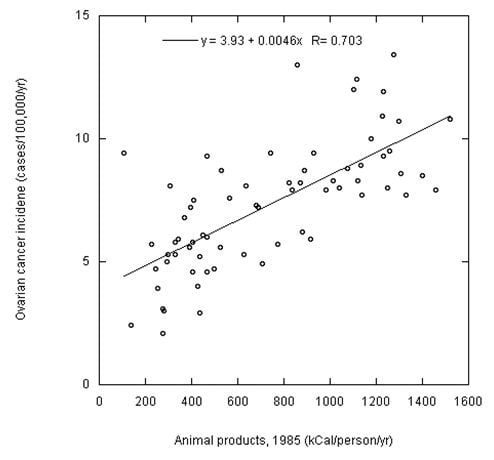
A Multicountry Ecological Study of Cancer Incidence Rates in 2008 with Respect to Various Risk-Modifying Factors
Abstract
:1. Introduction
- Tobacco use is the most important risk factor for cancer, causing 22% of global cancer deaths and 71% of global lung cancer deaths.
- Cancer-causing viral infections such as hepatitis B and C viruses and human papillomavirus are responsible for up to 20% of cancer deaths in low- and middle-income countries.
2. Data and Methods
3. Results
4. Discussion
| Factor | Animal Energy, 1980 | Animal Energy, 1985 (r, r2, p) | Animal Energy, 1990 (r, r2, p) | Animal Energy, 1993 (r, r2, p) | Animal Energy, 2000 (r, r2, p) | Animal Fat, 2000 (r, r2, p) | Cereals 1993 (r, r2, p) |
|---|---|---|---|---|---|---|---|
| Alcohol, 1993 | 0.62, 0.37 | 0.66, 0.43 | 0.71, 0.49 | 0.63, 0.38 | 0.65, 0.41 | 0.67, 0.44 | −0.51, 0.24 |
| Animal en, 1980 | 0.99, 0.98 | 0.96, 0.92 | 0.94, 0.87 | 0.94, 0.87 | 0.92, 0.85 | −0.59, 0.34 | |
| Animal en, 1985 | 0.98, 0.96 | 0.96, 0.92 | 0.94, 0.88 | 0.95, 0.90 | −0.61, 0.36 | ||
| Animal en, 1990 | 0.99, 0.97 | 0.95, 0.89 | 0.95, 0.90 | −0.61, 0.36 | |||
| Animal en, 1993 | 0.95, 0.91 | 0.95, 0.90 | −0.58, 0.33 | ||||
| Animal en 2000 | 0.99, 0.98 | −0.60, 0.36 | |||||
| Animal fat, 2000 | −0.60, 0.35 | ||||||
| Factor | Fat, 2000 (r, r2, p) | GDP 1999 (r, r2, p) | Latitude (r, r2, p) | Life Expectancy (r, r2, p) | Lung Cancer, M (r, r2, p) | Lung Cancer F (r, r2, p) | Sweeteners 1990 (r, r2, p) |
| Alcohol, 1993 | 0.63, 0.39 | 0.57, 0.31 | 0.51, 0.25 | 0.47, 0.21 | 0.53, 0.27 | 0.45, 0.19 | 0.13, 0.001, 0.30 |
| Animal en, 1980 | 0.81, 0.64 | 0.74, 0.54 | 0.79, 0.63 | 0.63, 0.38 | 0.61, 0.36 | 0.73, 0.53 | 0.29, 0.07, 0.02 |
| Animal en, 1985 | 0.84, 0.69 | 0.77, 0.59 | 0.79, 0.61 | 0.66, 0.43 | 0.63, 0.39 | 0.72, 0.51 | 0.28, 0.07, 0.02 |
| Animal en, 1990 | 0.85, 0.72 | 0.76, 0.57 | 0.77, 0.60 | 0.67, 0.43 | 0.65, 0.42 | 070, 0.49 | 0.23, 0.04, 0.06 |
| Animal en, 1993 | 0.85, 0.72 | 0.75, 0.55 | 0.67, 0.44 | 0.65, 0.42 | 0.51, 0.25 | 0.65, 0.41 | 0.17, 0.02, 0.16 |
| Animal en 2000 | 0.89, 0.79 | 0.79, 0.62 | 0.66, 0.43 | 0.66, 0.43 | 0.51, 0.25 | 0.67, 0.45 | 0.17, 0.02, 0.16 |
| Animal fat, 2000 | 0.89, 0.78 | 0.77, 0.59 | 0.66, 0.43 | 0.63, 0.39 | 0.51, 0.25 | 0.67, 0.45 | 0.17, 0.01, 0.18 |
| Cereals, 1993 | −0.53, 0.27 | −0.58, 0.33 | −0.14, 0.008, 0.19 | −0.62, 0.38 | −0.23, 0.04, 0.04 | −0.50, 0.24 | −0.35, 0.11, 0.003 |
| Fat, 2000 | 0.83, 0.68 | 0.53, 0.27 | 0.73, 0.53 | 0.46, 0.20 | 0.65, 0.41 | 0.14, 0.004, 0.27 | |
| GDP, 1999 | 0.41, 0.16 | 0.77, 0.59 | 0.26, 0.06, 0.02 | 0.67, 0.44 | 0.37, 0.12, 0.003 | ||
| Latitude | 0.24, 0.05, 0.02 | 0.63, 0.39 | 0.47, 0.21 | — | |||
| Life expectancy | 0.30, 0.08, 0.005 | 0.55, 0.29 | 0.29, 0.07, 0.02 | ||||
| Lung cancer, M, 2008 | 0.49, 0.23 | — | |||||
| Lung cancer, F, 2008 | 0.32, 0.09, 0.007 |
| Cancer | Sex | Number of Countries | Lung Cancer | Animal Energy | Latitude | GDP 1999 | Miscellaneous | Adjusted |
|---|---|---|---|---|---|---|---|---|
| (β, p) | (year; β, p) | (β, p) | (β, p) | (β, p) | r2, F, p | |||
| All | M | 82 | 0.40, * | 2000; 0.44, * | Alc93: 0.23, 0.003 | 0.77, 93, * | ||
| 84 | 0.38, * | 1993; 0.46, * | Alc93: 0.20, 0.997 | 0.77, 91, * | ||||
| F | 87 | 0.49, * | 1993; 0.33, * | Cer93: −0.20, 0.002 | 0.77, 99, * | |||
| 82 | 0.53, * | 1993; 0.30, * | Alc93: 0.30, 0.004 | 0.77, 93, * | ||||
| All Less Lung | M | 62 | 0.23, 0.01 | 1980; 0.54, * | 0.21, 0.04 | 0.74, 58, * | ||
| 87 | 0.28, * | 1993; 0.52, * | Cer93: −0.21, 0.006 | 0.69, 63, * | ||||
| F | 62 | 0.40, * | 1990; 0.44, * | 0.10, 0.38 | 0.72, 52, * | |||
| 82 | 0.43, * | 1993; 0.34, * | Alc93: 0.22, 0.004 | 0.71, 67, * | ||||
| 82 | 0.43, * | 0.22, 0.004 | Alc93: 0.20, 0.013Cer00: −0.25, 0.001 | 0.71, 51, * | ||||
| Bladder | M | 79 | 0.43, * | 0.30, 0.003 | 0.23, 0.006 | 0.58, 37, * | ||
| F | 80 | 0.48, * | 0.26, 0.003 | 0.17, 0.09 | 0.58, 37, * | |||
| Brain | M | 79 | 0.58, * | 0.26, 0.004 | 0.52, 44, * | |||
| 79 | 1993; 0.26, 0.004 | 0.58, * | 0.51, 41, * | |||||
| F | 85 | 0.33, 0.001 | 0.38, * | 0.35, 24, * | ||||
| Breast | F | 80 | 0.23, 0.009 | 1993; 0.41, * | 0.31, 0.002 | 0.72, 67, * | ||
| Cervix | F | 80 | −0.40, * | −0.33, 0.001 | 0.37, 24, * | |||
