Obesity and Dyslipidemia in South Asians
Abstract
:1. Introduction
2. Definitions
3. Search Strategy
4. Guidelines for Diagnosis of Obesity
| Variable | Consensus guidelines for Asian Indians a | Prevalent International Criteria |
|---|---|---|
| Generalized obesity | Normal: 18.0–22.9 | Normal: 18.5–24.9 b |
| (BMI cut-offs in kg/m2) | Overweight: 23.0–24.9 Obesity: >25 | Overweight: 25.0–29.9 b Obesity: >30 b |
| Abdominal obesity (Waist circumference cut-offs in cm) | Men: >90 c Women: >80 c | Men: >102 d Women: >88 d |
5. Prevalence
5.1. Generalized Obesity
| Studies | Location in India/urban/rural | Age (year) | Sample population (men) | Sample population (women) | Criteria (BMI in kg/m2 and WC in cm) | Prevalence (%) in men | Prevalence (%) in women |
|---|---|---|---|---|---|---|---|
| Dhurandhar et al., 1992 [35] | West India (Urban) | >15 | 791 | 791 | BMI: >30 | 4.8 | 7.8 |
| Gupta et al., 2003 [36] | North India (Urban) | ≥20 | 532 | 559 | WC: >102 (M), ≥88 (F) | 21.8 | 44.0 |
| Misra et al., 2001 [10] | North India(Urban) ** | >18 | 170 | 362 | BMI: >25 | 13.3 | 15.6 |
| Gupta et al., 2004 [37] | North India (Urban) | >20 | 960 | 840 | WC: ≥102 (M); ≥88 (F) | 25.6 | 44.0 |
| Prabhakaran et al., 2005 [38] | North Indian(industrial population) | 20–59 | 2935 * | BMI: >25 WC: ≥90 (M); ≥80 (F) | 35.0 *43.0 * | ||
| Misra et al., 2005 [39] | North India(Urban) | 38.9 | 640 * | WC: ≥90 (M); ≥80 (F) | 10.1 | 25.9 | |
| Gupta et al., 2007 [40] | North India(Urban) *** | Mean: 43.2 (M) 44.7 (F) | 226 | 232 | BMI: ≥30, WC: ≥102 (M); ≥88 (F) | 20.8 | 34.5 |
| Deepa et al., 2007 [41] | South India(Urban) | >20 | 2350 * | BMI: ≥25 WC: ≥90 (M); ≥80 (F) | 43.256.2 | 47.435.1 | |
| Wijewardene et al., 2008 [42] | Sri Lanka | 30–65 | 2692 | 3355 | BMI: ≥25 | 20.3 | 36.5 |
| Zaman et al., 2001 [43] | Bangladesh (Rural) | >18 | 238 | 272 | WC: ≥94 (M); ≥80 (F) | 2.9 | 16.8 |
| Nanan [44] | Pakistan | 25–64 | National Survey | BMI: >30 | 13 | 23 | |
| Vaidya et al., 2008 [45,46] | Kathmandu, Nepal (urban) | 21–57 | 341 | BMI: >25 | 33 | ||
| Chow et al., 2008 [47] | South India(rural) | 20–90 | 4535 * | BMI: >25 WC: ≥90 (M); ≥80 (F) | 32.4 | 41.4 | |
| Bhardwaj et al., 2011 [48] | North India (urban) | >18 | 217 | 242 | BMI: >25, WC: ≥90 (M); ≥80 (F) | 50.2 | 50 |
| Gupta et al., 2011 [49] | West India (Urban) | 35–70 | 4621 | WC: ≥102 (M); ≥88 (F) | 14.4 | ||
5.2. Urban
5.3. Rural
5.4. Abdominal Obesity
5.5. Obesity in Women
5.6. Obesity in Children/Adolescents
6. Phenotype of Obesity in South Asians
6.1. Body Fat
6.2. Truncal and Abdominal Adiposity
7. Prediction Equations for Insulin Resistance and Body Fat Depots for Asian Indians
7.1. For Insulin Resistance
7.2. For Body Fat Depots
| Variable predictive equation |
|---|
| %BF: 42.42 + 0.003 × age + 7.04 × gender + 0.42 × TR sf + 0.29 × WC + 0.22 × Wt − 0.42 × Ht |
| TAF: −47,657.00 + 1384.11 × gender + 1466.54 × BMI + 416.10 × WC |
