Prevalence of Hyperhomocysteinemia in China: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Experimental Section
2.1. Literature Searches
2.2. Study Selection and Inclusion Criteria
2.3. Data Extraction
2.4. Statistical Analysis
3. Results
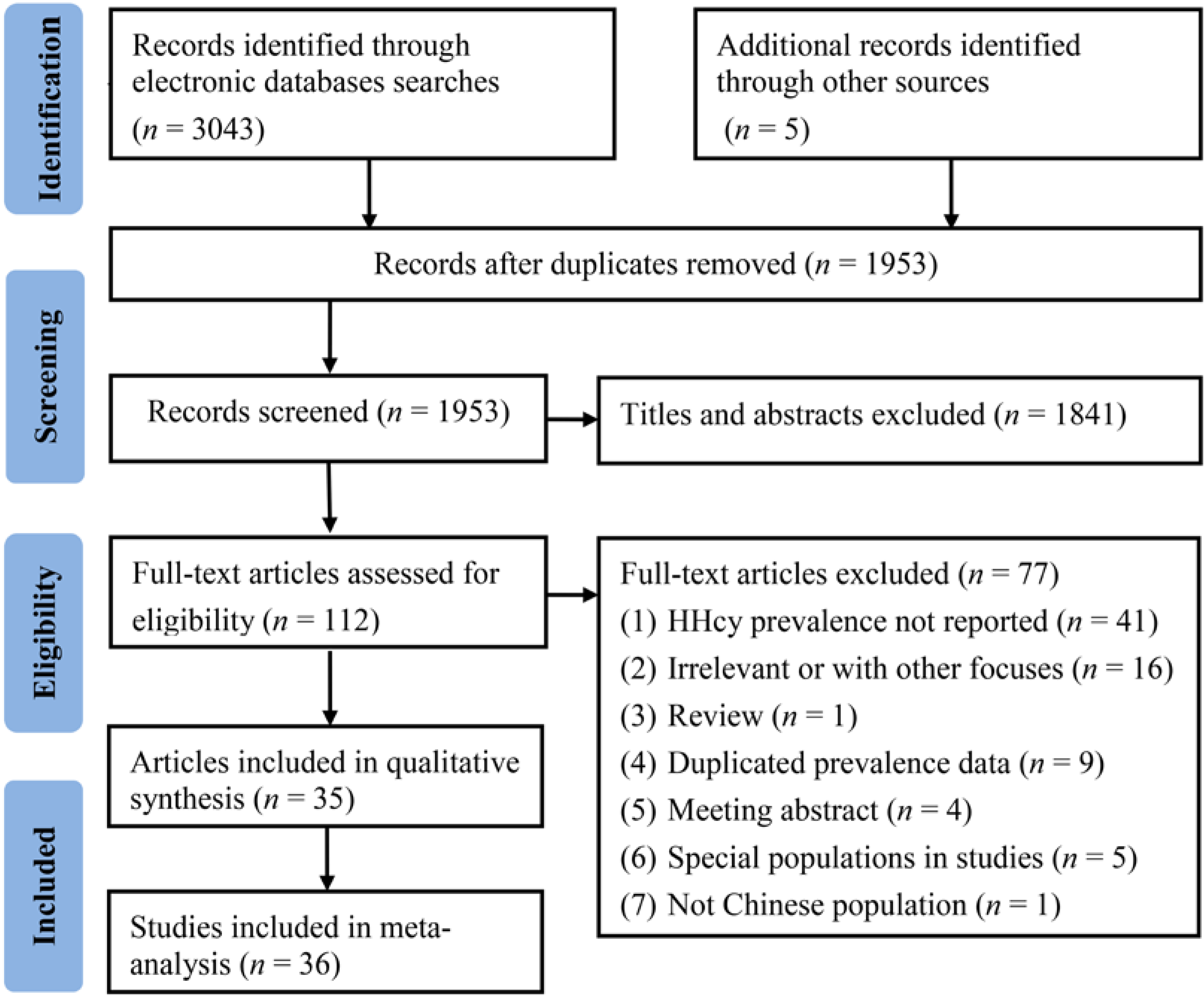
3.1. Study Selection Procedure and Characteristics
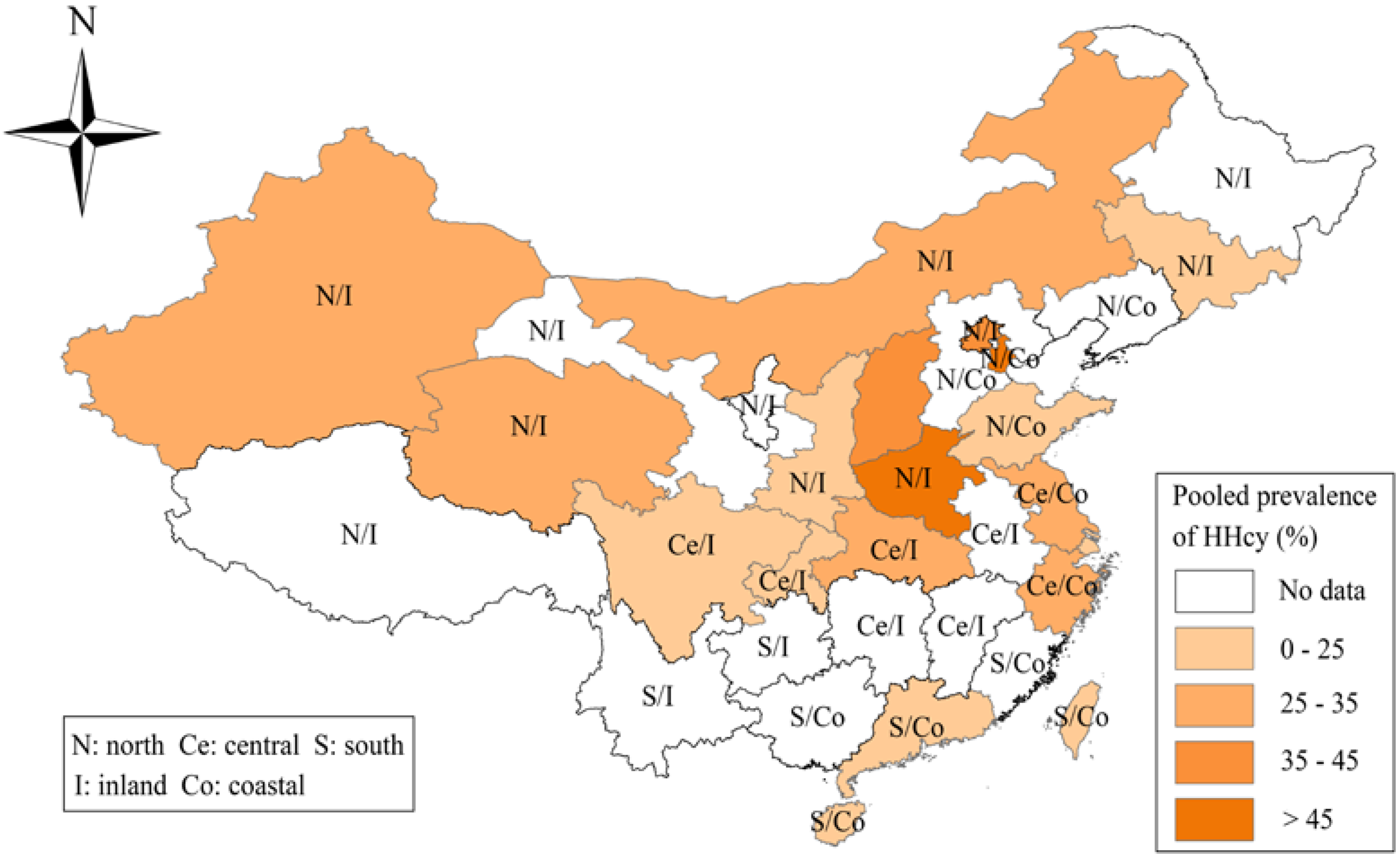
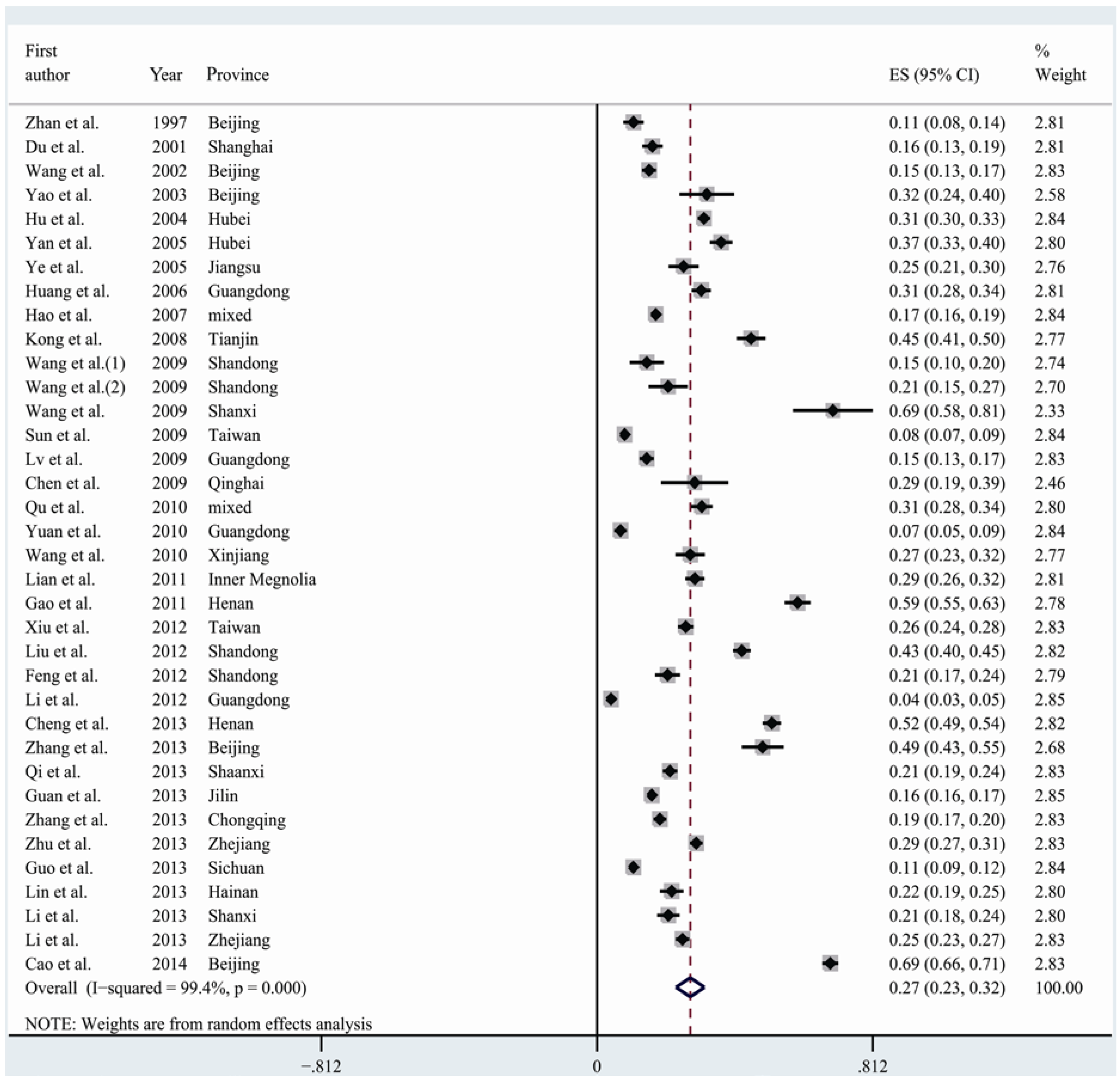
3.2. Meta-Analysis and Pooled Prevalence of HHcy
3.3. Sensitivity and Meta-Regression Analyses
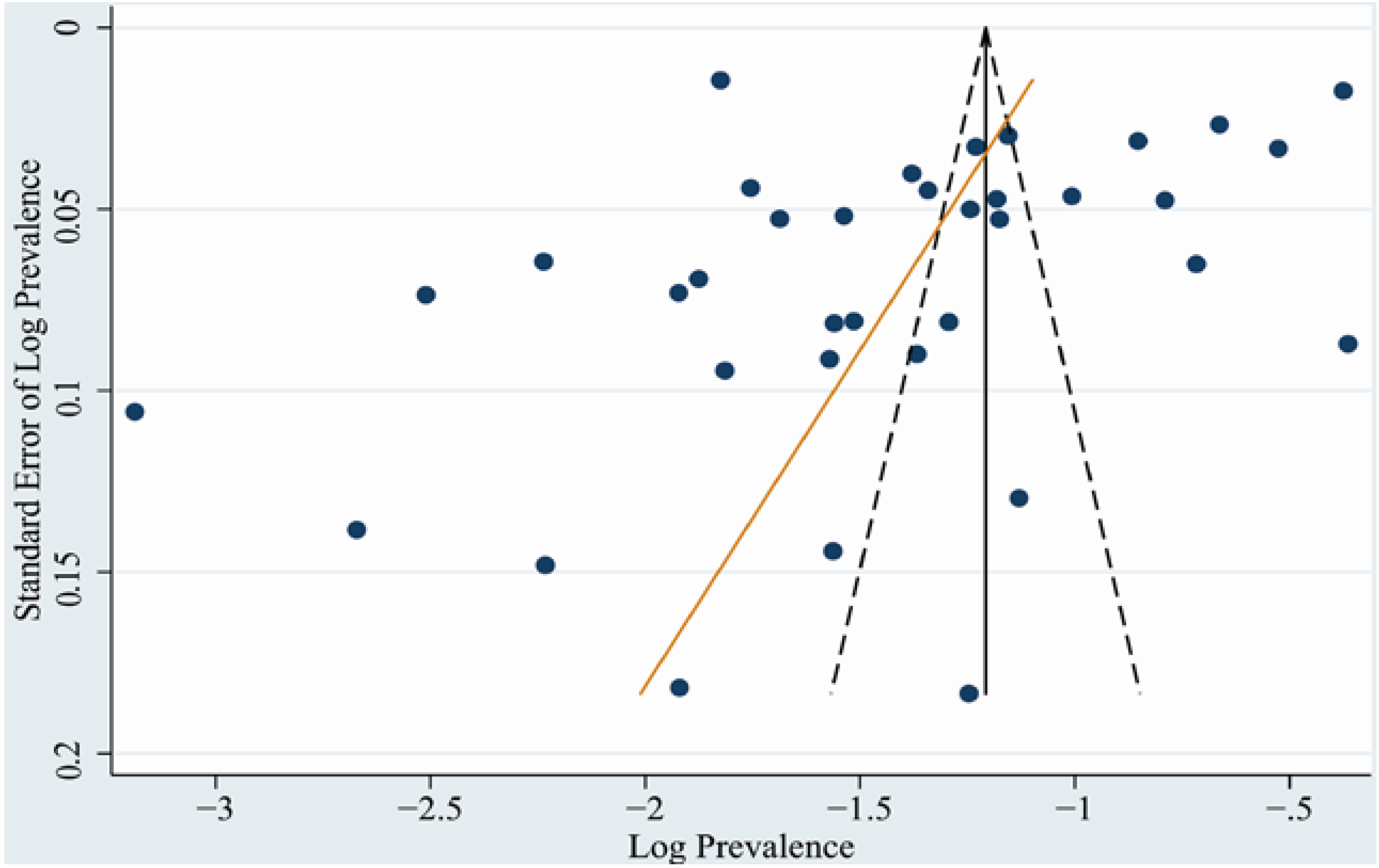
3.4. Publication Bias
| Subgroup | No. of Studies | No. of Total Participants | No. of Cases | Prevalence (%) | 95% CI | Heterogeneity Test | |
|---|---|---|---|---|---|---|---|
| I2 (%) | p Value | ||||||
| Total | 36 | 60,754 | 13,183 | 27.5 | (23.3–31.6) | 99.4 | <0.001 |
| Study Region | |||||||
| North | 21 | 37,678 | 8780 | 34.3 | (26.7–42.0) | 99.5 | <0.001 |
| Central | 10 | 13,588 | 3020 | 21.0 | 14.5–27.5 | 99.0 | <0.001 |
| South | 7 | 9080 | 1276 | 16.0 | (9.4–22.6) | 99.1 | <0.001 |
| Study Location | |||||||
| Inland | 19 | 42,703 | 9096 | 31.5 | (25.0–38.0) | 99.5 | <0.001 |
| Coastal | 15 | 14,777 | 3411 | 23.0 | (16.0–29.9) | 99.3 | <0.001 |
| Sex | |||||||
| Men | 19 | 27,982 | 7736 | 34.8 | (29.1–40.5) | 98.8 | <0.001 |
| Women | 20 | 19,700 | 2120 | 18.7 | (15.2–22.2) | 98.0 | <0.001 |
| Age | |||||||
| <45 | 10 | 20,333 | 3062 | 17.9 | (15.5–20.4) | 94.5 | <0.001 |
| 45–65 | 9 | 9164 | 1846 | 22.7 | (19.0–26.5) | 93.7 | <0.001 |
| >65 | 8 | 5039 | 974 | 35.2 | (22.0–48.4) | 99.2 | <0.001 |
| Sample Size | |||||||
| <1000 | 19 | 8770 | 2475 | 29.5 | (22.8–36.1) | 98.3 | <0.001 |
| ≥1000 | 17 | 51,984 | 10,708 | 25.4 | (19.4–31.3) | 99.7 | <0.001 |
| Year of Data Collection | |||||||
| 1990–2005 | 14 | 14,501 | 2690 | 22.7 | (16.8–28.5) | 99.1 | <0.001 |
| 2006–2012 | 20 | 44,700 | 9938 | 29.6 | (23.4–35.8) | 99.5 | <0.001 |
| Study Setting | |||||||
| Rural | 8 | 4650 | 1196 | 28.1 | (21.1–35.1) | 96.6 | <0.001 |
| Urban | 30 | 55,633 | 12,042 | 26.5 | (21.7–31.3) | 99.5 | <0.001 |
| Models | Available Studies for Analysis | Prevalence (%) (95% CI) | Heterogeneity Test | |
|---|---|---|---|---|
| I2 (%) | p Value | |||