| Colorectal | M | 77 | 0.40, * | 0.31, * | Alc93: 0.39, * | 0.75, 76, * | ||
| F | 67 | 0.37, * | 1985; 0.17, 0.12 | Alc93: 0.24, 0.003LE: 0.28, 0.001 | 0.77, 56, * | |||
| 82 | 0.33, * | 0.25, 0.001 | Alc93: 0.25, 0.001LE: 0.28, * | 0.74, 58, * | ||||
| Corpus uteri | F | 68 | 1985; 0.67, * | 0.44, 53, * | ||||
| Hodgkin’s Lymphoma | M | 82 | 0.25, 0.02 | Fat00: 0.48, * | 0.40, 28, * | |||
| F | 78 | 0.33, 0.005 | Fat00: 0.29, 0.01 | 0.26, 15, * | ||||
| Kidney | M | 87 | 0.27, 0.004 | 0.50, * | Cer93: −0.25, 0.001 | 0.60, 44, * | ||
| 87 | 0.27, 0.003 | 1993; 0.32, 0.001 | 0.32, 0.003 | 0.60, 43, * | ||||
| 87 | 0.40, * | 1993; 0.47, * | 0.56, 55, * | |||||
| F | 85 | 0.30, 0.001 | 0.52, * | Cer93: −0.17, 0.04 | 0.60, 42, * | |||
| 85 | 0.30, 0.002 | 1993; 0.23, 0.04 | 0.39, * | 0.60, 42, * | ||||
| Laryngeal | M | 85 | 0.86, * | 2000; −0.33, * | 0.55, 52, * | |||
| F | 78 | 0.48, 0.001 | 2000; −0.39, 0.008 | 0.12, 6, * | ||||
| Leukemia | M | 86 | 0.43, * | LE: 0.44, * | 0.47, 39, * | |||
| 80 | 0.46, * | 0.40, * | 0.46, 35, * | |||||
| F | 86 | 0.30, 0.006 | LE: 0.36, 0.001 | 0.32, 21 * | ||||
| Lip, oral | M | 87 | 0.53, * | 0.28, 34, * | ||||
| F | 86 | 0.51, * | −0.31, 0.007 | 0.19, 11, * | ||||
| Liver | M | 67 | 1980; −0.27, 0.03 | 0.06, 5, 0.03 | ||||
| F | 67 | 1980; −0.51, * | 0.25, 23, * | |||||
| Lung | M | 54 | CigM: 0.65, * | AF93; 0.36, * | 0.73, 72, * | |||
| 54 | CigM: 0.63, * | 0.35, * | 0.72, 67, * | |||||
| 54 | CigM: 0.79, * | 0.62, 89, * | ||||||
| 54 | Cig: 0.80, * | 0.62, 86, * | ||||||
| F | 54 | CigF: 0.65,* | AF93; 0.32, * | 0.77, 92, * | ||||
| 54 | CigF: 0.64, * | 010, 0.35 | AF93: 0.26, 0.018 | 0.77, 61, * | ||||
| 54 | CigF: 0.70, * | 0.26, 0.003 | 0.75, 81, * | |||||
| 54 | CigF: 0.85, * | 0.71, 131, * | ||||||
| Lat × Lat | ||||||||
| Melanoma | M | 53 ** | 0.41, * | 0.52, * | 0.72, 68, * | |||
| 54 *** | 0.48, * | 0.43, * | 0.69, 59, * | |||||
| 52 ** | 0.40, * | 0.53, * | 0.72, 67, * | |||||
| 53 *** | 0.45, * | 0.47, * | 0.70, 63, * | |||||
| Multiple myeloma | M | 64 | 85; 0.57, * | LE: 0.27, 0.02 | 0.59, 46, * | |||
| 58 | 85; 0.53, * | 0.29, 0.04 | 0.59, 42, * | |||||
| F | 77 | 0.44, * | LE: 0.37, 0.003 | 0.58, 54, * | ||||
| 77 | 0.74, * | 0.53, 88, * | ||||||
| Non-Hodgkin’s lymphoma | M | 80 | 0.75, * | 0.55, 97, * | ||||
| F | 80 | 0.73, * | 0.54, 91, * | |||||
| Ovarian | F | 68 | 85; 0.70, * | 0.49, 65, * | ||||
| Pancreatic | M | 86 | 0.71, * | 1993; 0.16, 0.04 | 0.64, 78, * | |||
| 86 | 0.80, * | 0.63, 144, * | ||||||
| F | 86 | 0.40, 0.001 | 1993; 0.31, 0.008 | 0.39, 29, * | ||||
| Prostate | M | 68 | −0.26, 0.014 | 80; 0.62, * | Cer93: −0.36, 0.001 | 0.59, 33, * | ||
| Testicular | M | 59 | 85; 0.66, * | 0.25, 0.023 | 0.74, 82, * | |||
| Thyroid | M | 82 | 1993; 0.74, * | −0.29, 0.023 | 0.33, 20, * | |||
| F | 84 | 1993; 0.58, * | −0.25, 0.07 | 0.19, 10, * |
| Cancer | Sex | Number of Countries | Lung Cancer | Animal Energy | Latitude | GDP 1999 | Miscellaneous | Miscellaneous | Adjusted |
|---|---|---|---|---|---|---|---|---|---|
| (β, p) | (year; β, p) | (β, p) | (β, p) | (β, p) | (β, p) | r2, F, p | |||
| Bladder | M | 140 | 0.40, * | 0.36, * | 0.19, 0.002 | 0.66, 91, * | |||
| F | 139 | 0.37, * | 0.25, 0.001 | 0.22, 0.009 | 0.52, 50, * | ||||
| Brain | M | 126 | 1990; 0.26, 0.007 | 0.42, * | LE: 0.27, * | 0.71, 101, * | |||
| 130 | 0.16, 0.036 | 1990; 0.37, * | 0.38, * | 0.68, 91, * | |||||
| 145 | 0.59, * | LE: 0.35, * | 0.67, 149, * | ||||||
| 130 | 1990; 0.44, * | 0.44, * | 0.67, 131, * | ||||||
| 126 | 1090; 0.58, * | LE: 0.27, 0.001 | 0.64, 111, * | ||||||
| F | 147 | 0.30, * | 0.43, * | Sweet93: 0.18, 0.006 | 0.54, 59, * | ||||
| 148 | 0.36, * | 0.47, * | 0.53, 83, * | ||||||
| Breast | F | 142 | 0.22, * | 1993; 0.49, * | 0.26, 0.001 | 0.76, 147, * | |||
| 142 | 0.28, * | 0.17, * | 0.51, * | 0.70, 111, * | |||||
| Cervix | F | 142 | −0.40, * | −0.30, * | 0.38, 44, * | ||||
| Colorectal | M | 122 | 0.38, * | 1985; 0.21, 0.013 | 0.25, * | Alc93: 0.22, * | 0.84, 160, * | ||
| 138 | 0.35, * | 0.17, 0.007 | 0.26, * | Alc93: 0.31, * | 0.80, 141, * | ||||
| F | 131 | 0.36, * | 1985; 0.31, * | LE: 0.14, 0.02 | Alc93: 0.21, * | 0.77, 112, * | |||
| 136 | 0.35, * | 1985; 0.40, * | Alc93: 0.23, * | 0.74, 127, * | |||||
| Corpus uteri | F | 137 | 0.38, * | 1990; 0.37, * | Sweet90: 0.21, 0.001 | 0.68, 97, * | |||
