Long Term Successful Weight Loss with a Combination Biphasic Ketogenic Mediterranean Diet and Mediterranean Diet Maintenance Protocol
Abstract
:1. Introduction
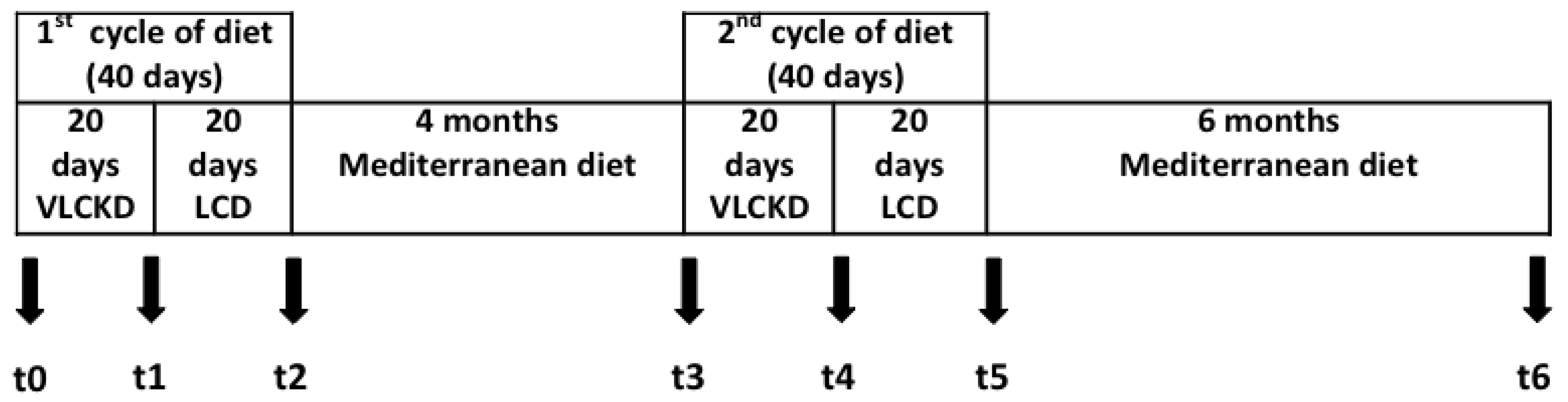
2. Subjects and Methods
- An initial 20 days of very low carbohydrate ketogenic diet (K1)
- Followed by 20 days of a low carbohydrate non ketogenic diet (stabilization) (LC1)
- A first period of 4 months of normal caloric Mediterranean diet (M1)
- A second 20 day very low carbohydrate ketogenic diet (K2)
- 20 Days of low carbohydrate non ketogenic diet (LC2)
- Final 6 months of normal caloric Mediterranean diet (M2)
2.1. Diet Protocols
| Macronutrients | Ketogenic Phase | Lowcarbohydrates Phase | Mediterranean Phase |
|---|---|---|---|
| Kcal/day | 976 ± 118 | 1111 ± 65 | 1800 ± 248 |
| Protein (% totaldaily Kcal) | 41 ± 2 | 27 ± 2 | 15 ± 2 |
| Fat (% totaldaily Kcal) | 46 ± 4 | 41 ± 2 | 27 ± 3 |
| Carbohydrate (% totaldaily Kcal) | 12 ± 2 | 33 ± 2 | 58 ± 4 |
| Protein (g/day) | 100 ± 11 | 74 ± 11 | 67.5 ± 9 |
| Fat (g/day) | 51 ± 9 | 50 ± 2 | 54 ± 6 |
| Carbohydrates (g/day) | 30 ± 0.2 | 91 ± 5 | 261 ± 18 |
2.2. Analysis
2.3. Statistical Analysis
3. Results
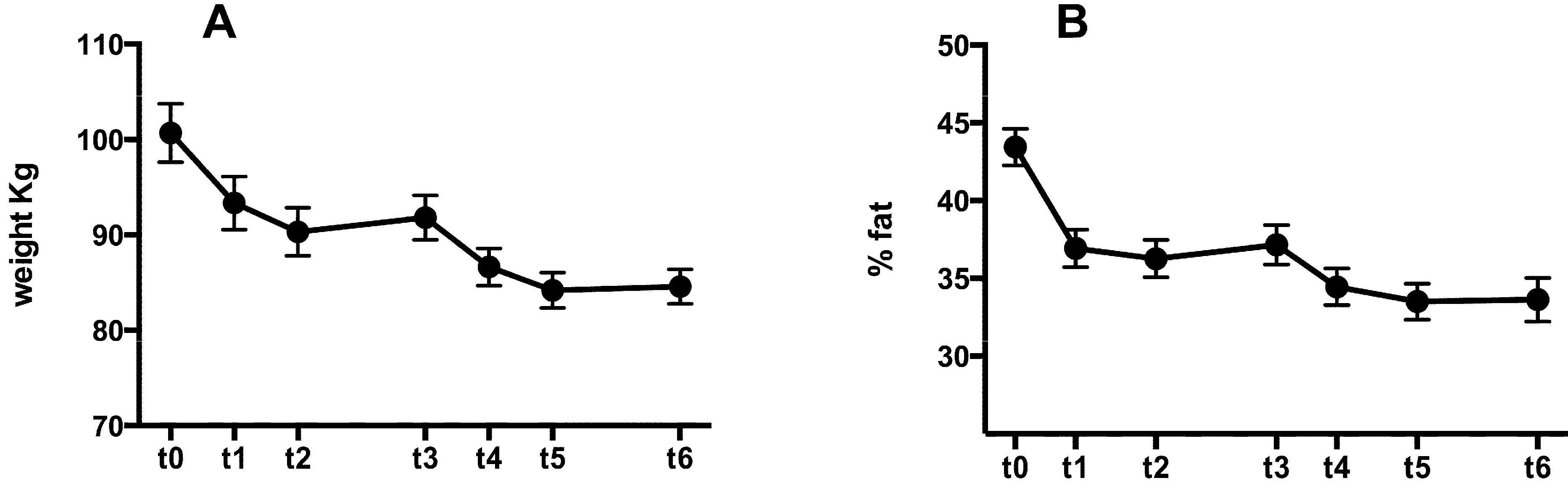
| Anthropometric Data | t0 | t1 | t2 | t3 | t4 | t5 | t6 |
|---|---|---|---|---|---|---|---|
| Body weight | 100.7 ± 16.54 | 93.34 ± 15.04 | 90.33 ± 13.57 | 91.81 ± 12.58 | 86.64 ± 10.56 | 84.2 ± 10.04 | 84.59 ± 9.71 |
| % Body fat | 43.44 ± 6.34 | 36.93 ± 6.49 | 36.26 ± 6.46 | 37.15 ± 6.82 | 34.46 ± 6.3 | 33.50 ± 6.18 | 33.63 ± 7.6 |
| Blood Parameters | t0 | t2 | t6 | t0 vs. t2 | t0 vs. t6 | t2 vs. t6 |