| 1. Exclude studies that cut-point for HHcy was not 15 μmol/L | 32 | 27.4 (23.0–31.8) | 99.5 | <0.001 |
| 2. Exclude studies with sample size <100 or >20,000 | 33 | 26.7 (21.5–31.9) | 99.5 | <0.001 |
| 3. Exclude studies that gender proportion was not reported | 32 | 28.1 (23.8–32.4) | 99.3 | <0.001 |
| 4. Exclude studies that year of data collection was not reported | 31 | 26.6 (22.1–31.0) | 99.5 | <0.001 |
| 5. Exclude studies that Hcy Measurement method was not reported | 33 | 28.6 (24.2–32.9) | 99.3 | <0.001 |
| 6. Exclude studies that gender proportion, year of date collection, study setting, and Hcy measurement method was not reported | 25 | 27.6 (22.7–32.5) | 99.4 | <0.001 |
| Variables | No. of study (n = 28) | Coefficient (95% CI) | P value |
|---|---|---|---|
| Univariate meta-regression | |||
| Year of publication | 28 | 0.03 (−0.03–0.08) | 0.288 |
| Male% in sample | 28 | 0.001 (−0.01–0.02) | 0.862 |
| Sample size | 28 | 0.00 (−0.00–0.00) | 0.435 |
| Study location | |||
| Inland | 19 | Reference | |
| Coastal | 9 | −0.35 (−0.84–0.14) | 0.153 |
| Study region | 0.054 | ||
| South | 5 | Reference | |
| Central | 6 | 0.54 (−0.15–1.22) | 0.119 |
| North | 17 | 0.71 (0.14–1.30) | 0.017 |
| Study period | |||
| 2006–2012 | 18 | Reference | |
| 1990–2005 | 10 | −0.11 (−0.60–0.38) | 0.656 |
| Study setting | 0.451 | ||
| Rural and urban | 4 | Reference | |
| Urban | 21 | 0.39 (−0.29–1.06) | 0.251 |
| Rural | 3 | 0.51 (−0.44–1.47) | 0.278 |
| Diagnostic criteria | |||
| 15 μmol/L | 25 | Reference | |
| Others | 3 | 0.16 (−0.60–0.92) | 0.671 |
| Methods of Hcy measurement | 0.798 | ||
| FPIA | 5 | Reference | |
| ECA | 9 | −0.10 (−0.81–0.62) | 0.783 |
| HPLC | 8 | 0.07 (−0.66–0.80) | 0.846 |
| Others | 6 | 0.22 (−0.55–1.00) | 0.555 |
| Multivariate meta-regression | 0.0511 | ||
| Year of publication | 28 | 0.04 (−0.01–0.09) | 0.242 |
| Study location | |||
| Inland | 19 | Reference | |
| Coastal | 9 | −0.16 (−0.70–0.38) | 0.614 |
| Study region | 0.294 | ||
| South | 5 | Reference | |
| Central | 6 | 0.48 (−0.25–1.21) | 0.185 |
| North | 17 | 0.66 (−0.01–1.33) | 0.052 |
4. Discussion
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wald, D.S.; Law, M.; Morris, J.K. Homocysteine and cardiovascular disease: Evidence on causality from a meta-analysis. BMJ 2002, 325. [Google Scholar] [CrossRef]