| Hodgkin’s lymphoma | M | 141 | 0.26, 0.003 | Fat00: 0.44, * | 0.41, 49, * | ||||
| F | 132 | 0.33, * | Fat00: 0.36, * | 0.39, 43, * | |||||
| Kidney | M | 153 | 0.32, * | 0.54, * | Cer93: −0.26, * | 0.70, 122, * | |||
| 152 | 0.25, * | 1993; 0.39, * | 0.30, * | 0.70, 119, * | |||||
| 154 | 0.37, * | 1993; 0.53, * | 0.67, 153, * | ||||||
| 153 | 0.36, * | 0.51, * | 0.64, 135, * | ||||||
| F | 153 | 0.24, * | 1993; 0,21, 0.018 | 0.45, * | Cer93: −0.15, 0.007 | 0.69, 84, * | |||
| 153 | 0.30, * | 0.57, * | Cer93: −0.21, * | 0.68, 106, * | |||||
| 153 | 0.24, * | 1993; 0.34, * | 0.35, * | 0.67, 105, * | |||||
| 153 | 0.37, * | 0.53, * | 0.64, 133, * | ||||||
| 154 | 0.28, * | 1993; 0.57, * | 0.62, 123, * | ||||||
| Laryngeal | M | 157 | 0.68, * | 0.46, 131, * | |||||
| F | 139 | 0.31, * | 0.09, 15, * | ||||||
| Leukemia | M | 147 | 0.32, * | 2000; 0.38, * | LE: 0.20, 0.016 | 0.62, 78, * | |||
| F | 151 | 0.37, * | LE: 0.45, * | 0.52, 81, * | |||||
| 146 | 0.32, * | 2000; 0.15, 0.15 | LE: 0.36, * | 0.52, 52, * | |||||
| Lip, oral | M | 157 | 0.45, * | −0.20, 0.06 | 0.11, 10, * | ||||
| F | 156 | 0.29, 0.002 | −0.29, 0.003 | 0.06, 6, 0.003 | |||||
| Liver | M | 136 | 1980; −0.37, * | 0.13, 21, * | |||||
| F | 136 | 1980; −0.44, * | 0.19, 33, * | ||||||
| Lung | M | 68 | CigM; 0.62, * | 0.33, * | 0.68, 72, * | ||||
| 68 | CigM; 0.63, * | AF00: 0.34, * | 0.67, 68, * | ||||||
| 68 | CigM; 0.60, * | 0.23, 0.04 | AF00: 0.14, 0.22 | 0.68, 49, * | |||||
| F | 71 | CigF; 0.69, * | AF93; 0.26, 0.001 | 0.78, 123, * | |||||
| 71 | CigF; 0.74, * | 0.21, 0.004 | 0.77, 118, * | ||||||
| Multiple myeloma | M | 112 | 1985; 0.61, * | 0.22, 0.05 | 0.64, 100, * | ||||
| 122 | 1985; 0.66, * | LE: 0.17, 0.05 | 0.61, 97, * | ||||||
| F | 110 | 1990; 0.33, 0.003 | 0.49, * | 0.61, 88, * | |||||
| 129 | 0.55, * | LE: 0.28, 0.001 | 0.60, 85, * | ||||||
| Non-Hodgkin’s lymphoma | M | 142 | 0.68, * | 0.46, 120, * | |||||
| F | 141 | 0.71, * | 0.50, 140, * | ||||||
| Ovarian | F | 138 | 0.25, 0.002 | 1990; 0.55, * | 0.55, 85, * | ||||
| Sweet90: | |||||||||
| Pancreatic | M | 132 | 0.42, * | 1985; 0.28, 0.001 | Alc93: 0.18, 0.005 | 0.15, 0.013 | 0.74, 93, * | ||
| 132 | 0.45, * | 1990; 0.39, * | 0.15, 0.013 | 0.73, 116, * | |||||
| F | 132 | 0.26, 0.001 | 1990; 0.46, * | 0.20, 0.003 | 0.64, 80, * | ||||
| Prostate | M | 137 | 1980; 0.52, * | Cer93: −0.23, * | Sweet90: 0.18, 0.019 | 0.56, 58, * | |||
| 132 | 0.45, * | 1990; 0.39, * | 0.15, 0.013 | 0.73, 116, * | |||||
| F | 132 | 0.26, 0.001 | 1990; 0.46, * | 0.20, 0.003 | 0.64, 80, * | ||||
| Testicular | M | 123 | 1985; 0.85, * | 0.72, 312, * | |||||
| Thyroid | M | 146 | 1993; 0.78, * | −0.26, 0.009 | 0.36, 42, * | ||||
| F | 152 | 1993; 0.60, * | −0.27, 0.014 | 0.18, 18, * |
| Factor | All Countries M | All Countries F | All Countries M/F | HQ Countries M | HQ Countries F | HQ Countries M/F |
|---|---|---|---|---|---|---|
| Direct correlation | ||||||
| Alcohol | 1 | 1 | 1 | 1 | 1 | 1 |
| Animal fat | 2 | 1 | 2 | 2 | 1 | 2 |
| Animal product energy | 9 | 7 | 12 | 5 | 6 | 10 |
| Fat | 1 | 1 | 1 | 1 | 1 | 1 |
| GDP | 4 | 4 | 5 | 5 | 3 | 7 |
| Latitude | 6 | 4 | 6 | 5 | 6 | 6 |
| Life expectancy | 1 | 2 | 3 | 2 | 3 | 3 |
| Lung cancer | 8 | 11 | 12 | 8 | 9 | 10 |
| Sweeteners | 1 | 2 | 3 | 0 | 0 | 0 |
| Inverse correlation | ||||||
| Animal product energy | 0 | 0 | 0 | 2 | 2 | 2 |
| Cereals | 2 | 1 | 2 | 2 | 1 | 2 |
| GDP | 0 | 1 | 1 | 0 | 1 | 1 |
| Latitude | 2 | 3 | 3 | 0 | 2 | 2 |
4.1. Latitude
4.2. GDP
4.3. Life Expectancy
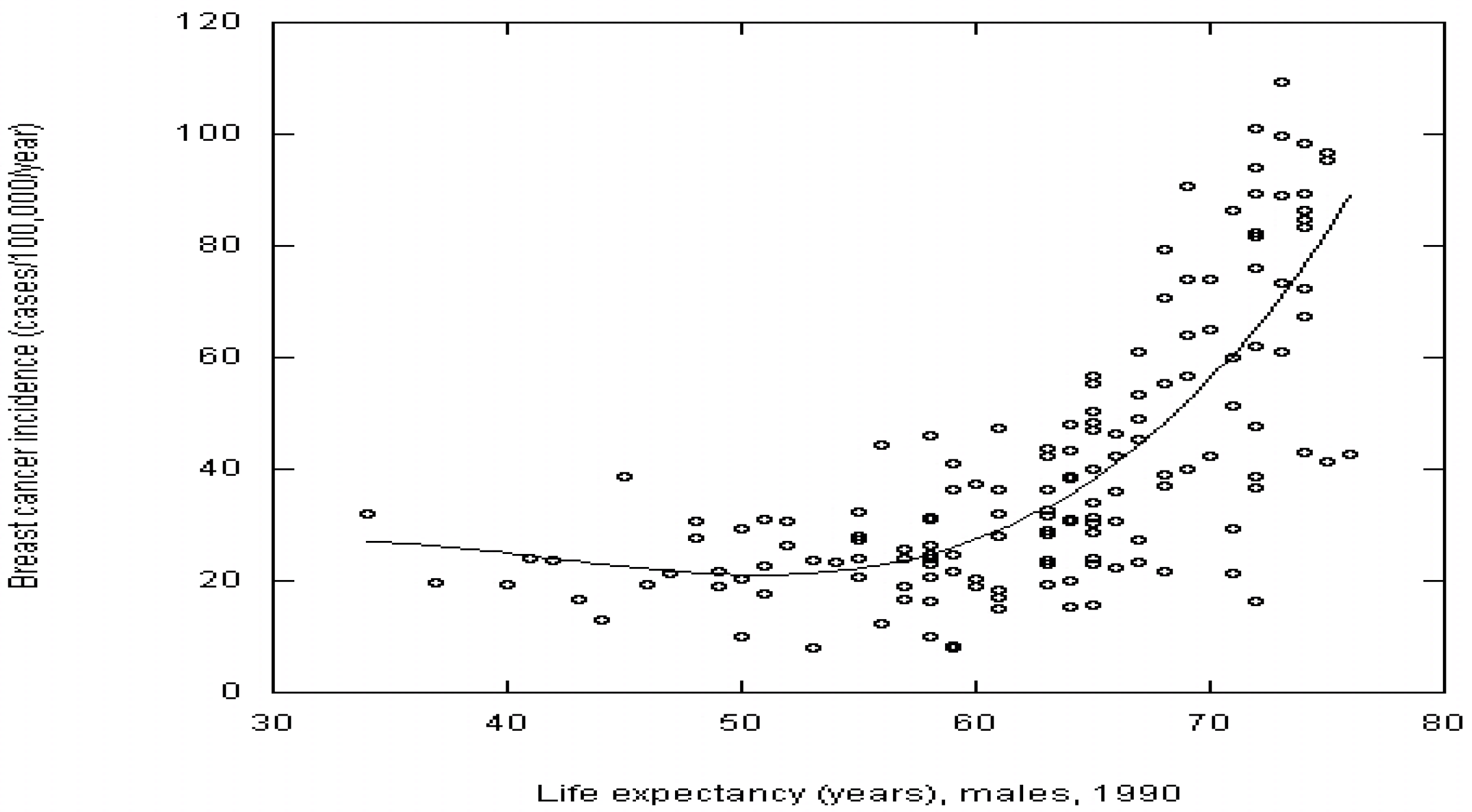
4.4. Individual Cancers
| Cancer | Animal Products | Smoking | GDP per Capita | Latitude * | Alcohol | Misccellaneous | Infection |
|---|---|---|---|---|---|---|---|
| Bladder | [78,79] | [80,81] | [82] | [46] UVB | |||
| Brain | [83] | [84] | [47,67] | [67,85] | |||
| Breast | [86,87] | [82,88] | UVB | ||||
| Cervix | [80] | [89] | |||||
| Colorectal | [78,90,91] | [80] | [49,82,88] | UVB | [92,93] | Sugar [94] | [89] |
| Corpus uteri | [78] | [95] | uvb | Sugar [96] | |||
| Esophageal | [90,91,97] | [80,81] | uvb | [92] | [98] | ||
| Gallbladder | uvb | ||||||
| Gastric | [81] | uvb | [99] | ||||
| Hodgkin’s lymphoma | [100] | uvb | [101] | ||||
| Kidney | [79] | [102] | [82] | [51,103] UVB | Cereals [104,105] | ||
| Laryngeal | [80,81] | uvb | [92] | [106] | |||
