The Role of Sweet Taste in Satiation and Satiety
Abstract
:1. Introduction
2. Taste and Its Function
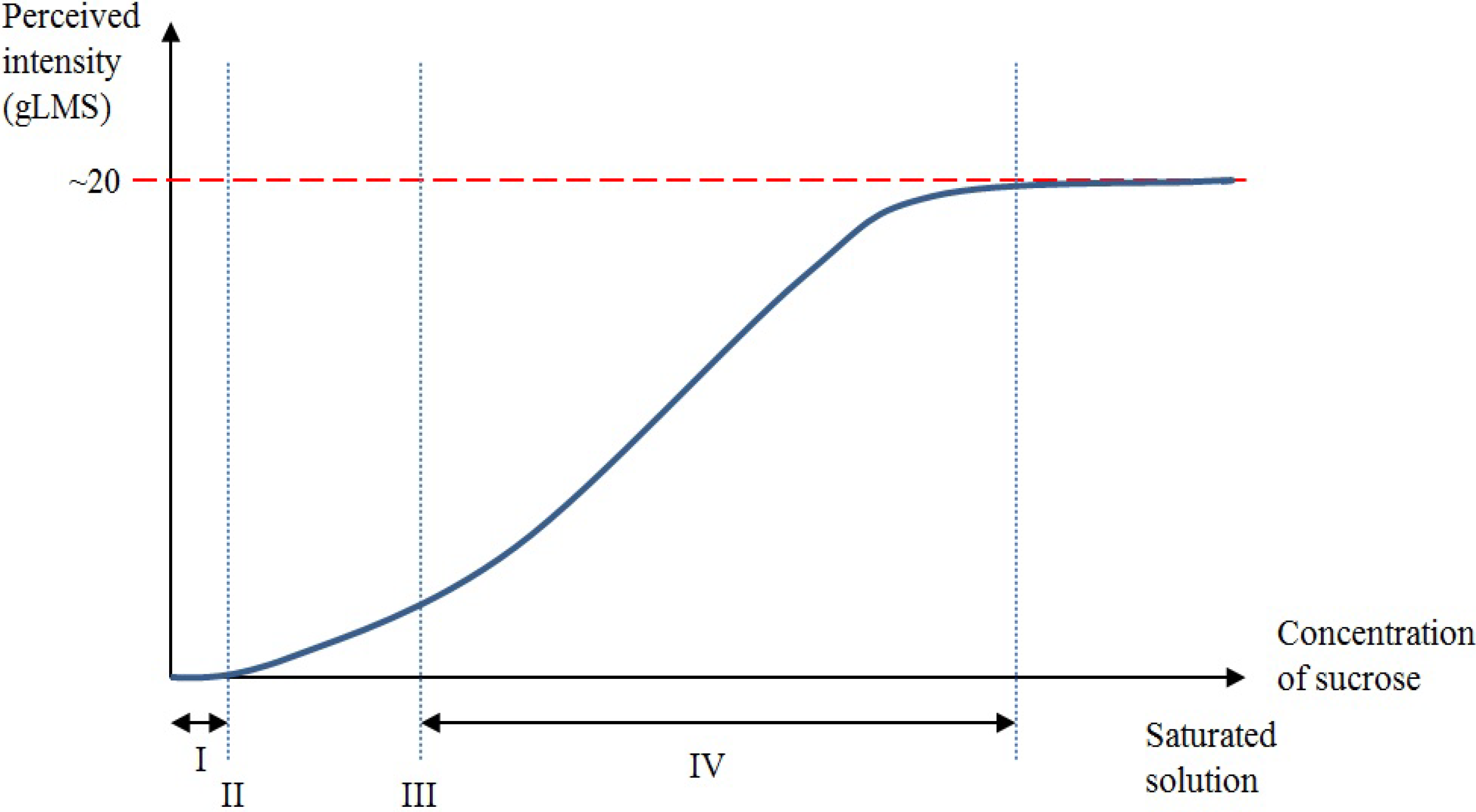
3. Peripheral and Central Mechanisms for Sweet Taste Detection
3.1. Peripheral Mechanisms for Sweet Taste Detection
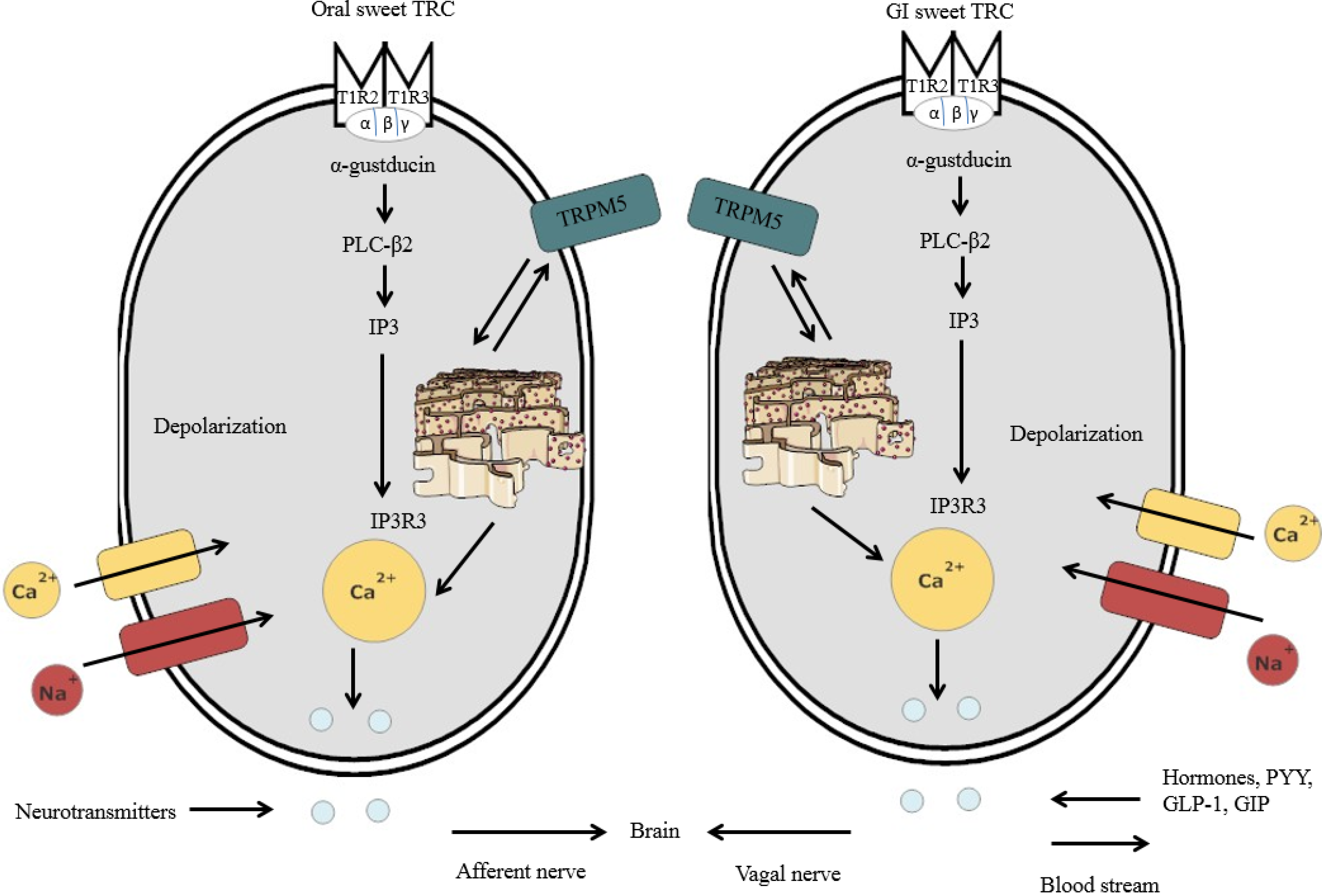
3.2. Sweet Taste Detection in the Oral Cavity
3.3. Sweet Taste Detection in the GIT
3.4. HIS and Their Role in the GIT
3.5. Sweet Taste Processing in the Brain
3.6. Are HIS Equally as Rewarding to the Brain as Caloric Sugar?
4. Possible Functions of Sweet Taste Perception
Individual Differences in Oral Sweet Taste Sensitivity
5. Implications of Decreased Oral and GI Sweet Taste Sensitivity and Perception
5.1. Implications of Oral Sweet Taste Sensitivity on Body Mass Index
5.2. Sweet Taste and Appetite
5.3. Links between Oral Sweet Taste Perception of Caloric Sweeteners and High Intensity Sweeteners and Satiation
5.4. Links between Attenuated Oral Sweet Taste Perception, the Gastrointestinal System and Satiety
6. Conclusions
Author Contributions
Conflicts of Interest
References
- World Health Organization. Obesity and Overweight. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/ (accessed on 30 May 2014).