|---|---|---|---|---|---|---|
| Chol-tot | 193.2 ± 37.87 | 171.9 ± 31.94 | 179.8 ± 32.42 | p < 0.0001 | p = 0.0003 | n.s. |
| HDL-C | 43.03 ± 6.09 | 49.59 ± 8 | 44.59 ± 8 | p < 0.0001 | n.s. | p < 0.001 |
| LDL-C | 144.5 ± 58.4 | 108.0 ± 42.66 | 122.9 ± 42.25 | p < 0.0001 | p = 0.0004 | p < 0.0001 |
| TG | 112.7± 61.02 | 88.62 ± 40.65 | 95.45 ± 39.99 | p = 0.0006 | p = 0.0106 | n.s. |
| Glu | 102.6 ± 11.5 | 90.31 ± 8.45 | 95.31 ± 8.45 | p < 0.0001 | p = 0.0004 | p < 0.0001 |
| ALT | 18.75 ± 11.6 | 16.53 ± 6.72 | 17.11 ± 9.3 | n.s. | n.s. | n.s. |
| AST | 18 ± 8.69 | 17.13 ± 7.2 | 17.76 ± 5.43 | n.s. | n.s. | n.s. |
| GGT | 20.68 ± 16.16 | 16.1 ± 5.3 | 17.8 ± 6.8 | p = 0.012 | p < 0.05 | n.s. |
| Creatinine | 0.79 ± 0.16 | 0.76 ± 0.07 | 0.77 ± 0.1 | n.s. | n.s. | n.s. |
| BUN | 15.87 ± 3.83 | 16.1 ± 85.29 | 15 ± 3.87 | n.s. | n.s. | n.s. |
| Uric acid | 4.56 ± 0.86 | 4.2 ± 0.64 | 4.01 ± 0.91 | p < 0.01 | p < 0.05 | n.s. |
| SBP | 125 ± 10 | 117 ± 6 | 118 ± 4 | p < 0.01 | p < 0.01 | n.s. |
| DBP | 86 ± 5 | 82 ± 8 | 82 ± 5 | n.s. | n.s. | n.s. |
4. Discussion
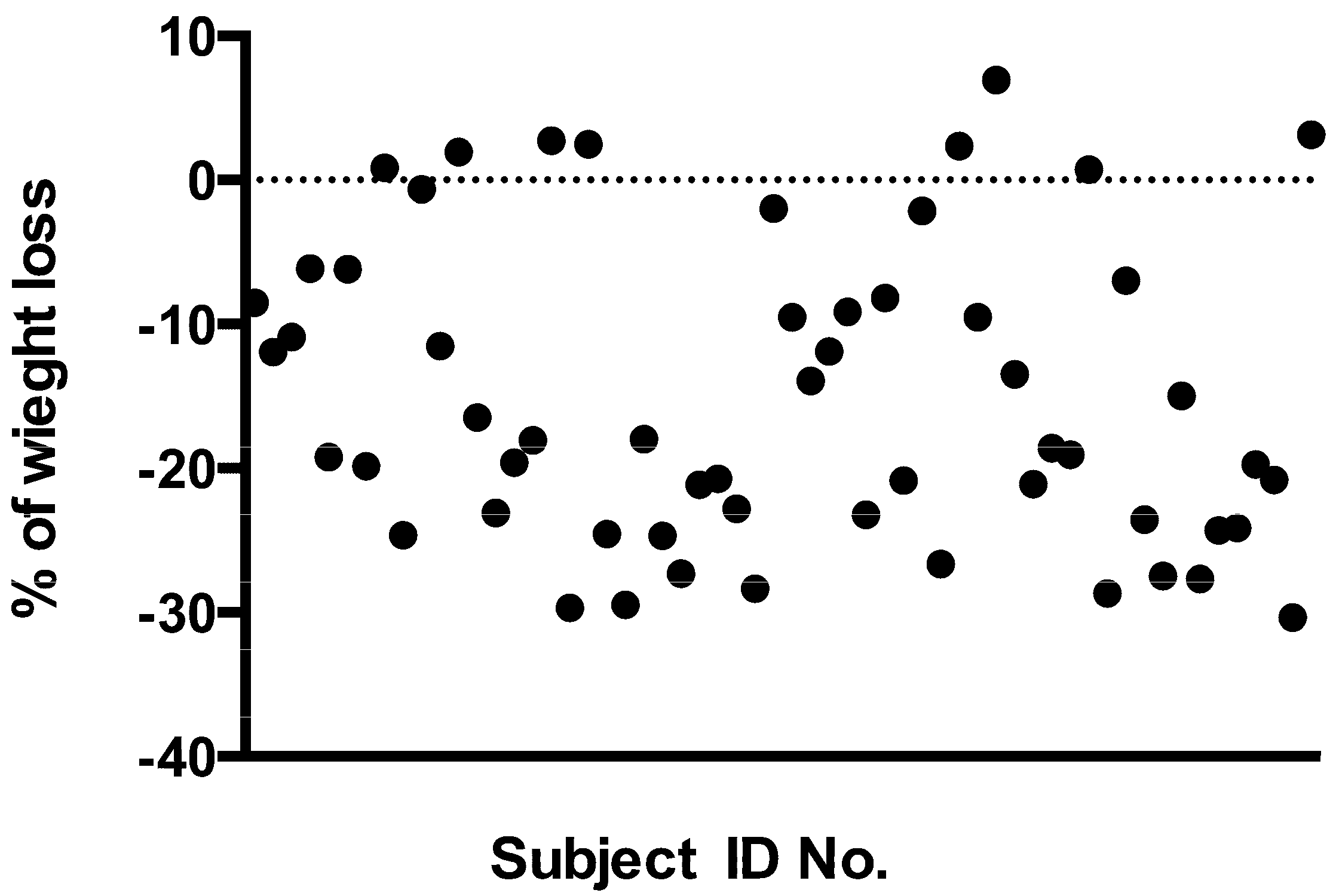
5. Conclusions
Ackowledgments
Conflicts of Interest
References
- Olshansky, S.J.; Passaro, D.J.; Hershow, R.C.; Layden, J.; Carnes, B.A.; Brody, J.; Hayflick, L.; Butler, R.N.; Allison, D.B.; Ludwig, D.S. A potential decline in life expectancy in the united states in the 21st century. N. Engl. J. Med. 2005, 352, 1138–1145. [Google Scholar] [CrossRef]
- Koh-Banerjee, P.; Wang, Y.; Hu, F.B.; Spiegelman, D.; Willett, W.C.; Rimm, E.B. Changes in body weight and body fat distribution as risk factors for clinical diabetes in us men. Am. J. Epidemiol. 2004, 159, 1150–1159. [Google Scholar] [CrossRef]