| Leukemia | [107] | [107] | [55] | ||||
| Lip | [108] | [108] (direct) | |||||
| Liver | [74,90] | [81,102] | [109] | ||||
| Lung | [87,91,110] | [81] | [88] | [52] uvb | |||
| Mult. Myel. | [100,111] | [112] | |||||
| Non-Hodgkin’s lymphoma | [113,114] | [82,88] | UVB | [115] | |||
| Ovarian | [78,116,117] | [118] | [82] | [53] UVB | |||
| Pancreatic | [78,87,119] | [80,81] | [88] | [54] UVB | [92,120] | Sugar [94] | |
| Prostate | [79] | [121] (inverse) | [82,88] | uvb | [122] | Sugar [94], rice [121] | |
| Testicular | [79] | Fat [123] | |||||
| Thyroid | [124] | uvb |
4.5. Strengths and Limitations of This Study
5. Conclusions
Conflicts of Interest
References
- Ferlay, J.; Shin, H.R.; Bray, F.; Forman, D.; Mathers, C.; Parkin, D.M. GLOBOCAN 2008, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 10. Int. J. Cancer. 2010, 127. Available online: http://globocan.iarc.fr/factsheet.asp (accessed on 29 August 2013). [Google Scholar]
- Siegel, R.; Ward, E.; Brawley, O.; Jemal, A. Cancer statistics, 2011: The impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J. Clin. 2011, 61, 212–236. [Google Scholar] [CrossRef]
- Hoyert, D.L.; Xu, J. Deaths: Preliminary data for 2011. Nat. Vital Stat. Rep. 2012, 61, 8. [Google Scholar]
- Cancer. Available online: http://www.who.int/mediacentre/factsheets/fs297/en/ (accessed on 4 November 2013).
- The American Institute for Cancer Research/World Cancer Research Fund. Policy and Action for Cancer Prevention. Food, Nutrition and Physical Activity: A Global Perspective; AICR: Washington, DC, USA, 2009. [Google Scholar]
- Armstrong, B.; Doll, R. Environmental factors and cancer incidence and mortality in different countries, with special reference to dietary practices. Int. J. Cancer 1975, 15, 617–631. [Google Scholar] [CrossRef]
- Garland, C.F.; Garland, F.C. Do sunlight and vitamin D reduce the likelihood of colon cancer? Int. J. Epidemiol. 1980, 9, 227–231. [Google Scholar] [CrossRef]
- Garland, C.F.; Garland, F.C.; Gorham, E.D.; Lipkin, M.; Newmark, H.; Mohr, S.B.; Holick, M.F. The role of vitamin D in cancer prevention. Am. J. Public Health 2006, 96, 252–261. [Google Scholar] [CrossRef]
- Grant, W.B.; Garland, C.F. The association of solar ultraviolet B (UVB) with reducing risk of cancer: Multifactorial ecologic analysis of geographic variation in age-adjusted cancer mortality rates. Anticancer Res. 2006, 26, 2687–2699. [Google Scholar]
- Grant, W.B. Ecological studies of the UVB—Vitamin D—Cancer hypothesis. Anticancer Res. 2012, 32, 223–236. [Google Scholar]
- Garland, C.F.; Gorham, E.D.; Mohr, S.B.; Garland, F.C. Vitamin D for cancer prevention: Global perspective. Ann. Epidemiol. 2009, 19, 468–483. [Google Scholar] [CrossRef]
- Moukayed, M.; Grant, W.B. Molecular link between vitamin D and cancer prevention. Nutrients 2013, 5, 3993–4021. [Google Scholar] [CrossRef]
- Hill, A.B. The environment and disease: Association or causation? Proc. Royal Soc. Med. 1965, 58, 295–300. [Google Scholar]
- Grant, W.B. How strong is the evidence that solar ultraviolet B and vitamin D reduce the risk of cancer? An examination using Hill’s criteria for causality. Dermato-Endocrinology 2009, 1, 17–24. [Google Scholar] [CrossRef]
- Mohr, S.B.; Gorham, E.D.; Alcaraz, J.E.; Kane, C.I.; Macera, C.A.; Parsons, J.K.; Wingard, D.L.; Garland, C.F. Does the evidence for an inverse relationship between serum vitamin D status and breast cancer risk satisfy the Hill criteria? Dermato-Endocrinology 2012, 4, 152–157. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. Food Balance Sheets, 1992–94 Average; FAO: Rome, Italy, 1997. [Google Scholar]
- Harashima, E.; Nakagawa, Y.; Urata, G.; Tsuji, K.; Shirataka, M.; Matsumura, Y. Time-lag estimate between dietary intake and breast cancer mortality in Japan. Asia Pac. J. Clin. Nutr. 2007, 16, 193–198. [Google Scholar]
- Zhang, J.; Dhakal, I.B.; Zhao, Z.; Li, L. Trends in mortality from cancers of the breast, colon, prostate, esophagus, and stomach in East Asia: Role of nutrition transition. Eur. J. Cancer Prev. 2012, 21, 480–489. [Google Scholar] [CrossRef]
- World Health Organization. Tables of Causes of Death and Disability Adjusted Life Years by Country 2004. Available online: http://www.who.int/healthinfo/statistics/bodgbddeathdalyestimates.xls (accessed on 5 November 2013).