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; AlMazroa, M.A.; Amann, M.; Anderson, H.R.; Andrews, K.G.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the global burden of disease study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef] [PubMed]
- Finucane, M.M.; Stevens, G.A.; Cowan, M.J.; Danaei, G.; Lin, J.K.; Paciorek, C.J.; Singh, G.M.; Gutierrez, H.R.; Lu, Y.; Bahalim, A.N.; et al. National, regional, and global trends in body-mass index since 1980: Systematic analysis of health examination surveys and epidemiological studies with 960 country-years and 9.1 million participants. Lancet 2011, 377, 557–567. [Google Scholar]
- Kelly, T.; Yang, W.; Chen, C.; Reynolds, K.; He, J. Global burden of obesity in 2005 and projections to 2030. Int. J. Obes. 2008, 32, 1431–1437. [Google Scholar] [CrossRef]
- Swinburn, B.A.; Sacks, G.; Hall, K.D.; McPherson, K.; Finegood, D.T.; Moodie, M.L.; Gortmaker, S.L. The global obesity pandemic: Shaped by global drivers and local environments. Lancet 2011, 378, 804–814. [Google Scholar] [CrossRef] [PubMed]
- Bellisle, F.; Drewnowski, A. Intense sweeteners, energy intake and the control of body weight. Eur. J. Clin. Nutr. 2007, 61, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A. Energy density, palatability, and satiety: Implications for weight control. Nutr. Rev. 1998, 56, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Bermudez, O.I.; Gao, X. Greater consumption of sweetened beverages and added sugars is associated with obesity among US young adults. Ann. Nutr. Metab. 2010, 57, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Malik, V.S.; Schulze, M.B.; Hu, F.B. Intake of sugar-sweetened beverages and weight gain: A systematic review. Am. J. Clin. Nutr. 2006, 84, 274–288. [Google Scholar] [PubMed]
- MacGregor, G.A.; Hashem, K.M. Action on sugar—Lessons from UK salt reduction programme. Lancet 2014, 383, 929–931. [Google Scholar] [CrossRef] [PubMed]
- Soenen, S.; Westerterp-Plantenga, M.S. No differences in satiety or energy intake after high-fructose corn syrup, sucrose, or milk preloads. Am. J. Clin. Nutr. 2007, 86, 1586–1594. [Google Scholar] [PubMed]
- Benton, D. Can artificial sweeteners help control body weight and prevent obesity? Nutr. Res. Rev. 2005, 18, 63–76. [Google Scholar] [CrossRef]
- Swithers, S.E.; Martin, A.A.; Davidson, T.L. High-intensity sweeteners and energy balance. Physiol. Behav. 2010, 100, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, L.B.; Møller, P.; Flint, A.; Martens, M.; Raben, A. Effect of sensory perception of foods on appetite and food intake: A review of studies on humans. Int. J. Obes. 2003, 27, 1152–1166. [Google Scholar]
- Johnson, R.J.; Segal, M.S.; Sautin, Y.; Nakagawa, T.; Feig, D.I.; Kang, D.-H.; Gersch, M.S.; Benner, S.; Sánchez-Lozada, L.G. Potential role of sugar (fructose) in the epidemic of hypertension, obesity and the metabolic syndrome, diabetes, kidney disease, and cardiovascular disease. Am. J. Clin. Nutr. 2007, 86, 899–906. [Google Scholar] [PubMed]
- Miller, P.; Perez, V. Low-calorie sweeteners and body weight and composition: A meta-analysis of randomized controlled trials and prospective cohorts (391.1). FASEB J. 2014, 28, 765–777. [Google Scholar]
- Mattes, R.D.; Popkin, B.M. Non-nutritive sweetener consumption in humans: Effects on appetite and food intake and their putative mechanisms. Am. J. Clin. Nutr. 2009, 89, 1–14. [Google Scholar] [PubMed]
- Bachmanov, A.A.; Beauchamp, G.K. Taste receptor genes. Annu. Rev. Nutr. 2006, 27, 389–414. [Google Scholar] [CrossRef]
- Keast, R.S.; Roper, J. A complex relationship among chemical concentration, detection threshold, and suprathreshold intensity of bitter compounds. Chem. Senses 2007, 32, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Simon, S.A.; de Araujo, I.E.; Gutierrez, R.; Nicolelis, M.A. The neural mechanisms of gustation: A distributed processing code. Nat. Rev. Neurosci. 2006, 7, 890–901. [Google Scholar] [CrossRef] [PubMed]
- Newman, L.; Haryono, R.; Keast, R. Functionality of fatty acid chemoreception: A potential factor in the development of obesity? Nutrients 2013, 5, 1287–1300. [Google Scholar] [CrossRef] [PubMed]
- Keast, R.S.; Breslin, P.A. An overview of binary taste-taste interactions. Food Qual. Prefer. 2003, 14, 111–124. [Google Scholar] [CrossRef]