| IAAT: −238.7 + 16.9 × age + 934.18 × gender + 578.09 × BMI − 441.06 × HC + 434.2 × WC |
| SCAT: −49,376.4 − 17.15 × age + 1016.5 × gender + 783.3 × BMI + 466 × HC |
8. Deposition of Fat at “Ectopic” Sites
8.1. Hepatic Fat
8.2. Skeletal Muscle Triglycerides
8.3. Other Ectopic Sites of Fat Deposition
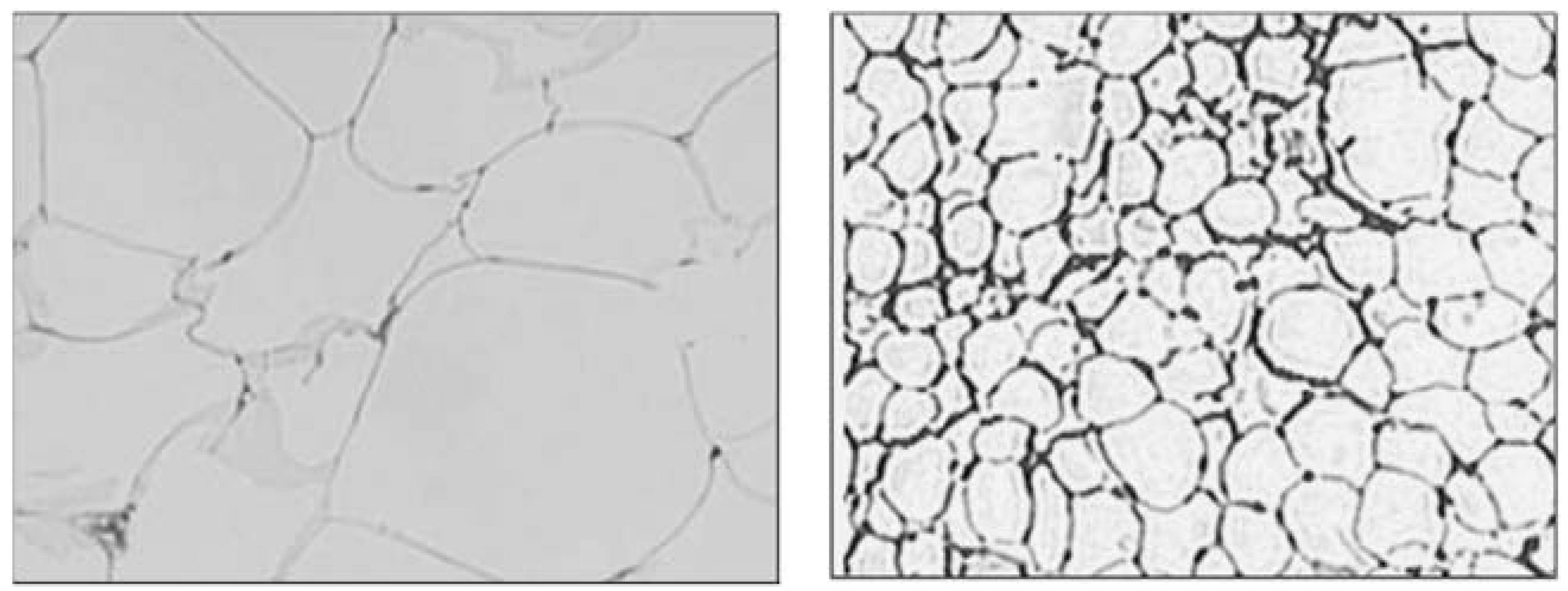
8.4. Adipocyte Size
9. Dyslipidemia
| Variable | Percentage |
|---|---|
| Obesity (BMI criteria) | 50.1 |
| Impaired Fasting Glucose | 24 |
| Diabetes | 8.5 |
| Hypercholesterolemia | 26.6 |
| Hypertriglyceridemia | 42.7 |
| LDL-C ≥ 100 mg/dL | 51.6 |
| HDL-C < 40 mg/dL (males) and <50 mg/dL (females) | 37 |
| Author | N | Parameter | South Asians/Asian Indians | Whites/Europeans |
|---|---|---|---|---|
| Adults | ||||
| Chandalia et al. [112] | 1031 AIs and 455 Whites a | Percent population j with low HDL | M, (42% m; 52% n); F, (56% m; 72% n) | M, 35%; F, 25% * |
| Ajjan et al. [113] | 245 SAs and 245 UK Whites | HDL (mmol/L) | 1.10 | 1.43 ** |
| Williams et al. [114] | 63 SAs and 42 Europeans b | HDL (mmol/L) | 1.27 | 1.20 |
| Smith et al. [115] | 82 AIs and 83 Caucasians c | HDL (mmol/L) | M, 0.97; F, 1.13 | M, 1.24; F, 1.51 * |
| Somani et al. [116] | 141 SAs and 121 Whites | HDL (mmol/L) | 1.1 | 1.5 |
| Bhalodkar et al. [117] | 119 AIs and 1752 Caucasians d | HDL (mmol/L) HDL size (nm) k | 1.378.9 | 1.379.4 |
| Forouhi et al. [118] | 113 SAs and Caucasians e | HDL (mmol/L) | M, 1.26, F, 1.51 | M, 1.39; F, 1.56 |
| Chambers et al. [119] | 518 AIs and 507 Whites f | HDL (mmol/L) | 1.22 | 1.33 ** |
| Enas et al. [120] | 1131 AI men and 557 AI women compared with Caucasians from FOS g | HDL (mmol/L) l | M, 0.98; F, 1.24 | M, 1.18 **; F, 1.45 ** |
| McKeigue et al. [121] | 1421 SAs and 1515 Europeans h | HDL (mmol/L) | 1.16 | 1.25 |
| McKeigue et al. [122] | 253 Bangladeshis and Europeans i | HDL (mmol/L)Percent of TC as HDL (%) | M, 1.13; F, 1.19M, 21.3; F, 22.4 | M, 1.43; F, 1.45M, 25.3; F, 25.2 |
| Children | ||||
| Ehtisham et al. [123] | 65 SAs and 64 European adolescents (14–17 years) | HDL (mmol/L) | M, 1.28; F, 1.49 | M, 1.39; F, 1.67 |
| Whincup et al. [124] | 73 SAs and 1287 Caucasian children (10–11 years) | HDL (mmol/L) | 1.38 | 1.43 |
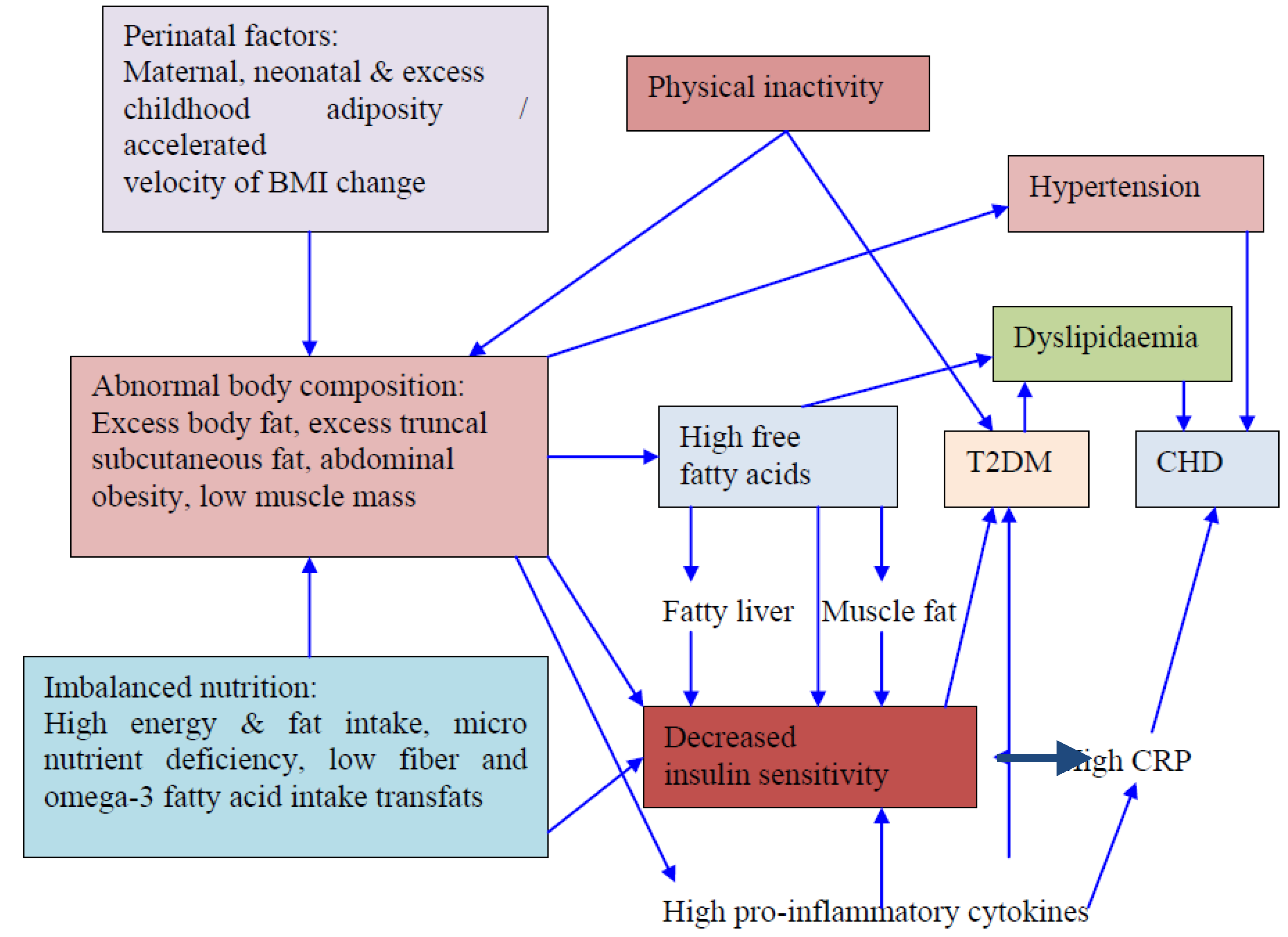
10. Determinants of Obesity and Dyslipidemia in South Asians
10.1. Nutritional Transition
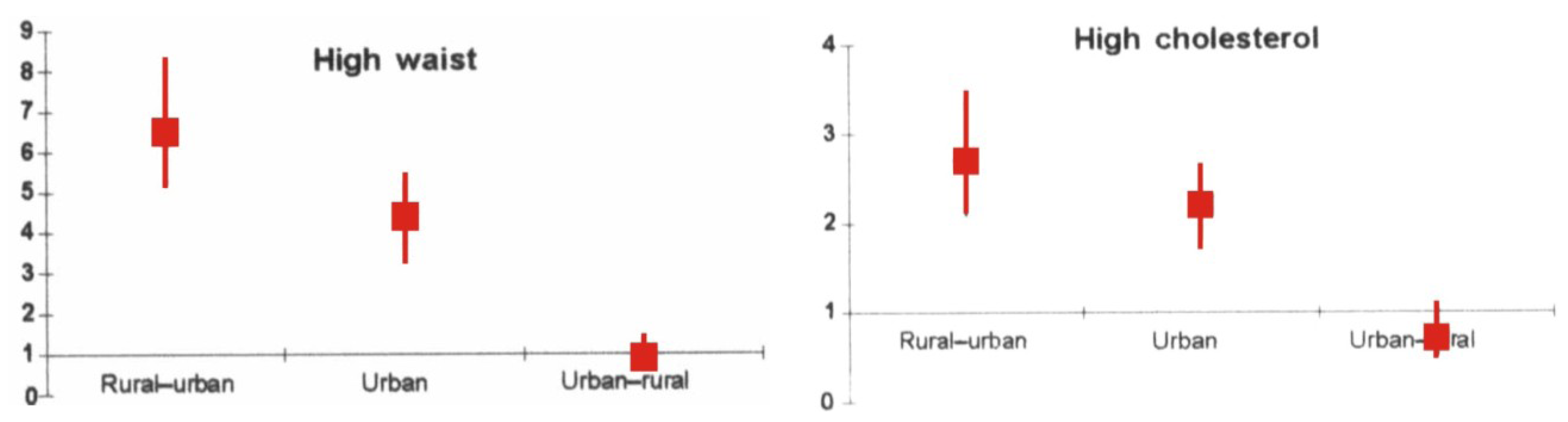
10.2. Urbanization, Demographic Transition and Rural-to-Urban Migration
10.3. Physical Inactivity
10.4. Socio-Economic and Cultural Factors
10.5. Genetics
11. Interventions
11.1. Adults
11.2. School Children
12. Conclusions
Conflict of Interest
References
- Bloom, D.E.; Rosenberg, L. The Future of South Asia: Population Dynamics, Economic Prospects, and Regional Coherence. Available online: http://www.hsph.harvard.edu/pgda/WorkingPapers/2011/PGDA_WP_68.pdf (accessed on 12 January 2013).