- Nordmann, A.J.; Nordmann, A.; Briel, M.; Keller, U.; Yancy, W.S., Jr.; Brehm, B.J.; Bucher, H.C. Effects of low-carbohydrate vs. low-fat diets on weight loss and cardiovascular risk factors: A meta-analysis of randomized controlled trials. Arch. Intern. Med. 2006, 166, 285–293. [Google Scholar] [CrossRef]
- Chahoud, G.; Aude, Y.W.; Mehta, J.L. Dietary recommendations in the prevention and treatment of coronary heart disease: Do we have the ideal diet yet? Am. J. Cardiol. 2004, 94, 1260–1267. [Google Scholar] [CrossRef]
- Paoli, A.; Rubini, A.; Volek, J.S.; Grimaldi, K.A. Beyond weight loss: A review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Eur. J. Clin. Nutr. 2013, 67, 789–796. [Google Scholar] [CrossRef]
- Veech, R.L. The therapeutic implications of ketone bodies: The effects of ketone bodies in pathological conditions: Ketosis, ketogenic diet, redox states, insulin resistance, and mitochondrial metabolism. Prostaglandins Leukot. Essent. Fat. Acids 2004, 70, 309–319. [Google Scholar] [CrossRef]
- Krebs, H.A. The regulation of the release of ketone bodies by the liver. Adv. Enzym. Regul. 1966, 4, 339–354. [Google Scholar] [CrossRef]
- Bueno, N.B.; de Melo, I.S.; de Oliveira, S.L.; da Rocha Ataide, T. Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: A meta-analysis of randomised controlled trials. Br. J. Nutr. 2013, 110, 1178–1187. [Google Scholar] [CrossRef]
- Janda, M.; Zeidler, D.; Bohm, G.; Schoberberger, R. An instrument to measure adherence to weight loss programs: The compliance praxis survey-diet (compass-diet). Nutrients 2013, 5, 3828–3838. [Google Scholar] [CrossRef]