- The Homocysteine Studies Collaboration. Homocysteine and risk of ischemic heart disease and stroke: A meta-analysis. JAMA 2002, 288, 2015–2022. [Google Scholar]
- De Ruijter, W.; Westendorp, R.G.; Assendelft, W.J.; den Elzen, W.P.; de Crean, A.J.; le Cessies, S.; Gussekloo, J. Use of Framingham risk score and new biomarkers to predict cardiovascular mortality in older people: Population based observational cohort study. BMJ 2009, 8. [Google Scholar] [CrossRef]
- Brustolin, S.; Giugliani, R.; Felix, T.M. Genetics of homocysteine metabolism and associated disorders. Braz. J. Med. Biol. Res. 2010, 43, 1–7. [Google Scholar] [CrossRef]
- Yakub, M.; Schulze, K.J.; Khatry, S.K.; Stewart, C.P.; Christian, P., Jr.; West, K.P. High plasma homocysteine increases risk of metabolic syndrome in 6 to 8 year old children in rural Nepal. Nutrients 2014, 6, 1649–1661. [Google Scholar] [CrossRef]
- Refsum, H.; Ueland, P.M.; Nygard, O.; Vollset, S.E. Homocysteine and cardiovascular disease. Annu. Rev. Med. 1998, 49, 31–62. [Google Scholar] [PubMed]
- Nygard, O.; Vollset, S.E.; Refsum, H.; Brattstrom, L.; Ueland, P.M. Total homocysteine and cardiovascular disease. J. Intern. Med. 1999, 246, 425–454. [Google Scholar]
- Schafer, J.H.; Glass, T.A.; Bressler, J.; Todd, A.C.; Schwartz, B.S. Blood lead is a predictor of homocysteine levels in a population-based study of older adults. Environ. Helath Prespect. 2005, 1, 31–35. [Google Scholar]
- Refsum, H.; Nurk, E.; Smith, A.D.; Ueland, P.M.; Gjesdal, C.G.; Bjelland, I.; Tverdal, A.; Tell, G.S.; Nygard, O.; Vollset, S.E. The Hordaland Homocysteine Study: A community-based study of homocysteine, its determinants, and associations with disease. J. Nutr. 2006, 136, 1731–1740. [Google Scholar]
- Carmel, R.; Green, R.; Jacobsen, D.W.; Rasmussen, K.; Fbrea, M.; Azen, C. Serum cobalamin, homocysteine, and methylmalonic acid concentrations in a multiethnic elderly population: Ethnic and sex differences in cobalamin and metabolite abnormalities. Am. J. Clin. Nutr. 1999, 70, 904–910. [Google Scholar] [PubMed]