- Beglinger, C.; Degen, L. Gastrointestinal satiety signals in humans—Physiologic roles for GLP-1 and PYY? Physiol. Behav. 2006, 89, 460–464. [Google Scholar]
- Dyer, J.; Salmon, K.; Zibrik, L.; Shirazi-Beechey, S. Expression of sweet taste receptors of the T1R family in the intestinal tract and enteroendocrine cells. Biochem. Soc. Trans. 2005, 33, 302–305. [Google Scholar] [CrossRef] [PubMed]
- Raybould, H.E. Gut chemosensing: Interactions between gut endocrine cells and visceral afferents. Auton. Neurosci. 2010, 153, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Sclafani, A. Sweet taste signaling in the gut. Proc. Natl. Acad. Sci. USA 2007, 104, 14887–14888. [Google Scholar] [CrossRef] [PubMed]
- Höfer, D.; Püschel, B.; Drenckhahn, D. Taste receptor-like cells in the rat gut identified by expression of alpha-gustducin. Proc. Natl. Acad. Sci. USA 1996, 93, 6631–6634. [Google Scholar]
- Bezençon, C.; le Coutre, J.; Damak, S. Taste-signaling proteins are coexpressed in solitary intestinal epithelial cells. Chem. Senses 2007, 32, 41–49. [Google Scholar]
- Kojima, I.; Nakagawa, Y. The role of the sweet taste receptor in enteroendocrine cells and pancreatic β-cells. Diabetes Metab. J. 2011, 35, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Rozengurt, E.; Sternini, C. Taste receptor signaling in the mammalian gut. Curr. Opin. Pharmacol. 2007, 7, 557–562. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.E.; Feinle-Bisset, C.; Keast, R.S. Fatty acid detection during food consumption and digestion: Associations with ingestive behavior and obesity. Prog. Lipid Res. 2011, 50, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Nelson, G.; Hoon, M.A.; Chandrashekar, J.; Zhang, Y.; Ryba, N.J.; Zuker, C.S. Mammalian sweet taste receptors. Cell 2001, 106, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Cui, M.; Jiang, P.; Maillet, E.; Max, M.; Margolskee, R.F.; Osman, R. The heterodimeric sweet taste receptor has multiple potential ligand binding sites. Curr. Pharm. Des. 2006, 12, 4591–4600. [Google Scholar] [CrossRef] [PubMed]
- Margolskee, R.F. Molecular mechanisms of bitter and sweet taste transduction. J. Biol. Chem. 2002, 277, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Nie, Y.; Vigues, S.; Hobbs, J.R.; Conn, G.L.; Munger, S.D. Distinct contributions of T1R2 and T1R3 taste receptor subunits to the detection of sweet stimuli. Curr. Biol. 2005, 15, 1948–1952. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Staszewski, L.; Tang, H.; Adler, E.; Zoller, M.; Li, X. Different functional roles of T1R subunits in the heteromeric taste receptors. Proc. Natl. Acad. Sci. USA 2004, 101, 14258–14263. [Google Scholar] [PubMed]
- Prinster, S.C.; Hague, C.; Hall, R.A. Heterodimerization of G protein-coupled receptors: Specificity and functional significance. Pharmacol. Rev. 2005, 57, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Pin, J.-P.; Galvez, T.; Prézeau, L. Evolution, structure, and activation mechanism of family 3/C G-protein-coupled receptors. Pharmacol. Ther. 2003, 98, 325–354. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Cui, M.; Zhao, B.; Liu, Z.; Snyder, L.A.; Benard, L.M.; Osman, R.; Margolskee, R.F.; Max, M. Lactisole interacts with the transmembrane domains of human T1R3 to inhibit sweet taste. J. Biol. Chem. 2005, 280, 15238–15246. [Google Scholar] [CrossRef] [PubMed]
- Morini, G.; Bassoli, A.; Temussi, P.A. From small sweeteners to sweet proteins: Anatomy of the binding sites of the human T1R2-T1R3 receptor. J. Med. Chem. 2005, 48, 5520–5529. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Ji, Q.; Liu, Z.; Snyder, L.A.; Benard, L.M.; Margolskee, R.F.; Max, M. The cysteine-rich region of T1R3 determines responses to intensely sweet proteins. J. Biol. Chem. 2004, 279, 45068–45075. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Cui, M.; Zhao, B.; Snyder, L.A.; Benard, L.M.; Osman, R.; Max, M.; Margolskee, R.F. Identification of the cyclamate interaction site within the transmembrane domain of the human sweet taste receptor subunit T1R3. J. Biol. Chem. 2005, 280, 34296–34305. [Google Scholar] [CrossRef] [PubMed]
- Wong, G.T.; Gannon, K.S.; Margolskee, R.F. Transduction of bitter and sweet taste by gustducin. Nature 1996, 381, 796–800. [Google Scholar] [CrossRef] [PubMed]