- Sumithran, P.; Prendergast, L.A.; Delbridge, E.; Purcell, K.; Shulkes, A.; Kriketos, A.; Proietto, J. Ketosis and appetite-mediating nutrients and hormones after weight loss. Eur. J. Clin. Nutr. 2013, 67, 759–764. [Google Scholar] [CrossRef]
- Jeffery, R.W. Does weight cycling present a health risk? Am. J. Clin. Nutr. 1996, 63, 452S–455S. [Google Scholar]
- Sumithran, P.; Proietto, J. The defence of body weight: A physiological basis for weight regain after weight loss. Clin. Sci. 2013, 124, 231–241. [Google Scholar] [CrossRef]
- Paoli, A.; Cenci, L.; Fancelli, M.; Parmagnani, A.; Fratter, A.; Cucchi, A.; Bianco, A. Ketogenic diet and phytoextracts comparison of the efficacy of mediterranean, zone and tisanoreica diet on some health risk factors. Agro Food Ind. Hi-Tech 2010, 21, 24–29. [Google Scholar]
- Paoli, A.; Cenci, L.; Grimaldi, K.A. Effect of ketogenic mediterranean diet with phytoextracts and low carbohydrates/high-protein meals on weight, cardiovascular risk factors, body composition and diet compliance in Italian council employees. Nutr. J. 2011, 10, 112. [Google Scholar] [CrossRef]
- Paoli, A.; Grimaldi, K.; D’Agostino, D.; Cenci, L.; Moro, T.; Bianco, A.; Palma, A. Ketogenic diet does not affect strength performance in elite artistic gymnasts. J. Int. Soc. Sports Nutr. 2012, 9, 34. [Google Scholar] [CrossRef]
- Toeller, M.; Buyken, A.; Heitkamp, G.; Milne, R.; Klischan, A.; Gries, F.A. Repeatability of three-day dietary records in the eurodiab iddm complications study. Eur. J. Clin. Nutr. 1997, 51, 74–80. [Google Scholar]