- Amouzou, E.K.; Chabi, N.W.; Adjalla, C.E.; Rodriguez-Gueant, R.M.; Feillet, F.; Villaume, C.; Sanni, A.; Gueant, J.L. High prevalence of hyperhomocysteinemia related to folate deficiency and the 677C→T mutation of the gene encoding methylenetetrahydrofolate reductase in coastal West Africa. Am. J. Clin. Nutr. 2004, 79, 619–624. [Google Scholar] [PubMed]
- Hao, L.; Ma, J.; Stampfer, M.J.; Ren, A.; Tian, Y.; Willett, W.C.; Li, Z. Geographical, seasonal and gender differences in folate status among Chinese adults. J. Nutr. 2003, 133, 3630–3635. [Google Scholar]
- Yang, B.; Liu, Y.; Li, Y.; Fan, S.; Zhi, X.; Lu, X.; Wang, D.; Zheng, Q.; Wang, Y.; Wang, Y.; et al. Geographical distribution of MTHFR C677T, A1298C and MTRR A66G Gene polymorphisms in China: Findings from 15357 adults of Han nationality. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- Wang, Y.F.; Pei, L.J.; Wang, J.F.; Zheng, X.Y. Is the prevalence of MTHFR C677T polymorphism associated with ultraviolet radiation in Eurasia? J. Hum. Genet. 2012, 57, 780–786. [Google Scholar] [CrossRef] [PubMed]
- The World Bank. Toward a Healthy and Harmonious Life in China: Stemming the Rising Tide of Non-Communicable Disease. Available online: http://www.worldbank.org/en/news/feature/2011/07/26/toward-health-harmonious-life-china-stemming-rising-tide-of-non-communicable-diseases (accessed on 4 September 2014).
- Ministry of Health of the People’s Republic of China. China Birth Defects Prevention Report (2012). Available online: http://www.moh.gov.cn/publicfiles/business/htmlfiles/mohfybjysqwss/s7901/201209/55840.htm (accessed on 4 September 2014).
- Li, X.H.; Zhu, J.; Wang, Y.P.; Mu, D.Z.; Zhou, G.X.; Li, Q. Geographic and urban-rural disparities in the total prevalence of neural tube defects and their subtypes during 2006–2008 in China: A study using the hospital-based birth defects surveillance system. BMC Public Health 2013, 13. [Google Scholar] [CrossRef] [PubMed]
- Zintzaras, E.; Ioannidis, J.P. Heterogeneity testing in meta-analysis of genome searches. Genet. Epidemiol. 2005, 28, 123–137. [Google Scholar] [CrossRef] [PubMed]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Control. Clin. Trials. 1986, 7, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Thompson, S.G.; Sharp, S.J. Explaining heterogeneity in meta-analysis: A comparison of methods. Stat. Med. 1999, 18, 2693–2708. [Google Scholar] [CrossRef] [PubMed]