- Rössler, P.; Kroner, C.; Freitag, J.; Noè, J.; Breer, H. Identification of a phospholipase C beta subtype in rat taste cells. Eur. J. Cell Biol. 1998, 77, 253–261. [Google Scholar]
- Pierce-Shimomura, J.T.; Faumont, S.; Gaston, M.R.; Pearson, B.J.; Lockery, S.R. The homeobox gene lim-6 is required for distinct chemosensory representations in C. elegans. Nature 2001, 410, 694–698. [Google Scholar]
- Prawitt, D.; Monteilh-Zoller, M.K.; Brixel, L.; Spangenberg, C.; Zabel, B.; Fleig, A.; Penner, R. TRPM5 is a transient Ca2+-activated cation channel responding to rapid changes in [Ca2+]i. Proc. Natl. Acad. Sci. USA 2003, 100, 15166–15171. [Google Scholar] [CrossRef] [PubMed]
- Just, T.; Pau, H.W.; Engel, U.; Hummel, T. Cephalic phase insulin release in healthy humans after taste stimulation? Appetite 2008, 51, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, Y.; Nagasawa, M.; Yamada, S.; Hara, A.; Mogami, H.; Nikolaev, V.O.; Lohse, M.J.; Shigemura, N.; Ninomiya, Y.; Kojima, I. Sweet taste receptor expressed in pancreatic β-cells activates the calcium and cyclic AMP signaling systems and stimulates insulin secretion. PLoS One 2009, 4, e5106. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.J.; Rother, K.I. Non-nutritive sweeteners and their role in the gastrointestinal tract. J. Clin. Endocrinol. Metab. 2012, 97, 2597–2605. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.-J.; Kokrashvili, Z.; Theodorakis, M.J.; Carlson, O.D.; Kim, B.-J.; Zhou, J.; Kim, H.H.; Xu, X.; Chan, S.L.; Juhaszova, M. Gut-expressed gustducin and taste receptors regulate secretion of glucagon-like peptide-1. Proc. Natl. Acad. Sci. USA 2007, 104, 15069–15074. [Google Scholar] [CrossRef] [PubMed]
- Egan, J.M.; Margolskee, R.F. Taste cells of the gut and gastrointestinal chemosensation. Mol. Interv. 2008, 8, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Renwick, A.G.; Molinary, S.V. Sweet-taste receptors, low-energy sweeteners, glucose absorption and insulin release. Br. J. Nutr. 2010, 104, 1415–1420. [Google Scholar] [CrossRef] [PubMed]
- Margolskee, R.F.; Dyer, J.; Kokrashvili, Z.; Salmon, K.S.; Ilegems, E.; Daly, K.; Maillet, E.L.; Ninomiya, Y.; Mosinger, B.; Shirazi-Beechey, S.P. T1R3 and gustducin in gut sense sugars to regulate expression of Na+-glucose cotransporter 1. Proc. Natl. Acad. Sci. USA 2007, 104, 15075–15080. [Google Scholar] [CrossRef] [PubMed]
- Mace, O.J.; Affleck, J.; Patel, N.; Kellett, G.L. Sweet taste receptors in rat small intestine stimulate glucose absorption through apical GLUT2. J. Physiol. 2007, 582, 379–392. [Google Scholar] [CrossRef] [PubMed]
- Whitehouse, C.R.; Boullata, J.; McCauley, L.A. The potential toxicity of artificial sweeteners. Am. Assoc. Occup. Health Nurses J. 2008, 56, 251–259. [Google Scholar]
- Chandrashekar, J.; Hoon, M.A.; Ryba, N.J.; Zuker, C.S. The receptors and cells for mammalian taste. Nature 2006, 444, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Fujita, Y.; Wideman, R.D.; Speck, M.; Asadi, A.; King, D.S.; Webber, T.D.; Haneda, M.; Kieffer, T.J. Incretin release from gut is acutely enhanced by sugar but not by sweeteners in vivo. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E473–E479. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Bellon, M.; Wishart, J.M.; Young, R.; Blackshaw, L.A.; Jones, K.L.; Horowitz, M.; Rayner, C.K. Effect of the artificial sweetener, sucralose, on gastric emptying and incretin hormone release in healthy subjects. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 296, G735–G739. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Chang, J.; Checklin, H.L.; Young, R.L.; Jones, K.L.; Horowitz, M.; Rayner, C.K. Effect of the artificial sweetener, sucralose, on small intestinal glucose absorption in healthy human subjects. Br. J. Nutr. 2010, 104, 803–806. [Google Scholar] [CrossRef] [PubMed]
- Ford, H.; Peters, V.; Martin, N.; Sleeth, M.; Ghatei, M.; Frost, G.; Bloom, S. Effects of oral ingestion of sucralose on gut hormone response and appetite in healthy normal-weight subjects. Eur. J. Clin. Nutr. 2011, 65, 508–513. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.A.; Akiba, Y.; Kaunitz, J.D. Recent advances in gut nutrient chemosensing. Curr. Med. Chem. 2012, 19, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Dyer, J.; Hosie, K.; Shirazi-Beechey, S. Nutrient regulation of human intestinal sugar transporter (SGLT1) expression. Gut 1997, 41, 56–59. [Google Scholar] [PubMed]