- Gaby, A.R. Natural approaches to epilepsy. Altern. Med. Rev. 2007, 12, 9–24. [Google Scholar]
- Zupec-Kania, B.; Zupanc, M.L. Long-term management of the ketogenic diet: Seizure monitoring, nutrition, and supplementation. Epilepsia 2008, 49, 23–26. [Google Scholar] [CrossRef]
- Phinney, S.D.; Horton, E.S.; Sims, E.A.; Hanson, J.S.; Danforth, E., Jr.; LaGrange, B.M. Capacity for moderate exercise in obese subjects after adaptation to a hypocaloric, ketogenic diet. J. Clin. Investig. 1980, 66, 1152–1161. [Google Scholar] [CrossRef]
- Shai, I.; Schwarzfuchs, D.; Henkin, Y.; Shahar, D.R.; Witkow, S.; Greenberg, I.; Golan, R.; Fraser, D.; Bolotin, A.; Vardi, H.; et al. Weight loss with a low-carbohydrate, mediterranean, or low-fat diet. N. Engl. J. Med. 2008, 359, 229–241. [Google Scholar] [CrossRef]
- Black, K.E.; Skidmore, P.M.; Brown, R.C. Energy intakes of ultraendurance cyclists during competition, an observational study. Int. J. Sport Nutr. Exerc. Metab. 2012, 22, 19–23. [Google Scholar]
- Piccoli, A.; Brunani, A.; Savia, G.; Pillon, L.; Favaro, E.; Berselli, M.E.; Cavagnini, F. Discriminating between body fat and fluid changes in the obese adult using bioimpedance vector analysis. Int. J. Obes. Relat. Metab. Disord. 1998, 22, 97–104. [Google Scholar]
- Saunders, N.H.; al-Zeibak, S.; Ryde, S.J.; Birks, J.L. The composition of weight loss in dieting obese females by electrical methods. Int. J. Obes. Relat. Metab. Disord. 1993, 17, 317–322. [Google Scholar]
- GraphPad Prism, version 6.00; GraphPad Software Inc.: San Diego, CA, USA, 2013.