- The Growing Danger of Non Communicable Diseases: Acting Now to Reverse Course. Available online: http://siteresources.worldbank.org/HEALTHNUTRITIONANDPOPULATION/Resources/Peer-Reviewed-Publications/WBDeepeningCrisis.pdf (accessed on 24 April 2012).
- Nikolic, I.A.; Stanciole, A.E.; Zaydman, M. Chronic Emergency: Why NCDs Matter. Available online: http://siteresources.worldbank.org/HEALTHNUTRITIONANDPOPULATION/Resources/281627–1095698140167/ChronicEmergencyWhyNCDsMatter.pdf (accessed on 2 September 2011).
- Key Facts. Obesity and Overweight. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/index.html (accessed on 26 January 2013).
- Global Status Report on Non-communicable Diseases 2010. Available online: http://www.who.int/nmh/publications/ncd_report2010/en/ (accessed on 26 July 2012).
- De Onis, M.; Blössner, M.; Borghi, E. Global prevalence and trends of overweight and obesity among preschool children. Am. J. Clin. Nutr. 2010, 92, 1257–1264. [Google Scholar] [CrossRef]
- Mathers, C.D.; Loncar, D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006, 3, e442. [Google Scholar] [CrossRef]
- Engelgau, M.; El-Saharty, S.; Kudesia, P.; Rajan, V.; Rosenhouse, S.; Okamoto, K. Capitalizing on the Demographic Transition: Tackling Non-Communicable Diseases in South Asia; World Bank: Washington, DC, USA, 2011. [Google Scholar]
- Misra, A.; Khurana, L. Obesity and the metabolic syndrome in developing countries. J. Clin. Endocrinol. Metab. 2008, 93, S9–S30. [Google Scholar] [CrossRef]
- Misra, A.; Pandey, R.M.; Devi, J.R.; Sharma, R.; Vikram, N.K.; Khanna, N. High prevalence of diabetes, obesity and dyslipidaemia in urban slum population in northern India. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 1722–1729. [Google Scholar] [CrossRef]
- Wasir, J.S.; Misra, A. The metabolic syndrome in Asian Indians: The impact of nutritional and socio-economic transition in India. Met. Syndr. Relat. Disord. 2004, 2, 14–23. [Google Scholar] [CrossRef]
- Misra, A.; Vikram, N.K. Insulin resistance syndrome (metabolic syndrome) and obesity in Asian Indians: Evidence and implications. Nutrition 2004, 20, 482–491. [Google Scholar] [CrossRef]
- Misra, A.; Sharma, R.; Pandey, R.M.; Khanna, N. Adverse profile of dietary nutrients, anthropometry and lipids in urban slum dwellers of northern India. Eur. J. Clin. Nutr. 2001, 55, 727–734. [Google Scholar] [CrossRef]
- Smith, D.G. Epidemiology of dyslipidemia and economic burden on the health care system. Am. J. Manag. Care 2007, 13, S68–S71. [Google Scholar]
- Goff, D.C.; Betoni, A.G.; Kramer, H.; Bonds, D.; Blumenthal, R.S.; Tsai, M.Y.; Psatsy, B.M. Dyslipidemia prevalence, treatment, and control in the multi-Ethnic study of atherosclerosis (MESA) gender, ethnicity, and coronary artery calcium. Circulation 2006, 113, 647–656. [Google Scholar] [CrossRef]
- Misra, A.; Luthra, K.; Vikram, N.K. Dyslipidemia in Asian Indians: Determinants and significance. J. Assoc. Physicians India 2004, 52, 137–142. [Google Scholar]
- Office of the Registrar General & Census Commissioner, India. Available online: www.censusindia.gov.in (accessed on 30 April 2013).
- WHO Expert Consultation, Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. In Lancet; 2004; 363, pp. 157–163.
- Gupta, R.; Sarna, M.; Thanvi, J.; Rastogi, P.; Kaul, V.; Gupta, V.P. High prevalence of multiple coronary risk factors in Punjabi Bhatia community. Jaipur Heart Watch-3. Indian Heart J. 2004, 56, 646–652. [Google Scholar]
- Deurenberg, P.; Yap, M.; van Staveren, W.A. Body mass index and percent body fat: A meta analysis among different ethnic groups. Int. J. Obes. Relat. Metab. Disord. 1998, 22, 1164–1171. [Google Scholar] [CrossRef]
- Deurenberg-Yap, M.; Chew, S.K.; Lin, V.F.; Tan, B.Y.; van Staveren, W.A.; Deurenberg, P. Relationships between indices of obesity and its co-morbidities in multi-ethnic Singapore. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 1554–1562. [Google Scholar] [CrossRef]
- Vikram, N.K.; Pandey, R.M.; Misra, A.; Sharma, R.; Devi, J.R.; Khanna, N. Non-obese (body mass index < 25 kg/m2) Asian Indians with normal waist circumference have high cardiovascular risk. Nutrition 2003, 19, 503–509. [Google Scholar] [CrossRef]
- Misra, A. We Need Ethnic-Specific Criteria for Classification of BMI. In Progress in Obesity Research; Medeiros-Neto, G., Halpern, A., Bouchrad, C., Eds.; John Libbey Eurotext Ltd.: London, UK, 2003; pp. 547–553. [Google Scholar]
- Misra, A. Revision of limits of body mass index to define overweight and obesity are needed for the Asian ethnic groups. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 1294–1296. [Google Scholar] [CrossRef]
- Stevens, J. Ethnic-specific cut points for obesity vs country-specific guidelines for action. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 287–288. [Google Scholar] [CrossRef]
- Asia-Pacific Steering Committee, The Asia-Pacific Perspective: Redefining Obesity and Its Treatment; International Diabetes Institute; Health Communications Australia: St Leonards, Australia, 2000.