- Reimann, F.; Habib, A.M.; Tolhurst, G.; Parker, H.E.; Rogers, G.J.; Gribble, F.M. Glucose sensing in L cells: A primary cell study. Cell Metab. 2008, 8, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Gribble, F.M.; Williams, L.; Simpson, A.K.; Reimann, F. A novel glucose-sensing mechanism contributing to glucagon-like peptide-1 secretion from the glutag cell line. Diabetes 2003, 52, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Moriya, R.; Shirakura, T.; Ito, J.; Mashiko, S.; Seo, T. Activation of sodium-glucose cotransporter 1 ameliorates hyperglycemia by mediating incretin secretion in mice. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E1358–E1365. [Google Scholar] [CrossRef] [PubMed]
- Sclafani, A.; Ackroff, K. Role of gut nutrient sensing in stimulating appetite and conditioning food preferences. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2012, 302, R1119–R1133. [Google Scholar] [CrossRef] [PubMed]
- Janssen, S.; Depoortere, I. Nutrient sensing in the gut: New roads to therapeutics? Trends Endocrinol. Metab. 2013, 24, 92–100. [Google Scholar]
- Tomchik, S.M.; Berg, S.; Kim, J.W.; Chaudhari, N.; Roper, S.D. Breadth of tuning and taste coding in mammalian taste buds. J. Neurosci. 2007, 27, 10840–10848. [Google Scholar] [CrossRef] [PubMed]
- O’Doherty, J.P.; Deichmann, R.; Critchley, H.D.; Dolan, R.J. Neural responses during anticipation of a primary taste reward. Neuron 2002, 33, 815–826. [Google Scholar]
- Kelley, A.E.; Berridge, K.C. The neuroscience of natural rewards: Relevance to addictive drugs. J. Neurosci. 2002, 22, 3306–3311. [Google Scholar] [PubMed]
- Zheng, H.; Berthoud, H.-R. Eating for pleasure or calories. Curr. Opin. Pharmacol. 2007, 7, 607–612. [Google Scholar] [CrossRef] [PubMed]
- Berthoud, H.-R.; Morrison, C. The brain, appetite, and obesity. Annu. Rev. Psychol. 2008, 59, 55–92. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, C. Tricking Taste Buds but Not the Brain: Artificial Sweeteners Change Brain’s Pleasure Response to Sweet. Available online: http://blogs.scientificamerican.com/mind-guest-blog/2013/09/05/tricking-taste-buds-but-not-the-brain-artificial-sweeteners-changes-brains-pleasure-response-to-sweet/ (accessed on 30 May 2014).
- Smeets, P.A.; Weijzen, P.; de Graaf, C.; Viergever, M.A. Consumption of caloric and non-caloric versions of a soft drink differentially affects brain activation during tasting. Neuroimage 2011, 54, 1367–1374. [Google Scholar] [CrossRef] [PubMed]
- Frank, G.K.; Oberndorfer, T.A.; Simmons, A.N.; Paulus, M.P.; Fudge, J.L.; Yang, T.T.; Kaye, W.H. Sucrose activates human taste pathways differently from artificial sweetener. Neuroimage 2008, 39, 1559–1569. [Google Scholar] [CrossRef] [PubMed]
- Green, E.; Murphy, C. Altered processing of sweet taste in the brain of diet soda drinkers. Physiol. Behav. 2012, 107, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Westerterp, K.R. Perception, passive overfeeding and energy metabolism. Physiol. Behav. 2006, 89, 62–65. [Google Scholar] [CrossRef] [PubMed]
- Bartoshuk, L.M.; Duffy, V.B.; Hayes, J.E.; Moskowitz, H.R.; Snyder, D.J. Psychophysics of sweet and fat perception in obesity: Problems, solutions and new perspectives. Philos. Trans. R. Soc. B Biol. Sci. 2006, 361, 1137–1148. [Google Scholar] [CrossRef]
- Duffy, V.B. Variation in oral sensation: Implications for diet and health. Curr. Opin. Gastroenterol. 2007, 23, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Cicerale, S.; Riddell, L.J.; Keast, R.S. The association between perceived sweetness intensity and dietary intake in young adults. J. Food Sci. 2012, 77, H31–H35. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Roth, R. Relationship between taste sensitivity and eating style in Japanese female university students. Food Nutr. Sci. 2012, 3, 302–309. [Google Scholar] [CrossRef]
- Lim, J.; Urban, L.; Green, B.G. Measures of individual differences in taste and creaminess perception. Chem. Senses 2008, 33, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Fushan, A.A.; Simons, C.T.; Slack, J.P.; Drayna, D. Association between common variation in genes encoding sweet taste signaling components and human sucrose perception. Chem. Senses 2010, 35, 579–592. [Google Scholar] [CrossRef] [PubMed]