- Mickey, R.M.; Greenland, S. The impact of confounder selection criteria on effect estimation. Am. J. Epidemiol. 1989, 129, 125–137. [Google Scholar] [PubMed]
- Trikalinos, T.A.; Salanti, G.; Zintzaras, E. Meta-analysis methods. Adv. Genet. 2008, 60, 311–334. [Google Scholar] [PubMed]
- Egger, M.; Davey, S.G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Mello, A.L.; Cunha, S.F.; Foss-Freitas, M.C.; Vannucchi, H. Evaluation of plasma homocysteine level according to the C677T and A1298C polymorphism of the enzyme MTHFR in type 2 diabetic adults. Arq. Bras. Endocrinol. Metab. 2012, 56, 428–434. [Google Scholar] [CrossRef]
- Mahfouz, R.A.; Cortas, N.K.; Charafeddine, K.M.; Abdul Khalik, R.N.; Sarieddine, D.S.; Kadi, R.H. Correlation of methylenetetrahydrofolate reductase polymorphisms with homocysteine metabolism in healthy Lebanese adults. Gene 2012, 504, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Marti, F.; Vollenweider, P.; Marques-Vidal, P.M.; Mooser, V.; Weaber, G.; Paccaud, F.; Bochud, M. Hyperhomocysteinemia is independently associated with albuminuria in the population-based Colaus study. BMC Public Health 2011, 11. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.K.; Ordovas, J.M.; Selhub, J.; Campos, H. B vitamins and plasma homocysteine concentrations in an urban and rural area of Costa Rica. J. Am. Coll. Nutr. 2003, 22, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.W.; Whang, D.H.; Ko, Y.J.; Joo, S.Y.; Yun, Y.M.; Hur, M.; Kim, J.Q. Reference interval and determinants of the serum homocysteine level in a Korean population. J. Clin. Lab. Anal. 2011, 5, 317–323. [Google Scholar]
- Fakhrzadeh, H.; Ghotbi, S.; Pourebrahim, R.; Nouri, M.; Heshmat, R.; Bandarian, F.; Shafaee, A.; Larijani, B. Total plasma homocysteine, folate, and vitamin B12 status in healthy Iranian adults: The Tehran homocysteine survey (2003–2004)/A cross-sectional population based study. BMC Public Health 2006, 6. [Google Scholar] [CrossRef]
- Hambaba, L.; Abdessemed, S.; Yahia, M.; Laroui, S.; Rouabah, F. Relationship between hyperhomocysteinemia and C677T polymorphism of methylenetetrahydrofolate reductase gene in a healthy Algerian population. Ann. Biol. Clin. (Paris) 2008, 66, 637–641. [Google Scholar]