- Misra, A.; Khurana, L. Obesity related non-communicable diseases: South Asians vs White Caucasians. Int. J. Obes. 2011, 35, 167–187. [Google Scholar] [CrossRef]
- Misra, A.; Chowbey, P.K.; Makkar, B.M.; Vikram, N.K.; Wasir, J.S.; Chadha, D.; Joshi, S.R.; Sadikot, S.; Gulati, S.; Munjal, Y.P. Consensus statement for diagnosis of obesity, abdominal obesity and the metabolic syndrome for Asian Indians and recommendations for physical activity, medical and surgical management. J. Assoc. Physicians India 2009, 57, 163–170. [Google Scholar]
- Grundy, S.M.; Brewer, H.B., Jr.; Cleeman, J.I.; Smith, S.C., Jr.; Lenfant, C. Definition of metabolic syndrome: Report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Circulation 2004, 109, 433–438. [Google Scholar] [CrossRef]
- Grundy, S.M.; Cleeman, J.I.; Daniels, S.R.; Donato, K.A.; Eckel, R.H.; Franklin, B.A.; Gordon, D.J.; Krauss, R.M.; Savage, P.J.; Smith, S.C., Jr.; et al. Diagnosis and management of the metabolic syndrome. An American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Executive Summary. Circulation 2005, 112, 2735–2752. [Google Scholar] [CrossRef]
- Einhorn, D.; Reaven, G.M.; Cobin, R.H.; Ford, E.; Ganda, O.P.; Handelsman, Y.; Hellman, R.; Jellinger, P.S.; Kendall, D.; Krauss, R.M.; et al. American College of Endocrinology position statement on the insulin resistance syndrome. Endocr. Pract. 2003, 9, 237–252. [Google Scholar]
- How Do We Define Obesity and Overweight? Obesity and Overweight. Available online: http://www.who.int/dietphysicalactivity/media/en/gsfs_obesity.pdf (accessed on 6 May 2013).
- IDF Worldwide Definition of the Metabolic Syndrome. Available online: http://www.idf.org/metabolic-syndrome (accessed on 29 November 2012).
- National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002, 106, 3143–3421.
- Dhurandhar, N.V.; Kulkarni, P.R. Prevalence of obesity in Bombay. Int. J. Obes. Relat. Metab. Disord. 1992, 16, 367–375. [Google Scholar]
- Gupta, A.; Gupta, R.; Sarna, M.; Rastogi, S.; Gupta, V.P.; Kothari, K. Prevalence of diabetes, impaired fasting glucose and insulin resistance syndrome in an urban Indian population. Diabetes Res. Clin. Pract. 2003, 61, 69–76. [Google Scholar] [CrossRef]
- Gupta, R.; Deedwania, P.C.; Gupta, A.; Rastogi, S.; Panwar, R.B.; Kothari, K. Prevalence of metabolic syndrome in an Indian urban population. Int. J. Cardiol. 2004, 97, 257–261. [Google Scholar] [CrossRef]
- Prabhakaran, D.; Shah, P.; Chaturvedi, V.; Ramakrishnan, L.; Manhapra, A.; Reddy, K.S. Cardiovascular risk factor prevalence among men in a large industry of northern India. Natl. Med. J. India 2005, 18, 59–65. [Google Scholar]
- Misra, A.; Wasir, J.S.; Pandey, R.M. An evaluation of candidate definitions of the metabolic syndrome in adult Asian Indians. Diabetes Care 2005, 28, 398–403. [Google Scholar] [CrossRef]
- Gupta, R.; Sarna, M.; Thanvi, J.; Sharma, V.; Gupta, V.P. Fasting glucose and cardiovascular risk factors in an urban population. J. Assoc. Physicians India 2007, 55, 705–709. [Google Scholar]
- Deepa, M.; Farooq, S.; Deepa, R.; Manjula, D.; Mohan, V. Prevalence and significance of generalized and central body obesity in an urban Asian Indian population in Chennai, India (CURES: 47). Eur. J. Clin. Nutr. 2009, 63, 259–267. [Google Scholar] [CrossRef]
- Wijewardene, K.; Mohideen, M.R.; Mendis, S.; Fernando, D.S.; Kulathilaka, T.; Weerasekara, D.; Uluwitta, P. Prevalence of hypertension, diabetes and obesity: Baseline findings of a population based survey in four provinces in Sri Lanka. Ceylon Med. J. 2005, 50, 62–70. [Google Scholar]
- Zaman, M.M.; Yoshiike, N.; Rouf, M.A.; Syeed, M.H.; Khan, M.R.; Haque, S.; Mahtab, H.; Tanaka, H. Cardiovascular risk factors: Distribution and prevalence in a rural population of Bangladesh. J. Cardiovasc. Risk 2001, 8, 103–108. [Google Scholar] [CrossRef]
- Nanan, D. Health status of the Pakistani population. Am. J. Public Health 2001, 91, 1545–1546. [Google Scholar] [CrossRef]
- Vaidya, A.; Pokharel, P.K.; Nagesh, S.; Karki, P.; Kumar, S.; Majhi, S. Association of obesity and physical activity in adult males of Dharan, Nepal. Kathmandu Univ. Med. J. 2006, 4, 192–197. [Google Scholar]
- WHO STEPS Surveillance, Non-Communicable Disease Risk Factor Survey 2008; Ministry of Health and Population: Kathmandu, Nepal, 2008.
- Chow, C.K.; Naidu, S.; Raju, K.; Raju, R.; Joshi, R.; Sullivan, D.; Celermajer, D.S.; Neal, B.C. Significant lipid, adiposity and metabolic abnormalities amongst 4535 Indians from a developing region of rural Andhra Pradesh. Atherosclerosis 2008, 196, 943–952. [Google Scholar] [CrossRef]
- Bhardwaj, S.; Misra, A.; Misra, R.; Goel, K.; Bhatt, S.P.; Rastogi, K.; Vikram, N.K.; Gulati, S. High prevalence of abdominal, intra-abdominal and subcutaneous adiposity and clustering of risk factors among urban Asian Indians in North India. PLoS One 2011, 6, e24362. [Google Scholar]
- Gupta, R.; Agrawal, A.; Misra, A.; Gupta, S.; Pandey, R.M.; Misra, P.; Vikram, N.K.; Dey, S.; Rao, S.; Menon, V.U.; et al. Migrating husbands and changing cardiovascular risk factors in the wife: A cross sectional study in Asian Indian women. J. Epidemiol. Community Health 2012, 66, 881–889. [Google Scholar] [CrossRef]
- Popkin, B.M.; Adair, L.S.; Ng, S.W. Global nutrition transition and the pandemic of obesity in developing countries. Nutr. Rev. 2012, 70, 3–21. [Google Scholar] [CrossRef]
- Misra, A.; Khurana, L.; Isharwal, S.; Bhardwaj, S. South Asian diets and insulin resistance. Br. J. Nutr. 2009, 101, 465–473. [Google Scholar]
- Chadha, S.L.; Gopinath, N.; Shekhawat, S. Urban-rural differences in the prevalence of coronary heart disease and its risk factors in Delhi. Bull. World Health Organ. 1997, 75, 31–38. [Google Scholar]
- Kaur, P.; Rao, S.R.; Radhakrishnan, E.; Rajasekar, D.; Gupte, M.D. Prevalence, awareness, treatment, control and risk factors for hypertension in a rural population in South India. Int. J. Public Health 2012, 57, 87–94. [Google Scholar] [CrossRef]
- Misra, P.; Upadhyay, R.P.; Krishnan, A.; Vikram, N.K.; Sinha, S. A community-based study of metabolic syndrome and its components among women of rural community in Ballabgarh, Haryana. Met. Syn. Relat. Disord. 2011, 10, 1–8. [Google Scholar]
- Misra, R.; Misra, A.; Vikram, N.K.; Gupta, N.S.; Sharma, S.S.; Patel, S.; Thakor, B.P. Difference in prevalence of diabetes, obesity, metabolic syndrome and associated cardiovascular risk factors in a rural area of Tamil Nadu and an urban area of Delhi. Int. J. Diabetes Dev. Ctries. 2011, 31, 82–90. [Google Scholar] [CrossRef]
- Misra, A.; Vikram, N.K. Clinical and patho-physiological consequences of abdominal adiposity and abdominal adipose tissue depots. Nutrition 2003, 19, 457–466. [Google Scholar] [CrossRef]
- India. National Family Health Survey (NFHS-2). Key Findings. Available online: www.measuredhs.com/pubs/pdf/SR81/SR81.pdf (accessed on 14 May 2012).