- Keskitalo, K.; Knaapila, A.; Kallela, M.; Palotie, A.; Wessman, M.; Sammalisto, S.; Peltonen, L.; Tuorila, H.; Perola, M. Sweet taste preferences are partly genetically determined: Identification of a trait locus on chromosome 16. Am. J. Clin. Nutr. 2007, 86, 55–63. [Google Scholar] [PubMed]
- Knaapila, A.; Tuorila, H.; Silventoinen, K.; Wright, M.J.; Kyvik, K.O.; Keskitalo, K.; Hansen, J.; Kaprio, J.; Perola, M. Environmental effects exceed genetic effects on perceived intensity and pleasantness of several odors: A three-population twin study. Behav. Genet. 2008, 38, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.-H.; Zhang, H.-Y.; Wang, X.-F.; Zhan, Y.-H.; Deng, S.-P.; Qin, Y.-M. The relationship between fungiform papillae density and detection threshold for sucrose in the young males. Chem. Senses 2009, 34, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Keller, K.L.; Olsen, A.; Cravener, T.L.; Bloom, R.; Chung, W.K.; Deng, L.; Lanzano, P.; Meyermann, K. Bitter taste phenotype and body weight predict children’s selection of sweet and savory foods at a palatable test-meal. Appetite 2014, 77, 115–123. [Google Scholar] [CrossRef]
- Turner-McGrievy, G.; Tate, D.F.; Moore, D.; Popkin, B. Taking the bitter with the sweet: Relationship of supertasting and sweet preference with metabolic syndrome and dietary intake. J. Food Sci. 2013, 78, S336–S342. [Google Scholar]
- Hellfritsch, C.; Brockhoff, A.; Stähler, F.; Meyerhof, W.; Hofmann, T. Human psychometric and taste receptor responses to steviol glycosides. J. Agric. Food Chem. 2012, 60, 6782–6793. [Google Scholar] [CrossRef] [PubMed]
- Wiet, S.G.; BEYTS, P.K. Sensory characteristics of sucralose and other high intensity sweeteners. J. Food Sci. 1992, 57, 1014–1019. [Google Scholar] [CrossRef]
- Umabiki, M.; Tsuzaki, K.; Kotani, K.; Nagai, N.; Sano, Y.; Matsuoka, Y.; Kitaoka, K.; Okami, Y.; Sakane, N.; Higashi, A. The improvement of sweet taste sensitivity with decrease in serum leptin levels during weight loss in obese females. Tohoku J. Exp. Med. 2010, 220, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Felsted, J.; O’Malley, S.; Nachtigal, D.; Gant, P.; Small, D. Relationships between BMI, perceived pleasantness and ad lib consumption of food in smokers and non-smokers. In List of Abstracts from the Twenty-Ninth Annual Meeting of the Association for Chemoreception Sciences; Chemical Senses: Oxford, UK, 2007; p. A17. [Google Scholar]
- Grinker, J.; Hirsch, J.; Smith, D.V. Taste sensitivity and susceptibility to external influence in obese and normal weight subjects. J. Personal. Soc. Psychol. 1972, 22, 320–325. [Google Scholar] [CrossRef]
- Malcolm, R.; O’Neil, P.; Hirsch, A.; Currey, H.; Moskowitz, G. Taste hedonics and thresholds in obesity. Int. J. Obes. 1979, 4, 203–212. [Google Scholar]
- Navabi, N.; Hamzeh, F. Investigation of the relationship between sweet taste sensitivity and body mass index. Daneshvar Med. 2008, 16, 57–62. [Google Scholar]
- Frijters, J.E.; Rasmussen-Conrad, E.L. Sensory discrimination, intensity perception, and affective judgment of sucrose-sweetness in the overweight. J. Gen. Psychol. 1982, 107, 233–247. [Google Scholar] [CrossRef] [PubMed]
- Rodin, J.; Moskowitz, H.R.; Bray, G.A. Relationship between obesity, weight loss, and taste responsiveness. Physiol. Behav. 1976, 17, 591–597. [Google Scholar] [CrossRef] [PubMed]
- Blundell, J.; Hill, A. Paradoxical effects of an intense sweetener (aspartame) on appetite. Lancet 1986, 327, 1092–1093. [Google Scholar] [CrossRef]
- Rogers, P.J.; Blundell, J.E. Separating the actions of sweetness and calories: Effects of saccharin and carbohydrates on hunger and food intake in human subjects. Physiol. Behav. 1989, 45, 1093–1099. [Google Scholar] [CrossRef] [PubMed]
- Rogers, P.J.; Carlyle, J.-A.; Hill, A.J.; Blundell, J.E. Uncoupling sweet taste and calories: Comparison of the effects of glucose and three intense sweeteners on hunger and food intake. Physiol. Behav. 1988, 43, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Tordoff, M.G.; Alleva, A.M. Oral stimulation with aspartame increases hunger. Physiol. Behav. 1990, 47, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Mattes, R. Effects of aspartame and sucrose on hunger and energy intake in humans. Physiol. Behav. 1990, 47, 1037–1044. [Google Scholar] [CrossRef] [PubMed]