- Jacques, P.F.; Selhub, J.; Bostom, A.G.; Wilson, P.W.; Rosenberg, I.H. The effect of folic acid fortification on plasma folate and total homocysteine concentrations. N. Engl. J. Med. 1999, 340, 1449–1454. [Google Scholar]
- Hickling, S.; Hung, J.; Knuiman, M.; Jamrozik, K.; McQuilan, B.; Beilby, J.; Thompson, P. Impact of voluntary folate fortification on plasma homocysteine and serum folate in Australia from 1995 to 2001: A population based cohort study. J. Epidemiol. Community Health 2005, 59, 371–376. [Google Scholar] [PubMed]
- Berry, R.J.; Li, Z.; Erickson, J.D.; Li, S.; Moore, C.A.; Wang, H.; Mulinare, J.; Zhao, P.; Wong, L.Y.; Gindler, J.; et al. Prevention of neural-tube defects with folic acid in China: China-U.S. Collaborative Project for Neural Tube Defect Prevetion. N. Engl. J. Med. 1999, 20, 1485–1490. [Google Scholar] [CrossRef]
- Debreceni, B.; Debreceni, L. The role of homocysteine-lowering B-vitamins in the primary prevention of cardiovascular disease. Cardiovasc. Ther. 2014, 3, 130–138. [Google Scholar]
- Rautiainen, S.; Akesson, A.; Levitan, E.B.; Morgenstern, R.; Mittleman, M.A.; Wolk, A. Multivitamin use and the risk of myocardial infarction: A population-based cohort of Swedish women. Am. J. Clin. Nutr. 2010, 5, 1251–1256. [Google Scholar] [CrossRef]
- Ji, Y.; Tan, S.; Xu, Y.; Chandra, A.; Shi, C.; Song, B.; Qin, J.; Gao, Y. Vitamin B supplementation, homocysteine levels, and the risk of cerebrovascular disease: A meta-analysis. Neurology 2013, 15, 1298–1307. [Google Scholar] [CrossRef]
- Qin, X.; Huo, Y.; Lanqman, C.B.; Hou, F.; Chen, Y.; Matossian, D.; Xu, X.; Wang, X. Folic acid therapy and cardiovascular disease in ESRD or advanced chronic kidney disease: A meta-analysis. Clin. J. Am. Soc. Nephrol. 2011, 3, 482–488. [Google Scholar] [CrossRef]
- Ebbing, M.; Bonna, K.H.; Nygard, O.; Arnesen, E.; Ueland, P.M.; Nordrehaug, J.K.; Rasmussen, K.; Njolstad, I.; Refsum, H.; Nilsen, D.W.; et al. Cancer incidence and mortality after treatment with folic acid and vitamin B12. JAMA 2009, 19, 2119–2126. [Google Scholar] [CrossRef]
- Qin, X.; Cui, Y.; Shen, L.; Sun, N.; Zhang, Y.; Li, J.; Xu, X.; Wang, B.; Xu, X.; Huo, Y.; et al. Folic acid supplementation and cancer risk: A meta-analysis of randomized controlled trials. Int. J. Cancer 2013, 5, 1033–1041. [Google Scholar]