- Mathers, C.D.; Bernard, C.; Iburg, K.; Inoue, M.; Ma Fat, D.; Shibuya, K.; Stein, C.; Tomijima, N. The Global Burden of Disease in 2002: Data Sources, Methods and Results. In GPE Discussion Paper No. 54; World Health Organization: Geneva, Swizerland, 2003. [Google Scholar]
- Food and Agriculture Organization of the United Nations. Food Balance Sheets. Available online: http://faostat.fao.org/site/368/default.aspx#ancor (accessed on 4 November 2013).
- Grant, W.B. Role of solar UVB irradiance and smoking in cancer as inferred from cancer incidence rates by occupation in Nordic countries. Dermato-Endocrinology 2012, 4, 203–211. [Google Scholar] [CrossRef]
- Grant, W.B. An ecologic study of cancer mortality rates in Spain with respect to indices of solar UVB irradiance and smoking. Int. J. Cancer 2007, 120, 1123–1128. [Google Scholar] [CrossRef]
- Bilal, U.; Fernandez, E.; Beltran, P.; Navas-Acien, A.; Bolumar, F.; Franco, M. Validation of a method for reconstructing historical rates of smoking prevalence. Am. J. Epidemiol. 2013. [Google Scholar] [CrossRef]
- Afzal, S.; Bojesen, S.E.; Nordestgaard, B.G. Low plasma 25-hydroxyvitamin D and risk of tobacco-related cancer. Clin. Chem. 2013, 59, 771–780. [Google Scholar]
- Data on Tobacco Consumption. Available online: http://www.who.int/tobacco/media/en/Poland.pdf (accessed on 20 December 2013).
- International Monetary Fund. List of Countries by Past and Future GDP (PPP) per Capita. Available online: http://en.wikipedia.org/w/index.php?title=List_of_countries_by_pastand_futur_GDP_(PPP)_per_capita&oldid=574426146 (accessed on 4 November 2013).
- Central Intelligence Agency. The CIA World Fact Book 2008; Central Intelligence Agency: Washington, DC, USA, 2008. [Google Scholar]
- KaleidaGraph, version 4.2; Synergy Software: Reading, PA, USA, 2005.
- Tsuji, K.; Harashima, E.; Nakagawa, Y.; Urata, G.; Shirataka, M. Time-lag effect of dietary fiber and fat intake ratio on Japanese colon cancer mortality. Biomed. Environ. Sci. 1996, 9, 223–228. [Google Scholar]
- Andersson, S.O.; Baron, J.; Wolk, A.; Lindgren, C.; Bergström, R.; Adami, H.O. Early life risk factors for prostate cancer: A population-based case-control study in Sweden. Cancer Epidemiol. Biomark. Prev. 1995, 4, 187–192. [Google Scholar]
- John, E.M.; Koo, J.; Schwartz, G.G. Sun exposure and prostate cancer risk: Evidence for a protective effect of early-life exposure. Cancer Epidemiol. Biomark. Prev. 2007, 16, 1283–1286. [Google Scholar] [CrossRef]
- Hirayama, T. Mortality in Japanese with life-styles similar to Seventh-Day Adventists: Strategy for risk reduction by life-style modification. Nat. Cancer Inst. Monogr. 1985, 69, 143–153. [Google Scholar]
- Fraser, G.E. Associations between diet and cancer, ischemic heart disease, and all-cause mortality in non-Hispanic white California Seventh-day Adventists. Am. J. Clin. Nutr. 1999, 70, 532–538. [Google Scholar]
- Thygesen, L.C.; Hvidt, N.C.; Hansen, H.P.; Hoff, A.; Ross, L.; Johansen, C. Cancer incidence among Danish Seventh-day Adventists and Baptists. Cancer Epidemiol. 2012, 36, 513–518. [Google Scholar]
- Cancer Increasing among Meat Eaters. New York Times. Sep 24. 1907. Available online: http://query.nytimes.com/gst/abstract.html?res=9502E1D81331E733A25757C2A96F9C946697D6CF (accessed on 24 December 2013).
- Patterson, J.T. The Dread Disease; Cancer and Modern Culture; Harvard University Press: Cambridge, MA, USA, 1987; pp. 44–45. [Google Scholar]
- McCarty, M.F. Mortality from Western cancers rose dramatically among African-Americans during the 20th century: Are dietary animal products to blame? Med. Hypotheses 2001, 57, 169–174. [Google Scholar] [CrossRef]
- Grant, W.B. Trends in diet and Alzheimer’s disease during the nutrition transition in Japan and developing countries. J. Alzheimerʼs Dis. 2013, 38, 611–620. [Google Scholar]
- Kaaks, R. Nutrition, insulin, IGF-1 metabolism and cancer risk: A summary of epidemiological evidence. Novartis Found. Symp. 2004, 262, 247–260; discussion 260–268. [Google Scholar] [CrossRef]
- Grant, W.B. An ecological study of cancer mortality rates including indices for dietary iron and zinc. Anticancer Res. 2008, 28, 1955–1963. [Google Scholar]
- McCarty, M.F. Insulin and IGF-I as determinants of low “Western” cancer rates in the rural third world. Int. J. Epidemiol. 2004, 33, 908–910. [Google Scholar] [CrossRef]
- Fontana, L.; Weiss, E.P.; Villareal, D.T.; Klein, S.; Holloszy, J.O. Long-term effects of calorie or protein restriction on serum IGF-1 and IGFBP-3 concentration in humans. Aging Cell 2008, 7, 681–687. [Google Scholar] [CrossRef]
- Allen, N.E.; Appleby, P.N.; Davey, G.K.; Kaaks, R.; Rinaldi, S.; Key, T.J. The associations of diet with serum insulin-like growth factor I and its main binding proteins in 292 women meat-eaters, vegetarians, and vegans. Cancer Epidemiol. Biomark. Prev. 2002, 11, 1441–1448. [Google Scholar]
- Dewell, A.; Weidner, G.; Sumner, M.D.; Barnard, R.J.; Marlin, R.O.; Daubenmier, J.J.; Chi, C.; Carroll, P.R.; Ornish, D. Relationship of dietary protein and soy isoflavones to serum IGF-1 and IGF binding proteins in the Prostate Cancer Lifestyle Trial. Nutr. Cancer 2007, 58, 35–42. [Google Scholar] [CrossRef]
- Mohr, S.B.; Garland, C.F.; Gorham, E.D.; Grant, W.B.; Garland, F.C. Ultraviolet B irradiance and incidence rates of bladder cancer in 174 countries. Am. J. Prev. Med. 2010, 38, 296–302. [Google Scholar] [CrossRef]
- Mohr, S.B.; Gorham, E.D.; Garland, C.F.; Grant, W.B.; Garland, F.C. Low ultraviolet B and increased risk of brain cancer: an ecological study of 175 countries. Neuroepidemiology 2010, 35, 281–290. [Google Scholar] [CrossRef]
- Mohr, S.B.; Garland, C.F.; Gorham, E.D.; Grant, W.B.; Garland, F.C. Relationship between low ultraviolet B irradiance and higher breast cancer risk in 107 countries. Breast J. 2008, 14, 255–260. [Google Scholar] [CrossRef]
- Cuomo, R.; Mohr, S.B.; Gorham, E.D.; Garland, C.F. What is the relationship between ultraviolet B and global incidence rates of colorectal cancer? Dermato-Endocrinology 2013, 5, 181–185. [Google Scholar] [CrossRef]
- Mohr, S.B.; Garland, C.F.; Gorham, E.D.; Grant, W.B.; Garland, F.C. Is ultraviolet B irradiance inversely associated with incidence rates of endometrial cancer: An ecological study of 107 countries. Prev. Med. 2007, 45, 327–331. [Google Scholar]
- Mohr, S.B.; Gorham, E.D.; Garland, C.F.; Grant, W.B.; Garland, F.C. Are low ultraviolet B and high animal protein intake associated with risk of renal cancer? Int. J. Cancer 2006, 119, 2705–2709. [Google Scholar] [CrossRef]
- Mohr, S.B.; Garland, C.F.; Gorham, E.D.; Grant, W.B.; Garland, F.C. Could ultraviolet B irradiance and vitamin D be associated with lower incidence rates of lung cancer? J. Epidemiol. Community Health 2008, 62, 69–74. [Google Scholar] [CrossRef]
- Garland, C.F.; Mohr, S.B.; Gorham, E.D.; Grant, W.B.; Garland, F.C. Role of ultraviolet B irradiance and vitamin D in prevention of ovarian cancer. Am. J. Prev. Med. 2006, 31, 512–514. [Google Scholar] [CrossRef]
- Mohr, S.B.; Garland, C.F.; Gorham, E.D.; Grant, W.B.; Garland, F.C. Ultraviolet B irradiance and vitamin D status are inversely associated with incidence rates of pancreatic cancer worldwide. Pancreas 2010, 39, 669–674. [Google Scholar] [CrossRef]
- Mohr, S.B.; Garland, C.F.; Gorham, E.D.; Grant, W.B.; Garland, F.C. Ultraviolet B and incidence rates of leukemia worldwide. Am. J. Prev. Med. 2011, 41, 68–74. [Google Scholar] [CrossRef]
- Mizoue, T. Ecological study of solar radiation and cancer mortality in Japan. Health Phys. 2004, 87, 532–538. [Google Scholar]
- Astbury, A. Non Uniformity in Cancer Mortality in the USA and Australia Appears to Share a Common Pathway. Triumf Report, TRI-PP-05–01, 2005. Available online: http://www.triumf.ca/publications/pub/arch05/pp-05–01.pdf (accessed on 21 November 2011).