- Schwarzfuchs, D.; Golan, R.; Shai, I. Four-year follow-up after two-year dietary interventions. N. Engl. J. Med. 2012, 367, 1373–1374. [Google Scholar] [CrossRef]
- Maclean, P.S.; Bergouignan, A.; Cornier, M.A.; Jackman, M.R. Biology’s response to dieting: The impetus for weight regain. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 301, R581–R600. [Google Scholar] [CrossRef]
- Wing, R.R.; Hill, J.O. Successful weight loss maintenance. Annu. Rev. Nutr. 2001, 21, 323–341. [Google Scholar] [CrossRef]
- Thomas, P.R. Weighing the Options: Criteria for Evaluating Weight-Management Programs; National Academies Press: Washington, DC, USA, 1995. [Google Scholar]
- Feinman, R.D.; Fine, E.J. Nonequilibrium thermodynamics and energy efficiency in weight loss diets. Theor. Biol. Med. Model. 2007, 4, 27. [Google Scholar] [CrossRef]
- Westerterp-Plantenga, M.S.; Nieuwenhuizen, A.; Tome, D.; Soenen, S.; Westerterp, K.R. Dietary protein, weight loss, and weight maintenance. Annu. Rev. Nutr. 2009, 29, 21–41. [Google Scholar] [CrossRef]
- Paoli, A.; Grimaldi, K.; Bianco, A.; Lodi, A.; Cenci, L.; Parmagnani, A. Medium term effects of a ketogenic diet and a Mediterranean diet on resting energy expenditure and respiratory ratio. BMC Proc. 2012, 6, 37. [Google Scholar]
- Veldhorst, M.; Smeets, A.; Soenen, S.; Hochstenbach-Waelen, A.; Hursel, R.; Diepvens, K.; Lejeune, M.; Luscombe-Marsh, N.; Westerterp-Plantenga, M. Protein-induced satiety: Effects and mechanisms of different proteins. Physiol. Behav. 2008, 94, 300–307. [Google Scholar] [CrossRef]
- Johnstone, A.M.; Horgan, G.W.; Murison, S.D.; Bremner, D.M.; Lobley, G.E. Effects of a high-protein ketogenic diet on hunger, appetite, and weight loss in obese men feeding ad libitum. Am. J. Clin. Nutr. 2008, 87, 44–55. [Google Scholar]
- Wing, R.R.; Phelan, S. Long-term weight loss maintenance. Am. J. Clin. Nutr. 2005, 82, 222S–225S. [Google Scholar]
- Sumithran, P.; Prendergast, L.A.; Delbridge, E.; Purcell, K.; Shulkes, A.; Kriketos, A.; Proietto, J. Long-term persistence of hormonal adaptations to weight loss. N. Engl. J. Med. 2011, 365, 1597–1604. [Google Scholar] [CrossRef]
- Tagliabue, A.; Bertoli, S.; Trentani, C.; Borrelli, P.; Veggiotti, P. Effects of the ketogenic diet on nutritional status, resting energy expenditure, and substrate oxidation in patients with medically refractory epilepsy: A 6-month prospective observational study. Clin. Nutr. 2012, 31, 246–249. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Psaltopoulou, T.; Orfanos, P.; Hsieh, C.C.; Trichopoulos, D. Low-carbohydrate-high-protein diet and long-term survival in a general population cohort. Eur. J. Clin. Nutr. 2007, 61, 575–581. [Google Scholar]
- Jia, Y.; Hwang, S.Y.; House, J.D.; Ogborn, M.R.; Weiler, H.A.; O, K.; Aukema, H.M. Long-term high intake of whole proteins results in renal damage in pigs. J. Nutr. 2010, 140, 1646–1652. [Google Scholar] [CrossRef]