- India. National Family Health Survey (NFHS-3). 2005–06. Key Findings. Available online: www.measuredhs.com/pubs/pdf/SR128/SR128.pdf (accessed on 4 May 2013).
- Bharati, S.; Pal, M.; Bhattacharya, B.N.; Bharati, P. Prevalence and causes of chronic energy deficiency and obesity in Indian women. Hum. Biol. 2007, 79, 395–412. [Google Scholar] [CrossRef]
- Bhardwaj, S.; Misra, A.; Khurana, L.; Gulati, S.; Shah, P.; Vikram, N.K. Childhood obesity in Asian Indians: A burgeoning cause of insulin resistance, diabetes and sub-clinical inflammation. Asia Pac. J. Clin. Nutr. 2008, 17, 172–175. [Google Scholar]
- Misra, A.; Vikram, N.K.; Arya, S.; Pandey, R.M.; Dhingra, V.; Chatterjee, A.; Dwivedi, M.; Sharma, R.; Luthra, K.; Guleria, R.; Talwar, K.K. High prevalence of insulin resistance in post-pubertal Asian Indian children is associated with adverse truncal body fat patterning, abdominal adiposity and excess body fat. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 1217–1226. [Google Scholar] [CrossRef]
- Misra, A.; Khurana, L.; Vikram, N.K.; Goel, A.; Wasir, J.S. Metabolic syndrome in children: Current issues and South Asian perspective. Nutrition 2007, 23, 895–910. [Google Scholar] [CrossRef]
- Singhal, N.; Misra, A.; Shah, P.; Rastogi, K.; Vikram, N.K. Secular trends in obesity, regional adiposity and metabolic parameters among Asian Indian adolescents in North India: A comparative data analysis of two selective samples 5 years apart (2003, 2008). Ann. Nutr. Metab. 2010, 56, 176–181. [Google Scholar] [CrossRef]
- Misra, A.; Madhavan, M.; Vikram, N.K.; Pandey, R.M.; Dhingra, V.; Luthra, K. Simple anthropometric measures identify fasting hyperinsulinemia and clustering of cardiovascular risk factors in Asian Indian adolescents. Metabolism 2006, 55, 1569–1573. [Google Scholar] [CrossRef]
- Raj, M.; Sundaram, K.R.; Paul, M.; Deepa, A.S.; Kumar, R.K. Obesity in Indian children: Time trends and relationship with hypertension. Natl. Med. J. India 2007, 20, 288–293. [Google Scholar]
- Misra, A.; Shah, P.; Goel, K.; Hazra, D.K.; Gupta, R.; Seth, P.; Tallikoti, P.; Mohan, I.; Bhargava, R.; Bajaj, S.; et al. The high burden of obesity and abdominal obesity in Urban Indian school children: A multi-centric study of 38,296 children. Ann. Nutr. Metab. 2011, 58, 203–211. [Google Scholar] [CrossRef]
- Deurenberg-Yap, M.; Schmidt, G.; van Staveren, W.A.; Deurenberg, P. The paradox of low body mass index and high body fat percentage among Chinese, Malays and Indians in Singapore. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 1011–1017. [Google Scholar] [CrossRef]
- Deurenberg-Yap, M.; Schmidt, G.; van Staveren, W.A.; Hautvast, J.G.; Deurenberg, P. Body fat measurement among Singaporean Chinese, Malays and Indians: A comparative study using a four-compartment model and different two-compartment models. Br. J. Nutr. 2001, 85, 491–498. [Google Scholar] [CrossRef]
- Banerji, M.A.; Faridi, N.; Alturi, R.; Chaiken, R.L.; Lebovitz, H.E. Body composition, visceral fat, leptin and insulin resistance in Asian Indian men. J. Clin. Endocrinol. Metab. 1999, 84, 137–144. [Google Scholar] [CrossRef]
- Dudeja, V.; Misra, A.; Pandey, R.M.; Devina, G.; Kumar, G.; Vikram, N.K. BMI does not accurately predict overweight in Asian Indians in northern India. Br. J. Nutr. 2001, 86, 105–112. [Google Scholar] [CrossRef]
- Misra, A.; Athiko, D.; Sharma, R.; Pandey, R.M.; Khanna, N. Non-obese hyperlipidemic Asian northern Indian males have adverse anthropometric profile. Nutr. Metab. Cardiovasc. Dis. 2002, 12, 178–183. [Google Scholar]
- Chandalia, M.; Abate, N.; Garg, A.; Stray-Gundersen, J.; Grundy, S.M. Relationship between generalized and upper body obesity to insulin resistance in Asian Indian men. J. Clin. Endocrinol. Metab. 1999, 84, 2329–2335. [Google Scholar]
- Misra, A.; Vikram, N.K.; Gupta, R.; Pandey, R.M.; Wasir, J.S.; Gupta, V.P. Waist circumference cutoff points and action levels for Asian Indians for identification of abdominal obesity. Int. J. Obes. 2006, 30, 106–111. [Google Scholar] [CrossRef]
- Chandalia, M.; Lin, P.; Seenivasan, T.; Livingston, E.H.; Snell, P.G.; Grundy, S.M.; Abate, N. Insulin resistance and body fat distribution in South Asian men compared to Caucasian Men. PLoS One 2007, 2, e812. [Google Scholar] [CrossRef]
- Jonathan, C.K. Wells commentary: Why are South Asians susceptible to central obesity? The El Niño hypothesis. Int. J. Epidemiol. 2007, 36, 226–227. [Google Scholar]
- Hughes, K.; Aw, T.; Kuperan, P.; Choo, M. Central obesity, insulin resistance, syndrome X, lipoprotein (a) and cardiovascular risk in Indians, Malays, and Chinese in Singapore. J. Epidemiol. Community Health 1997, 51, 394–399. [Google Scholar] [CrossRef]
- Kamath, S.K.; Hussain, E.A.; Amin, D.; Mortillaro, E.; West, B.; Peterson, C.T.; Aryee, F.; Murillo, G.; Alekel, D.L. Cardiovascular disease risk factors in 2 distinct ethnic groups: Indian and Pakistani compared with American premenopausal women. Am. J. Clin. Nutr. 1999, 69, 621–631. [Google Scholar]
- De Koning, L.; Merchant, A.T.; Pogue, J.; Anand, S.S. Waist circumference and waist-to-hip ratio as predictors of cardiovascular events: Meta-regression analysis of prospective studies. Eur. Heart J. 2007, 28, 850–856. [Google Scholar] [CrossRef]
- Raji, A.; Seely, E.W.; Arky, R.A.; Simonson, D.C. Body fat distribution and insulin resistance in healthy Asian Indians and Caucasians. J. Clin. Endocrinol. Metab. 2001, 86, 5366–5371. [Google Scholar] [CrossRef]
- Vega, G.L. Obesity, the metabolic syndrome, and cardiovascular disease. Am. Heart J. 2001, 142, 1108–1116. [Google Scholar] [CrossRef]
- Misra, A.; Misra, R.; Wijesuriya, M.; Banerjee, D. The metabolic syndrome in South Asians: Continuing escalation & possible solutions. Indian J. Med. Res. 2007, 125, 345–354. [Google Scholar]