- Chen, W.; Clements, M.; Rahman, B.; Zhang, S.; Qiao, Y.; Armstrong, B.K. Relationship between cancer mortality/incidence and ambient ultraviolet B irradiance in China. Cancer Causes Control 2010, 21, 1701–1709. [Google Scholar] [CrossRef]
- Fioletov, V.E.; McArthur, L.J.; Mathews, T.W.; Marrett, L. Estimated ultraviolet exposure levels for a sufficient vitamin D status in North America. J. Photochem. Photobiol. 2010, 100, 57–66. [Google Scholar] [CrossRef]
- Jablonski, N.G.; Chaplin, G. The evolution of human skin coloration. J. Hum. Evol. 2000, 39, 57–106. [Google Scholar]
- Jablonski, N.G.; Chaplin, G. Colloquium paper: Human skin pigmentation as an adaptation to UV radiation. Proc. Natl. Acad. Sci. USA 2010, 107, 8962–8968. [Google Scholar] [CrossRef]
- Hagenau, T.; Vest, R.; Gissel, T.N.; Poulsen, C.S.; Erlandsen, M.; Mosekilde, L.; Vestergaard, P. Global vitamin D levels in relation to age, gender, skin pigmentation and latitude: An ecologic meta-regression analysis. Osteoporos Int. 2009, 20, 133–140. [Google Scholar] [CrossRef]
- Van Schoor, N.M.; Lips, P. Worldwide vitamin D status. Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 671–680. [Google Scholar] [CrossRef]
- Hilger, J.; Friedel, A.; Herr, R.; Rausch, T.; Roos, F.; Wahl, D.A.; Pierroz, D.D.; Weber, P.; Hoffmann, K. A systematic review of vitamin D status in populations worldwide. Br. J. Nutr. 2013. [Google Scholar] [CrossRef]
- Kurtzke, J.F. Epidemiologic evidence for multiple sclerosis as an infection. Clin. Microbiol. Rev. 1993, 6, 382–427. [Google Scholar]
- Lucas, R.M.; Ponsonby, A.L.; Dear, K.; Valery, P.C.; Pender, M.P.; Taylor, B.V.; Kilpatrick, T.J.; Dwyer, T.; Coulthard, A.; Chapman, C.; et al. Sun exposure and vitamin D are independent risk factors for CNS demyelination. Neurology 2011, 76, 540–548. [Google Scholar] [CrossRef]
- Thomas, F.; Lafferty, K.D.; Brodeur, J.; Elguero, E.; Gauthier-Clerc, M.; Misse, D. Incidence of adult brain cancers is higher in countries where the protozoan parasite Toxoplasma gondii is common. Biol. Lett. 2012, 8, 101–103. [Google Scholar] [CrossRef]
- Basu, S.; Yoffe, P.; Hills, N.; Lustig, R.H. The relationship of sugar to population-level diabetes prevalence: An econometric analysis of repeated cross-sectional data. PLoS One 2013, 8, e57873. [Google Scholar]
- Bosdriesz, J.R.; Witvliet, M.I.; Visscher, T.L.; Kunst, A.E. The influence of the macro-environment on physical activity: A multilevel analysis of 38 countries worldwide. Int. J. Behav. Nutr. Phys. Act. 2012, 9, 110. [Google Scholar] [CrossRef]
- Hawkes, C. The role of foreign direct investment in the nutrition transition. Public Health Nutr. 2005, 8, 357–365. [Google Scholar] [CrossRef]
- Renton, A.; Wall, M.; Lintott, J. Economic growth and decline in mortality in developing countries: An analysis of the World Bank development datasets. Public Health 2012, 126, 551–560. [Google Scholar] [CrossRef]
- Huicho, L.; Trelles, M.; Gonzales, F.; Mendoza, W.; Miranda, J. Mortality profiles in a country facing epidemiological transition: An analysis of registered data. BMC Public Health 2009, 9, 47. [Google Scholar] [CrossRef]
- Shuman, E.K. Global climate change and infectious diseases. Int. J. Occup. Environ. Med. 2011, 2, 11–19. [Google Scholar]
- Freedman, N.D.; Cross, A.J.; McGlynn, K.A.; Abnet, C.C.; Park, Y.; Hollenbeck, A.R.; Schatzkin, A.; Everhart, J.E.; Sinha, R. Association of meat and fat intake with liver disease and hepatocellular carcinoma in the NIH-AARP cohort. J. Natl. Cancer Inst. 2010, 102, 1354–1365. [Google Scholar] [CrossRef]
- Fedirko, V.; Trichopolou, A.; Bamia, C.; Duarte-Salles, T.; Trepo, E.; Aleksandrova, K.; Nothlings, U.; Lukanova, A.; Lagiou, P.; Boffetta, P.; et al. Consumption of fish and meats and risk of hepatocellular carcinoma: The European Prospective Investigation into Cancer and Nutrition (EPIC). Ann. Oncol. 2013, 24, 2166–2173. [Google Scholar] [CrossRef]
- Bentham, G.; Aase, A. Incidence of malignant melanoma of the skin in Norway, 1955–1989: associations with solar ultraviolet radiation, income and holidays abroad. Int. J. Epidemiol. 1996, 25, 1132–1138. [Google Scholar] [CrossRef]
- Agredano, Y.Z.; Chan, J.L.; Kimball, R.C.; Kimball, A.B. Accessibility to air travel correlates strongly with increasing melanoma incidence. Melanoma Res. 2006, 16, 77–81. [Google Scholar]
- Tavani, A.; La Vecchia, C.; Gallus, S.; Lagiou, P.; Trichopoulos, D.; Levi, F.; Negri, E. Red meat intake and cancer risk: A study in Italy. Int. J. Cancer 2000, 86, 425–428. [Google Scholar] [CrossRef]
- Oplander, C.; Volkmar, C.M.; Paunel-Gorgulu, A.; van Faassen, E.E.; Heiss, C.; Kelm, M.; Halmer, D.; Murtz, M.; Pallua, N.; Suschek, C.V. Whole body UVA irradiation lowers systemic blood pressure by release of nitric oxide from intracutaneous photolabile nitric oxide derivates. Circ. Res. 2009, 105, 1031–1040. [Google Scholar] [CrossRef]
- Thun, M.J.; Henley, S.J.; Calle, E.E. Tobacco use and cancer: An epidemiologic perspective for geneticists. Oncogene 2002, 21, 7307–7325. [Google Scholar] [CrossRef]
- Yun, Y.H.; Jung, K.W.; Bae, J.M.; Lee, J.S.; Shin, S.A.; Min Park, S.; Yoo, T.; Yul Huh, B. Cigarette smoking and cancer incidence risk in adult men: National Health Insurance Corporation Study. Cancer Detect. Prev. 2005, 29, 15–24. [Google Scholar]
- Bray, F.; Jemal, A.; Grey, N.; Ferlay, J.; Forman, D. Global cancer transitions according to the Human Development Index (2008–2030): A population-based study. Lancet Oncol. 2012, 13, 790–801. [Google Scholar] [CrossRef]
- Kaplan, S.; Novikov, I.; Modan, B. Nutritional factors in the etiology of brain tumors: Potential role of nitrosamines, fat, and cholesterol. Am. J. Epidemiol. 1997, 146, 832–841. [Google Scholar] [CrossRef]