- Chen, P.; Li, C.; Li, X.; Li, J.; Chu, R.; Wang, H. Higher dietary folate intake reduces the breast cancer risk: A systematic review and meta-analysis. Br. J. Cancer 2014, 9, 2327–2338. [Google Scholar] [CrossRef]
- Lin, H.L.; An, Q.Z.; Wang, Q.Z.; Liu, C.X. Folate intake and pancreatic cancer risk: An overall and dose-response meta-analysis. Public Health 2013, 7, 607–613. [Google Scholar] [CrossRef]
- Dai, W.M.; Yang, B.; Chu, X.Y.; Wang, Y.Q.; Zhao, M.; Chen, L.; Zhang, G.Q. Association between folate intake, serum folate levels and the risk of lung cancer: A systematic review and meta-analysis. Chin. Med. J. (Engl.) 2013, 10, 1957–1964. [Google Scholar]
- Wang, Y.; Li, X.; Qin, X.; Cai, Y.; He, M.; Sun, L.; Li, J.; Zhang, Y.; Tang, G.; Wang, B.; et al. Prevalence of hyperhomocysteinaemia and its major determinants in rural Chinese hypertensive patients aged 45–75 years. Br. J. Nutr. 2013, 109, 1284–1293. [Google Scholar] [CrossRef] [PubMed]
- Craig, S.A. Betaine in human nutrition. Am. J. Clin. Nutr. 2004, 80, 539–549. [Google Scholar] [PubMed]
- Vogiatzoglou, A.; Smith, A.D.; Nurk, E.; Berstad, D.; Drevon, C.A.; Ueland, P.M.; Vollset, S.E.; Tell, G.S.; Refsum, H. Dietary sources of vitamin B-12 and their association with plasma vitamin B-12 concentrations in the general population: The Hordaland Homocysteine Study. Am. J. Clin. Nutr. 2009, 89, 1078–1087. [Google Scholar] [CrossRef] [PubMed]
- Jacques, P.F.; Rosenberg, I.H.; Rogers, G.; Selhub, J.; Bowman, B.A.; Gunter, E.W.; Wright, J.D.; Johnson, C.L. Serum total homocysteine concentrations in adolescent and adult Americans: Results from the third National Health and Nutrition Examination Survey. Am. J. Clin. Nutr. 1999, 69, 482–489. [Google Scholar] [PubMed]
- Refsum, H.; Smith, A.D.; Ueland, P.M.; Nexo, E.; Clarke, R.; Mcpartlin, J.; Johnson, C.; Enqbeak, F.; Schneede, J.; McPartlin, C.; et al. Facts and recommendations about total homocysteine determinations: An expert opinion. Clin. Chem. 2004, 50, 3–32. [Google Scholar]
- Brattstrom, L.; Lindgren, A.; Israelsson, B.; Andersson, A.; Hultberg, B. Homocysteine and cysteine: Determinants of plasma levels in middle-aged and elderly subjects. J. Intern. Med. 1994, 236, 633–641. [Google Scholar] [CrossRef]