- Goel, R.; Misra, A.; Kondal, D.; Pandey, R.M.; Vikram, N.K.; Wasir, J.S.; Dhingra, V.; Luthra, K. Identification of insulin resistance in Asian Indian adolescents: Classification and regression tree (CART) and logistic regression based classification rules. Clin. Endocrinol. (Oxf.) 2009, 70, 717–724. [Google Scholar] [CrossRef]
- Goel, K.; Gupta, N.; Misra, A.; Poddar, P.; Pandey, R.M.; Vikram, N.K.; Wasir, J.S. Predictive equations for body fat and abdominal fat with DXA and MRI as reference in Asian Indians. Obesity (Silver Spring) 2008, 16, 451–456. [Google Scholar] [CrossRef]
- Goel, K.; Misra, A.; Vikram, N.K.; Poddar, P.; Gupta, N. Subcutaneous abdominal adipose tissue is associated with the metabolic syndrome in Asian Indians independent of intra abdominal and total body fat of body. Heart 2010, 96, 579–583. [Google Scholar] [CrossRef]
- See, R.; Abdullah, S.M.; McGuire, D.K.; Khera, A.; Patel, M.J.; Lindsey, J.B.; Grundy, S.M.; de Lemos, J.A. The association of differing measures of overweight and obesity with prevalent atherosclerosis: The Dallas Heart Study. J. Am. Coll. Cardiol. 2007, 50, 752–759. [Google Scholar] [CrossRef]
- Victor, R.G.; Haley, R.W.; Willett, D.L.; Peshock, R.M.; Vaeth, P.C.; Leonard, D.; Basit, M.; Cooper, R.S.; Iannacchione, V.G.; Visscher, W.A.; et al. The Dallas Heart Study: A population-based probability sample for the multidisciplinary study of ethnic differences in cardiovascular health. Am. J. Cardiol. 2004, 93, 1473–1480. [Google Scholar] [CrossRef]
- Ian, J.; Neeland, I.J.; Turer, A.T.; Colby, R.; Ayers, M.S.; Tiffany, M.; Wiley, P.; Gloria, L.; Farzaneh-Far, R.; Grundy, S.M.; et al. Dysfunctional adiposity and the risk of pre-diabetes and type 2 diabetes in obese adults. JAMA 2012, 308, 1150–1159. [Google Scholar] [CrossRef]
- Vega, G.L.; Adams-Huet, B.; Peshock, R.; Willett, D.; Shah, B.; Grundy, S.M. Influence of body fat content and distribution on variation in metabolic risk. J. Clin. Endocrinol. Metab. 2006, 29, 1414–1416. [Google Scholar]
- Liu, J.; Grundy, S.M.; Wang, W.; Smith, S.C., Jr.; Gloria, L.V.; Zhaosa, W.; Zhechun, Z.; Wenhua, W.; Dong, W. Ten-year risk of cardiovascular incidence related to diabetes, prediabetes, and the metabolic syndrome. Am. Heart J. 2007, 153, 552–558. [Google Scholar]
- Yusuf, S.; Hawken, S.; Ounpuu, S.; Bautista, L.; Franzosi, M.G.; Commerford, P.; Lang, C.C.; Rumboldt, Z.; Onen, C.L.; Lisheng, L.; et al. Obesity and the risk of myocardial infarction in 27,000 participants from 52 countries: A case-control study. Lancet 2005, 366, 1640–1649. [Google Scholar] [CrossRef]
- Garg, A.; Misra, A. Hepatic steatosis, insulin resistance, and adipose tissue disorders. J. Clin. Endocrinol. Metab. 2002, 87, 3019–3022. [Google Scholar] [CrossRef]
- Luthra, A.; Nigam, P.; Misra, A. Metabolic correlates of non-alcoholic fatty liver disease and management issues: A perspective for Asian Indians. Diabetes Metab. Syndr. Res. Rev. 2007, 1, 279–285. [Google Scholar] [CrossRef]
- Bajaj, S.; Nigam, P.; Luthra, A.; Pandey, R.M.; Kondal, D.; Bhatt, S.P.; Wasir, J.S.; Misra, A. Insulin resistance, metabolic co-variates and prediction score in non-alcoholic fatty liver disease: A Case-control study. Indian J. Med. Res. 2009, 129, 285–292. [Google Scholar]
- Sharma, R.; Sinha, S.; Danishad, K.A.; Vikram, N.K.; Gupta, A.; Ahuja, V.; Jagannathan, N.R.; Pandey, R.M.; Misra, A. Investigation of hepatic gluconeogenesis pathway in non-diabetic Asian Indians with non-alcoholic fatty liver disease using in vivo (31P) phosphorus magnetic resonance spectroscopy. Atherosclerosis 2009, 203, 291–297. [Google Scholar] [CrossRef]
- Petersen, K.F.; Dufour, S.; Feng, J.; Befroy, D.; Dziura, J.; Dalla Man, C.; Cobelli, C.; Shulman, G.I. Increased prevalence of insulin resistance and non-alcoholic fatty liver disease in Asian Indian men. Proc. Natl. Acad. Sci. USA 2006, 103, 18273–18277. [Google Scholar] [CrossRef]
- Petersen, K.F.; Hendler, R.; Price, T.; Perseghin, G.; Rothman, D.L.; Held, N.; Amatruda, J.M.; Shulman, G.I. 13C/31P NMR studies on the mechanism of insulin resistance in obesity. Diabetes 1998, 47, 381–386. [Google Scholar] [CrossRef]
- Perseghin, G.; Scifo, P.; de Cobelli, F.; Pagliato, E.; Battezzati, A.; Arcelloni, C.; Vanzulli, A.; Testolin, G.; Pozza, G.; del Maschio, A.; et al. Intra-myocellular triglyceride content is a determinant of in vivo insulin resistance in humans: A 1H-13C nuclear magnetic resonance spectroscopy assessment in offspring of type 2 diabetic parents. Diabetes 1999, 48, 1600–1606. [Google Scholar] [CrossRef]
- Misra, A.; Sinha, S.; Kumar, M.; Jagannathan, N.R.; Pandey, R.M. Proton magnetic resonance spectroscopy study of soleus muscle in non-obese healthy and type 2 diabetic Asian Northern Indian males: High intra-myocellular lipid content correlates with excess body fat and abdominal obesity. Diabet. Med. 2003, 20, 361–367. [Google Scholar] [CrossRef]
- Sinha, S.; Misra, A.; Kumar, V.; Jagannathan, N.R.; Bal, C.S.; Pandey, R.M.; Singhania, R.; Deepak. Proton magnetic resonance spectroscopy and single photon emission computed tomography study of the brain in asymptomatic young hyperlipidaemic Asian Indians in North India show early abnormalities. Clin. Endocrinol. (Oxf.) 2004, 61, 182–189. [Google Scholar] [CrossRef]
- Sinha, S.; Misra, A.; Rathi, M.; Kumar, V.; Pandey, R.M.; Luthra, K.; Jagannathan, N.R. Proton magnetic resonance spectroscopy and biochemical investigation of type 2 diabetes mellitus in Asian Indians: Observation of high muscle lipids and C-reactive protein levels. Magn. Reson. Imaging 2009, 27, 94–100. [Google Scholar] [CrossRef]
- Garg, A.; Misra, A. Lipodystrophies: Rare disorders causing metabolic syndrome. Endocrinol. Metab. Clin. N. Am. 2004, 33, 305–331. [Google Scholar] [CrossRef]
- Mallon, P.W.; Wand, H.; Law, M.; Miller, J.; Cooper, D.A.; Carr, A. Buffalo hump seen in HIV-associated lipodystrophy is associated with hyperinsulinemia but not dyslipidemia. J. Acquir. Immune Defic. Syndr. 2005, 38, 156–162. [Google Scholar] [CrossRef]