- Mandelzweig, L.; Novikov, I.; Sadetzki, S. Smoking and risk of glioma: A meta-analysis. Cancer Causes Control 2009, 20, 1927–1938. [Google Scholar]
- Alibek, K.; Kakpenova, A.; Baiken, Y. Role of infectious agents in the carcinogenesis of brain and head and neck cancers. Infect. Agents Cancer 2013, 8, 7. [Google Scholar] [CrossRef]
- Ishimoto, H.; Nakamura, H.; Miyoshi, T. Epidemiological study on relationship between breast cancer mortality and dietary factors. Tokushima J. Exp. Med. 1994, 41, 103–114. [Google Scholar]
- Hu, J.; La Vecchia, C.; DesMeules, M.; Negri, E.; Mery, L. Meat and fish consumption and cancer in Canada. Nutr. Cancer 2008, 60, 313–324. [Google Scholar] [CrossRef]
- Knaul, F.M.; Adami, H.O.; Adebamowo, C.; Arreola-Ornelas, H.; Berger, A.J.; Bhadelia, A.; Cleary, J.; Hunter, D.J.; Keating, N.; Mebewu, A.; et al. The Global Cancer Divide: An Equity Imperative. In Closing the Cancer Divide: An Equity Imperative; Harvard University: Cambridge, MA, USA, 2012. [Google Scholar]
- Zur Hausen, H. Papillomaviruses in the causation of human cancers—A brief historical account. Virology 2009, 384, 260–265. [Google Scholar] [CrossRef]
- Cross, A.J.; Leitzmann, M.F.; Gail, M.H.; Hollenbeck, A.R.; Schatzkin, A.; Sinha, R. A prospective study of red and processed meat intake in relation to cancer risk. PLoS Med. 2007, 4, e325. [Google Scholar] [CrossRef]
- Aune, D.; de Stefani, E.; Ronco, A.; Boffetta, P.; Deneo-Pellegrini, H.; Acosta, G.; Mendilaharsu, M. Meat consumption and cancer risk: A case-control study in Uruguay. Asian Pac. J. Cancer Prev. 2009, 10, 429–436. [Google Scholar]
- Haas, S.L.; Ye, W.; Lohr, J.M. Alcohol consumption and digestive tract cancer. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 457–467. [Google Scholar] [CrossRef]
- Nan, H.; Lee, J.E.; Rimm, E.B.; Fuchs, C.S.; Giovannucci, E.L.; Cho, E. Prospective study of alcohol consumption and the risk of colorectal cancer before and after folic acid fortification in the United States. Ann. Epidemiol. 2013, 23, 558–563. [Google Scholar] [CrossRef]
- Hu, J.; La Vecchia, C.; Augustin, L.S.; Negri, E.; de Groh, M.; Morrison, H.; Mery, L.; Canadian Cancer Registries Epidemiology Research Group. Glycemic index, glycemic load and cancer risk. Ann. Oncol. 2013, 24, 245–251. [Google Scholar] [CrossRef]
- Al-Zoughool, M.; Dossus, L.; Kaaks, R.; Clavel-Chapelon, F.; Tjonneland, A.; Olsen, A.; Overvad, K.; Boutron-Ruault, M.C.; Gauthier, E.; Linseisen, J.; et al. Risk of endometrial cancer in relationship to cigarette smoking: Results from the EPIC study. Int. J. Cancer 2007, 121, 2741–2747. [Google Scholar] [CrossRef]
- Zemla, B.; Guminski, S.; Franek, K.; Kolosza, Z.; Banasik, R. Etiological factors in invasive corpus uteri carcinoma. Neoplasma 1991, 38, 157–163. [Google Scholar]
- Choi, Y.; Song, S.; Song, Y.; Lee, J.E. Consumption of red and processed meat and esophageal cancer risk: Meta-analysis. World J. Gastroenterol. 2013, 19, 1020–1029. [Google Scholar] [CrossRef]
- Zheng, S.; Vuitton, L.; Sheyhidin, I.; Vuitton, D.A.; Zhang, Y.; Lu, X. Northwestern China: A place to learn more on oesophageal cancer. Part one: Behavioural and environmental risk factors. Eur. J. Gastroenterol. Hepatol. 2010, 22, 917–925. [Google Scholar] [CrossRef]
- Crew, K.D.; Neugut, A.I. Epidemiology of gastric cancer. World J. Gastroenterol. 2006, 12, 354–362. [Google Scholar]
- Tavani, A.; Pregnolato, A.; Negri, E.; Franceschi, S.; Serraino, D.; Carbone, A.; La Vecchia, C. Diet and risk of lymphoid neoplasms and soft tissue sarcomas. Nutr. Cancer 1997, 27, 256–260. [Google Scholar] [CrossRef]
- Hjalgrim, H. On the aetiology of Hodgkin lymphoma. Dan. Med. J. 2012, 59, B4485. [Google Scholar]
- Ray, G.; Henson, D.E.; Schwartz, A.M. Cigarette smoking as a cause of cancers other than lung cancer: An exploratory study using the Surveillance, Epidemiology, and End Results Program. Chest 2010, 138, 491–499. [Google Scholar] [CrossRef]
- Karami, S.; Boffetta, P.; Stewart, P.; Rothman, N.; Hunting, K.L.; Dosemeci, M.; Berndt, S.I.; Brennan, P.; Chow, W.H.; Moore, L.E. Occupational sunlight exposure and risk of renal cell carcinoma. Cancer 2010, 116, 2001–2010. [Google Scholar] [CrossRef]
- La Vecchia, C.; Chatenoud, L.; Negri, E.; Franceschi, S. Session: Whole cereal grains, fibre and human cancer wholegrain cereals and cancer in Italy. Proc. Nutr. Soc. 2003, 62, 45–49. [Google Scholar] [CrossRef]
- Daniel, C.R.; Park, Y.; Chow, W.H.; Graubard, B.I.; Hollenbeck, A.R.; Sinha, R. Intake of fiber and fiber-rich plant foods is associated with a lower risk of renal cell carcinoma in a large US cohort. Am. J. Clin. Nutr. 2013, 97, 1036–1043. [Google Scholar] [CrossRef]
- Li, X.; Gao, L.; Li, H.; Gao, J.; Yang, Y.; Zhou, F.; Gao, C.; Li, M.; Jin, Q. Human papillomavirus infection and laryngeal cancer risk: A systematic review and meta-analysis. J. Infect. Dis. 2013, 207, 479–488. [Google Scholar] [CrossRef]
- Ma, X.; Park, Y.; Mayne, S.T.; Wang, R.; Sinha, R.; Hollenbeck, A.R.; Schatzkin, A.; Cross, A.J. Diet, lifestyle, and acute myeloid leukemia in the NIH-AARP cohort. Am. J. Epidemiol. 2010, 171, 312–322. [Google Scholar] [CrossRef]
- Maruccia, M.; Onesti, M.G.; Parisi, P.; Cigna, E.; Troccola, A.; Scuderi, N. Lip cancer: A 10-year retrospective epidemiological study. Anticancer Res. 2012, 32, 1543–1546. [Google Scholar]
- Ringelhan, M.; Heikenwalder, M.; Protzer, U. Direct effects of hepatitis B virus-encoded proteins and chronic infection in liver cancer development. Dig. Dis. 2013, 31, 138–151. [Google Scholar] [CrossRef]