- Rauh, M.; Verwied, S.; Knerr, I.; Dorr, H.G.; Sonnichsen, A.; Koletzko, B. Homocysteine concentrations in a German cohort of 500 individuals: Reference ranges and determinants of plasma levels in healthy children and their parents. Amino Acids 2001, 20, 409–418. [Google Scholar] [CrossRef]
- Giltay, E.J.; Hoogeveen, E.K.; Elbers, J.M.; Gooren, L.J.; Asscheman, H.; Stehouwer, C.D. Effects of sex steroids on plasma total homocysteine levels: A study in transsexual males and females. J. Clin. Endocrinol. Metab. 1998, 83, 550–553. [Google Scholar] [CrossRef] [PubMed]
- Mijatovic, V.; Kenemans, P.; Jakobs, C.; van Baal, W.M.; Peter-Muller, E.R.; vander Mooren, M.J. A randomized controlled study of the effects of 17 beta-estradiol-dydrogesterone on plasma homocysteine in postmenopausal women. Obstet. Gynecol. 1998, 91, 432–436. [Google Scholar] [CrossRef] [PubMed]
- Guttormsen, A.B.; Ueland, P.M.; Svarstad, E.; Refsum, H. Kinetic basis of hyperhomocysteinemia in patients with chronic renal failure. Kidney. Int. 1997, 52, 495–502. [Google Scholar] [CrossRef]
- Selhub, J.; Jacques, P.F.; Wilson, P.W.; Rush, D.; Rosenberg, I.H. Vitamin status and intake as primary determinants of homocysteinemia in an elderly population. JAMA 1993, 270, 2693–2698. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, B.; Fan, S.; Zhi, X.; Wang, Y.; Wang, Y.; Zheng, Q.; Sun, G. Prevalence of Hyperhomocysteinemia in China: A Systematic Review and Meta-Analysis. Nutrients 2015, 7, 74-90. https://doi.org/10.3390/nu7010074
Yang B, Fan S, Zhi X, Wang Y, Wang Y, Zheng Q, Sun G. Prevalence of Hyperhomocysteinemia in China: A Systematic Review and Meta-Analysis. Nutrients. 2015; 7(1):74-90. https://doi.org/10.3390/nu7010074
Chicago/Turabian StyleYang, Boyi, Shujun Fan, Xueyuan Zhi, Yinuo Wang, Yanxun Wang, Quanmei Zheng, and Guifan Sun. 2015. "Prevalence of Hyperhomocysteinemia in China: A Systematic Review and Meta-Analysis" Nutrients 7, no. 1: 74-90. https://doi.org/10.3390/nu7010074
APA StyleYang, B., Fan, S., Zhi, X., Wang, Y., Wang, Y., Zheng, Q., & Sun, G. (2015). Prevalence of Hyperhomocysteinemia in China: A Systematic Review and Meta-Analysis. Nutrients, 7(1), 74-90. https://doi.org/10.3390/nu7010074