- Haque, W.A.; Oral, E.A.; Dietz, K.; Bowcock, A.M.; Agarwal, A.K.; Garg, A. Risk factors for diabetes in familial partial lipodystrophy, Dunnigan variety. Diabetes Care 2003, 26, 1350–1355. [Google Scholar] [CrossRef]
- Misra, A.; Jaiswal, A.; Shakti, D.; Wasir, J.; Vikram, N.K.; Pandey, R.M.; Kondal, D.; Bhushan, B. Novel phenotypic markers and screening score for the metabolic syndrome in adult Asian Indians. Diabetes Res. Clin. Pract. 2008, 79, e1–e5. [Google Scholar]
- Weyer, C.; Foley, J.E.; Bogardus, C.; Tataranni, P.A.; Pratley, R.E. Enlarged subcutaneous abdominal adipocyte size, but not obesity itself, predicts type II diabetes independent of insulin resistance. Diabetologia 2000, 43, 1498–1506. [Google Scholar] [CrossRef]
- Grundy, S.M.; Vega, G.L. Two different views of the relationship of hyper-triglyceridemia to coronary heart disease: Implications for treatment. Arch. Intern. Med. 1992, 152, 28–34. [Google Scholar] [CrossRef]
- Vega, G.L. Management of atherogenic dyslipidemia of the metabolic syndrome: Evolving rationale for combined drug therapy. Endocrinol. Metab. Clin. N. Am. 2004, 33, 525–544. [Google Scholar] [CrossRef]
- Chandalia, M; Deedwania, P.C. Coronary heart disease and risk factors in Asian Indians. Adv. Exp. Med. Biol. 2001, 498, 27–34. [Google Scholar] [CrossRef]
- Kulkarni, K.R.; Markovitz, J.H.; Nanda, N.C.; Segrest, J.P. Increased prevalence of smaller and denser LDL particles in Asian Indians. Arterioscler. Thromb. Vasc. Biol. 1999, 19, 2749–2755. [Google Scholar] [CrossRef]
- Abate, N.; Chandalia, M.; Snell, P.G.; Grundy, S.M. Adipose tissue metabolites and insulin resistance in non diabetic Asian Indian men. J. Clin. Endocrinol. Metab. 2004, 89, 2750–2755. [Google Scholar] [CrossRef]
- Abbasi, J.W.; Kulkarni, K.R.; Lamendola, C.; Mc Laughlin, T.L. Multiple lipoprotein abnormalities associated with insulin resistance in healthy volunteers are identified by the vertical auto profile-II methodology. Clin. Chem. 2003, 49, 1014–1017. [Google Scholar] [CrossRef]
- Chandalia, M.; Mohan, V.; Adams-Huet, B.; Deepa, R.; Abate, N. Ethnic difference in sex gap in high-density lipoprotein cholesterol between Asian Indians and Whites. J. Investig. Med. 2008, 56, 574–580. [Google Scholar]
- Ajjan, R.; Carter, A.M.; Somani, R.; Kain, K.; Grant, P.J. Ethnic differences in cardiovascular risk factors in healthy Caucasian and South Asian individuals with the metabolic syndrome. J. Thromb. Haemost. 2007, 5, 754–760. [Google Scholar] [CrossRef]
- Williams, E.D.; Kooner, I.; Steptoe, A.; Kooner, J.S. Psychosocial factors related to cardiovascular disease risk in UK South Asian men: A preliminary study. Br. J. Health Psychol. 2007, 12, 559–570. [Google Scholar] [CrossRef]
- Smith, J.; Al-Amri, M.; Sniderman, A.; Cianflone, K. Leptin and adiponectin in relation to body fat percentage, waist to hip ratio and the apoB/apoA1 ratio in Asian Indian and Caucasian men and women. Nutr. Metab. (Lond.) 2006, 3, 18. [Google Scholar] [CrossRef]
- Somani, R.; Grant, P.J.; Kain, K.; Catto, A.J.; Carter, A.M. Complement C3 and C-reactive protein are elevated in South Asians independent of a family history of stroke. Stroke 2006, 37, 2001–2006. [Google Scholar]
- Bhalodkar, N.C.; Blum, S.; Rana, T.; Kitchappa, R.; Bhalodkar, A.N.; Enas, E.A. Comparison of high-density and low-density lipoprotein cholesterol subclasses and sizes in Asian Indian women with Caucasian women from the Framingham Offspring Study. Clin. Cardiol. 2005, 28, 247–251. [Google Scholar] [CrossRef]
- Forouhi, N.G.; Sattar, N.; McKeigue, P.M. Relation of C-reactive protein to body fat distribution and features of the metabolic syndrome in Europeans and South Asians. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 1327–1331. [Google Scholar] [CrossRef]
- Chambers, J.C.; Eda, S.; Bassett, P.; Karim, Y.; Thompson, S.G.; Gallimore, J.R.; Pepys, M.B.; Kooner, J.S. C-reactive protein, insulin resistance, central obesity, and coronary heart disease risk in Indian Asians from the United Kingdom compared with European whites. Circulation 2001, 104, 145–150. [Google Scholar] [CrossRef]
- Enas, E.A.; Garg, A.; Davidson, M.A.; Nair, V.M.; Huet, B.A.; Yusuf, S. Coronary heart disease and its risk factors in first-generation immigrant Asian Indians to the United States of America. Indian Heart J. 1996, 48, 343–353. [Google Scholar]
- McKeigue, P.M.; Shah, B.; Marmot, M.G. Relation of central obesity and insulin resistance with high diabetes prevalence and cardiovascular risk in South Asians. Lancet 1991, 337, 382–386. [Google Scholar] [CrossRef]
- McKeigue, P.M.; Marmot, M.G.; Court, Y.D.S.; Cottier, D.E.; Rahman, S.; Riemersma, R.A. Diabetes, hyperinsulinaemia, and coronary risk factors in Bangladeshis in east London. Br. Heart J. 1988, 60, 390–396. [Google Scholar] [CrossRef]
- Ehtisham, S.; Crabtree, N.; Clark, P.; Shaw, N.; Barrett, T. Ethnic differences in insulin resistance and body composition in United Kingdom adolescents. J. Clin. Endocrinol. Metab. 2005, 90, 3963–3969. [Google Scholar]
- Whincup, P.H.; Gilg, J.A.; Papacosta, O.; Seymour, C.; Miller, G.J.; Alberti, K.G.; Cook, D.G. Early evidence of ethnic differences in cardiovascular risk: Cross sectional comparison of British South Asian and white children. BMJ 2002, 324, 635. [Google Scholar] [CrossRef]
- Bhardwaj, S.; Passi, S.J.; Misra, A. Overview of trans fatty acids: Biochemistry and health effects. Diabetes Metab. Syndr. 2011, 5, 161–164. [Google Scholar] [CrossRef]
- Chapter 4 Trends in Urbanisation. Available online: http://www.censusindia.gov.in/2011-prov-results/paper2-vol2/data_files/Mizoram/Chapter_4.pdf (accessed on 15 July 2011).
- Family Welfare Statistics in India. 2011. Available online: http://mohfw.nic.in/WriteReadData/l892s/972971120FW%20Statistics%202011%20Revised%2031%2010%2011.pdf (accessed on 17 January 2013).