- Rolls, B.J.; Kim, S.; Fedoroff, I.C. Effects of drinks sweetened with sucrose or aspartame on hunger, thirst and food intake in men. Physiol. Behav. 1990, 48, 19–26. [Google Scholar] [PubMed]
- Canty, D.J.; Chan, M.M. Effects of consumption of caloric vs. noncaloric sweet drinks on indices of hunger and food consumption in normal adults. Am. J. Clin. Nutr. 1991, 53, 1159–1164. [Google Scholar]
- Drewnowski, A.; Massien, C.; Louis-Sylvestre, J.; Fricker, J.; Chapelot, D.; Apfelbaum, M. Comparing the effects of aspartame and sucrose on motivational ratings, taste preferences, and energy intakes in humans. Am. J. Clin. Nutr. 1994, 59, 338–345. [Google Scholar] [PubMed]
- Holt, S.H.; Sandona, N.; Brand-Miller, J.C. The effects of sugar-free vs. sugar-rich beverages on feelings of fullness and subsequent food intake. Int. J. Food Sci. Nutr. 2000, 51, 59–71. [Google Scholar]
- Monneuse, M.-O.; Bellisle, F.; Louis-Sylvestre, J. Responses to an intense sweetener in humans: Immediate preference and delayed effects on intake. Physiol. Behav. 1991, 49, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Pérez, C.; Dalix, A.-M.; Guy-Grand, B.; Bellisle, F. Human responses to five concentrations of sucrose in a dairy product: Immediate and delayed palatability effects. Appetite 1994, 23, 165–178. [Google Scholar]
- Griffioen-Roose, S.; Hogenkamp, P.S.; Mars, M.; Finlayson, G.; de Graaf, C. Taste of a 24-h diet and its effect on subsequent food preferences and satiety. Appetite 2012, 59, 1–8. [Google Scholar]
- Rolls, B.J.; Rolls, E.T.; Rowe, E.A.; Sweeney, K. Sensory specific satiety in man. Physiol. Behav. 1981, 27, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Rolls, B.J.; Hetherington, M.; Laster, L.J. Comparison of the effects of aspartame and sucrose on appetite and food intake. Appetite 1988, 11, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Appleton, K.; Rogers, P.; Blundell, J. Effects of a sweet and a non-sweet lunch on short-term appetite: Differences in female high and low consumers of sweet/low-energy beverages. J. Hum. Nutr. Diet. 2004, 17, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Appleton, K.; Blundell, J. Habitual high and low consumers of artificially-sweetened beverages: Effects of sweet taste and energy on short-term appetite. Physiol. Behav. 2007, 92, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Perry, B.; Wang, Y. Appetite regulation and weight control: The role of gut hormones. Nutr. Diabetes 2012, 2, e26. [Google Scholar] [CrossRef] [PubMed]
- Le Roux, C.W.; Welbourn, R.; Werling, M.; Osborne, A.; Kokkinos, A.; Laurenius, A.; Lönroth, H.; Fändriks, L.; Ghatei, M.A.; Bloom, S.R. Gut hormones as mediators of appetite and weight loss after Roux-en-Y gastric bypass. Ann. Surg. 2007, 246, 780–785. [Google Scholar]
- Karamanakos, S.N.; Vagenas, K.; Kalfarentzos, F.; Alexandrides, T.K. Weight loss, appetite suppression, and changes in fasting and postprandial ghrelin and peptide-YY levels after Roux-en-Y gastric bypass and sleeve gastrectomy: A prospective, double blind study. Ann. Surg. 2008, 247, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Borg, C.; le Roux, C.; Ghatei, M.; Bloom, S.; Patel, A.; Aylwin, S. Progressive rise in gut hormone levels after Roux-en-Y gastric bypass suggests gut adaptation and explains altered satiety. Br. J. Surg. 2006, 93, 210–215. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Low, Y.Q.; Lacy, K.; Keast, R. The Role of Sweet Taste in Satiation and Satiety. Nutrients 2014, 6, 3431-3450. https://doi.org/10.3390/nu6093431
Low YQ, Lacy K, Keast R. The Role of Sweet Taste in Satiation and Satiety. Nutrients. 2014; 6(9):3431-3450. https://doi.org/10.3390/nu6093431
Chicago/Turabian StyleLow, Yu Qing, Kathleen Lacy, and Russell Keast. 2014. "The Role of Sweet Taste in Satiation and Satiety" Nutrients 6, no. 9: 3431-3450. https://doi.org/10.3390/nu6093431
APA StyleLow, Y. Q., Lacy, K., & Keast, R. (2014). The Role of Sweet Taste in Satiation and Satiety. Nutrients, 6(9), 3431-3450. https://doi.org/10.3390/nu6093431