- Wakefield, A.P.; House, J.D.; Ogborn, M.R.; Weiler, H.A.; Aukema, H.M. A diet with 35% of energy from protein leads to kidney damage in female sprague-dawley rats. Br. J. Nutr. 2011, 106, 656–663. [Google Scholar] [CrossRef]
- Martin, W.F.; Armstrong, L.E.; Rodriguez, N.R. Dietary protein intake and renal function. Nutr. Metab. 2005, 2, 25. [Google Scholar] [CrossRef]
- Skov, A.R.; Haulrik, N.; Toubro, S.; Molgaard, C.; Astrup, A. Effect of protein intake on bone mineralization during weight loss: A 6-month trial. Obes. Res. 2002, 10, 432–438. [Google Scholar] [CrossRef]
- Welle, S.; Nair, K.S. Relationship of resting metabolic rate to body composition and protein turnover. Am. J. Physiol. 1990, 258, E990–E998. [Google Scholar]
- Poplawski, M.M.; Mastaitis, J.W.; Isoda, F.; Grosjean, F.; Zheng, F.; Mobbs, C.V. Reversal of diabetic nephropathy by a ketogenic diet. PLoS One 2011, 6, e18604. [Google Scholar]
- Cahill, G.F., Jr. Fuel metabolism in starvation. Annu. Rev. Nutr. 2006, 26, 1–22. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Kouris-Blazos, A.; Wahlqvist, M.L.; Gnardellis, C.; Lagiou, P.; Polychronopoulos, E.; Vassilakou, T.; Lipworth, L.; Trichopoulos, D. Diet and overall survival in elderly people. BMJ 1995, 311, 1457–1460. [Google Scholar] [CrossRef]
- Perona, J.S.; Cabello-Moruno, R.; Ruiz-Gutierrez, V. The role of virgin olive oil components in the modulation of endothelial function. J. Nutr. Biochem. 2006, 17, 429–445. [Google Scholar] [CrossRef]
- Richard, C.; Couture, P.; Desroches, S.; Lamarche, B. Effect of the mediterranean diet with and without weight loss on markers of inflammation in men with metabolic syndrome. Obesity 2013, 21, 51–57. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Paoli, A.; Bianco, A.; Grimaldi, K.A.; Lodi, A.; Bosco, G. Long Term Successful Weight Loss with a Combination Biphasic Ketogenic Mediterranean Diet and Mediterranean Diet Maintenance Protocol. Nutrients 2013, 5, 5205-5217. https://doi.org/10.3390/nu5125205
Paoli A, Bianco A, Grimaldi KA, Lodi A, Bosco G. Long Term Successful Weight Loss with a Combination Biphasic Ketogenic Mediterranean Diet and Mediterranean Diet Maintenance Protocol. Nutrients. 2013; 5(12):5205-5217. https://doi.org/10.3390/nu5125205
Chicago/Turabian StylePaoli, Antonio, Antonino Bianco, Keith A Grimaldi, Alessandra Lodi, and Gerardo Bosco. 2013. "Long Term Successful Weight Loss with a Combination Biphasic Ketogenic Mediterranean Diet and Mediterranean Diet Maintenance Protocol" Nutrients 5, no. 12: 5205-5217. https://doi.org/10.3390/nu5125205
APA StylePaoli, A., Bianco, A., Grimaldi, K. A., Lodi, A., & Bosco, G. (2013). Long Term Successful Weight Loss with a Combination Biphasic Ketogenic Mediterranean Diet and Mediterranean Diet Maintenance Protocol. Nutrients, 5(12), 5205-5217. https://doi.org/10.3390/nu5125205