- Misra, A.; Ganda, O.P. Migration and its impact on adiposity and type 2 diabetes. Nutrition 2007, 23, 696–708. [Google Scholar] [CrossRef]
- Hayes, L.; White, M.; Unwin, N.; Bhopal, R.; Fischbacher, C.; Harland, J.; Alberti, K.G. Patterns of physical activity and relationship with risk markers for cardio vascular disease and diabetes in Indian, Pakistani, Bangladeshi and European adults in a UK population. J. Public Health Med. 2002, 24, 170–178. [Google Scholar] [CrossRef]
- Gulati, S.; Misra, A.; Colles, S.L.; Kondal, D.; Gupta, N.; Goel, K.; Bansal, S.; Mishra, M.; Madkaikar, V.; Bhardwaj, S. Dietary intakes and familial correlates of overweight/obesity: A four cities study in India. Ann. Nutr. Metab. 2013, 62, 279–290. [Google Scholar] [CrossRef]
- Bhatt, S.P.; Nigam, P.; Misra, A.; Guleria, R.; Luthra, K.; Jain, S.K.; Qadar Pasha, M.A. Association of the Myostatin gene with obesity, abdominal obesity and low lean body mass and in non-diabetic Asian Indians in North India. PLoS One 2012, 7, e40977. [Google Scholar]
- Sharma, M.; Misra, A.; Vikram, N.; Bhatt, S.; Chhabra, S.P.; Garg, N.; Pandey, RM.; Upadhyay, A.D.; Luthra, K. Genotype of the LMNA 1908C>T variant is associated with generalized obesity in Asian Indians in North India. Clin. Endocrinol. (Oxf.) 2011, 75, 642–649. [Google Scholar]
- Tabassum, R.; Jaiswal, A.; Chauhan, G.; Dwivedi, O.P.; Ghosh, S.; Marwaha, R.K.; Tandon, N.; Bhardwaj, D. Genetic variant of AMD1 is associated with obesity in Urban Indian Children. PLoS One 2012, 7, e33162. [Google Scholar] [CrossRef]
- Bhatt, S.P.; Nigam, P.; Misra, A.; Guleria, R.; Luthra, K.; Vidya, M. SREBP-2 1784 G/C genotype is associated with non-alcoholic fatty liver disease in north Indians. Dis. Markers 2011, 31, 371–377. [Google Scholar] [CrossRef]
- Bhatt, S.P.; Nigam, P.; Misra, A.; Guleria, R.; Luthra, K.; Pandey, R.M.; Pasha, M.A.Q. Association of peroxisome proliferator activated receptor-γ gene with non-alcoholic fatty liver disease in Asian Indians residing in north India. Gene 2013, 512, 143–147. [Google Scholar] [CrossRef]
- Tabassum, R.; Mahajan, A.; Chauhan, G.; Dwivedi, O.P.; Ghosh, S.; Tandon, N.; Bhardwaj, D. Evaluation of DOK5 as a susceptibility gene for type 2 diabetes and obesity in North Indian population. BMC Med. Genet. 2010, 11, 35. [Google Scholar]
- Kumar, P.; Luthra, K.; Dwivedi, M.; Behl, V.K.; Pandey, R.M.; Misra, A. Apolipoprotein E gene polymorphisms in patients with premature myocardial infarction: A case-controlled study in Asian Indians in North India. Ann. Clin. Biochem. 2003, 40, 382–387. [Google Scholar] [CrossRef]
- Miller, M.; Rhyne, J.; Chen, H.; Beach, V.; Ericson, R.; Luthra, K.; Dwivedi, M.; Misra, A. APOC3 promoter polymorphisms C-482T and T-455C are associated with the metabolic syndrome. Arch. Med. Res. 2007, 38, 444–451. [Google Scholar] [CrossRef]
- Meena, K.; Misra, A.; Vikram, N.; Ali, S.; Pandey, R.M.; Luthra, K. Cholesterol ester transfer protein and apolipoprotein E gene polymorphisms in hyperlipidemic Asian Indians in North India. Mol. Cell. Biochem. 2011, 352, 189–196. [Google Scholar] [CrossRef]
- Misra, A.; Sharma, R.; Gulati, S.; Joshi, S.R.; Sharma, V.; Ghafoorunissa; Ibrahim, A.; Joshi, S.; Laxmaiah, A.; Kurpad, A.; et al. Consensus dietary guidelines for healthy living and prevention of obesity, the metabolic syndrome, diabetes, and related disorders in Asian Indians. Diabetes Technol. Ther. 2011, 13, 683–694. [Google Scholar] [CrossRef]
- Misra, A.; Nigam, P.; Hills, A.P.; Chadha, D.S.; Sharma, V.; Deepak, K.K.; Vikram, N.K.; Joshi, S.; Chauhan, A.; Khanna, K.; et al. Consensus physical activity guidelines for Asian Indians. Diabetes Technol. Ther. 2012, 14, 83–98. [Google Scholar] [CrossRef]
- Ramachandran, A.; Snehalatha, C.; Yamuna, A.; Mary, S.; Ping, Z. Cost-effectiveness of the interventions in the primary prevention of diabetes among asian indians within-trial results of the Indian Diabetes Prevention Programme (IDPP). Diabetes Care 2007, 30, 2548–2552. [Google Scholar]
- Ramachandran, A.; Snehalatha, C. Diabetes prevention programs. Med. Clin. N. Am. 2011, 95, 353–372. [Google Scholar] [CrossRef]
- Shah, P.; Misra, A.; Gupta, N.; Hazra, D.K.; Gupta, R.; Seth, P.; Agarwal, A.; Gupta, A.K.; Jain, A.; Kulshreshta, A.; et al. Improvement in nutrition-related knowledge and behaviour of urban Asian Indian school children: Findings from the ‘Medical education for children/Adolescents for Realistic prevention of obesity and diabetes and for healthy aGeing’ (MARG) intervention study. Br. J. Nutr. 2010, 104, 427–436. [Google Scholar] [CrossRef]
- Singhal, N.; Misra, A.; Shah, P.; Gulati, S. Effects of controlled school-based multi-component model of nutrition and lifestyle interventions on behavior modification, anthropometry and metabolic risk profile of urban Asian Indian adolescents in North India. Eur. J. Clin. Nutr. 2010, 64, 364–373. [Google Scholar]
- Singhal, N.; Misra, A.; Shah, P.; Gulati, S.; Bhatt, S.P.; Sharma, S.; Pandey, R.M. Impact of intensive school-based nutrition education and lifestyle interventions on insulin resistance, β-cell function, disposition index, and subclinical inflammation among Asian Indian adolescents: A controlled intervention study. Metab. Syndr. Relat. Disord. 2011, 9, 143–150. [Google Scholar] [CrossRef]
- Singhal, N.; Misra, A. A school-based intervention for diabetes risk reduction. N. Engl. J. Med. 2010, 363, 1769–1770. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Misra, A.; Shrivastava, U. Obesity and Dyslipidemia in South Asians. Nutrients 2013, 5, 2708-2733. https://doi.org/10.3390/nu5072708
Misra A, Shrivastava U. Obesity and Dyslipidemia in South Asians. Nutrients. 2013; 5(7):2708-2733. https://doi.org/10.3390/nu5072708
Chicago/Turabian StyleMisra, Anoop, and Usha Shrivastava. 2013. "Obesity and Dyslipidemia in South Asians" Nutrients 5, no. 7: 2708-2733. https://doi.org/10.3390/nu5072708
APA StyleMisra, A., & Shrivastava, U. (2013). Obesity and Dyslipidemia in South Asians. Nutrients, 5(7), 2708-2733. https://doi.org/10.3390/nu5072708