- Kubik, A.; Zatloukal, P.; Tomasek, L.; Dolezal, J.; Syllabova, L.; Kara, J.; Kopecky, P.; Plesko, I. A case-control study of lifestyle and lung cancer associations by histological types. Neoplasma 2008, 55, 192–199. [Google Scholar]
- Vlajinac, H.D.; Pekmezovic, T.D.; Adanja, B.J.; Marinkovic, J.M.; Kanazir, M.S.; Suvajdzic, N.D.; Colovic, M.D. Case-control study of multiple myeloma with special reference to diet as risk factor. Neoplasma 2003, 50, 79–83. [Google Scholar]
- McShane, C.M.; Murray, L.J.; Engels, E.A.; Landgren, O.; Anderson, L.A. Common community-acquired infections and subsequent risk of multiple myeloma: A population-based study. Int. J. Cancer 2013. [Google Scholar] [CrossRef]
- Zheng, T.; Holford, T.R.; Leaderer, B.; Zhang, Y.; Zahm, S.H.; Flynn, S.; Tallini, G.; Zhang, B.; Zhou, K.; Owens, P.H.; et al. Diet and nutrient intakes and risk of non-Hodgkin’s lymphoma in Connecticut women. Am. J. Epidemiol. 2004, 159, 454–466. [Google Scholar]
- Van Leeuwen, M.T.; Turner, J.J.; Falster, M.O.; Meagher, N.S.; Joske, D.J.; Grulich, A.E.; Giles, G.G.; Vajdic, C.M. Latitude gradients for lymphoid neoplasm subtypes in Australia support an association with ultraviolet radiation exposure. Int. J. Cancer 2013, 133, 944–951. [Google Scholar] [CrossRef]
- Liu, Y.C.; Yang, Y.H.; Hsiao, H.H.; Yang, W.C.; Liu, T.C.; Chang, C.S.; Yang, M.Y.; Lin, P.M.; Hsu, J.F.; Chang, P.Y.; et al. Herpes zoster is associated with an increased risk of subsequent lymphoid malignancies—A nationwide population-based matched-control study in Taiwan. BMC Cancer 2012, 12, 503. [Google Scholar] [CrossRef]
- Risch, H.A.; Jain, M.; Marrett, L.D.; Howe, G.R. Dietary fat intake and risk of epithelial ovarian cancer. J. Natl. Cancer Inst. 1994, 86, 1409–1415. [Google Scholar] [CrossRef]
- Kolahdooz, F.; Ibiebele, T.I.; van der Pols, J.C.; Webb, P.M. Dietary patterns and ovarian cancer risk. Am. J. Clin. Nutr. 2009, 89, 297–304. [Google Scholar]
- Beral, V.; Gaitskell, K.; Hermon, C.; Moser, K.; Reeves, G.; Peto, R. Ovarian cancer and smoking: Individual participant meta-analysis including 28,114 women with ovarian cancer from 51 epidemiological studies. Lancet Oncol. 2012, 13, 946–956. [Google Scholar] [CrossRef]
- Ghadirian, P.; Thouez, J.P.; PetitClerc, C. International comparisons of nutrition and mortality from pancreatic cancer. Cancer Detect. Prev. 1991, 15, 357–362. [Google Scholar]
- Anderson, M.A.; Zolotarevsky, E.; Cooper, K.L.; Sherman, S.; Shats, O.; Whitcomb, D.C.; Lynch, H.T.; Ghiorzo, P.; Rubinstein, W.S.; Vogel, K.J.; et al. Alcohol and tobacco lower the age of presentation in sporadic pancreatic cancer in a dose-dependent manner: A multicenter study. Am. J. Gastroenterol. 2012, 107, 1730–1739. [Google Scholar] [CrossRef]
- Severson, R.K.; Nomura, A.M.; Grove, J.S.; Stemmermann, G.N. A prospective study of demographics, diet, and prostate cancer among men of Japanese ancestry in Hawaii. Cancer Res. 1989, 49, 1857–1860. [Google Scholar]
- Sawada, N.; Inoue, M.; Iwasaki, M.; Sasazuki, S.; Yamaji, T.; Shimazu, T.; Tsugane, S. Alcohol and smoking and subsequent risk of prostate cancer in Japanese men: The Japan Public Health Center—Based prospective study. Int. J. Cancer 2014, 134, 971–978. [Google Scholar] [CrossRef]
- Sigurdson, A.J.; Chang, S.; Annegers, J.F.; Duphorne, C.M.; Pillow, P.C.; Amato, R.J.; Hutchinson, L.P.; Sweeney, A.M.; Strom, S.S. A case-control study of diet and testicular carcinoma. Nutr. Cancer 1999, 34, 20–26. [Google Scholar] [CrossRef]
- Markaki, I.; Linos, D.; Linos, A. The influence of dietary patterns on the development of thyroid cancer. Eur. J. Cancer 2003, 39, 1912–1919. [Google Scholar] [CrossRef]
- Grant, W.B. Update on evidence that support a role of solar ultraviolet-B irradiance in reducing cancer risk. Anti-Cancer Agents Med. Chem. 2013, 13, 140–146. [Google Scholar] [CrossRef]
- Bellack, N.R.; Koehoorn, M.W.; MacNab, Y.C.; Morshed, M.G. A conceptual model of water’s role as a reservoir in Helicobacter pylori transmission: A review of the evidence. Epidemiol. Infect. 2006, 134, 439–449. [Google Scholar] [CrossRef]
- Ferreccio, C.; Smith, A.H.; Duran, V.; Barlaro, T.; Benitez, H.; Valdes, R.; Aguirre, J.J.; Moore, L.E.; Acevedo, J.; Vasquez, M.I.; et al. Case-control study of arsenic in drinking water and kidney cancer in uniquely exposed Northern Chile. Am. J. Epidemiol. 2013, 178, 813–818. [Google Scholar] [CrossRef]
- Lopez-Abente, G.; Garcia-Perez, J.; Fernandez-Navarro, P.; Boldo, E.; Ramis, R. Colorectal cancer mortality and industrial pollution in Spain. BMC Public Health 2012, 12, 589. [Google Scholar]
- Petti, S.; Masood, M.; Scully, C. The magnitude of tobacco smoking-betel quid chewing-alcohol drinking interaction effect on oral cancer in South-East Asia. A meta-analysis of observational studies. PLoS One 2013, 8, e78999. [Google Scholar] [CrossRef]
Appendix
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Grant, W.B. A Multicountry Ecological Study of Cancer Incidence Rates in 2008 with Respect to Various Risk-Modifying Factors. Nutrients 2014, 6, 163-189. https://doi.org/10.3390/nu6010163
Grant WB. A Multicountry Ecological Study of Cancer Incidence Rates in 2008 with Respect to Various Risk-Modifying Factors. Nutrients. 2014; 6(1):163-189. https://doi.org/10.3390/nu6010163
Chicago/Turabian StyleGrant, William B. 2014. "A Multicountry Ecological Study of Cancer Incidence Rates in 2008 with Respect to Various Risk-Modifying Factors" Nutrients 6, no. 1: 163-189. https://doi.org/10.3390/nu6010163
APA StyleGrant, W. B. (2014). A Multicountry Ecological Study of Cancer Incidence Rates in 2008 with Respect to Various Risk-Modifying Factors. Nutrients, 6(1), 163-189. https://doi.org/10.3390/nu6010163