on Glucose Tolerance or Insulin Resistance. Nutrients, 7(10), 8830-8845. https://doi.org/10.3390/nu7105430




